Abstract
Objectives:
The primary treatment for varicose ulcers is to address the underlying cause of venous hypertension, additionally, split-thickness skin transplantation (ST) is an effective option for large ulcers. The aim of this study was to compare the efficacy of venous surgery with or without split-thickness ST in the management of varicose ulcers (diameter ≥6 cm).
Methods:
Patients with varicose ulcers between 2013 and 2023 were included in the real-world retrospective cohort study. The endovenous microwave ablation/endovenous laser ablation-high ligation-foam sclerotherapy-compression therapy + ST group (EMA/EVLA-CT-HL-FS + ST) consisted of 32 patients, while the EMA/EVLA-CT-HL-FS group consisted of 48 patients.
Results:
A difference in ulcer healing time was found between the two groups (the hazard ratio [HR] was 4.4, 95% CI [2.3–8.2], P < 0.0001). However, the healing rate did not differ between the two groups at 6 and 12 months postoperatively.
Conclusion:
ST has been shown to expedite ulcer healing and ameliorate the symptoms of VVs in the early stages of treatment, while its intermediate to long-term efficacy for ulcer healing and overall patient quality of life may be limited.
Keywords
Introduction
Varicose ulcers of the lower extremities are prevalent chronic conditions that pose a significant public health concern. The long-term incurability of many cases results in substantial economic burdens on both society and affected families, making it a pressing issue for health care systems worldwide.1–3 Venous hypertension is the underlying pathophysiological mechanism of varicose ulcers. Surgery and compression therapy (CT) of various veins (VVs) aimed at mitigating venous hypertension in the lower extremities present opportunities for promoting the healing of varicose ulcers.4–7 However, extensive ulcers have prolonged healing times following venous surgery.8,9 The implementation of skin transplantation (ST) on an ulcerated surface is believed to facilitate ulcer healing.10,11
The primary conservative approach for reducing venous hypertension in the lower extremities is CT, which involves the use of gradient compression stockings or bandages to augment venous return and mitigate oedema.4,12–14 Venous surgical techniques include high ligation (HL) and stripping of the great saphenous vein (GSV)5,15; endovenous ablation using techniques such as laser, microwave or radiofrequency16–20; and foam sclerotherapy (FS) for VVs.6,21 Additionally, comprehensive surgical methods have been further developed to better manage VVs.15,22,23 Venous surgery with simultaneous ST has been reported as a treatment approach for varicose ulcers. 24 However, there is currently no literature reporting on the combined approach of HL of the GSV, thermal ablation of the main trunk of the GSV, FS for superficial VVs, and subsequent ST. In this study, endovenous microwave ablation/endovenous laser ablation-high ligation-foam sclerotherapy-compression therapy + skin transplantation (EMA/EVLA-HL-FS-CT + ST, ST group) and EMA/EVLA-HL-FS-CT (non-ST group) were utilized for the management of varicose ulcers measuring ≥6 cm, and the rate of ulcer healing within 1 year posttreatment were compared.
Methods
Patient population
We retrospectively analysed the 1-year follow-up data of a real-world cohort study of EMA/EVLA-HL-FS-CT + ST versus EMA/EVLA-HL-FS-CT for varicose ulcers. This retrospective cohort study was conducted at a regional medical centre, a 3200-bed general university-affiliated hospital, and was approved by the ethics committee and institutional review board of the Ganzhou People's Hospital (Approval Number: TY-ZKY2024-011-01). All procedures performed in studies were in accordance with the Helsinki Declaration of 1975 as revised in 2013. The reporting of this study conformed to the STROBE guideline. 25 Consecutive patients with varicose ulcers who were treated with EMA/EVLA-HL-FS-CT + ST or EMA/EVLA-HL-FS-CT at Ganzhou People's Hospital between 2013 and 2023 were included in this retrospective study and all the patients’ details have been de-identified. They were divided into two groups based on whether skin grafting was conducted: the ST group and the non-ST group. The baseline characteristics of the patients included sex, age, course of varicose veins, ulcer duration, ulcer area, ulcer location, reflux vessel, number of perforators and baseline VCSS. The exclusion criterion was an ulcer diameter <6 cm and patients with cellulitis or local infection.
