Abstract
The absence of improvement in survival rates across various cancers, including laryngeal cancer, has led to an increasing interest in understanding the immune response to cancer. In head and neck cancers, immune modulatory mechanisms such as immune microenvironment and immune infiltration are important in cancer pathogenesis. This study aims to explore the distribution of tumor-infiltrating lymphocyte (TIL) subgroups in the immune microenvironment and evaluate their impact on tumor histopathological characteristics and prognosis. The study included 50 patients who underwent laryngectomy for laryngeal squamous cell carcinoma, in Istanbul University - Cerrahpaşa, Faculty of Medicine Department of Otorhinolaryngology, between January 2016 and January 2018. Pathology specimens were evaluated using immunohistochemistry to assess the expressions of the CD3, CD20, CD8, CD4, CD25, and FoxP3 markers, identifying subgroups of TILs. The investigation aimed to uncover how these subgroups influence tumor histopathological features and survival outcomes. The high infiltration of CD3, CD20, and CD4 had a positive impact on disease-specific survival, disease-free survival, and recurrence-free survival. In addition, overall survival was positively affected by high CD3 and CD4 infiltrations. However, no significant relationship was observed between the expressions of CD8, FoxP3, and CD25 and any of the survival parameters. The infiltration of CD3, CD20, and CD4 positive cells indicative of a robust antitumoral immune response—emerged as favorable prognostic factors in laryngeal cancer. These findings suggest that enhancing the infiltration of CD3, CD20, and CD4 lymphocytes could be a therapeutic strategy worth exploring in clinical trials.
Keywords
Introduction
Laryngeal cancer is one of the most prevalent head and neck cancers (HNCs) with an overall 5-year survival rate of 67%. 1 Despite diagnostic and therapeutic advances over the past half-century, the mortality rates for many malignancies, including laryngeal cancer, have not significantly decreased. This has prompted interest in understanding the immune mechanisms involved in cancer pathogenesis. 2
Cancer cells, constituting around 30% of a tumor, rely on the remaining cells and stromal components for survival and proliferation. These stromal elements, together with components of the immune system, form the tumor microenvironment (TME), which can either support or inhibit tumor growth. 3 The interplay between cancer cells and immune microenvironment elements at the receptor level serves as the basis for immune system escape routes. Examples of these mechanisms include decreased expression of markers that facilitate immune recognition of cancer cells and increased expression of ligands that inhibit effector immune responses.4,5 In the area of HNCs, the concept of immunosuppression is gaining credibility, supported by increasing evidence of the involvement of immunomodulatory mechanisms in cancer pathogenesis. 6
Tumor-infiltrating lymphocytes (TILs) are the components of the TME responsible for regulating the antitumor immune response. T lymphocytes (identified by CD3 positivity) particularly cytotoxic T lymphocytes (CD8+ T lymphocytes, Tcyt) are crucial part of this response. 7 T helper 1 (Th1) cells, a subset of CD4+ T lymphocytes, attract and activate cytotoxic T lymphocytes through various cytokines, especially IL-2. 8 On the contrary, regulatory T lymphocytes (Treg), another subset of CD4+ T lymphocytes that express CD25 (interleukin 2 receptor alpha) and FoxP3 (Forkhead box P3), suppress immune responses. 9 Although B lymphocytes, identified by CD20 expression, typically play a limited role in the immune response against cancer cells, their presence in the TME may suggest a positive prognostic effect. 10
HNCs with increased levels of CD8+ T lymphocyte infiltration have been associated with favorable clinical outcomes and prognosis. 11 Conversely, tumors with a higher CD4+ to CD8+ T lymphocyte ratio are typically more aggressive. 12 However, the precise role of CD4+ T lymphocytes in cancer immunology is still contraversial13,14; the complexity of their involvement can be attributed to the diversity of CD4+ lymphocyte subtypes present in the TME, such as Tregs. 15
Research on the TME, focusing on the TIL distribution is steadily increasing. Studies have focused on different subtypes of HNC, with particular attention paid to more immune-infiltrated tumors, notably oropharyngeal carcinoma. Consequently, our aim was to explore the association of TIL distribution and Treg density with the clinical and histopathological features of laryngeal tumors and their impact on survival parameters.
Materials and methods
In this retrospective cohort study, we adhered to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines in the design, execution, and reporting. 16
Ethical considerations
A Clinical Research Ethics Committee Approval Form was obtained from Cerrahpaşa Faculty of Medicine at Istanbul University - Cerrahpaşa [Document Number: 2019-122461, Document Date: 08.08.2019] prior to the commencement of the study. Written informed consent forms were obtained from all patients before surgery. This study was funded by the Research Projects Coordination Unit of the faculty.
