Abstract
Plants benefit from plant-associated microorganisms, of which endophytes are of particular interest as they are transmitted from generation to generation. This study characterises endophytes from maize roots and determines their biocontrol potential against toxigenic fungi in Nigerian maize. Maize roots were collected from farms in Lafia, and stored grain samples were collected from the six Northern States of Nigeria, from which endophytes and toxigenic fungal strains were isolated. Molecular identification employing 16SrRNA/internal transcribed spacer (ITS) sequences for isolated fungal endophytes was carried out, and mycotoxins produced by fungi were determined by high-performance liquid chromatography analysis. Biocontrol activity of the endophytes was determined using the dual culture confrontation test. Aspergillus and Fusarium genera were the prevalent isolated fungal species. Eight fungal endophytes were identified of which Trichoderma harzianum, Dichotomopilus erectus and Burkholderia spp. were the isolates with biocontrol activities, while 12 Aspergillus spp. were found to produce varying amounts of ochratoxin A and aflatoxin B1, respectively. T. harzianum showed the best inhibition (74%), followed by D. erectus (50%) and Burkholderia spp. (48%). T. harzianum showed poor inhibition of Aspergillus flavus (B7) at 30%. However, results from the Pakdaman Biological Control Index showed that T. harzianum has the best antifungal biocontrol activity of the three endophytes. The study concludes that antifungal biocontrol agents can be sourced from endophytes to obtain indigenous control activities that can check mycotoxin contamination of food and livestock feed, as well as elucidate possible metabolites for agricultural and industrial applications, which will help improve plant performance, increase crop yield and sustainability.
Introduction
In various forms, maize is a common food source for Nigerian homes, households and livestock. It is the primary raw material for the feed industry, and its cultivation guarantees food security for homes in Nigeria as well as employment and regular income for the enormous labour force scattered throughout the nation. Maize production in Nigeria increased to 12.76 million tons in 2019, the highest production volume in recent years. 1 Maize is a versatile crop that can grow in a variety of agroecological zones. In Nigeria, fungi that produce mycotoxins heavily infect maize, which is a risk factor to human and animal health and food security in Africa.2,3 Food security in the West African region is threatened by contamination by fungi and mycotoxins, which causes farmers to experience immeasurable losses and frustration. In Nigeria, 617 billion Naira were lost in 2017 because of mycotoxin production and fungal diseases. 4
Mycotoxins are naturally occurring secondary metabolites with low molecular weight, produced by certain filamentous fungi under conducive environmental conditions. These fungi grow on different foodstuffs including cereals, nuts, spices, dried fruits, apples and coffee beans. 5 The mycotoxins produced cause a variety of health problems ranging from acute poisoning to long-term effects such as immunodeficiency and cancer. They are structurally diverse. 6 The tropical African climate coupled with poor agricultural management practices aids fungal proliferation and contamination of crops.
Endophytes are endosymbiotic microorganisms inhabiting plants and aiding in promoting the development and enhancing the survival of the plant host. Some are capable of producing novel bioactive secondary metabolites with anti-microbial, anti-insect or anti-cancer properties, while also enhancing plant growth and ameliorating abiotic and biotic stresses in the affected plant hosts. 7 Endophytes live together with their plant host without causing apparent disease conditions. These unique microorganisms access the host plant through germinating radicles, secondary roots, stomata or wounds and subsequently reside within cells, intercellular spaces, or the vascular system of plants.
Mycotoxins negatively affect the health of humans and livestock. The adoption of pre-harvest approaches has been proposed and tested.8,9 The need to find a practical, cost-effective, and non-toxic antifungal alternative to prevent fungal and mycotoxin contamination in stored farm products makes this study imperative. Using microorganisms and their products will avoid the risk associated with using chemicals as preservatives and fungicidal agents. The application of endophytes and their metabolites in preventing toxin production and contamination will go a long way in increasing cereal crop production for export. It will drastically reduce costs due to rejection at ports, and ensure safe and healthy food for Nigeria's large population and Africa as a whole.
Materials and methods
Collection of maize root samples: Maize roots were collected randomly from 15 different plants in Lafia Nasarawa State, Nigeria from which 50 g of roots per plant was used in the study.
Collection and preparation of maize grains: Maize grains stored for up to 6 weeks were collected from the six states making up North Central Nigeria, namely Kogi, Benue, Nasarawa, Plateau, Niger, and Kwara. Six composite samples of grains were randomly collected each of which was made up of 10 sub-samples (2 kg per sub-sample). One kilogram was separated from each sample, packaged in polythene bags, and labelled.
