Abstract
Background
The level of vitamin D in follicular fluid (FF) according to the ovarian reserve has never been investigated, and the effect of FF vitamin D on the outcome of assisted reproductive technology (ART) remains controversial. The aim of this study is to evaluate the association between FF vitamin D levels and baseline anti-Müllerian hormone (AMH) / ART outcomes.
Methods
Forty-seven patients who underwent controlled ovarian stimulation at the fertility clinic of an academic tertiary care center were enrolled for a prospective observational study. FF was collected from the first aspirated leading follicle of each ovary and assayed by an enzyme-linked immunosorbent assay. Multivariable linear regression analysis was used to assess the association between baseline AMH and FF vitamin D levels with adjustment for basal FSH and serum vitamin D levels.
Results
Both the AMH and serum vitamin D were significant predictors for FF vitamin D. The estimated marginal mean of FF vitamin D level was higher in women with decreased ovarian reserve (DOR) than those with normal ovarian reserve (24.1 ± 2.1 vs. 18.8 ± 1.4 ng/ml, p = 0.048). However, FF vitamin D did not demonstrate any significant associations with cycle outcomes, including fertilization rate and the number and proportion of good embryos at day three.
Conclusion
We observed significantly higher FF vitamin D levels in women with DOR. However, FF vitamin D did not demonstrate any significant associations with the outcome of ART. A larger prospective study is needed to investigate the effect of FF vitamin D on the clinical pregnancy rate and live birth rate.
Introduction
Vitamin D plays an important role in calcium and phosphorus homeostasis and bone mineralization, 1 and these biological actions of vitamin D are exerted through vitamin D receptors. Vitamin D receptors are identified in calcium-regulating tissues as well as in the reproductive endocrine system, including ovary, uterus, pituitary gland, and hypothalamus,2–5 and its distribution suggests a much wider role of vitamin D in female reproductive physiology.6–8 A recent meta-analysis regarding infertility reported that women with a normal level of serum vitamin D were more likely to achieve pregnancies or live births after assisted reproductive treatment (ART) than women with vitamin D deficiency. 9
However, serum levels of biomarkers might not represent an intrafollicular microenvironment in women who undergo ART. Follicular growth is largely influenced by an intrafollicular microenvironment which provides the essential functions for the oocyte developmental competence through somatic cell-oocyte signaling. 10 Therefore, follicular fluid (FF) may be considered the best specimen to evaluate this microenvironment. Previous studies have investigated the association between FF vitamin D levels and ART outcomes in infertile women; however, the results were inconclusive.11–14 These inconsistent findings might be because FF vitamin D levels according to ovarian reserve were not considered in the studies. Indeed, an altered follicular cell metabolism or metabolites were found in women with decreased ovarian reserve (DOR). 15
In this study, we evaluated FF vitamin D levels according to the status of ovarian reserve and the association between FF vitamin D levels and the development of embryos.
Materials and methods
Patient population
Infertile couples who visited the fertility clinic of Seoul National University Hospital from June 2017 to December 2019 were recruited and provided written informed consent to participate in this prospective observational study. The study participants were aged between 29–43 years and underwent a controlled ovarian stimulation (COS) cycle for fresh embryo transfer or fertility preservation.
Initially, a total of 58 women participated in this study. The exclusion criteria were as follows: [1] COS cycles were canceled, or FF could not be collected (n = 7); [2] cycles where oocyte was not retrieved (n = 4). Finally, forty-seven women with at least one oocyte retrieved in whom both serum and FF samples were available for data analysis. The Institutional Review Board of Seoul National University Hospital approved the study protocol (IRB No. 1706-052-858).