Statistical analysis
SPSS 19.0 software was used for statistical analysis. Descriptive statistics were presented as numbers and percentages for categorical data and as means, standard deviations, median values, minimum, and maximum values for numerical data. The comparison of categorical variables between groups was performed using Pearson Chi-Square Analysis. For numerical variables not satisfying the normal distribution, the Mann–Whitney U Test was used for two independent groups, and the Kruskal–Wallis Test for more than two independent groups. The statistical alpha significance level was accepted as p < 0.05. As the parametric test requirement for correlation analysis between numerical variables could not be satisfied, Spearman Correlation Analysis was applied.
The Kaplan–Meier method was employed to assess the overall survival (OS), disease-specific survival (DSS), disease-free survival (DFS), and recurrence-free survival (RFS). P-value calculated with the Breslow (Generalized Wilcoxon) method was considered significant if <0.05. Hazards Ratio was calculated by Cox Regression Analysis, and again, a p < 0.05 value was considered significant.
Patient selection and clinical evaluation
Fifty patients diagnosed with laryngeal squamous cell carcinoma, who underwent partial or total laryngectomy in Istanbul University - Cerrahpaşa, Cerrahpaşa Faculty of Medicine Department of Otorhinolaryngology, between January 2016 and January 2018, were enrolled in the study consecutively, unless they met one of the exclusion criteria. Patients were excluded if they had a history of another malignancy, prior chemotherapy or radiation treatment, or if they had hepatitis or HIV infection. Lateral neck dissection was performed based on the location of the tumor and lymph node status. All surgeries performed by a specialized team in HNC treatment.
The 8th edition of the American Joint Committee on Cancer (AJCC) Tumor Node Metastasis (TNM) staging was used for both pathological and clinical staging. Pathology specimens were examined by a single pathologist specialized in HNC pathology.
Demographic data, histopathological characteristics of the tumors, such as the tumor location, TNM stage, histological grade, the presence of vascular/lymphatic/perineural/cartilage invasion, and extranodal extension, were assessed (Table 1). The type and the date of the surgery, the presence of recurrence and/or metastases, and the date of event were noted. Surveillance parameters as OS, DSS, DFS, and RFS were calculated. Immunohistochemistry for CD3 (T lymphocyte), CD4 (Th), CD8 (Tcyt), CD20 (B lymphocyte), CD25 (Treg), and FoxP3 (Treg) expressions were used to evaluate the extent of the inflammatory response in the tumor tissue and the distribution of TIL subtypes.
Summary of clinical and histopathological data.

Immunohistochemical staining
After evaluating hematoxylin-eosin (Biognost, Zagreb, Croatia) stained slides of the cases under a light microscope (Olympus, Tokyo, Japan), one paraffin block was selected for each case, containing sufficient tumor tissue and suitable for immunohistochemical study. To determine the immunohistochemical staining properties, seven positively charged slides (Epiglass, Istanbul, Turkey) were obtained from the selected paraffin blocks in 2-micron sections, using a microtome knife Patho-Cutter II (Erma, Tokyo, Japan). The two-micron sections on a positively charged slide were kept in an oven (Hybaid, UK) set at 56 degrees for 2 h.
An automated staining device (Ventana Benchmark XT, Tuscon, AZ, USA) was used for immunohistochemical staining. For the procedure, a ready kit, UltraView Universal DAB Detection Kit (Ventana, Basel, Switzerland), containing biotin-free HRP multimeter-based hydrogen peroxide and 3,3-diaminobenzidine tetrachloride (DAB) chromogen suitable for the device, was used.
Monoclonal antibodies including CD3 (T3) [LN10] RTU Bond 7 mL (LEICA/Novocastra, Illinois, USA), CD4 (T4) [4B12] RTU Bond 7 mL (LEICA/Novocastra, Illinois, USA), CD8 (T8) [Clone:4B11] RTU Bond 7 mL (LEICA/Novocastra, Illinois, USA), CD20 (B1) [L26] RTU Bond 7 mL (LEICA/Novocastra, Illinois, USA), CD25 (IL2R.1) [4C9], (LEICA/Novocastra, Illinois, USA), and FoxP3 [EP340] Conc. 0.1 mL (1:100-200) (Epitomics, Nanterre, France) were applied to the slides.
Counterstaining was performed with hematoxylin solution (Biognost, Zagreb, Croatia) in the dyeing device and the process was completed after dehydration and xylene stages. The slide was sealed with entellan and made ready for evaluation under the light microscope.
