Abstract
The present study conducted an experimental investigation to impede the degradation of peanut (Arachis hypogaea L.) seeds and enhance their quality while being stored. The efficacy of eco-friendly chemicals such as ascorbic acid, salicylic acid, acetic acid, and propionic acid in seed preservation was evaluated over a period of six months. After a period of six months of storage in a greenhouse, an examination was conducted on peanut seeds that had undergone treatment. Rhizoctonia was observed after Cephalothorax, whereas Aspergillus, Fusarium, and Penicillium were the prevailing fungi throughout the storage period. The optimal outcomes were obtained from the conversion of acetic acid to propionic acid. The study observed a decline in seed oil, protein, carbohydrates, germination percentage, energy, index, length, vigour index, dead and rotten seeds, rotted seedlings, and surviving healthy seedlings, with an increase in storage duration ranging from zero to six months. The application of 100% propionic acid to peanut seeds throughout the storage duration resulted in decreased occurrences of deceased seeds, decaying seeds, and deteriorated seedlings. Peanut seeds that underwent treatment with green chemical agents of moderate and high intensity were found to be free of aflatoxin B1. The highest levels of chlorophylls a and b, carotenoids, and total phenols were observed in seeds stored in greenhouses and treated with 100% propionic acid and acetic acid extract. The application of propionic acid 100%, acetic acid 100%, salicylic acid 4 g/l and ascorbic acid 4 g/l proved to be the most effective treatments for peanut seeds, exhibiting the lowest total aflatoxin level of 0.40. The correlation coefficient between shoot fresh weight and shoot dry weight was found to be 0.99, whereas the correlation coefficient between root dry weight and shoot length was 0.67. The seed chemical analysis, seedling characteristics, and germination characteristics were subjected to clustering analysis, resulting in the formation of two distinct groups. The first group consisted of germination percentage and energy levels across all time points (0–6 months), while the second group consisted of the remaining characteristics. The findings of this research propose the utilisation of 100% propionic acid as a viable method for preserving peanut seeds and preventing their deterioration during storage. The application of 100% acetic acid has been found to be effective in enhancing the quality of seeds and minimising losses.
Keywords
Introduction
The viability and germination potential of seeds is reduced rapidly during storage, particularly in environments with high levels of moisture. The probability of a fungal infection interacting with harvested seed is heightened, thereby expediting the rate of seed deterioration during storage.1,2 Seed ageing is primarily associated with detrimental effects resulting from oxidative reactions. 3 These reactions can lead to the degradation of various cellular components such as proteins, including enzymes, 4 lipids, and cellular membranes, 5 RNA, 6 and DNA. 7 According to Kranner, 8 the various factors discussed have an adverse effect on the cellular and metabolic integrity of both seeds and seedlings. Arachis hypogaea L., commonly known as peanuts, holds significant value as an export crop in Egypt. Peanut seeds contain a significant proportion of oil, approximately 45–53%, which is characterised by a notable concentration of monounsaturated fatty acids and is devoid of cholesterol. This information has been reported in previous studies.9,10 Furthermore, it contains a substantial proportion of protein, ranging from 25% to 32%, and its byproducts are extensively acknowledged as a source of nourishment. It is widely accessible in numerous countries, especially those with a deficit of protein. Additionally, the existence of bioactive compounds in peanuts could potentially mitigate the likelihood of chronic ailments such as cancer, diabetes, and coronary heart disease.
Peanuts have been found to contain isoflavones and trans-resveratrol, as evidenced by prior identification and quantification efforts. 11 The principal cause of low crop productivity can be attributed to the incidence of bacterial infections.12,13 Peanut seed is susceptible to over 55 diseases caused by various pathogens such as fungi, bacteria, viruses, mycoplasma, nematodes, and parasitic flowering plants. Among these, fungal infections are the primary cause of output losses, as reported in the literature. 14 The susceptibility of peanuts to Aspergillus spp. is attributed to the presence of peanut pods in the soil, especially during pre- and post-harvest periods. As a result of infesting peanuts, A. flavus generates four variations of aflatoxins, which are identified as B1, B2, G1, and G2. According to Sun et al., 15 the subsequent isotopes with the highest fatality rates are G1, 132, and G2, respectively.
