Abstract
A non-suppurative inflammatory disease of the middle ear with middle ear effusion and conductive hearing loss is called otitis media with effusion. Poor eustachian tube function, immunological factors, viral factors, and other factors are primarily involved in its pathophysiology, which has not yet been completely understood. Several researcher scholars have recently studied platelet-activating factor and they found that platelet-activating factor is closely associated to the occurrence, development, and outcome of otitis media with effusion. Platelet-activating factor is a significant element contributing to the extension of otitis media with effusion. In order to give a reference for further investigation into the mechanism and clinical management of this illness, the research status of platelet-activating factor and otitis media with effusion during the past two decades is reviewed in this study, along with the mechanisms of otitis media with effusion leading to otitis media with effusion.
Keywords
Introduction
Otitis media with effusion (OME) is a condition characterized by the presence of fluid in the middle ear without signs or symptoms of infection. Patients with OME may present with different grades of conductive hearing loss depending on the type of fluid or effusion present. OME episodes can persist for at least three months and may be associated with hearing loss, balance problems, and poor school performance. OME is believed to affect 90% preschool children. 1 The cause of OME is unknown, however, it is now thought to have a variety of etiologies, including eustachian tube dysfunction, craniofacial abnormalities, bacterial and fungal infections, gastric reflux, and middle ear immune response.2,3 Despite the fact that certain efficacy has been achieved through various clinical approaches to treat OME, Many OME are still intractable, pversistent, and develop into chronic secretory otitis media, leading to sequelae like tympanic chamber adhesions, tympanosclerosis, and middle ear cholesterol granuloma.4,5 In recent years, several researchers have discovered that platelet-activating factor (PAF) can produce OME causing middle ear effusion, and they have hypothesized that PAF may play a significant role in the development of OME into chronic secretory otitis media. The molecular traits of PAF and the mechanisms involved in the pathophysiology of OME will be succinctly described in this review.
Biological characteristics of PAF
PAF is an endogenous biologically active phospholipid that is produced by a variety of cells, including neutrophils, platelets, mast cells, endothelial cells, and macrophages. It's a strong inflammatory mediator that not only activates platelets but also neutrophils, monocytes, and macrophages, making them aggregating and becoming chemotactic. This releases leukotrienes and oxygen radicals, inhibits cellular value added, stimulates B-cell synthesis of IgE and IgG, reduces cardiac contractility, and widens capillary permeability. PAF also binds to G proteins to activate phospholipase C, which releases Ca2+ from the storage pool and rearranges the platelet cytoskeleton. This activates platelets, resulting in vasoconstriction, increased permeability, abnormalities of the microcirculation, and inflammatory reactions.6–8 PAF is recognized to be directly linked to the development and occurrence of allergic diseases, eye diseases, cardiovascular diseases, reproductive system, asthma, cirrhosis, acute pancreatitis, gastrointestinal ulcers, endotoxic shock, and trauma (Figure 1).6,9