Isolation of fungal and bacterial endophytes from maize roots: Maize roots were cleaned and treated according to the method of Orole and Adejumo. 10 Roots were surface sterilized with 0.8% NaOCl for 2 min followed by a 30 s dip in 70% ethanol and 2 rinses with distilled water. One gram root sample was weighed, grounded and homogenized with 195 mL of sterile 0.1% peptone water. Dilutions of 10−3 were made for fungi and 10−6 for bacteria and plated on Potato Dextrose Agar (PDA) supplemented with 1.00 g/L streptomycin, and nutrient agar, respectively. Plates were incubated at 28°C (fungi) and 27°C (bacteria) for 5 to 7 days.
Identification of endophytes of maize roots: Identification of fungal endophytes was performed by using the 16SrRNA/internal transcribed spacer (ITS) sequence. DNA was extracted using lysis buffer (400 mM Tris-HCl [pH 8.0], 60 mM ethylenediaminetetraacetic acid (EDTA), 150 mM NaCl, 1% sodium dodecyl sulfate). After 10 min at room temperature, 150 µL of potassium acetate (pH 4.8) were added, mixed and centrifuged at 10,000 g for 1 min. The supernatant was transferred to a new tube and the process was repeated. An equal volume of isopropyl alcohol was added and the tube was mixed by inversion. The tube was spun at 10,000 g for 2 min, and the supernatant was discarded. The DNA pellet was washed with 300 µL 70% ethanol, spun at 10,000g for 1 min, and the supernatant discarded. The DNA pellet was air-dried and dissolved in 50 µL H2O. Extractions were done in duplicate for each sample. One microliter of the purified DNA was used in 25 to 50 µL of polymerase chain reaction (PCR) mixture. Fungal DNA was amplified using ITS1 (5’-CTTGGTCATTTAGAGGAAGTAA-3’) and ITS4 (5’- TCCTCCGCTTATTGATATGC-3’) primers. Bacterial DNA was extracted by adopting the first three steps in the extraction of fungal DNA. The obtained supernatant was treated with 300 µL each of phenol and chloroform isoamyl (24:1), mixed by inversion, centrifuged at 14,000g for 1 min, and the upper layer was dispensed into a new tube. To the mixture was added chloroform (500 µL) and centrifuged at 14,000g for 1 min, after which 500 µL isopropanol was added, and inverted again. The withdrawn upper phase was incubated on ice at −20°C for 10 min, centrifuged at 12,000g for 2 min, and the supernatant dispensed off. To the obtained DNA fragments was added 300 µL 70% ethanol and centrifuged at 10,000g for 1.5 min. The DNA fragment was dried for 20 min and 30 µL double distilled (dd) water was added and PCR amplified using primers F27 (5’- AGAGTTTGATCMCTGGCTCAG-3’) and R1492 (5’- TACGGYTACCTTGTTACGACTT −3’).
Sequencing was performed by Microsynth Seqlab GmbH (Göttingen, Germany). Sequences were edited and aligned using Geneious bioinformatics software (version 8.1). Sequences were used for homology search using the Basic Local Alignment Search Tool (BLAST) software algorithm at NCBI GenBank (http://www.ncbi.nlm.nih.gov.blast). Data obtained were deposited at NCBI (Table 1).
Isolation and identification of fungi from maize grains: Two approaches were adopted in isolating fungi from maize grains. (i) Fungi on maize grains were isolated by direct plating of grains on Malt Extract Agar and incubated at 25°C for 7 to 10 days according to the method of Al-Kahtani 11 to obtain different fungal species peculiar to the maize grains. (ii) Fungi suspected of producing mycotoxins (aflatoxin B1 (AFB1) and ochratoxin A (OTA)) were isolated on Czapek Dox Agar (CDA). One gram of maize grains was washed aseptically with 10 successive 10 mL volumes of sterile distilled water, surface sterilized using 10% KOH and rinsed with sterile distilled water. The grains were inoculated three times per sample randomly onto culture plates containing CDA to which streptomycin (0.50 g/L). Incubation was at 28°C and examination was carried out daily for 5 days for observation of growth and sporulation. After 5 days of incubation, different fungal colonies obtained were transferred onto fresh plates. The isolates’ morphological, phenotypical and spore-forming characteristics were employed in the identification as valid means.