The presence of aflatoxigenic fungal propagules during peanut harvest can significantly diminish the quality and economic worth of the product. The optimal moisture range for harvesting peanut seed is reported to be between 18% and 25%, following a drying process that reduces the moisture content to approximately 12–14%. At times, the mechanism exhibits inefficiency, leading to the rapid production of aflatoxin due to environmental factors. 16 A correlation has been established between aflatoxins 17 and the susceptibility to primary liver cancer. The impact of the Aspergillus group flavi inoculum, tissue type, sampling time, antioxidant therapy, and their interactions on the fungal population was found to be significant.18,19 The most prevalent genera observed on peanuts were Penicillium, Aspergillus, and Fusarium. The species Aspergillus flavus exhibited the highest frequency of occurrence, and a significant disparity was observed in its incidence between peanut seeds that had undergone treatment and those that had not. Aflatoxins were not detected in either the treated or control samples during the storage period. Seed viability during storage is primarily determined by temperature and moisture content, as stated in the work by Vijay et al. 20
The presence of fungal colonisation in peanut seeds and high seed lipid content has been found to reduce the germination requirements and vigour index of seeds, ultimately hastening seed decomposition during storage. 21 The implementation of environmental modifications and chemical interventions for the purpose of mitigating plant diseases can be financially burdensome and may result in the eradication of beneficial microorganisms. The employment of safe pesticides for the management of plant diseases has been increasing steadily. 22 Several scholarly articles assert that chemical substrates have the potential to consistently induce resistance. 23 Induced disease resistance refers to the phenomenon whereby biotic or abiotic factors within the host plant give rise to physical or chemical barriers that confer resistance against disease.24,25 Therefore, it is necessary to implement alternative management strategies to alleviate the potential hazards that fungicides pose to both human health and the environment. Several authors have posited that chemical substrates that depend on physical or chemical barriers within the host plant can systematically enhance resistance.23–25
The implementation of biocontrol has sparked significant discourse across various domains, including technology, economics, and politics.26,27 This is aimed at promoting sustainable agricultural practices while minimising environmental degradation at a reduced cost. As a result, several countries have developed protective measures that can potentially decrease pesticide utilisation by up to 50%. 28 The signals presented unequivocally indicate a thorough comprehension of the buildup of dangerous substances in the ecosystem and the various interrelationships within the food web. Additionally, these findings demonstrate the limited alternatives available for reducing the agricultural sector's reliance on pesticides. In order to enhance the efficacy of biocontrols in this particular scenario, further research is required. 27
The aforementioned indicators collectively imply that the scientific inquiry pertaining to a biological control technique based on microbial inoculum is advancing satisfactorily. Integrate the aforementioned strategy with the previously discussed ones to formulate a proficient approach towards combating plant ailments. Currently, the primary purpose of seed treatment is to safeguard seedlings against fungal pathogens that originate from the soil and seeds during the initial weeks following sowing. A correlation has been established between conventional seed vigour metrics and the synthesis of secondary metabolites, as reported by Randhir and Shetty. 29
Water-soluble antioxidants such as ascorbic acid, propionic acid, acetic acid and salicylic acid have the ability to donate protons to the cytosol. Additionally, it has been observed that they serve as efficient growth regulators that augment seed vigour and induce the production of phenolic compounds. 30 Furthermore, it has been established that all instances of evaporation are associated with stress-induced activity in the pentose phosphate pathway and may serve as a passive stimulant for the biosynthesis of proline. 31
The objective of this research was to investigate the inhibitory effects of antioxidants and other evaporation factors on the formation of aflatoxins in peanut seeds during storage.
