Abstract
Background
Chronic otitis media with or without cholesteatoma progresses with various degrees of bone resorption and remodeling. Estrogen mediates osteoprotective effects through the receptor activator of NF-κB ligand (RANKL) pathway, which is mainly mediated by estrogen receptor-alpha (ER-α).
Objectives
The present study investigated the expression patterns of receptor activator of NF-κB (RANK), osteoprotegerin (OPG), RANKL, and ER-α in pathological tissue from patients with chronic otitis media to determine the roles of those factors in osteolytic mechanisms underlying the pathogenesis of chronic otitis media.
Methods
Normal and pathological specimens from 18 patients with chronic otitis media were examined.
Results
There were no significant differences in RANK, OPG, RANKL, or ER-α mRNA expression between normal and pathological specimens of epithelial tissue.
Conclusions
Our findings suggested that RANK, OPG, RANKL, and ER-α are not associated with the bone destruction in chronic otitis media; other cytokines may directly activate the osteoclasts in chronic otitis media.
Introduction
Chronic otitis media, characterized by recurrent otorrhea and tympanic membrane perforation, can be divided into two subtypes according to the presence or absence of cholesteatoma: chronic otitis media with cholesteatoma (COM-C) and chronic otitis media without cholesteatoma (COM), respectively. Both subtypes of chronic otitis media cause various degrees of bone destruction; COM-C is usually much more widespread and aggressive. Although bone destruction is an important disease manifestation that causes diverse symptoms (e.g. hearing loss, facial palsy, dizziness, and otalgia) in COM and COM-C, the underlying mechanism is not fully understood. Conventional treatment for chronic otitis media comprises surgical resection of pathological tissue and involved bone, rather than inhibition of bone remodeling.
The results of previous studies have suggested possible mechanisms for bone erosion in COM-C. The pressure exerted by the expanding cholesteatoma is an important factor that results in bone resorption. 1 Enzymes including collagenase, 2 acid phosphatase, proteases, and cytokines from various inflammatory cells also participate in bone resorption. Similar to other diseases involving bone destruction, the activation of osteoclasts by the receptor activator of NF-κB ligand (RANKL) pathway is regarded as an important mediator of bone erosion in COM-C.3–7 Osteoprotegerin (OPG), a mediator in the RANKL pathway, plays an inhibitory role in osteoclastogenesis. COM also involves bony destructive lesions, and osteoclasts are presumably associated with its pathogenesis.8–10
Estrogen plays a role in the regulation of osteoporosis-related bone remodeling.11–13 Experimental and clinical findings suggest that estrogen exerts its osteoprotective effects through the RANKL pathway. 13 Previous studies have demonstrated that estrogen receptor-alpha (ER-α) is the main mediator of the osteoprotective effects of estrogen.14–17
Because chronic otitis media progresses with varying degrees of bone resorption and remodeling, we hypothesized that the expression patterns of receptor activator of NF-κB (RANK), OPG, RANKL, and ER-α differ between normal and pathological tissue in both COM and COM-C. First, to confirm the suitability of our experimental conditions, we examined the differentiation of RANKL-treated RAW 264.7 cells into osteoclasts and evaluated the expression patterns of ER-α in differentiated osteoclasts. Then, we investigated the expression patterns of RANK, OPG, RANKL, and ER-α in pathological tissue from patients with chronic otitis media to explore the potential effects of estrogen on osteolytic pathogenesis in affected tissue. To our knowledge, this is the first study regarding the role of ER-α in chronic otitis media.
Materials and methods
Specimens were obtained from 18 patients undergoing tympanomastoidectomy for COM or COM-C at the Department of Otorhinolaryngology, Inje University, Busan Paik Hospital, Busan, South Korea from June 2021 to June 2022. Initial 21 patients who underwent tympanomastoidectomy for COM or COM-C in our hospital were included in this study, and specimens from three patients were excluded, one specimen due to extreme numerical value of densitometry, and two specimens due to extremely small specimen size to analyze. This study design was prospective, and we obtained written informed consent from all participants prior to study initiation. Specimens of normal meatal skin and pathological epithelium surrounding destructive bony lesions were obtained from each patient.