Immunohistochemical evaluation
Initially, five areas with over 70% cellularity, minimal necrosis, and more than 50% tumor parenchyma were chosen from hematoxylin-eosin-stained tumor sections for each patient. These same areas were identified in slides subjected to immunohistochemistry with CD3, CD4, CD8, CD20, CD25, and FoxP3 monoclonal antibodies. Evaluations were conducted under a light microscope (Olympus, Tokyo, Japan) at 200x magnification (20x objective lens), with TILs stained using the mentioned dyes manually counted by a pathologist experienced in HNC pathology. As lymphocytes infiltrating the tumor parenchyma have demonstrated a closer association with prognosis, only these were considered. 17 Specifically, B lymphocytes (CD20+ cells), which typically do not infiltrate the tumor parenchyma, were counted in the tumor stroma.
Classification of tumor-infiltrating lymphocytes
CD3+, CD4+, CD8+, CD20+, CD25+, and FoxP3+ TILs were counted in five 200x magnification fields in a 0.05 mm diameter area and average counts were used for evaluation. Additionally, CD4/CD8 and FoxP3/CD8 ratios were calculated. Following the approach of similar studies,9,18 median values were computed for each marker, and tumors with counts higher than the median for a specific marker were categorized as the high infiltration group (e.g. CD3-high); while those with lower counts were classified as the low infiltration group (e.g. CD3-low). For each marker, these two groups were compared in terms of clinical, pathological, and survival parameters.
Results
Fifty patients (48 male, 2 female) who underwent laryngectomy for laryngeal squamous cell carcinoma were included in this study. Mean age was 62.46 ± 8.79 years (from 45 to 84). The mean follow-up period was 44.40 ± 16.20 months (min: 4 months, max: 64 months). Data considering the clinical and histopathological characteristics are summarized in Table 1.
The mean CD3+ cell count was 461.18 ± 273.73 (min: 60, max: 1496), with a median value of 443.5. The mean count of CD20+ cells was 145.45 ± 131.18 (min: 6, max: 612), and the median value was 63. For T lymphocyte subgroups, the mean count of CD4+ cells was 159.12 ± 129.01 (min: 12, max: 616), with a median value of 119. The mean CD8+ cell count was 300.18 ± 194.60 (min: 42, max: 972), and the median value was 223. The mean count of CD25+ cells was 70.36 ± 49.92 (min: 14, max: 188), with a median value of 52.5. The mean count of FoxP3+ cells was 35.72 ± 33.16 (min: 6, max: 69), and the median value was 28. Additionally, the mean CD8/CD4 ratio was 2.66 ± 1.73 (min: 0.36, max: 8.50), with a median value of 2.43. The mean CD8/FoxP3 ratio was 11.13 ± 7.29 (min: 3.42, max: 44.63), and the median value was 9.94. Samples of immunohistochemistry images are shown in Figure 1, and descriptive data for immunohistochemistry results is summarized in Figure 2.