Therefore, they were extensively employed in the food industry as an antimicrobial preservative or acidulant in a range of citric, salicylic, propionic, and acetic acids, which impede multiple strains of peanut seed degradation across diverse storage durations.
Materials and methods
A storage experiment was conducted by the Plant Pathology Department of the Faculty of Agriculture at Mansoura University in Egypt. The experiment was conducted from November 2017 to May 2018. The present study investigated the inhibitory effects of propionic, acetic, ascorbic, and salicylic acids, as well as different concentrations of ascorbic and salicylic acids (2, 3 and 4 g/l), and propionic and acetic acids (25, 50 and 100%), on the deterioration of peanut seeds caused by multiple species during storage periods of 0, 2, 4 and 6 months. Results indicate that these acids exhibit significant inhibitory effects on peanut seed deterioration. A pot experiment was conducted at the Plant Pathology Department, Faculty of Agriculture, Mansoura University, Egypt, during the summer of 2018. The experiment lasted for 21 days and aimed to evaluate the response of peanut seeds to the treatments that were previously tested, following a storage period of 6 months.
Seed treatment
Subsequently, two filter papers that were saturated at each concentration were introduced into individual containers, specifically cloth bags, that held 500 g of seeds. Whatman Individual No. 1 filter papers with a diameter of 11 cm were immersed in evaporation matters, namely ascorbic and salicylic acids, acetic acids, and propionic acids, at different concentrations of 25%, 50% and 100% until they reached saturation. Control seeds that were not subjected to any treatment were used. Following the process of evaporation, peanut seeds were subjected to storage in a laboratory setting for varying durations of 0, 2, 4 and 6 months. The initial moisture content of the seeds was recorded at 9%.
Laboratory experiments
Seed health testing
The techniques utilised for the detection of seed-borne mycoflora were in accordance with the guidelines established by the International Seed Testing Association. Two hundred seeds from each sample and the control group were subjected to testing using the standard blotter method. A total of ten seeds were sown in a petri dish measuring 11 cm in diameter, which was layered with three blotters soaked in water. The water used was sterile tap water. The plates were subjected to a temperature of 202 °C and exposed to alternating cycles of cold white fluorescent light and darkness for a duration of seven days. The plates were scrutinised for the purpose of detecting and analysing seed-borne fungus using a stereoscopic binocular microscope with a magnification range of 6–50×. The morphology of conidia and conidiophores was investigated, and confirmation of identification was conducted using the compound microscope as required. Fungal presence on seeds was identified through a comparative analysis with descriptions provided by the Danish Government Institute of Seed Pathology (DGISP) and the Commonwealth Mycological Institute located in Kew, Surrey, England (CMI).32–35
Disease assessment
For each storage time, the percentages of dead and rotting seeds (non-germinated seed), as well as rotted and survivor seedlings, were recorded.
Germination test
In accordance with the Regulation of the International Seed Testing Association, an assessment was conducted on both treated and untreated seeds to determine their respective germination rates. During the initial 10-day period following the onset of germination, regular tallies of the number of developing seeds were recorded. Germination energy (GE) was calculated by determining the proportion of germinated seeds in relation to the total number of seeds assessed on the fourth day following sowing.
Germination percentage and seedling characters
In order to assess the germination percentage and seedling characteristics, both treated and untreated seeds were sown in sterilised sand and left for a period of 10 days. The percentage of germination was ascertained by considering the total count of viable seedlings at the end of the experiment. The length of ten seedlings was measured in centimetres (cm), and their dry weight in grams (g) was determined by subjecting them to a 24-h drying process at 105 °C in a forced-air oven. The Seedling Vigour Index (SVI) was formulated using the equation that has been suggested in the literature.