Procedures
The procedures included EMA/EVLA-HL-FS-CT + ST or EMA/EVLA-HL-FS-CT. The same procedures used for the two groups were HL of the GSV, laser or microwave thermal ablation of the GSV trunk, FS injection of the VVs and CT after the operation. In the ST group, a split-thickness ST was concomitantly performed during venous surgery. The surgery was performed by a dedicated surgical team. Patients who met the inclusion criteria were thoroughly informed about the procedure and provided written consent for these specific interventions.
All VVs were preoperatively identified and labelled. After epidural or general anaesthesia, an oblique incision was made in the groin to ligate all collateral branches of the GSV, and HL was performed at the great saphenous femoral junction. 5 The sclerosing agent foam, prepared with a 1:4 ratio of 1% lauromacrogol to CO2 using the Tessari technique, was injected into the VVs. 26 FS was injected into the vessel lumen surrounding the ulcer and perforator vessels, with ultrasound guidance for accurate placement. 27 The maximum recommended dosage of lauromacrogol administered to a single limb typically does not exceed 10 ml.
EMA procedure
The GSV was punctured at the medial malleolus, and a microwave ablation needle (Shanxi Danhui Biotechnology Company, Shanxi, China) was inserted into the GSV. The ablation needle was advanced up the GSV to the groin. The distal end of the needle was linked to the microwave generator (HBS-B, Nanjing Huabai Electronic Medical, Nanjing, China). Saline solution for swelling infiltration was injected around the GSV from 10 cm below the knee to the groin, ensuring that there was at least a 1 cm distance between the vein and the skin. At a power of 40 W and a withdrawn speed of 1 cm/6 s, the microwave generator was activated to close the GSV trunk from 10 cm below the knee to the groin. The microwave wire was withdrawn 1 cm each time. For patients who were unable to undergo cephalad advancement of the microwave needle from the ankle, we elected to proceed with a retrograde approach by inserting the microwave needle downwards through the inguinal incision, followed by cauterization of the GSV. Local vein resections were performed for GSV and VVs larger than 1 cm in diameter or with tumor-like expansion. 28
EVLA procedure
The trunk of the GSV was punctured at the medial malleolus, and the laser fibre was inserted into the trunk of the GSV. At a power of 15 W and a withdrawn speed of 0.3 cm/sec with manual pressure being applied to achieve venous wall apposition around the laser fibre tip, 29 the 810 nm laser (AngioDynamics, Germany) was activated to close the GSV trunk from 10 cm below the knee to the groin. For those patients in whom the guidewire could not be inserted upwards from the ankle, such as those with local infection or stenosis of the GSV, we chose to insert the guidewire downwards into the GSV at the inguinal incision and retrogradely or segmentally cauterize the GSV using a laser. 26 Local veins resections were also performed like EMA. 28
For patients with ulcer ST, split-thickness skin grafts of matching dimensions were harvested from the anterior upper thigh on the same side and meticulously prepared into 5 mm skin grafts for subsequent application. After debridement of the ulcer, the wound bed was completely covered with skin grafts. Grafted and nongrafted ulcers were dressed with vaseline gauze and sterile dressing. Following the operation, the limbs were enveloped with a 4-layer compression bandage. Three days later, they were replaced with grade II (30–40 mmHg) compression stockings, with the wound covered with thin dressing. We suggested the CT of stockings should be maintained for more than 1 year.
All patients received postoperative prophylactic anticoagulant therapy with low molecular weight heparin. Sensitive antibiotics were administered to some patients with positive cultures of ulcer secretions and enlarged inguinal lymph nodes to prevent surgical site infection.
Discharge criteria
The patient could be mobile after 6 h of bed rest following spinal or general anaesthesia. If there were no complications such as wound bleeding, infection, DVT, etc. which require prolonged hospitalization, the patient was discharged 3 days after the procedure.