Cell line
The RAW 264.7 mouse macrophage cell line was purchased from American Type Culture Collection (Manassas, VA, USA). Cells were cultured in alpha-minimum essential medium (Cat# 12571-063; Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (HyClone, Logan, UT, USA), 100 U/mL penicillin, and 100 μg/mL streptomycin. The cells were incubated in a humid atmosphere at 37°C with 5% CO2. Subculturing was performed every 3 days, and the cells were detached by digestion with 0.25% trypsin.18,19
Osteoclast differentiation and tartrate-resistant acid phosphatase staining
RAW 264.7 cells were seeded at 1 × 103 cells per well in 24-well plates. At 24 h after seeding, RANKL (30 ng/mL) was added to induce osteoclastic differentiation 19 ; the cells were then incubated for 3 or 5 days. Tartrate-resistant acid phosphatase (TRAP) staining (Cat# MK300; TaKaRa, Kyoto, Japan) was performed in accordance with the manufacturer's protocol. Briefly, cells were washed several times with phosphate-buffered saline, fixed with 200 μL of fixation solution, and washed twice with sterile distilled water. Acid phosphatase substrate solution was added to the wells and the cells were incubated at 37°C for 30 min. Next, the solution was removed, and the cells were washed twice with sterile distilled water. For alkaline phosphatase staining, the substrate solution was added to wells containing cells for 30 min. The solution was removed, and the cells were washed twice with sterile distilled water.
Reverse transcription polymerase chain reaction and densitometry
Total RNA was extracted from cells using TRIzol, and cDNA was synthesized. The primers used to detect mRNA expression were as follows: OPG (forward 5′-TGGAACCCCAGAGCGAAATA-3′, and reverse 5′-TGTGAGCTGTGTTGCCGTTT-3′, 342 bp), RANKL (forward 5′-CACCATCAGCTGAAGATAGT-3′, and reverse 5′-CCAAGATCTCTAACATGACG-3′, 150 bp), and RANK (forward 5′-CAGGGATCGATCGGTACAGT-3′, and reverse 5′-GTTTGAGACCAGGCTGGGTA-3′, 592 bp).20,21 The polymerase chain reaction (PCR) protocol consisted of an initial denaturation step at 94°C for 10 min, followed by cycles of 94°C for 30 s, 60°C for 30 s, and 72°C for 30 s. β-Actin was used as an internal control. Densitometric analysis was performed using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Statistics and analysis
The Kolmogorov–Smirnov and Shapiro–Wilk tests were used to assess the normality of the data. All data exhibited normal distributions despite the small sample size. Paired t-tests were used to compare densitometry data regarding the expression levels of RANK, OPG, RANKL, and ER-α. The nonparametric Wilcoxon signed-rank test was used to compare the densitometry data among subgroups because the subgroup data did not exhibit normal distributions. Statistical analyses were performed using SPSS version 11 (SPSS Inc., Chicago, IL, USA). In all analyses, p < 0.05 was considered indicative of statistical significance.
Results
Cell culture
RAW 264.7 cells were successfully differentiated into osteoclasts by treatment with RANKL (30 ng/mL). TRAP-positive cells were visualized by light microscopy (Figure 1). Cathepsin K mRNA, which exhibited negligible expression in non-RANKL-treated RAW 264.7 cells, was expressed at higher levels in differentiated osteoclasts (Figure 2). The level of ER-α mRNA expression was lower in differentiated osteoclasts than in non-RANKL-treated RAW 264.7 cells. These findings indicated that our experimental conditions were appropriate for osteoclast differentiation.