36
Seed chemical analysis
Seed samples from each treatment were subjected to an oven-drying process and subsequently pulverised to a fine consistency for the purpose of a chemical examination. The AOAC 37 method was employed to determine the proportion of seed oil extracted using the Soxhlet apparatus and petroleum hexane 30 as an organic solvent. The seed nitrogen percentage was determined using the Micro Kjeldahl instrument and then multiplied by the conversion factor of 6.25.
Greenhouse experiment
The objective of this study was to investigate the effects of two different evaporation materials on the morphological and physiological characteristics of seedlings. To achieve this, samples of each treatment, along with untreated control seeds, were gathered and planted in plastic pots for a duration of 21 days following a 6-month storage period.
Morphological characters
The experimental units for each treatment were cultivated in plastic containers measuring 25 cm in diameter and filled with a soil mixture weighing 2 kg, comprised of a ratio of 2 parts sand to 1 part clay. Ten peanut seeds were sown in each pot under controlled environmental conditions of 21.3 °C and 58% relative humidity. Following the germination period of 10 days, the plants underwent pruning, resulting in the retention of only three seedlings. Equal amounts of water were used for each pot during every watering session in the experiment. Upon completion of the experiment, assessments were conducted on the shoot and root lengths (cm), fresh weights (g), and drying weights (g) after 21 days of planting.
Physiological characters
At 21 days after planting, the third leaf from the tip's blade was cut off to examine the presence of photosynthetic pigments (chlorophyll a, b, and carotenoids), which were extracted with methanol and measured. The folin-ciocalteau reagent was used to determine the total phenols in early shoots.
Determination of aflatoxins
The present study employed the AFB1, B2, G1, and G2 standards from Sigma, a chemical business based in the United States. The aflatoxins were subjected to removal, cleaning and identification in accordance with the guidelines set forth by the AOAC. 37 The recovery of AFs from desiccated S. platensis biomass was achieved through the utilisation of CB, as reported by AOAC. 37 A 50-g sample was introduced into a 500 ml Erlenmeyer flask along with 25 g of diatomaceous earth, 250 ml of chloroform, and 25 ml of distilled water. The mixture was subjected to horizontal shaking for a duration of 30 min using a shaker. The extract was subjected to filtration using Whatman No. 4 filter paper. The initial 50 cc of the extract were purified using a silica gel column. A quantity of 5 g of anhydrous sodium sulphate was introduced into a glass column measuring 22 by 300 mm for the purpose of creating chromatographic columns. The slurry composed of chloroform and 10 g of silica gel was utilised to coat the chromatographic column. The stopcock was opened to allow for the settling of the silica gel packing during the process of draining chloroform. In order to avoid desiccation, a quantity of 10 g of anhydrous sodium sulphate was introduced to the silica gel whilst draining.
Statistical analysis
Analysis of variance was used to assess the study's data. Differences in treatment means were statistically assessed using Duncan's multiple tests with a p-value of 0.05 using the CoStat V6.4 tool. R software was used to calculate the Pearson correlation and cluster heatmap.
Results
Laboratory experiments for peanuts
Seed health testing
According to Table S1, peanut seed mycology revealed 15 filamentous fungi from 11 taxa. Alternaria, Aspergillus, Cephalosporium, Fusarium, Macrophomina, Penicillium, Rhizoctonia, Rhizopus, Verticillium, Trichothecium, and Stemphylium participated. Alternaria, Aspergillus, Penicillium, Rhizopus, and Trichothecium populations grew with storage time, while other species decreased in the untreated control. Fusarium, Aspergillus, and Penicillium predominated. Rhizoctonia, Stemphlium, and Cephalosporium came later. Fusarium solani, Penicillium sp., Aspergillus flavus, and Rhizopus sp. predominated throughout storage. During storage, ascorbic, salicylic, acetic, and propionic acids suppress fungal genera. This effect increased as both evaporations concentrated from 25% to 100%. Propionic and acetic acids suppressed fungus populations the most. The high percentage (100%) and salicylic and ascorbic acid content (4 g/I) were more effective, except for A. flavus and A. niger.