Determination of mycotoxin production: Qualitative detection of mycotoxins was as described by Abo Dahab et al. 12 Aspergillus and Penicillium spp. were inoculated onto plates containing coconut milk agar (CMA) and yeast extract sucrose (YES) medium, and incubated at 28°C for 7 days. A fluorescent ring around colonies under ultraviolet (UV) light (365 nm) was taken as evidence of aflatoxin production. 13 Cultures with the fluorescent ring were inoculated onto fresh Czapek yeast extract agar and incubated at 27°C and 25°C for 7 days in the dark. From each culture, three 6 mm agar plugs with a fungal colony were removed and extracted with 1 mL methanol and analyzed using high-performance liquid chromatography (HPLC) with UV detector.
HPLC parameters: The protocol of Saleemi et al. 14 was adopted. For the determination of AFB1, a mixture of acetonitrile: methanol: water (22.5:22.5:55) as mobile phase was used with a flow rate of 1.0 mL/min, a C18 column and an oven temperature of 30°C. Excitation and emission wavelengths were 360 and 440 nm, respectively. For the measurement of OTA, a mixture of acetonitrile: water: acetic acid (57:41:2) as mobile phase at the flow rate of 1.0 mL/min and an oven temperature of 40°C was used. Excitation and emission wavelengths were 333 and 477 nm, respectively.
Dual culture confrontation test: A modified method of Taechowisan et al.
15
was employed to test for antagonistic effects of isolated endophytes and their metabolites on the radial growth of toxigenic fungi. Mycelium agar plugs (5 mm) of 5-day-old growing endophytic cultures and that of the toxigenic fungi were inoculated at a distance of 10 mm from the edge of a 90 mm Petri dish at opposite ends. Incubation was at 28°C for 5 days. The inhibition zones were determined by measuring the distance between the endophytic fungi and toxigenic fungal colonies. Plates were made in triplicates. Antifungal inhibitory activity was determined by employing the methods of Nuangmek et al.,
16
and the Pakdaman Biological Control Index (PBCI) to estimate pathogen resistance (R) against antitoxigenic endophytes calculated. The percentage inhibition in radial growth was calculated for each of the endophytes: Percentage of inhibition = (A – B/A) x 100
where A = radius of toxigenic fungi on the control plate; B = radius of toxigenic fungi in the dual culture plate.
PBCI and R-values were calculated using the formula: Pathogen resistance (R) =Z/M PBCI (mm) = M/Z × P
where Z = period after inoculation until the endophyte fully covers the pathogen (days); M = period after inoculation until the biological control agent (endophyte) fully grows on the plate in the absence of the pathogen (days); P = radial distance (mm) of the pathogen colony between the edge of the inoculation disc and the marginal point of the colony (pathogen) located on the presumed diagonal line connecting the centre of the disc in the plate.
R-value ˃ 1 shows that the pathogen is resisting the biological control mechanism(s) adopted by the control agent in confrontation with a pathogen. R-value = 1 indicates the absence of any resistance by the pathogen, and R-values < 1 indicates that a pathogen does not resist, but promotes the growth and development of a biological control agent. 17
Agar poison assay: Dichotomopilus erectus, Trichoderma harzianum and Burkholderia spp. growing cultures were separately inoculated into 20 mL sterile potato dextrose broth and incubated at 28°C for 14 days on a rotary shaker. Samples were filter sterilized using 0.45-micron syringe filters and 1 mL of the filtrate was added to 20 mL sterile molten PDA, then poured into sterile Petri dishes and allowed to solidify. The growing edge of a 3-day-old colony (6 mm) of a toxigenic fungus was placed in the centre of the prepared agar plates. Plates were incubated at 28°C and radial growth of the fungi was measured daily for 3 days. The control was pure isolates of toxigenic fungi grown on PDA.
Standardisation of endophytic bacterial and fungal inocula for Minimum Inhibitory Concentration: Burkholderia spp. was grown in 10 mL Mueller Hilton Broth (MHB) medium for 24 h at 37°C. The obtained suspension was diluted with MHB medium to obtain a suspension of 1.5 × 108 CFU/mL. The bacterial suspension (1 mL) was mixed with molten PDA in a 90 mm Petri dish to which 6 mm of freshly grown toxigenic fungi was inoculated in the centre and the colonial radius was measured daily for 3 days. As described by Petrikkou et al. 18 fungal cultures of D. erectus and T. harzianum were grown on Petri dishes containing PDA and inocula prepared from 4 to 5 days old cultures. The spore density of each fungus was adjusted to 2 × 106 spores/mL using a haemocytometer.
Minimum Inhibitory Concentration: Extracts of the antifungal endophytic isolates were analysed to determine the minimum inhibitory concentration against toxigenic fungi. Prepared extracts of the endophytic isolates were prepared in MHB to obtain a concentration of 250 mg/mL obtained by dissolving 100 g extract in 400 mL MHB. And 100 μL were dispensed into all wells of the microtitre plate. Toxigenic isolate's suspension corresponding to 0.5 McFarland standard was prepared and 5 μL of suspension was added into wells in columns 1 to 11 except column 12, which was the broth sterility control and blank for reading plates. The plates were incubated at 28°C for 18 h after which the reading of results was manually read.