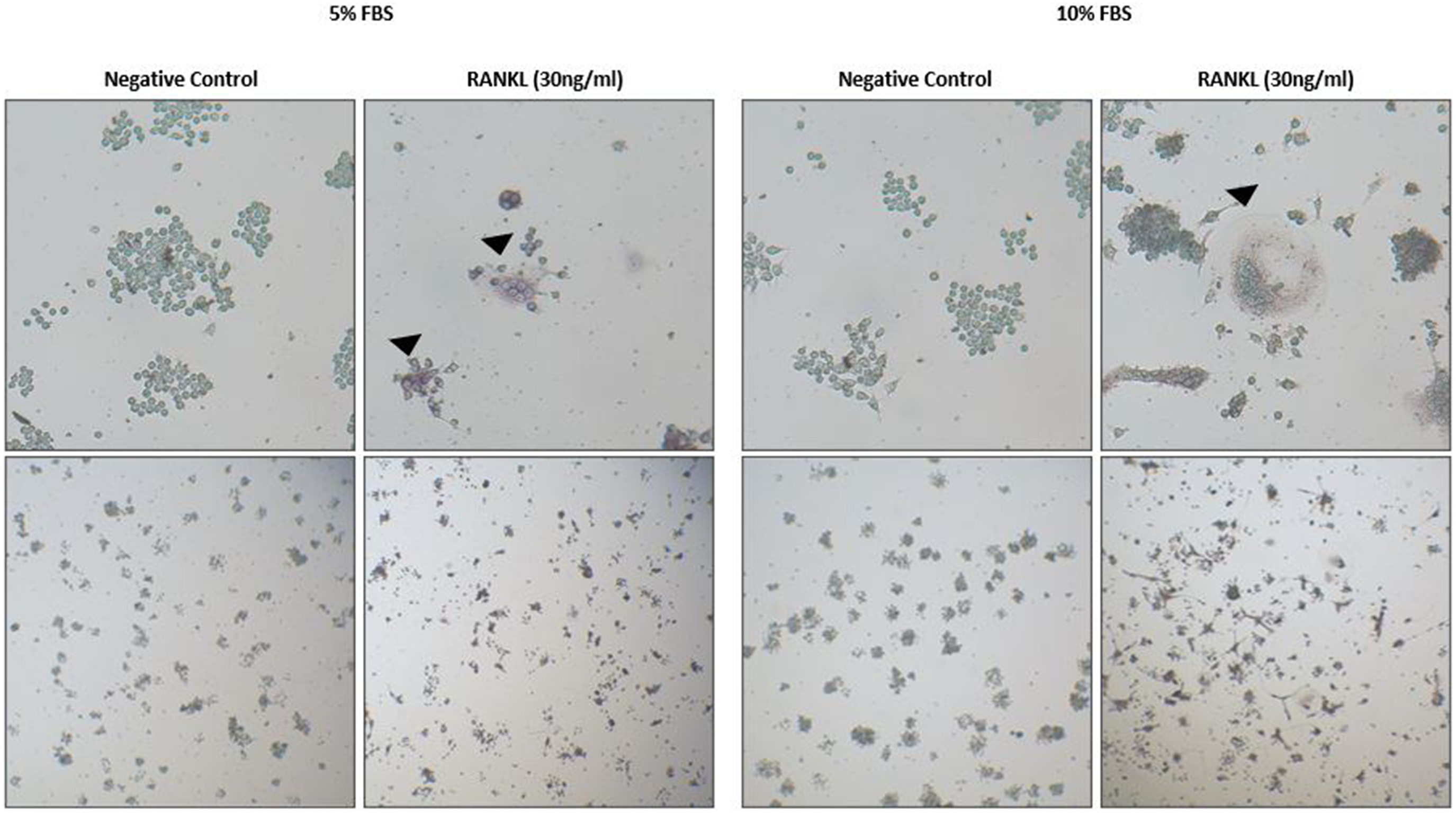
Osteoclast differentiated from RAW 264.7 cell in RANKL. Black arrow indicates TRAP-positive cells visualized via light microscope.
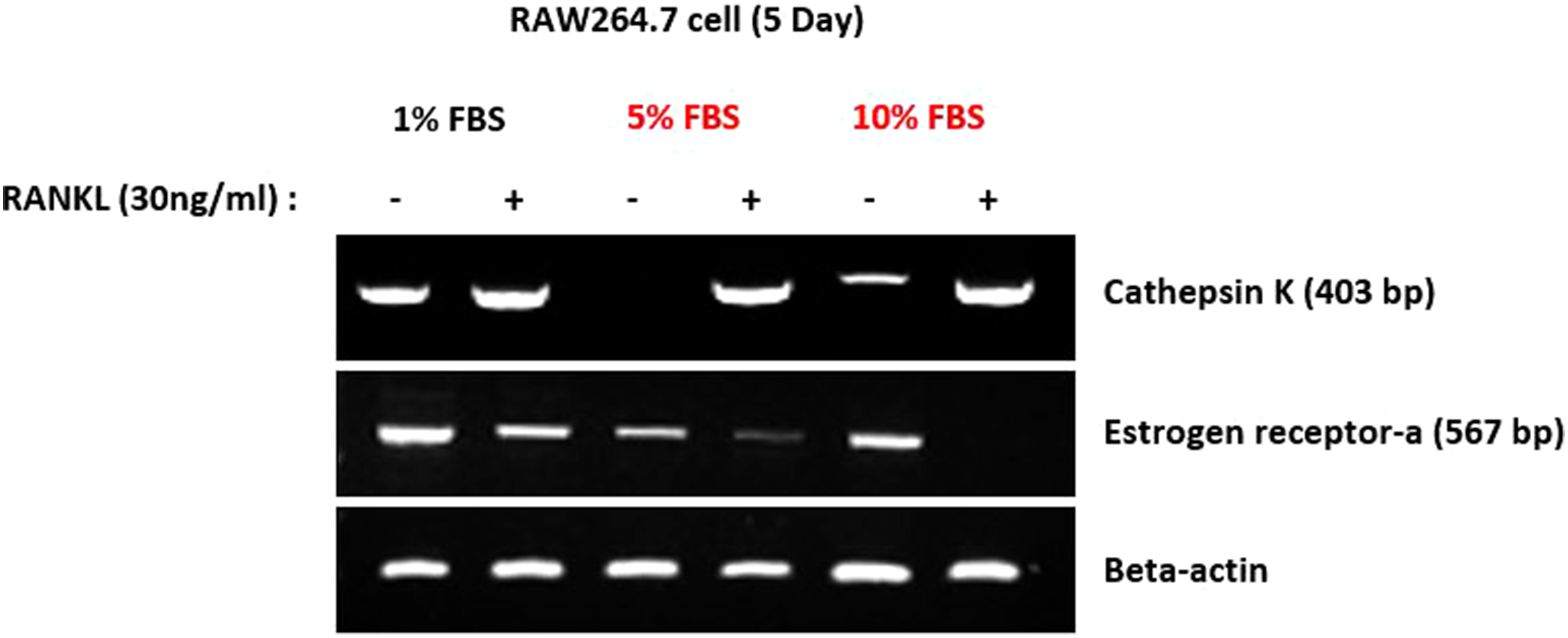
RAW264.7cell in RANKL expressed Cathepsin K, and scarcely expressed ER-α.
Patient characteristics
The study population consisted of 18 patients aged 49–72 years (nine patients with COM-C and nine patients with COM). Detailed patient characteristics are presented in Table 1. The study population included 11 women, all of whom were postmenopausal and had no history of hormone replacement therapy. Wullstein classification was used to determine tympanoplasty type (Table 1). 22
Patient characteristics.

P: patient; R: right; L: left; HL: hearing loss; Chole. : cholesteatoma; CWU: canal wall up mastoidectomy; CWD: canal wall down mastoidectomy; T1: tympanoplasty type 1; T3: tympanoplasty type 3; T4: tympanoplasty type 4; Rev.: revision.
RANK, OPG, RANKL, and ER-α expression (reverse transcription polymerase chain reaction)
Qualitative analysis of RANK, OPG, RANKL, and ER-α mRNA bands via reverse transcription polymerase chain reaction (RT-PCR) revealed no differences between normal epithelial tissue and pathological tissue (Figure 3). Densitometry was performed for semi-quantitative analysis.

mRNA expression of RANK, OPG, RAMKL, ER-α in normal tissue and pathologic tissue N; Normal tissue, P; Pathologic tissue.
Densitometry