Disease assessment
In each storage period, disease was shown by dead seeds, rotten seeds (seeds that never grew), rotten seedlings, and survival seedlings. Table 1 reveals that the number of dead and decaying seeds and seedlings increased dramatically with storage duration. Hence, surviving seedlings offered the reverse. At six months, the untreated control had the fewest healthy seedlings and the most dead and rotten seeds and seedlings. The four acids (ascorbic, salicylic, acetic, and propionic) cut down on the number of dead and rotting seeds and seedlings and made the ones that survived better. These impacts increased as evaporation concentrated the ingredients.
The percentage of dead seeds, rotted seeds, rotted seedlings, and survival seedlings for peanuts treated with evaporation and antioxidant treatment at various storage times.

At p ≤ 0.05, values with different letters indicate there are significant differences between the treatments.
Germination characters
The duration of storage resulted in a decrease in both the germination percentage and energy. The enhancement of peanut seed energy and germination was observed through the influence of evaporation factors and the presence of acetic, propionic, ascorbic, and salicylic acids. The germination rates of propionic and acetic acids were observed to be 100%. However, no significant association was found between the storage durations and evaporation factors, as indicated in Table S2.
Seedling characters
Length of the seedling, dry weight of 10 seedlings, vigour index, and storage times are all negatively related to each other. Seed treatment with any concentration of both evaporation materials considerably improved the metrics compared to the control. Acetic and propionic acids performed better; evaporation materials reduced seedling dry weight losses from storage periods. The length of time the seeds were kept in storage and the treatments did not affect the vigour index or length of the seedlings (Table S3).
Seed chemical analysis
Table 2 shows that as storage time increased, peanut seed oil and protein content declined dramatically. The seeds were held for a total of six months before their base values were used to create these characters. After 2, 4, and 6 months of storage, oil and seed quality were better in seeds that had been treated with peanut seed protein. It was discovered that propionic acid worked the best. Chemical studies of the seeds showed no effect of the interaction between storage time and any of the treatments.
The percentages of oil and protein for peanut seeds treated with evaporation and antioxidant treatments at various storage times.
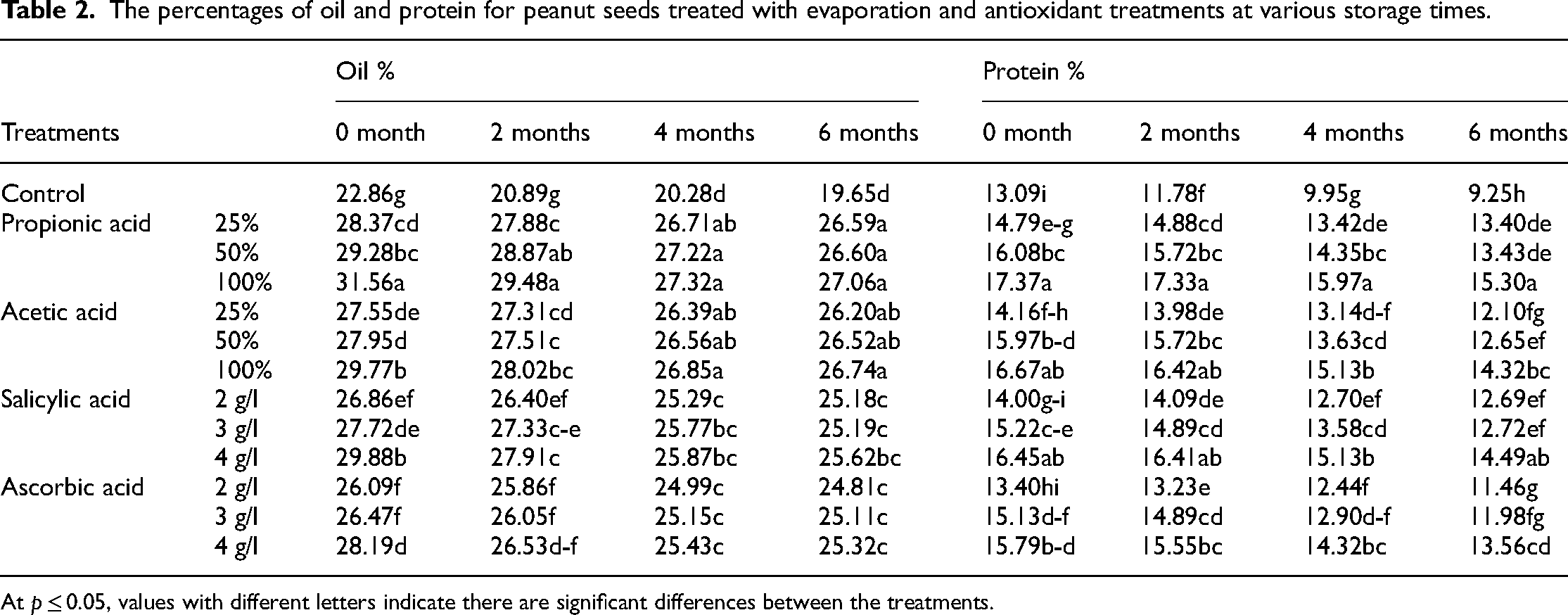
At p ≤ 0.05, values with different letters indicate there are significant differences between the treatments.
Greenhouse experiment
Morphological characteristics of seedlings