Results
Eight fungal endophytes were isolated from maize roots in the study (Table 1) and six bacterial isolates (Supplemental Table A) were identified. ITS sequences of the identified fungal isolates were deposited at NCBI and have been allotted accession numbers (https://submit.ncbi.nlm.nih.gov/subs/?search = SUB6216550).
List of identified fungal endophytes.

*Supplemental file.
Ninety-seven isolates were isolated from maize grains in the 6 states sampled, and 80 of those were identified as shown in Table 2. The genus Aspergillus had the highest number of isolates (50), while the genera Chrysosporium, Mortierella, and Madurella all had one isolate each. The result reflects the level and type of fungal contamination harboured in maize grains consumed in the respective states.
Isolated fungal species from maize grains and state of the source.
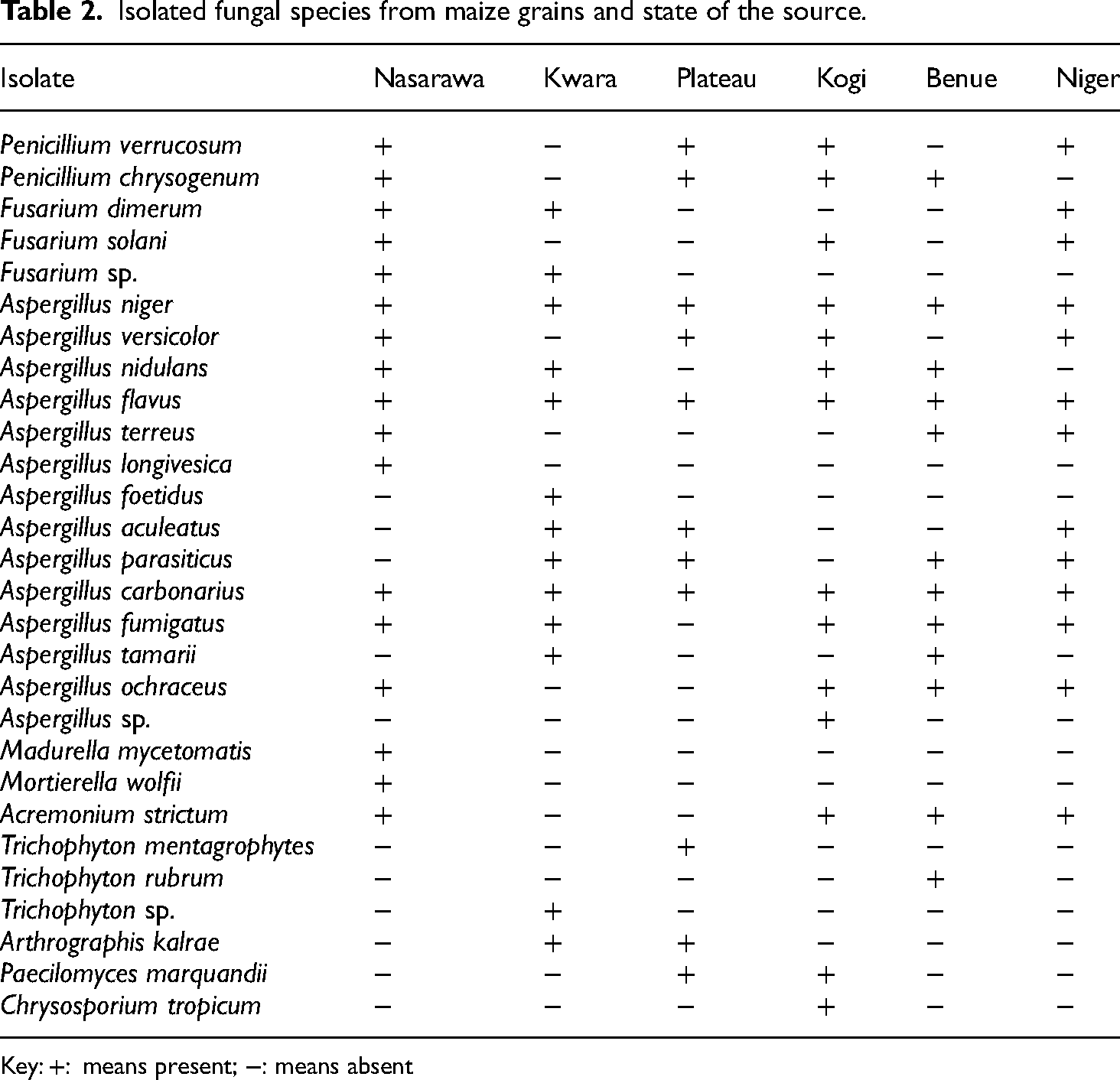
Key: +: means present; −: means absent
Identification of toxigenic fungi was based on preliminary tests carried out using CMA and YES agar, and validation with HPLC. Twenty-four of the isolates originally showed fluorescence (green and blue). Mycotoxin production was recorded in 13 of the fungal species identified in the study (Table 3) while the remaining 11 isolates did not produce toxins in quantifiable amounts.