Densitometry revealed no significant differences in terms of RANK, OPG, RANKL, and ER-α expression patterns between normal epithelial tissue and pathological tissue among patients overall. Furthermore, there were no significant differences between normal epithelial tissue and pathological tissue in any subgroup (i.e. male patients, female patients, COM-C group, or COM group) (Figure 4).

Densitometry result (A) Densitometry result in total patient, (B) Densitometry result in COM-C, (C) Densitometry result in COM, (D) Densitometry result in male, (E) Densitometry result in female N; Normal tissue, P; Pathologic tissue, n; the number of patient in each subgroup, p; p-value.
Discussion
Multiple studies have shown that bone destruction in cholesteatoma is mediated by the RANKL pathway.3,4,23 Conversely, some studies have revealed potential mechanisms of bone resorption in cholesteatoma that are independent of the RANKL pathway or osteoclast activation24,25; the results of the present study were consistent with those findings. Notably, estrogen plays an osteoprotective role in osteoporosis by regulating the RANKL pathway. Thus, we initially expected lower levels of ER-α expression in pathological tissue surrounding bony erosive lesions in patients with chronic otitis media. However, none of the mediators (e.g. RANK, OPG, RANKL, and ER-α) was associated with bone destruction in both COM-C and COM. Further studies with larger sample sizes are needed to determine the effects of estrogen on chronic otitis media.