Follow-up
The follow-up was conducted by the attending physicians involved in the clinical management of the patients. Patients were scheduled for follow-up appointments at the outpatient clinic 1 month, 6 months, and 1 year after the intervention. During these visits, data on ulcer healing time and ulcer recurrence were meticulously documented. The follow-up methods included physical examination, and questionnaire survey. Follow-up personnel meticulously documented the patients’ reported time to ulcer healing or recurrence during the follow-up period. The assessment of the impact of venous disease on quality of life (QoL) was conducted using the venous clinical severity score (VCSS). 30 The VCSS is a validated and sensitive tool for evaluating the severity of venous insufficiency, with scores ranging from 0 (indicating no significant venous lesions) to 30.
Outcomes
The primary outcome was the ulcer healing time. The ulcer healing time was operationally defined as the interval from the initiation of the intervention to complete re-epithelialization of the ulcer. The complete healing of leg ulcers is characterized by epithelialization. 26 The secondary outcomes included the ulcer healing rate at 1, 6, and 12 months after the operation, the ulcer recurrence rate at 12 months after surgery and the VCSSs of patients at 1, 6, and 12 months following surgery, as well as the complications of ST. The recurrence of ulcers refers to the reappearance of ulceration at the site of previous healing, presenting as defects in the skin tissue.
Statistical analysis
All the data were meticulously validated and entered into Excel sheets to establish a robust database. The homogeneity of variance in the measurement data was assessed using the two independent samples t test, while the heterogeneity of variance was evaluated using the rank sum test (also known as the Mann‒Whitney U test). Count data were analysed through the χ2 (chi-squared) test, a statistical method commonly used for categorical data analysis. Kaplan-Meier survival curve analysis was utilized to compare the difference in ulcer healing time between the two groups, and GraphPad Prism 8 was used for data visualization. The tests were conducted using a two-tailed approach, with a significance level set at 0.05. Statistical analyses were carried out utilizing IBM SPSS Statistics software, version 26.
Results
Patient characteristics
From 2013 to 2023, a total of 515 patients with varicose ulcers underwent surgical treatment at our hospital. Among them, 435 patients had ulcers <6 cm, while the remaining 80 patients were divided into two groups: the ST group with 32 patients and the non-ST group with 48 patients. After 1 year of follow-up, one patient in the ST group was lost to follow-up, and two patients in the non-ST group were lost to follow-up (Figure 1). The mean ages of the patients in the two groups were 61.7 ± 10.1 years and 62.1 ± 9.7 years, respectively. The average durations of the ulcers were 37.6 ± 87.2 months and 58.9 ± 116.4 months, while the mean ulcer area was 57.3 ± 26.4 cm2 and 57.4 ± 27.4 cm2, respectively. The baseline VCSSs were 15.2 ± 3.0 and 15.0 ± 3.3, respectively. There were no statistically significant differences in the baseline characteristics between the two groups (Table 1).

Patient flowchart.
Baseline characteristics in the in the two groups.

Note: EMA/EVLA-HL-FS-CT + ST, endovenous microwave ablation/endovenous laser ablation-high ligation-high ligation – foam sclerotherapy -compression therapy + skin transplantation; y, year; mo, month; VCSS, Venous Clinical Severity Score.
*Independent-sample t-test.
Pearson Chi-Square.
Mann–Whitney U test.
Primary outcome
The median time to ulcer healing was 26.3 days (range: 15.0–24.5 days) in the ST group, while the median time to ulcer healing was 69.2 days (range: 31.0- 87.0 days) in the non-ST group (Table 2). A difference in ulcer healing time was detected between the two groups (the hazard ratio [HR]for ulcer healing was 4.4, 95% CI [2.3–8.2], P < 0.0001) (Figure 2).