Mycotoxin production by isolated fungi in maize grains.

Key: ×××: strong fluorescent blue-green colour; ××: medium fluorescent blue-green colour; ×: weak fluorescent blue-green colour; +++: strong fluorescent blue colour; ++: medium fluorescent blue colour; +: weak fluorescent blue colour; −: no fluorescence; AFB1: aflatoxin B1; HPLC: high-performance liquid chromatography; nd: not detected; NID: not identified); OTA: ochratoxin A; YES: yeast extract sucrose.
Dual culture assays to determine the antifungal potential of the isolated endophytes (bacteria and fungi) against toxigenic fungal species showed poor activity. Three fungal endophytes D. erectus strain hk2619, T. harzianum strain kk073, and Burkholderia spp. gave appreciable inhibitory activities and were used for further studies. Figure 1 presents the inhibitory activities of the endophytes tested. T. harzianum strain kk073 had the best inhibitory activity against the fungal pathogens tested, except for Aspergillus flavus (B7) where inhibition was relatively low at 29.3%.
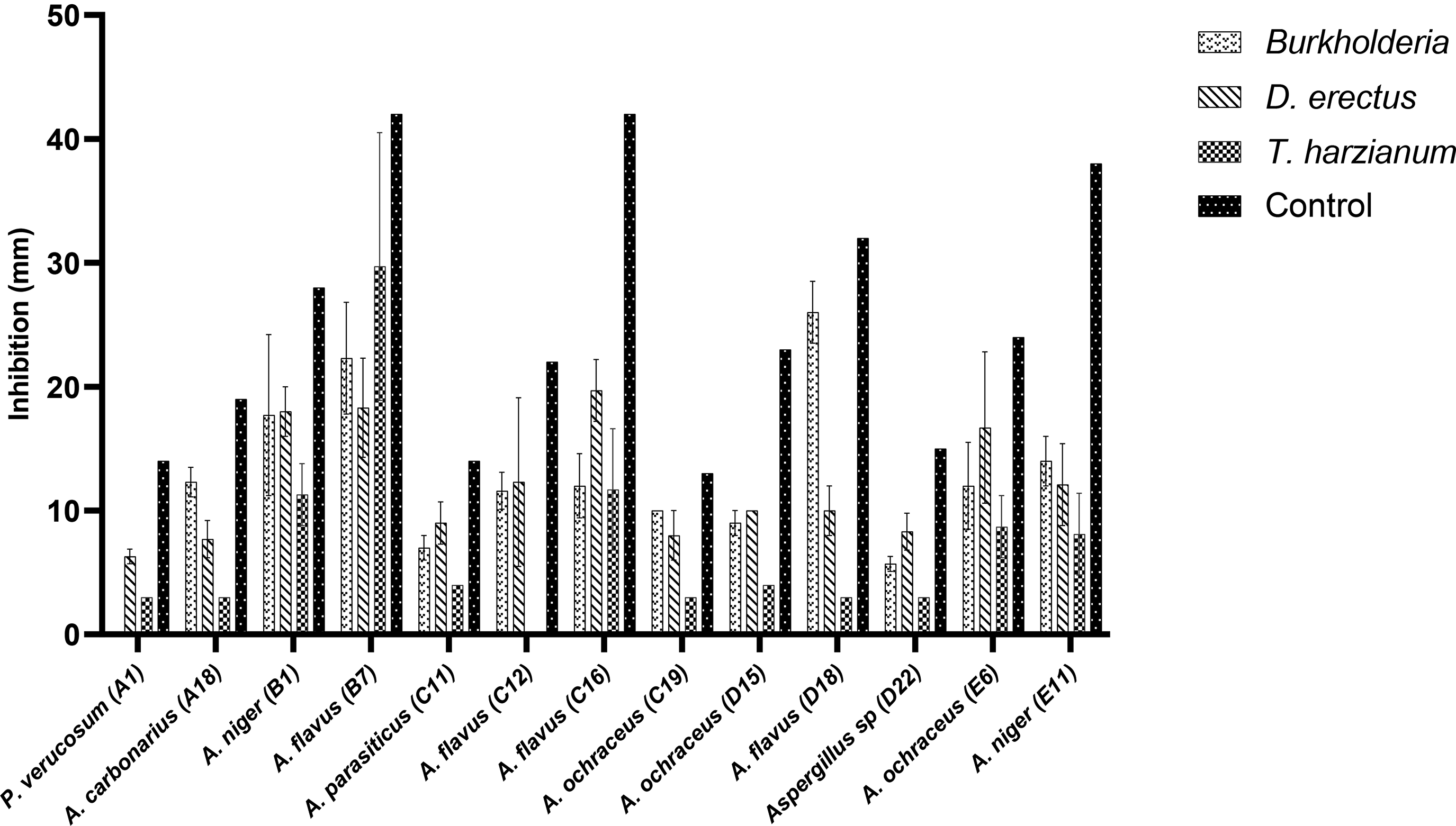
Inhibitory activity of extracts of endophytes against toxigenic fungi of maize.