Raynov et al. reported that the expression patterns of sex hormone receptors (e.g. progesterone receptor and estrogen receptor) differed between male and female patients with cholesteatoma, suggesting that estrogen has an effect on keratinocyte proliferation and differentiation in cholesteatoma. 26 In contrast to the study by Raynov et al., we examined manifestations of bone destruction in chronic otitis media. We focused on ER-α, which is assumed to have a close relationship with bone destruction in chronic otitis media, in contrast to the role of the progesterone receptor. Furthermore, we investigated both COM-C and COM, because both exhibit similar manifestations of bone resorption and bone remodeling. Nevertheless, our results did not show any significant sex-related differences in the incidence of cholesteatoma; this finding may have been influenced by the inclusion of only postmenopausal female patients in this study.
The mechanisms underlying bone destruction in COM and COM-C have not been determined, although the results of multiple experimental studies have suggested possible pathophysiological mechanisms. The RANKL pathway may be involved in bone destruction in cholesteatoma, but there remains insufficient data to support a clear conclusion. Our findings suggested that mechanisms other than the RANKL pathway are associated with the bone destruction in chronic otitis media. For example, experimental analyses have indicated that tumor necrosis factor-alpha may stimulate osteoclast differentiation in a RANKL-independent manner.27,28 Further studies are needed to identify cytokines other than RANKL that are associated with the mechanism of osteoclast differentiation in chronic otitis media, then determine why RANKL is not involved.
The present study had some limitations. Notably, the sample size was small and semi-quantitative densitometry was used, rather than quantitative RT-PCR, because of the small specimen size. Although this study was initially planned to include both premenopausal and postmenopausal female patient, the participants have become to be composed of postmenopausal female patient, as chronic otitis media tends to develop in advanced age. We regard it as limitation of this study. To overcome these limitations, we examined the differentiation of RANKL-treated RAW 264.7 cells into osteoclasts and characterized the expression patterns of ER-α in differentiated osteoclasts. RAW 264.7 cells were successfully differentiated into osteoclasts, and ER-α mRNA expression was reduced in the differentiated osteoclasts. Our cell culture findings supported the widely accepted hypothesis regarding the role of ER-α in osteoclasts, and our experimental conditions were confirmed to be appropriate for analysis of the roles of RANKL and ER-α in chronic otitis media. Therefore, our experimental results were biologically meaningful, although statistical significance could not be inferred.
Conclusion
The results of the present study suggest that RANK, OPG, RANKL, and ER-α are not associated with bone destruction in COM-C and COM; other cytokines may directly stimulate osteoclasts. Further studies are needed to identify the cytokines other than RANKL that are associated with osteoclast activation in chronic otitis media.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by Busan Paik hospital's institutional review board on 3 June 2021 (BP IRB 2021-05-017).
Funding
This work was supported by 2021 Inje University Busan Paik Hospital research grant.
Author biographies
Kyung Wook Heo, MD, PhD, is Professor of Otorhinolaryngology-Head and Neck Surgery, Inje University Busan Paik Hospital. Graduated from Inje University majoring in medicine and got PhD in Otology. He has accumulated plenty of research achievements.
MinHye Noh, PhD, is Postdoctoral researcher in Anatomy and Research Center for Tumor Immunology Inje University College of Medicine, Busan Paik Hospital. Graduated from Inje University majoring in biology and got PhD in Tumor Immunology.
Dae Young Hur, MD, PhD, is Professor of Anatomy, Inje University Busan Paik Hospital. Graduated from Inje University majoring in medicine and got PhD in Anatomy. He has accumulated plenty of research achievements.
Tae Ui Hong, MD, is an Assistant Professor of Otorhinolaryngology-Head and Neck Surgery, Inje University Busan Paik Hospital. Graduated from Inje University majoring in medicine, and got Master's degree in Otorhinolaryngology.
Sung Yool Park, MD, is Clinical Fellow doctor of Otorhinolaryngology-Head and Neck Surgery, Samsung Medical Center (SMC). Graduated from Inje University majoring medicine, and got master's degree in Otorhinolaryngology.
Woo Jin Kim, MD, is an Assistant Professor of Otorhinolaryngology-Head and Neck Surgery, Inje University Busan Paik Hospital. Graduated from Jeju national University majoring in medicine, and got master's degree in Otorhinolaryngology.