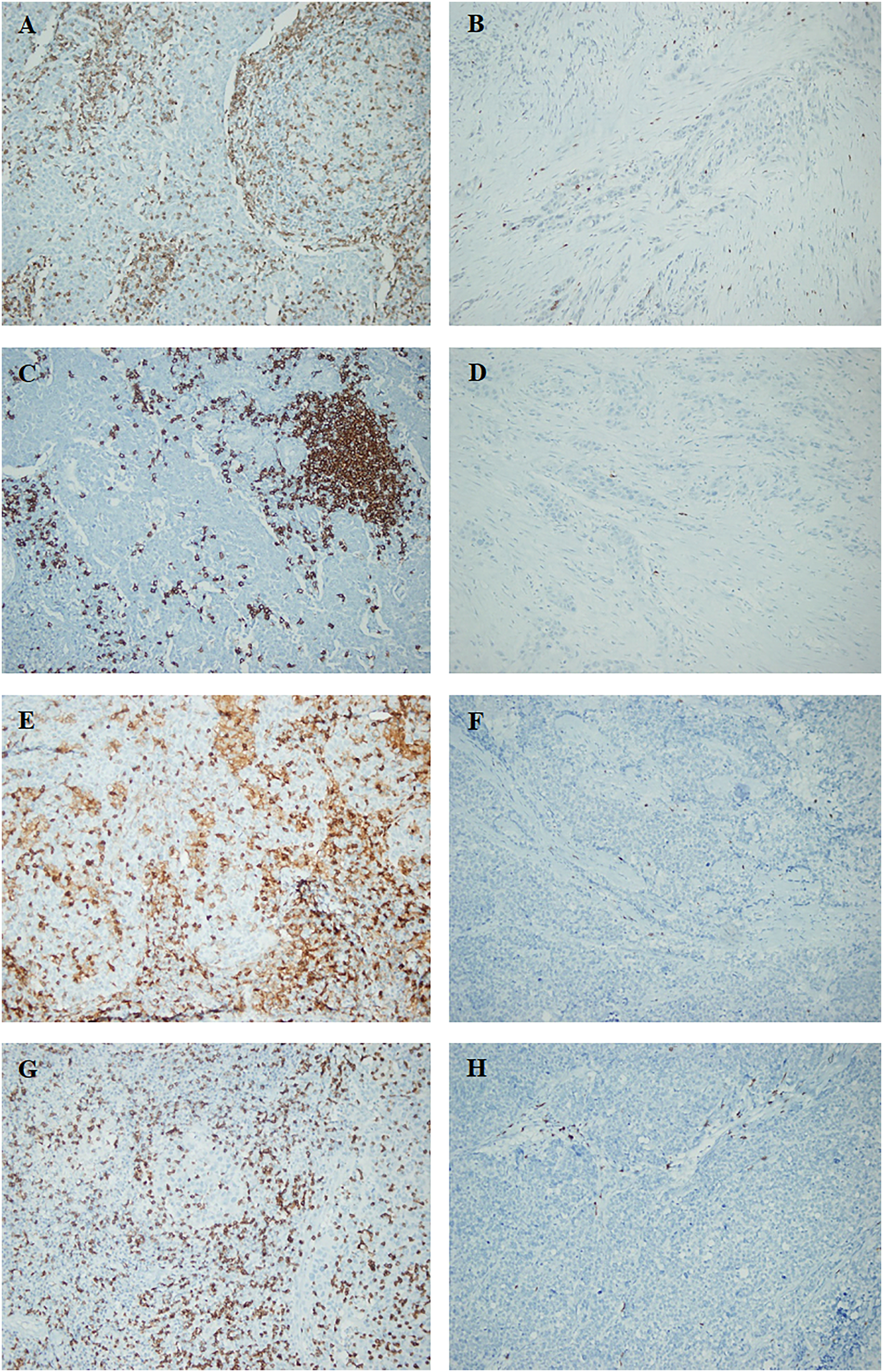
Display of CD3+ (T lymphocytes), CD20+ (B lymphocytes), CD4+ (helper T lymphocytes), and CD8+ (cytotoxic T lymphocytes) lymphocytes by immunohistochemistry (200x magnification). (a) High CD3 infiltration, (b) low CD3 infiltration, (c) high CD20 infiltration, (d) low CD20 infiltration, (e) high CD4 infiltration, (f) low CD4 infiltration, (g) high CD8 infiltration, and (h) low CD8 infiltration.

Diagram demonstrating CD3+, CD20+, CD4+, CD8+, CD25+, and FoxP3+ cell counts (per 0,05 mm dia) along with CD8/CD4 and CD8/FoxP3 ratios.
In the CD3-high group, PNI was significantly lower than in the CD3-low group, with a significant negative correlation (p = 0.010; r = −0.365). Recurrence and death due to disease-specific causes were also significantly lower in the CD3-high group (p = 0.005; r = −0.400). In the CD20-high group, BVI occurred at a lower rate (p = 0.005; r = −0.405). Additionally, CD20 infiltration was significantly lower in poorly differentiated tumors (p = 0.049; r = −0.274). Relapse and disease-related death occurred less frequently in the CD20-high group (p = 0.011; r = −0.361), than in the CD20-low group. There was no significant relationship between CD8 infiltration and histopathological or clinical features. However, PNI and BVI were observed less frequently in the CD4-high group, and a significant negative correlation was present (p = 0.002; r = −0.042 and p = 0.019; r = −0.319, respectively). Similar to CD3 and CD20, relapse and disease-related death were less common in the CD4-high group, with a significant negative correlation (p = 0.008; r = −0.380) (Table 2). CD8/CD4 ratio did not show any significant relationship with histopathological parameters (p > 0.05).
Relationship between infiltrations of CD3, CD20, CD4, CD8, CD25, and FoxP3 positive lymphocytes with histopathological and clinical parameters. The number of patients and corresponding p-values calculated using chi-square analysis, along with r correlation coefficients calculated using Spearman correlation analysis, are provided. Statistically significant p-values and r coefficients are shown in bold.