Confrontation tests were carried out using the two endophytes with the best antifungal activities. Most of the toxigenic fungi resisted the endophytes with values higher than 1 as shown in Table 4. Aspergillus ochraceus (C19) and Aspergillus niger (B1) both did not resist the growth of the endophytic fungi with values less than 1.
Resistance of toxigenic fungi against endophytic fungi.
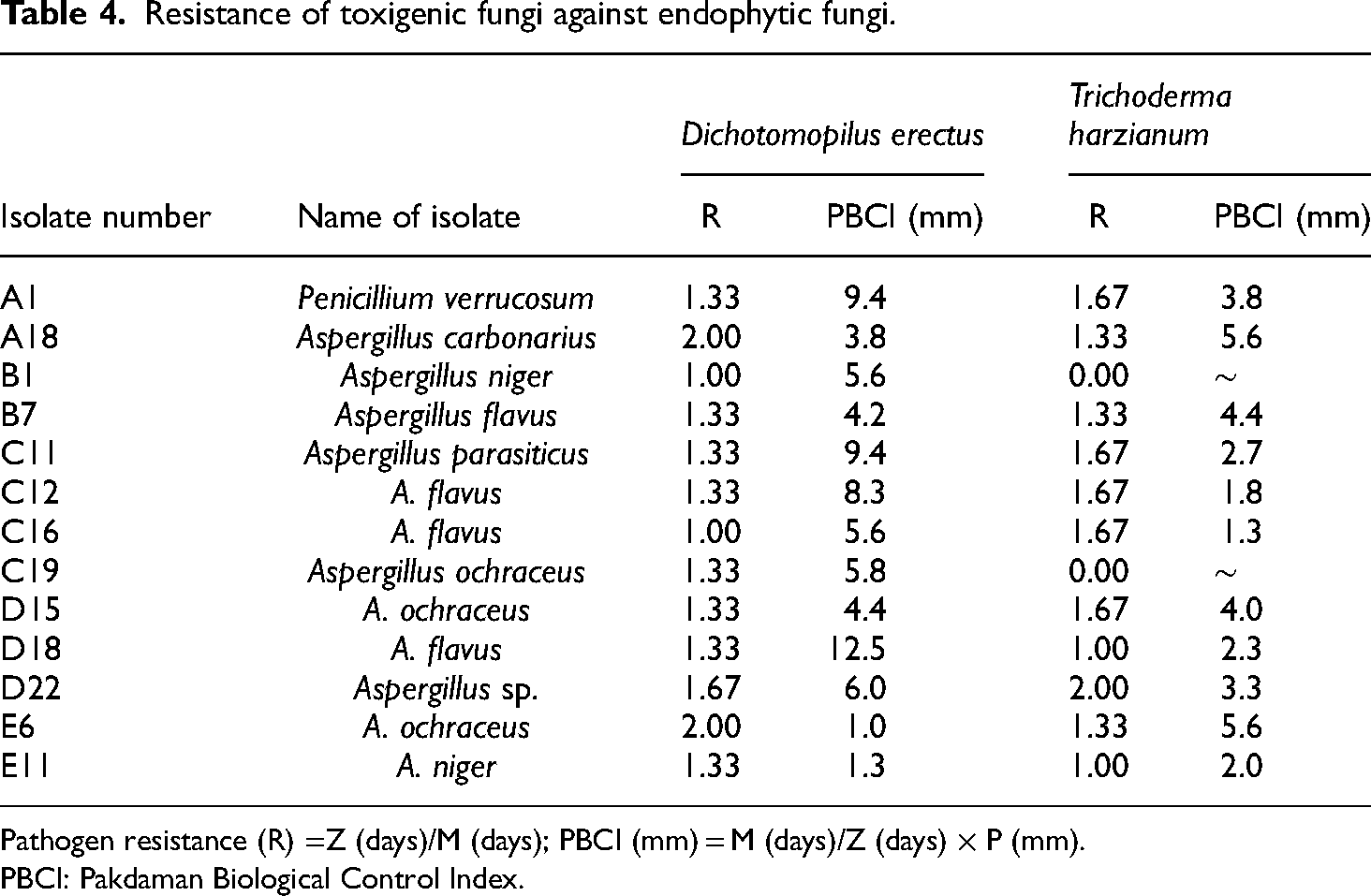
Pathogen resistance (R) =Z (days)/M (days); PBCI (mm) = M (days)/Z (days) × P (mm).