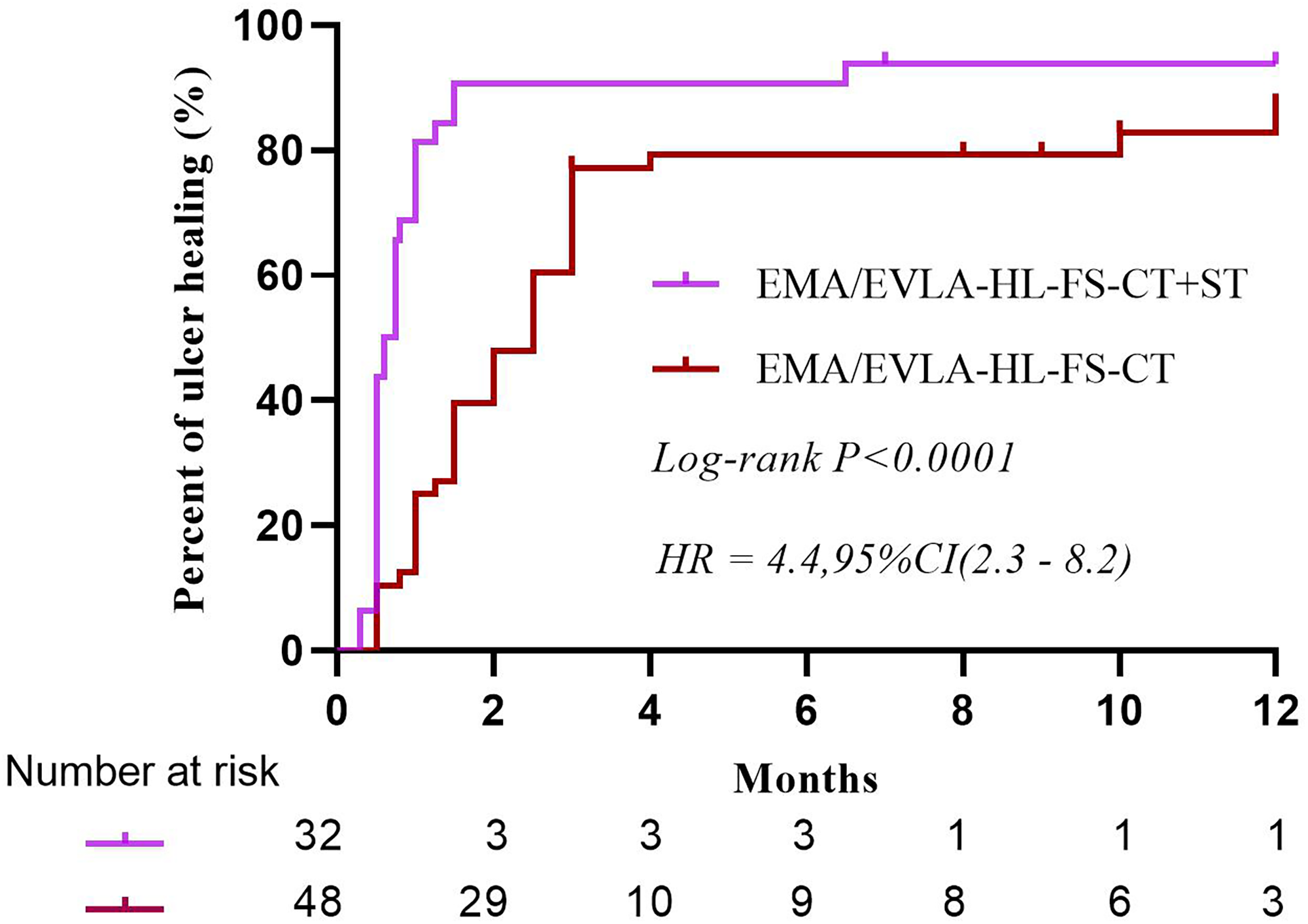
Ulcer healing time of the two groups.
The follow-up outcomes in the two groups.