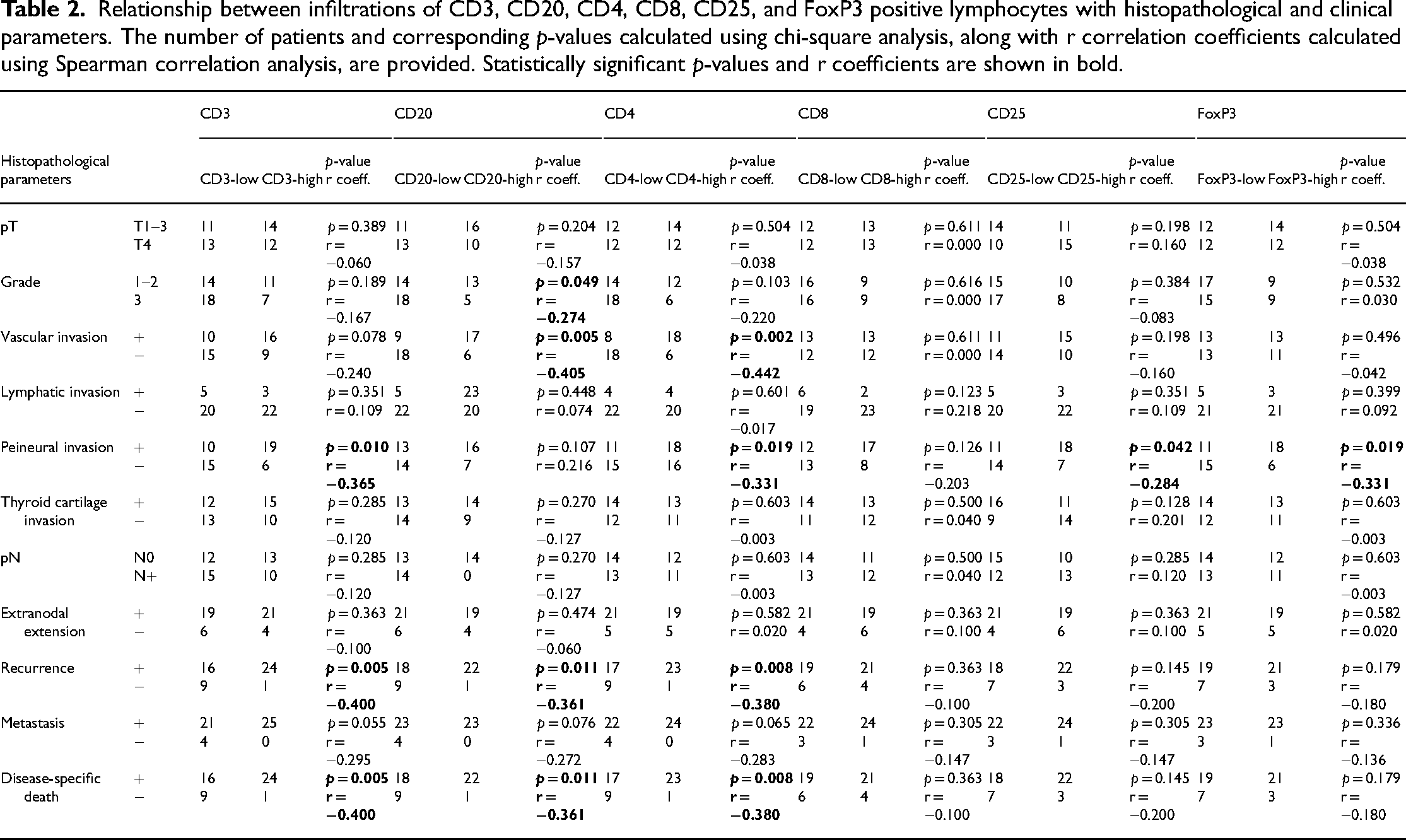
Considering CD25 and FoxP3 expression, PNI was significantly less observed in both CD25-high and FoxP3-high groups, with a negative correlation (p = 0.042; r = −0.284 and p = 0.019; r = −0.331, respectively). Apart from this, no significant relationship was found between CD25 and FoxP3 and other histopathological characteristics and clinical parameters (Table 2). CD8/FoxP3 ratio did not show any significant relationship with histopathological parameters (p > 0.05). There was a correlation between FoxP3 infiltration and CD3 and CD4 infiltrations in the tumor, with a significance level of p < 0.001. Again, CD25 demonstrated a significant correlation with FoxP3 and CD4 levels (p < 0.001).