According to the data presented in Table S4, the application of propionic and acetic acid at varying concentrations resulted in increased shoot and root length, as well as greater fresh and dry weight of seedlings, compared to the untreated control group, at the 21-day mark after planting. The relationship between the concentration of substances and the resulting outcomes is direct. The optimal outcomes for the morphological characteristics of seedlings are observed when utilising a propionic acid solution with a concentration of 100%, succeeded by an acetic acid solution with a concentration of 100%.
Physiological characteristics of seedlings
On the 21st day post-sowing, an analysis was conducted on the seedlings to determine the levels of chlorophyll a, b, and carotenoids, in addition to total phenols. The application of propionic and acetic acids resulted in a statistically significant increase in both total phenols and photosynthetic pigments in peanut seedlings, as evidenced by the data presented in Table S5. The optimal values are attained by initially employing a 100% concentration of propionic acid, followed by a 100% concentration of acetic acid. It is imperative to acknowledge that augmenting the concentration of propionic acid enhanced its efficacy. In contrast, acetic acid exhibited a contrary impact. Table 3 displays the most effective treatments for achieving the absence of various aflatoxins (AF G1, AF B1, AF G2, and AF B2) in peanut seeds, with a total aflatoxin concentration as low as 0.40. The treatments that yielded the best results were propionic acid at 100%, acetic acid at 100%, salicylic acid at 4 g/l, and ascorbic acid at 4 g/l.
The total content of aflatoxins and the percentage of their different types in control and treated peanut seeds.

Data analysis
The Pearson correlation between the morphological and physiological characteristics of the peanut plant is illustrated in Figure 1, both under normal conditions and following treatment with antioxidants. The study found a strong correlation of 0.99 between shoot fresh weight and shoot dry weight, while a weaker correlation of 0.67 was observed between root dry weight and shoot length. The data was subjected to a clustered heatmap analysis, which facilitated the classification of seed chemical analysis, seedling characters, and germination characters into two distinct clusters. The first cluster comprised germination percentage and GE across all time intervals (0–6 months), while the second cluster encompassed the remaining characteristics, as illustrated in Figure 2.

Pearson correlation between morphological and physiological characteristics of peanut plants at control and after treatment with antioxidants. The diameter of the circle indicates how strong the correlation is; the larger the circle, the stronger the association.