Note: EMA/EVLA-HL-FS-CT + ST, endovenous microwave ablation/ endovenous laser ablation-high ligation-high ligation – foam sclerotherapy -compression therapy + skin transplantation; IQR, interquartile range; VCSS, Venous Clinical Severity Score; NA, not applicable.
*Independent-sample t-test.
Fisher's Exact Test.
Mann–Whitney U test.
Secondary outcomes
The ulcer healing rates of the two groups at 1, 6 and 12 months after the operation were 87.5%, 96.9%, 96.9% and 25.0%, 85.4%, 89.6%, respectively. There was a significant difference in the ulcer healing rate between the two groups at 1 month after the operation (P < 0.001), but there was no significant difference at 6 months or 12 months after the operation. One year after surgery, ulcers recurred in 1 patient and in 3 patients in the two groups, respectively, with no statistically significant difference detected (Table 2). Additionally, two patients of skin donor site infection emerged, and they achieved scarred healing within a brief period; Necrosis of the grafting skin emerged in four patients.
Disease-specific QoL assessment
The VCSS (Figure 3) at 1 month after the operation was significantly lower than that before the operation in the respective groups (p = 0.0007). However, there were no differences in VCSS between the two groups at 6 and 12 months (p = 0.051 and 0.522, respectively).

VCSS of the two groups at different time.
Discussion
Skin transplantation can significantly expedite the healing of varicose ulcers and ameliorate the symptoms of varicose veins in the early stages of treatment. However, after six months of treatment, compared with the non-ST group, the ST group exhibited no improvement in terms of ulcer healing or QoL. This suggests that while ST may offer initial benefits, its intermediate to long-term efficacy for ulcer healing and overall patient QoL may be limited.
ST is a commonly performed surgical procedure for the management of recalcitrant skin wounds involving the placement of skin or skin substitutes at the wound site to replace and facilitate the regeneration of damaged tissue. 31 Medical professionals have long performed ST on varicose ulcers as a means to promote ulcer healing.32,33 Currently, some studies still support the efficacy of ST in treating refractory varicose ulcers and have demonstrated favourable outcomes.34,35
However, towards the end of the previous century, plastic surgeons conducted a comparative study on the effectiveness of ST for different types of lower extremity ulcers. They discovered that patients with varicose ulcers had poorer healing outcomes because the underlying cause was not addressed. 33 Clinical guidelines 29 and reviews 30 did not advocate ST as the primary intervention for varicose ulcers. The key to successful management of varicose ulcers lies in identifying and addressing the underlying aetiology, such as chronic venous insufficiency or venous hypertension. 36
The primary aetiology of varicose ulcers is chronic venous insufficiency, which results from various factors, such as valvular incompetence and calf muscle pump dysfunction. This leads to elevated ambulatory venous pressure, particularly in the ankle region, causing microcirculatory disturbances and subsequent tissue damage, ultimately culminating in ulceration.9,37 If venous hypertension is not alleviated, the ulcer will persist, progress in depth, and ultimately develop into a refractory ulcer. 38 Various surgical interventions, such as endovenous ablation, FS, and phlebectomy, along with CT aimed at reducing venous hypertension in the lower extremities, facilitate the healing of varicose ulcers.6,39,40 However, despite the implementation of lower extremity venous pressure reduction, we still frequently encounter chronic ulcers, particularly large ulcers with a diameter exceeding 6 cm. Ulcers with a diameter >6 cm were also assigned the highest VCSS of 3 points 30 ; therefore, they were selected for this study.