The two factors that had a significant positive effect on OS were high CD3 infiltration (p = 0.006) and high CD4 infiltration (p = 0.043). However, in terms of DSS, CD3 (p = 0.002), CD20 (p = 0.007) and CD4 (p = 0.004) levels were found to be significant positive prognostic factors (Figures 3–5). Although inflammatory response did not show a significant relationship with OS and DSS; it was a favorable prognostic factor in terms of DFS (p = 0.02) and RFS (p = 0.044). DFS was also significantly associated with CD3 (p < 0.001), CD20 (p = 0.001), and CD4 (p = 0.001) infiltration (Figures 3–5). Similarly, high CD3, C20, and CD4 levels were positive prognostic factors in terms of RFS (Figures 3–5). Although there was a tendency towards better survival in the patient groups with high CD8, CD25, and FoxP3 expression compared to the groups with low expression, this difference did not reach the level of significance. CD8/CD4 and CD8/FoxP3 ratios did not show a significant effect on any of the survival parameters.

Kaplan–Meier diagrams depicting the relationship between CD3+ lymphocyte infiltration and (a) overall survival (OS), (b) disease-specific survival (DSS), (c) disease-free survival (DFS), and (d) recurrence-free survival (RFS) (Tick marks on the curves represent the censored data).

Kaplan–Meier diagrams depicting the relationship between CD20+ lymphocyte infiltration and (a) overall survival (OS), (b) disease-specific survival (DSS), (c) disease-free survival (DFS), and (d) recurrence-free survival (RFS) (Tick marks on the curves represent the censored data).

Kaplan–Meier diagrams depicting the relationship between CD4+ lymphocyte infiltration and (a) overall survival (OS), (b) disease-specific survival (DSS), (c) disease-free survival (DFS), and (d) recurrence-free survival (RFS) (Tick marks on the curves represent the censored data).
Discussion
Despite advancements in cancer research, laryngeal cancer still lacks significant improvements in means of survival, mortality, and morbidity prompting an interest towards immune mechanisms in cancer pathogenesis, as in many other cancers. 19 Numerous studies have established a positive correlation between lymphocyte infiltration in the TME and improved outcomes.9,20 Even use of immunoscore in TNM staging was suggested for certain cancers. 21 HNCs, known for high immune infiltration and reliance on immune tolerance mechanisms, have been a focal point for TME studies. 22 Previous studies on HNCs revealed a positive association between immune infiltration and better prognosis. 23 A recent meta-analysis on laryngeal cancer, demonstrated superior clinical outcomes in terms of OS and DFS in patients with higher TIL levels, by evaluating CD3, CD4, and CD8 infiltrations. 24 However, the study did not include evaluations of CD20, CD25, and FoxP3 infiltrations, nor did it assess histopathological parameters. Our study in contrast included evaluation of CD3, CD8, CD4, CD20, CD25, and FoxP3 positive TILs in laryngeal cancer by immunohistochemistry and their correlation with both histopathological and survival parameters. While lymphocyte distribution can either be evaluated by flow cytometry or by immunohistochemistry in tumor tissue; flow cytometry provides the total lymphocyte count without distinguishing between those in peripheral vascular structures and those infiltrating the tumor parenchyma.17,25 That is why, in this study, we used microscopic evaluation with immunohistochemistry staining rather than flow cytometry, allowing for a specific assessment of TILs as lymphocytes infiltrating the tumor parenchyma correlate more strongly with the outcomes.
High CD3+ cell density is associated with improved survival in many solid tumors. 20 Studies on oral cavity malignancies have indicated that low lymphocyte infiltration correlated with higher recurrence rates. 17 In a meta-analysis, CD3+ TIL infiltration was associated with better OS and DFS in laryngeal cancer. 24 In our study, CD3 infiltration in laryngeal cancer was a crucial favorable predictive indicator confirming previous studies. In the CD3-high group, PNI in the tumor as well as recurrence and disease-related deaths were less frequent. The CD3-high group exhibited significantly better OS (p = 0.006), DSS (p = 0.002), DFS (p < 0.001), and RFS (p = 0.002). The CD3-T cell receptor complex is the primary receptor system on T cells, delivering the first signal essential for T cell activation and dictating the specificity of the immune response. The CD3 subunits are responsible for the signal transduction required for T cell activation. 26 This is why CD3+ cell infiltration is crucial for triggering the antitumoral immune response. Consequently, high CD3 levels are strongly associated with better survival.