Clustered heatmap between seed chemical analysis, seedling characteristics, and germination characters of peanut plants at control and after treatment with antioxidants. The variable level is shown by the colour scale, which goes from 2 (deep red) to −2 (deep blue).
Discussion
Seed treatment is a crucial aspect of plant conservation, primarily used to safeguard nascent seedlings from soil and seedborne fungal infections post-planting. Seed treatment is known to impede or halt the degradation of seeds, particularly those of the oily variety, subsequent to the process of harvesting. The present investigation revealed that the application of salicylic acid, acetic acid, propionic acid, and ascorbic acid exhibited fungistatic and disease-control properties in peanut seeds for a duration of six months. Furthermore, the treatment resulted in enhanced germination standards, seed quality, and seedling physiology and morphology. The spoilage of seeds during storage can be attributed to various reasons that can be categorised into four groups, namely: (a) nutritional variables inherent to the seed; (b) external factors affecting the seed; (c) processing-related factors; and (d) implicit factors, 38 all of which are caused by fungi. The population of fungi is subject to fluctuations during the storage period due to various factors.
A prevalent strain of Aspergillus, known as A. flavus, is among the most frequently encountered types. According to Mahmoud et al., 39 A. flavus is more prevalent in peanut seed samples compared to A. parasiticus. The aggressive nature of A. flavus and the biochemical structure of seeds make peanut seeds an optimal substrate for the growth of this fungus, resulting in the production of aflatoxin.40–42 The severity of pre- and post-emergence damping-off, which is caused by various fungi such as Cephalosporium sp., F. verticillioids, F. oxysporum, F. solani, R. solani, and V. dahliae, was significantly decreased when the faba bean seeds were treated with a water suspension containing 109 CFU mL-1 of S. eerevisiae before planting.
The efficacy of a root-colonising microbe as a biocontrol agent against soil-borne diseases and its consequent positive impact on plant growth were also demonstrated by Youssef et al. 43 and Bleve et al. 44 According to El-Mehalawy et al., 45 the primary factor contributing to the proliferation of microorganisms in the rhizosphere is their capacity for survival and growth. The symbiotic relationship between plants and microbes facilitates plant growth through various mechanisms such as siderophores production, solubilisation of phosphate, plant regulation, and increased nutrient availability. 46 The application of environmentally friendly chemicals, namely salicylic acid, acetic acid, propionic acid, and ascorbic acid, has been found to effectively decrease the overall fungal count on peanut seeds and promote resistance to damping-off. 47 The chemicals in question operate through the process of inducing the synthesis of pathogenesis-related proteins, thereby resulting in partial resistance to pathogens. Additionally, they serve to enhance the activity of oxidative enzymes, specifically peroxidase and polyphenol oxidase, while also increasing the content of phenol compounds. Phenolics have gained popularity as plant chemicals due to their antifungal, antibacterial, and antiviral properties.