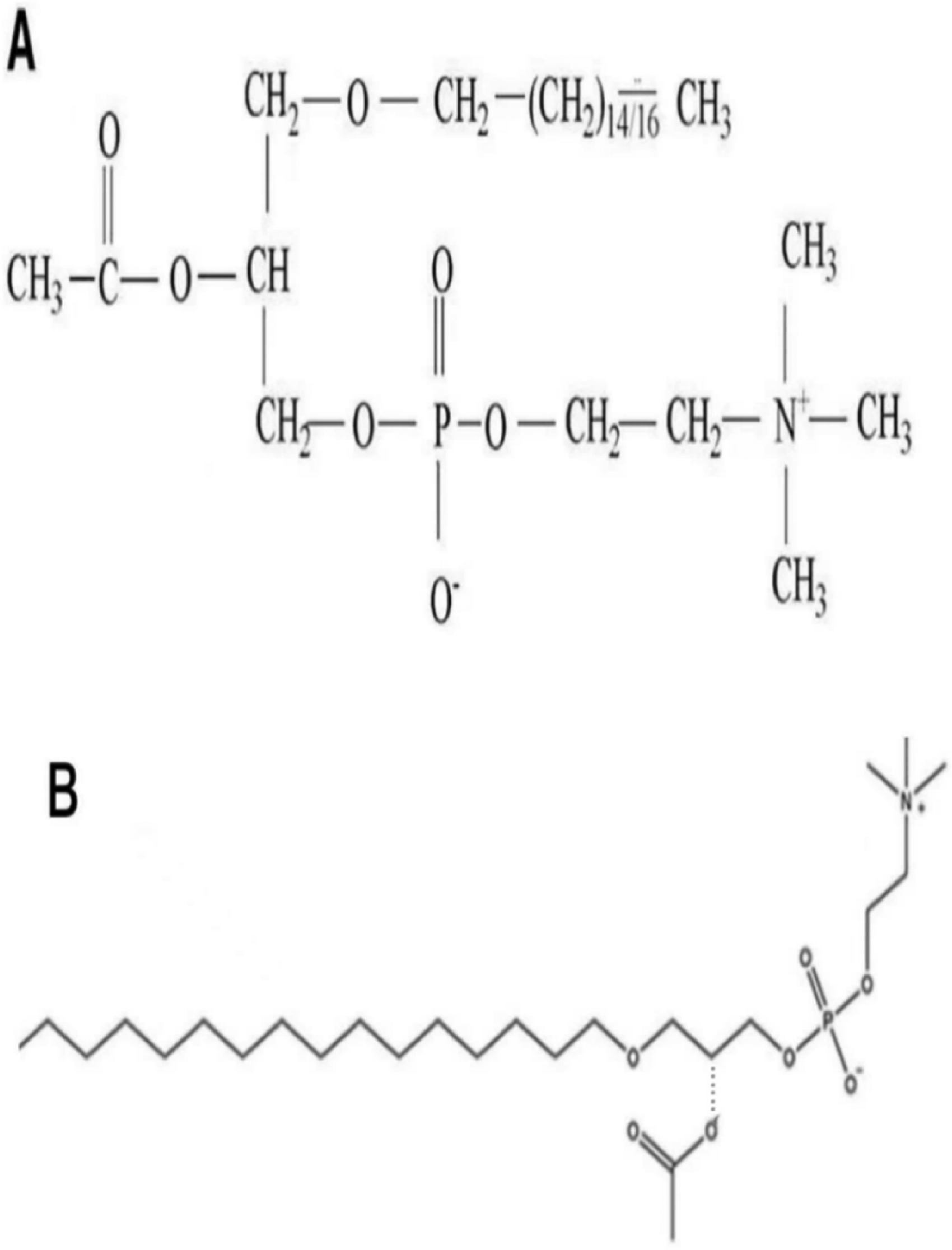
The structure of platelet-activating factor (PAF): (A) chemical structure of PAF; (B) spatial structure model of PAF.
PAF is present in middle ear effusion of OME patients
Numerous cells, including neutrophils, platelets, mast cells, endothelial cells, and macrophages, generate PAF. 10 This endogenous physiologically active phospholipidis is a strong inflammatory mediator that not only activates platelets but also neutrophils, monocytes, and macrophages, making them clump together and chemotactic. This releases leukotrienes and oxygen radicals, inhibits cellular value-added, stimulates B-cell synthesis of IgG and IgE, reduces cardiac contractility, and widens capillary permeability. Since these cells also produce significant amounts of inflammatory factors, including PAF, it was hypothesized that PAF is also present in the middle ear effusion. Benveniste 7 was the first to observe PAF in the middle ear effusion of otitis media patients; By using high performance liquid chromatography. Chen Liang et al. 11 examined the PAF level in the middle ear effusion of 56 patients with OME and discovered that the PAF level in the middle ear effusion of mucus was significantly higher than the PAF level in plasma effusion; In order to test the PAF level in the serum of 26 patients with OME, Wen Fengni et al. 11 employed an enzyme-linked immunosorbent assay and discovered that the PAF concentration in the serum of OME patients was significantly greater than that of the control group. It is clear that PAF, a potent lipid inflammatory mediator, is crucial to both the pathogenesis of OME and the persistence of the disease.
By increasing middle ear vascular permeability, PAF induces middle ear effusion
PAF dramatically increased capillary permeability, even 1000–10,000 times more than the typical inflammatory mediator histamine. 12 This is likely due to the fact that PAF acts on endothelial cells to disrupt the endothelial barrier or because PAF activates various inflammatory cells, particularly platelets and neutrophils, which leads to secondary endothelial cell damage.
According to Brunner et al., 13 the interplay between calciumions and their activated channels in the pathophysiology of OME was a significant element affecting the PAF-induced release of histamine, which increased middle ear mucosal capillary permeability and resulted in middle ear effusion. Additionally, PAF increased middle ear mucosal vascular permeability, acting on the cells that generated it (such as neutrophils, monocytes, and macrophages) to further promote the metabolism of arachidonic acid. The arachidonic acid then acted on itself to further increase mucosal vascular permeability and promoted the development of middle ear effusion. In agreement with Brunner's findings, Rhee et al. 14 discovered that PAF-induced middle ear effusion contained both cyclooxygenase and lipoxygenase after injecting PAF into the tympanic chamber of chinchillas without eustachian tube obstruction. They proposed that the first 24 hours of PAF leading to middle ear effusion may be achieved by causing an increase in middle ear mucosal vascular permeability, whereas the next 24–48 hours may be achieved by the interaction of inflammatory mediators during the initial period and thus chemotaxis of PAF, which further confirms the close relationship between PAF and arachidonic acid metabolism in human middle ear effusion.