HNCs with a higher CD8+ T lymphocyte ratio are associated with improved clinical parameters and prognosis.11,12,27 For hypopharyngeal cancers, the group with increased CD8 infiltration exhibited superior OS, DFS, and DSS. 9 In a retrospective immunohistochemistry study on recurrent laryngeal cancer, a high CD8+ lymphocyte count was significantly associated with better DFS and DSS. 28 In contrast to these findings, Wolf et al. 17 identified a higher recurrence rate for tumors with a higher CD8+ cell count in oral cavity cancers. In our study, there was a tendency towards better survival in the high CD8 group without significance, which may be due to complex interplay of factors such as tumor heterogeneity, variations in the TME, genetic diversity, and treatment strategies.
The role of CD4+ T lymphocytes in the TME remains unclear, as studies have reported both antitumor and pro-tumor effects.13,14 While certain studies indicated that a significant infiltration of CD4+ T lymphocytes in HNCs correlated with a favorable prognosis 29 ; others suggested that tumors with an elevated CD4 ratio displayed more aggressive clinical and histological characteristics. 12 In a study on laryngeal cancer, CD4+ lymphocyte infiltration was significantly associated with improved DFS and DSS, but not OS. 28 However, this study failed to show any significant relationship between CD4+ TILs and histopathological characteristics. According to Wang et al., 9 CD4+ T lymphocyte infiltration was not an independent prognostic factor, but a high CD8/CD4 ratio was associated with better DFS. Oral cavity cancers with a high CD8/CD4 ratio exhibited lower grade, the absence of metastases, and better prognosis.30,31 Yet, our analysis revealed that CD4 infiltration alone was a significant favorable predictive factor for laryngeal cancer, while CD8/CD4 ratio did not show any significance. In addition, our study showed that BVI and PNI were significantly lower in the CD4-high group, and recurrence and disease-related deaths were lower. OS (p = 0.043), DSS (p = 0.004), DFS (p = 0.001), and RFS (p = 0.002) were significantly better in the CD4-high group than in the CD4-low group. The significance of CD4+ T cells in the antitumor response has been increasingly recognized recently. 32 The association between high CD4 infiltration and improved survival is likely due to the orchestrating role of CD4+ lymphocytes in coordinating antitumoral immune responses.
Tregs, suppresses the immune response through various mechanisms and the transcription factor FoxP3 (Forkhead Box Protein-3), enhances their immunosuppressive function by upregulating certain markers.15,33 The prevailing view is that Tregs should be associated with poor prognosis and reduced survival,23,34 although conflicting results exist, with some studies linking Treg infiltration to a better survival.34,35 In HNC, Tregs play a complex role in the TME and prognosis. Studies show that while higher Treg ratios within the TME suggest local immunosuppression and potentially aggressive tumors, higher Treg counts in peripheral blood may indicate a systemic immune response associated with a better prognosis.25,36
The presence of FoxP3+ Tregs within the tumor has shown conflicting effects on survival outcomes, with some studies suggesting better survival11,37 and others showing negative effects.3,38 In a genome atlas study of HNCs, FoxP3+ cell infiltration was associated with better survival parameters, suggesting that Treg infiltration is a natural consequence of the intense immune response against the tumor, balancing cytotoxicity and not serving as an independent prognostic factor. 39 This study did not specify the subgroups of HNC and did not provide data specific to laryngeal cancer. Our investigation revealed that FoxP3+ cell infiltration did not correlate with any negative prognostic factors, but the FoxP3-high group exhibited lower levels of PNI. Although our study did not reach statistical significance in certain outcome measures like OS, DSS, DFS, and RFS trends suggest a potential association between higher Treg infiltration and improved prognosis. We propose that increased Treg infiltration represented a natural response to an intense immune reaction against the tumor. This proposal was supported by the correlation between CD3 or CD4 infiltration and FoxP3 infiltration in the tumor, with a significance level of p < 0.001.
The significance of CD8/FoxP3 and CD8/CD4 ratios in HNC has been highlighted by some authors over individual cell counts. 9 While higher levels of FoxP3+ cell infiltration have been observed in early stage cancers, a high CD8/FoxP3 ratio has been linked to advanced tumor stages in oral cavity cancers. 17 However, other studies suggest that a high CD8/FoxP3 ratio generally indicates a positive prognostic factor. 3 While high FoxP3 infiltration is individually associated with better OS and DFS in some studies29,40; a high CD8/FoxP3 ratio is also linked to better DFS in hypopharyngeal, 9 tonsillar, 37 and oral tongue cancers. 41 Fridman et al. 34 conducted a large-scale meta-analysis and found CD8/FoxP3 ratio as the most significant lymphocyte parameter in terms of survival, attributing this to unbalanced cytotoxic cellular immunity against the tumor. However, our investigation failed to demonstrate a significant relationship between the CD8/FoxP3 ratio and histopathological features and survival parameters. Variations in methodology, sample size, the complex immune landscape in HNCs, and possibly additional immune interactions may have contributed to the lack of a clear association.