PBCI: Pakdaman Biological Control Index.
The lowest concentrations at which metabolites of the endophytic antifungal agents were recorded were Burkholderia spp. at 15.63 mg/mL (against A. flavus (C12) and A. ochraceus (D15)), D. erectus at 7.81 mg/mL (against A. ochraceus (D15)) and T. harzianum at 15.63 mg/mL (against A. ochraceus (C19) and A. flavus (D18)). The result of the minimum inhibitory concentration of antifungal endophytes against toxigenic fungal isolates of maize grains showed that T. harzianum had the best activity at a concentration of 62.50 mg/mL (Supplemental Table B).
Discussion
This study describes the isolation of endophytes from maize roots and the determination of their biocontrol activity against toxigenic fungi in Nigerian maize. The isolated fungi were high in number, and the species obtained were similar in the states. These could be attributed to shared climatic conditions (rainfall, temperature and humidity) in the collection region, and maize being a food source, rich in carbohydrates and other food nutrients that encourage fungal invasion.19,20 Poor storage facilities in the respective states where samples were collected may also have contributed to the high fungal contamination observed. Government policies and regulations concerning the storage of agricultural products are not strictly adhered to in these states, which might be another reason for the high fungal growth and diversity of species. Fusarium, Aspergillus and Penicillium spp. have been reported as contaminants of cereals, fruits and vegetable crops under poor storage conditions. 21
The capacity of Aspergillus spp. to invade growing maize plants and during storage, when conditions are favourable for producing toxins, may account for the high occurrence of Aspergillus spp. identified as mycotoxin producers in the study. 22 According to Mounjouenpou et al. 23 and Chen et al., 24 these fungi produced varying amounts of OTA in contaminated cocoa bean seeds and other crops, and the production of OTA by A. niger, A. ochraceus, Aspergillus carbonarius, and Penicillium chrysogenum observed in this study is consistent with their findings. The values of OTA and AFB1 produced by the fungi exceeded the tolerable recommended levels of 2 and 4 ng/g, respectively, set by the European Commission 25 in food sources to be consumed. This raises concern as the two mycotoxins are designated as carcinogenic metabolites by IARC. Utilising effective post-harvest techniques that can significantly minimise fungal colonisation and maintain the quality of crops and their nutritional state is essential for preventing fungal growth and the creation of toxins. 26 The values of AFB1 found in this investigation were greater than those found by Mwalwayo and Thole, 27 who found the highest value of 10 ng/g in maize seed from Malawi, but they were in agreement with Perrone et al. 28 who evaluated samples of maize from Nigeria. We found high concentrations of mycotoxins in grains which were higher than the tolerable values approved for consumed grains (aflatoxins – 10 ng/g and ochratoxins – 5 ng/g). 29
The choice of maize root endophytes as biocontrol agents were based on the beneficial behaviours of the strains isolated from the rhizosphere of maize plants. The endophytes had the ability to stimulate development and protect maize against infection. 30 When compared to imported strains for use as biocontrol agents, other reports from Nigeria indicated increased maize root mass. 31 while Clonostachys rosea, an endophytic fungus had reported antifungal properties, 32 Trichoderma spp. stimulate plant growth and development, induce plant resistance to diseases and enhance tolerance to abiotic stress in the host plant. Trichoderma spp. use one or more of mycoparasitism, hydrolytic enzyme synthesis and antifungal metabolite formation pathways to carry out their biocontrol actions. 33 Trichoderma and Acremonium spp. benefit plants by promoting root growth and enhancing resistance to root rot disease. The endophytes may also secrete saponins and other beneficial compounds such as indole-3-acetic acid and jasmonic acid which are involved in the biosynthetic pathway in host plants. 34
The dual culture assay was done in this study as a standard confrontation test to assess the inhibitory action of a biocontrol agent. 17 T. harzianum obtained the best antifungal activity, which is in agreement with the results of Pierre et al. 35 who got similar results in their study. Trichoderma spp. use a variety of tactics to combat diseases, including outgrowing their pathogenic competitors for nutrition and space, producing antibiotic chemicals, or turning on the host's defensive mechanisms. 36 According to Samuels, 37 the nature, quality and quantity of released metabolites against the pathogen determine how effective they are. Trichoderma spp. apart from its biocontrol potential also enhances plant growth 38 and elicits exudation of phytohormones, siderophores, enzymes and plant growth stimulator defence, which promotes plant vigour and development. Our result on the activity of Acremonium spp. contradicted that of Demirci et al. 36 who reported that the endophytic fungus had parasitic and antibiotic activities against crop pathogenic fungi. The PBCI shows how successful an endophyte is. The success of the anti-microbial potential of an endophyte increases with an increase in pathogen resistance to endophyte control and an increase in pathogen colony growth.