Alterations in oxidative enzymes and phenolic concentration enhance resistance, a phenomenon that is analogous to the manifestation of resistance in diverse host-pathogen interactions. 48 The involvement of peroxidase activity has been observed in the pathogenesis of various diseases. Plants promptly deposit phenols at the infection site as an initial defence mechanism against pathogens. 49 According to Chen et al., 50 the accumulation of salicylic acid, acetic acid, propionic acid, and ascorbic acid in close proximity to incompatible infection sites plays a crucial role in regulating defence responses. They constitute an integral component of the innate immune system and contribute to the development of adaptive immunity. The catalytic activity of catalase can be altered from dismutation to peroxidation through the utilisation of substrates that donate a single electron, such as ascorbic acid, salicylic acid, acetic acid, and propionic acid. 51 Consequently, the enzyme becomes entrapped in a state of reduced activity, leading to a decreased elimination of H2O2. Kumar 52 have proposed that altering the configuration of certain natural phytoalexins to enhance the stability of potential radical intermediates and equip them with antioxidant properties is crucial in mitigating infection during storage conditions. Salicylic acid, acetic acid, propionic acid, and ascorbic acid exhibit antimicrobial properties. However, it is worth noting that oxidised chemicals could potentially have an impact on membrane lipids due to their ability to dissolve in said lipids. This interference could potentially disrupt important membrane functions such as the transport of nutrients and the synthesis of proteins, RNA, and DNA. 53
The application of salicylic acid, acetic acid, propionic acid, and ascorbic acid to seeds has been observed to enhance the morphological characteristics of peanut seedlings. In contrast to full doses, the utilisation of 25% concentrations of salicylic acid, acetic acid, propionic acid, and ascorbic acid in this investigation exhibited a comparatively reduced impact. One plausible explanation could be the adverse impact on the physiological processes of plants when exposed to high levels of dosage. The reduction of transmembrane electrochemical potential in mitochondria resulted in a decrease in the uptake of potassium and phosphorus, leading to a deceleration in ATP synthesis. Modifications in the conductivity of stomata result in reduced transpiration through the foliage. The graphical representation of the hierarchical cluster heatmap is a useful tool for depicting the interrelationships and associations among various experimental conditions and sample parameters.
The integration of heatmaps with hierarchical categories that are based on similarity or distance has been identified as a valuable approach.54,55 The present study's heatmap cluster illustrates the resemblance between the attributes of seedlings and those of germinating seeds.
Conclusion
Overall, the storage of seeds resulted in a decrease in their oil, protein, and carbohydrate levels across all experimental conditions. Following a storage period of six months, the chemical compounds exhibited their minimum levels. The observed loss pattern may be attributed to various factors such as accelerated deterioration of stored seeds, heightened respiratory activity, or oxidation of amino acids. Therefore, prolonged seed storage would lead to heightened metabolic activity, concomitant with reduced dry matter weight and reserve substance concentration. The present investigation revealed that the use of eco-friendly chemical agents enhanced the sustainability of peanut seeds during storage, resulting in a reduction in aflatoxin B1 levels and an elevation in seed oil, protein, and carbohydrate content. It is suggested that the application of salicylic acid, acetic acid, propionic acid, and ascorbic acid as seed treatments may improve seed quality and mitigate peanut seed degradation during storage.
Supplemental Material
sj-docx-1-sci-10.1177_00368504231176165 - Supplemental material for Impact of green antioxidants on decreasing the aflatoxins percentage in peanut oil seed (Arachis hypogaea L.) during storage
Supplemental material, sj-docx-1-sci-10.1177_00368504231176165 for Impact of green antioxidants on decreasing the aflatoxins percentage in peanut oil seed (Arachis hypogaea L.) during storage by Esraa Mohsen, Mohamed A El-Metwally, Amira A Ibrahim and Magda I Soliman in Science Progress
Footnotes
Author contributions
AAI and EM conceived and designed the experiments; AAI and ME analysed the data; AAI and MS summarised the data; AAI and ME wrote the manuscript; all authors revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Author biographies
Esraa Mohsen is MSc candidate and she interested in microbiology, molecular biology and genetics.
Mohamed A El-Metwally is Professor of Mycology in Plant Pathology Research Institute, Egypt. He interested in seed borne fungi and biotic stress in plants and biotechnology.
Amira A Ibrahim is Assistant Professor of Cytology and Genetics, Botany Department, Faculty of Science, Arish University. She received her PhD in 2018 from Mansoura University, Egypt. She has published more than 30 research/review and chapters in high quality international and local journals. She interested in DNA barcoding, plant breeding, karyotype, genome editing and molecular biology.
Magda I Soliman is Professor of Cytology and Genetics in Faculty of Science, Mansoura University, Egypt and she interested in Chromosome, karyotype, cytogenetics and molecular biology. Prof. Magda has published more than 70 research/review and chapters in high quality international and local journals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