CD25, while expressed in various lymphocytes, holds particular significance for Treg. Therefore, it's expected that CD25 expression would correlate with Treg infiltration. Loose et al. 42 conducted a study demonstrating that higher CD25 levels were associated with improved locoregional control and survival rates in HNCs. Similarly, Badoual et al. 29 investigated the prognostic impact of CD4+ CD25+ T cells in HNCs, finding a trend towards better locoregional control and longer survival in those with high CD25 infiltration, although not statistically significant. In our study, using the median value as the cut-off, we observed a trend towards improved survival in the CD25-high group, although it did not reach statistical significance. This finding may be influenced by the methodology used to determine CD25 positivity, suggesting that employing the best predictive value could enhance accuracy in cut-off determination. Notably, CD25 demonstrated a significant correlation with FoxP3 and CD4 levels (p < 0.001), suggesting that both CD25 and FoxP3 serve as natural indicators of extensive immune infiltration and may be associated with a favorable prognosis.
B lymphocytes, identified by CD20 positivity, traditionally lack a defined role in antitumoral immune response. However, Fucikova et al. 43 demonstrated that CD20 infiltration was associated with favorable prognosis in ovarian cancers. Likewise, Hladíková et al. 10 observed aggregates of CD20+ lymphocytes in tumor tissue, associating high tumor-infiltrating B lymphocyte density with improved prognosis in oropharyngeal cancers. Similarly, our study revealed that the presence of B lymphocytes in tumors, particularly in aggregates, was correlated with better DSS (p = 0.007), DFS (p = 0.001), and RFS (p = 0.006). Additionally, elevated CD20 levels were associated with lower grade and decreased likelihood of BVI (p = 0.049; r = −0.274 and p = 0.05; r = −0.405, respectively). Although the conventional perspective underestimates the role of CD20+ lymphocytes in antitumoral immune responses, our findings, along with those of the mentioned studies, necessitate further research to reveal the exact mechanisms.
There are few studies that have evaluated the TME and their correlation with prognostic factors in laryngeal cancer, which provides our study with an important contribution to the field. Although flow cytometry might have improved the study's quantitative aspects of cell isolation, it is limited in its ability to distinguish TILs from lymphocytes in the vascular compartment. Despite the apparent drawback of a small patient cohort in some statistical evaluations, studies with a similar number of patients, especially for laryngeal cancers, are limited. Another limitation is the lack of standardized criteria for identifying positivity for certain markers. Using the best predictive value measurement with a larger patient population could yield more accurate results, aiding in establishing cut-off values for each marker and guiding further investigations.
Conclusion
The TME plays a crucial role in tumorigenesis. In our study, we investigated the distribution of TIL subsets and demonstrated that higher infiltration of CD3, CD20, and CD4 cells were favorable prognostic factors and were associated with better survival outcomes. However, CD8, CD25 and FoxP3 infiltrations did not have a significant effect on prognosis. These findings suggest that enhancing the infiltration of CD3, CD20, and CD4 lymphocytes could be a therapeutic strategy worth exploring in clinical trials.
Footnotes
Author contributions
All authors listed; made a substantial contribution to the concept or design of the work, or acquisition, analysis or interpretation of data, drafted the article or revised it critically, approved the version to be published, take public responsibility for appropriate portions of the content.
Fırat Tevetoğlu: Conceptualization (equal), data curation (equal), formal analysis (equal), funding acquisition (equal), investigation (equal), methodology (equal), project administration (equal), software (equal), visualization (equal), and writing—original draft (equal).
Nil Çomunoğlu: Conceptualization (equal), investigation (equal), methodology (equal), resources (equal), supervision (equal), validation (equal), visualization (equal), and writing—review & editing (equal).
Haydar Murat Yener: Conceptualization (equal), formal analysis (equal), funding acquisition (equal), methodology (equal), project administration (equal), software (equal), supervision (equal), validation (equal), writing – review & editing (equal).
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Istanbul University - Cerrahpasa Scientific Research Projects Coordination Unit (Grant Number 34821).
Ethics declaration
A Clinical Research Ethics Committee Approval was obtained prior to the commencement of the study from the Cerrahpaşa Faculty of Medicine at Istanbul University - Cerrahpaşa [Document Number: 2019-122461, Document Date: 08.08.2019]. Written informed consent was obtained from all of the patients. There is no disclosure of personal information and images of patients in this paper.