Biological control agents are taking the centre stage in disease management and control because they have numerous advantages over synthetic agents. They do not leave toxic substances after use; they act by promoting plant growth and development and maintaining balance in the environment. 39 Most of the toxigenic fungi resisted the two biocontrol endophytes tested at values ˃ 1. Toxigenic A. niger (B1) and A. ochraceus (C19) both showed no resistance to T. harzianum but promoted the growth and development of the endophytic fungus. The effectiveness of the antifungal endophytes might be due to their isolation from the same host as the toxigenic fungal isolates and so were competent indigenous competitors. The minimum inhibitory concentration of the tested biocontrol agents showed that the endophytic fungi mostly had inhibitory activities at a concentration of 31.25 mg/mL crude extract though Burkholderia spp. and D. erectus recorded lower values of 15.63 and 7.81 mg/mL crude extract concentration, respectively. The report of the inhibitory activity showed that a few endophytes had inhibitory activities while the majority did not show such potential.
Conclusion
Endophytes isolated from maize roots possess significant antifungal activity against some maize pathogens. The endophytes T. harzianum, Burkholderia spp. and D. erectus could inhibit the growth of toxigenic fungi from maize grains. Our study provides a list of endophytes resident in maize roots in Nigeria. These endophytes might be developed into biocontrol agents, which are expected to contribute to profitability and good quality of maize production. Indigenous biocontrol agents promote competitiveness and perform better at suppressing local pathogens and controlling diseases that can lead to the production of more food and reduce environmental pollution. Further work aimed at determining metabolites produced by the endophytes responsible for inhibition, and determining the interrelationships between the endophytes and the toxigenic fungi to manipulate them for beneficial uses are recommended.
Supplemental Material
sj-docx-1-sci-10.1177_00368504231186514 - Supplemental material for Molecular identification of endophytes from maize roots and their biocontrol potential against toxigenic fungi of Nigerian maize
Supplemental material, sj-docx-1-sci-10.1177_00368504231186514 for Molecular identification of endophytes from maize roots and their biocontrol potential against toxigenic fungi of Nigerian maize by Olukayode Olugbenga Orole, Timothy Olubisi Adejumo, Tobias Link and Ralf Thomas Voegele in Science Progress
Footnotes
Acknowledgement
The authors would like to thank Heike Popovitch, University of Hohenheim, for technical assistance.
Authors’ contributions
OOO designed the study, collected and analyzed samples, data and wrote the first draft of the manuscript. TOA analysed samples and data obtained, while TL and RTV were involved in genomic aspect of the study. RTV edited the final draft. All the authors read and approved the final copy of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by a research grant from the Alexander von Humboldt Foundation, Jean-Paul-Str. 12 D-53173 Bonn Germany.
Supplemental material
Supplemental material for this article is available online.
Author biographies
Olukayode Olugbenga Orole is a Lecturer and Research Scientist. His area of research is Plant Pathology, Biological Control Mechanisms and Public Health.
Timothy Olubisi Adejumo is a Professor in Environmental and Agricultural Microbiology. His area of research is Phytopathology and Microbial Ecology.
Tobias Link is Research Scientist. His area of research is Fungal Metabolism, Fungal Plant Pathology and Molecular Plant Pathology.
Ralf Thomas Voegele is a Professor of Phytopathology. His research areas span Phytopathology, Molecular Biology, and Fungal metabolism.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