By inducing increased cytokine secretion, PAF induces middle ear effusion
Numerous experimental studies conducted in recent years have revealed that the levels of cytokines in middle ear effusions of OME patients are significantly higher than those in plasma which may play a significant role in the development of a chronic course or persistence of OME. IL-1, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12, tumor necrosis factor (TNF), interferon (IFN), transforming growth factor (TGF), and granulocyte-monocyte colony-stimulating factor (GM-CSF) are cytokines that have been demonstrated to be present in middle ear effusion. 15 Tachibana F demonstrated PAF was involved in the onset of AOM and COM on the acute exacerbation, and that leukotrienes (LTs) played an important role in OME by measureing PAF and LTs in middle ear fluid or in otorrhea of children with otitis media. 16 Additionally, longer illness duration was associated with higher levels of IL-8 in middle ear effusions. According to Hamel’s studie, 17 PAF can stimulate IL-4, IL-6, IL-8, TNF–a, and other inflammatory factors in vivo. Therefore, several academics hypothesized that PAF might cause middle ear effusion formation by inducing local cytokine production. In a dose-dependent manner, Ogura et al.15 discovered that PAF and PAF stable analog (C-PAF) significantly increased IL-8 levels in the middle ear cavity. Inflammatory mediators like IL-8 have been shown to increase middle ear vascular mucosal permeability and encourage middle ear effusion production. In conclusion, it is clear that PAF may influence the development of OME by enhancing cytokine expression in the middle ear cavity and affecting the prognosis.
By promoting the expression of mucus glycoproteins, PAF induces middle ear effusion
The secretory proteins in middle ear epithelial cells, which are produced by secretory cells and released extracellularly, include mucous glycoprotein, has been demonstrated to be a significant component of middle ear mucus. When the middle ear cavity is stimulated by inflammation, the submucosal cup cells aggregate and mucin secretion increases, which weakens the activity of middle ear mucous cilia and makes the accumulation of fluid difficult to drain. Therefore, it is important to understand the reason that OME persists and develops into chronic secretory otitis media. Kubba H 18 found releasing of inflammatory mediators will cause secretion of a mucin-rich effusion by up-regulating mucin genes. This would induce prolonged stimulation of the inflammatory response and poor mucociliary clearance leading to persistence of the middle ear fluid. In vitro research by Nadel JA revealed that PAF not only promptd mucous glycoprotein secretion but also increased goblet cell hyperplasia and mucin gene expression in a dose- and time-dependent manner. 19 Additionally, PAF increases the synthesis of arachidonic acid metabolites, which in turn enhances mucin secretion, causing a vicious cycle that simultaneously stimulates mucus glycoprotein secretion. 20
By affecting the function of the cilia that carry middle ear mucus, PAF induces middle ear effusion
The regular unidirectional oscillatory movement of middle ear mucus cilia from the middle ear to the opening of the eustachian tube facilitates the clearance of secretions from the ear cavity. It has been demonstrated that PAF weaken the ciliary activity of human sinus mucosa. hence it is presumed that PAF similarly affects the middle ear's mucous cilia. 21 Ohashi's research shown that cilia activity was reduced to 60% in mucinous OME while it was reduced to 80% in secretory OME, supporting the idea that the high expression of PAF in mucinous OME inhibits cilia function and is one of the causes of the prolonged OME (Figure 2).

A simplified schematic illustrating the four main PAF cell signaling pathways in secretory otitis media.
Conclusions
PAF is an unique cytokine with a broad range of biological activities, participating in a variety of physiopathological processes in vivo. It was discovered that PAF expression was significantly higher in middle ear effusion in OME. This increases middle ear exudation by increasing vascular permeability, exacerbates the inflammatory response by increasing cytokines in middle ear effusion, increases middle ear effusion viscosity by encouraging mucin production, and prolongs OME by interfering with the ciliary transport system's function. Based on the above mechanism, it is hypothesized that inhibiting PAF may decrease IL-8 expression, lower middle ear mucin production,improve mucus cilia activity, and thus reduce middle ear mucosal inflammation and fluid production. This theory offers a new theoretical foundation for the clinical treatment of persistent OME.
Footnotes
Authors contribution
Material preparation and literature search were done by L.W. Writing the original draft preparation was done by L.W. Writing the review and editing was done by S.L. Funding acquisition was done by S.L. Article idea innovation and figure production were done by F.Y.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Liaoning Provincial Education Department Fund (Project no.: JYTMS20230575).