Shortening the healing time of varicose ulcers has been our longstanding objective. In our previous study, we compared the clinical efficacy of CT alone with that of combined EVLA in the treatment of active varicose ulcers and found that combined EVLA resulted in a reduced ulcer healing time. 26 Later, we also conducted a comparative analysis of the clinical efficacy of traditional HL and EVLA of the GSV in the treatment of varicose ulcers and subsequently concluded that EVLA has the potential to expedite ulcer healing. 41 Some researchers have also conducted comparative studies on the efficacy of EMA and HL therapy in the treatment of varicose ulcers and have observed that the ulcer healing time was significantly shorter in the EMA group than in the HL group. 7
EMA/EVLA is a thermal ablation technique used to treat the GSV, causing damage to the reflux endothelium and leading to contraction and subsequent fibrosis until complete occlusion of the vein.17,42 FS has favourable effects on superficial VVs, periulcer varicose veins, and perforating veins.6,21,43 HL further blocked the reflux of the GSV.5,44 These interventions can synergistically inhibit venous reflux in the lower extremities, reduce venous pressure, mitigate the deleterious effects of high venous pressure on the skin, and facilitate the healing of lower extremity varicose ulcers. 45 Importantly, HL can prevent the detachment of thrombus formed in the proximal GSV after EVLA or EMA ablation, thus reducing the risk of endovenous heat-induced thrombosis (EHIT) 46 and pulmonary embolism. 47
This time, we implemented autologous split-thickness ST for varicose ulcers with a diameter ≥6 cm while maintaining consistency in the other treatments. By comparison, we observed a significantly shorter healing period in the ST group than in the non-ST group. Additionally, there was a marked improvement in the patients’ QoL following the surgical procedures. It seemed that the incorporation of ST confers a conspicuous advantage. However, these advantages were predominantly evident in the initial month following treatment. There were no discernible discrepancies in the ulcer healing rate or VCSS between the two groups at the 6-month or 1-year follow-ups. In other words, ST does not appear to confer any benefit to patients with varicose ulcers in the medium-term following intervention and may even exacerbate complications at the donor site and the recipient site. 34 In this sense, some clinicians are opposed to utilizing skin grafts for varicose ulcers and instead advocate prioritizing aetiological treatment.36,48 There is no guideline in which ST is recommended as the first-line treatment for varicose ulcers. However, split-thickness ST with continued compression is recommended for selected large venous leg ulcers that have failed to show signs of healing with 4 to 6 weeks of standard care. 49
Our study has several limitations. First, this was a single-centre retrospective study, and the sample size appeared to be small. Second, the follow-up period was restricted to only 12 months. It is imperative to conduct an extended follow-up to substantiate and validate the observed benefits in both cohorts. Furthermore, the utilization of a combination of methods to reduce venous pressure in the lower extremities, such as HL for GSV and FS injection for VVs, raises concerns about potential overmanipulation. Further research is required to elucidate the impact of combining ST with surgical techniques on the long-term prognosis of patients with varicose ulcers.
Conclusion
Nevertheless, based on comparative analyses, it can be inferred from existing data that in patients with recalcitrant large varicose ulcers, traditional surgical interventions aimed at mitigating lower limb venous hypertension coupled with ST may accelerate ulcer resolution and improve patients’ short-term QoL. However, protracted follow-up is necessary to ascertain its sustained efficacy.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241283352 - Supplemental material for Comparison of the effect of combination therapies in the treatment of large varicose ulcers: A real-world cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504241283352 for Comparison of the effect of combination therapies in the treatment of large varicose ulcers: A real-world cohort study by Fangfang Wen, Hailiang Xie, Minggui Lai, Guofu Zheng, Yuancai Xie and Xiaochun Liu in Science Progress
Footnotes
Acknowledgements
Thanks to American Journal Experts (AJE) for the English polishing of the paper.
Author contributions
Fangfang Wen and Xiaochun Liu conceived the study, made the design, performed the statistical analyses, and drafted the manuscript. Hailiang Xie and Xiaochun Liu participated in the design and statistic work. Hailiang Xie, Guofu Zheng and Minggui Lai collected data. Guofu Zheng, Yuancai Xie and Xiaochun Liu revised the paper. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author XCL. The data are not publicly available due to the containing information that could compromise the privacy of research participants.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Bureau of Science and Technology of Ganzhou Municipality Research Foundation (No: GZ2023ZSF014) and the Science and Technology Program of Jiangxi Health Commission (No: 202410094). The funders provided financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