Ovarian stimulation
For the GnRH agonist (GnRHa) long protocol, triptorelin 0.1 mg (Decapeptyl®; Ferring Pharmaceuticals, SaintPrex, Switzerland) was initiated during the midluteal phase of the previous cycle after documentation of ovulation (serum progesterone level > 3 ng/mL). Daily injections of recombinant follicle-stimulating hormone (FSH; Gonal-F®, Serono, Geneva, Switzerland) commenced on cycle day 2 or 3 until human chorionic gonadotropin (hCG) was triggered. The dosage of gonadotropin was determined based on the age, antral follicle count (AFC), and the baseline level of anti-Müllerian hormone (AMH) of each woman, and ranged from 75 IU to 450 IU per day. For the flexible GnRH antagonist (GnRHant) protocol, cetrorelix 0.25 mg (Cetrotide®, Serono) was added when the mean diameter of the leading follicle had reached 14 mm to prevent premature luteinizing hormone (LH) surge. Patients who planned fertility preservation (n = 3) started COS with the flexible GnRHant protocol regardless of the menstrual cycle. Oocyte maturation was triggered by 250 µg of recombinant hCG (Ovidrel®, Serono) or 10,000 IU of urinary hCG (IVF-C®, LG Chemical, Seoul, Korea) when the leading follicle had reached the mean diameter of 18 mm, or at least two follicles attained a diameter of 17 mm. Transvaginal ultrasound-guided oocyte retrieval was performed 34–36 h after the hCG trigger. On the day of aspiration, FF was collected from the first aspirated leading follicle in each ovary. The fluid was collected in a separate tube without Dulbecco's phosphate-buffered saline (D-PBS).
Fertilization and embryo transfer policy
Insemination or injection of the sperm was executed 4–6 h after oocyte retrieval. Intracytoplasmic sperm injection was performed in infertile couples with a history of severe male factor or previous fertilization failure. The presence of 2 pronuclei (2PN) was assessed 16–18 h after the insemination or injection of the sperm. We cultured embryos with 2PN in Continuous single culture complete (FUJIFILM Irvine Scientific, CA) overlaid with Hypure oil heavy (Kitazato IVF, Shizuoka, Japan) in culture dishes at 37°C under 6% CO2 and 5% O2 until the day of embryo transfer. The grade of embryos was evaluated on day 3. A good embryo was defined as an embryo that cleaved to more than 6 cells with fragmentation less than 20% and equally sized blastomeres without multinucleation. 16
Anti-Müllerian hormone (AMH) measurement
The level of serum AMH was determined using an AMH Gen II enzyme-linked immunosorbent assay (ELISA) (Beckman Coulter Inc., La Brea, CA) before December 2018 and using an automated Access AMH Chemiluminescent immunoassay (CLIA) (Beckman Coulter) after December 2018. The conversion equation was AMH (CLIA) = 0.868 × AMH (Gen II) – 0.039, and the correlation coefficient between the two methods was 0.996. Although there is a lack of standardization in AMH measurement, these two methods are provided by the same manufacturer and the results of the two assay types were highly correlated. 17
Vitamin D measurement
Blood samples were collected in a lithium heparin tube on the day of the trigger, and each blood sample was centrifuged at 1500 g for 10 min. FF samples were centrifuged at 300 g for 15 min immediately after the confirmation of the presence of oocytes. Supernatants of blood and FF samples were separated and stored at −196°C liquid-nitrogen tank and −80°C deep freezer, respectively, until vitamin D was assayed.
The level of vitamin D was determined by the Human 25-(OH) vitamin D3 ELISA Kit (CUSABIO, Houston, TX, USA). The lower limit of measurement was 4.8 ng/ml. As described in the instructions, precision was evaluated in accordance with a modified protocol based on CLSI EP-5A2. 18 The total coefficients of variation ranged from 0.021% to 11.714%. Intraassay and interassay coefficient variations of the kit were less than 10%, and 15%, respectively.
Data collection and analysis
Baseline characteristics were collected through medical records review. Body mass index (BMI) and basal FSH, estradiol, and AMH were measured before COS in all women as a routine clinical practice. DOR was defined as a serum level of AMH < 1 ng/ml, according to previous studies.19–21 Fertilization rate was calculated as the number of 2PN divided by the number of mature oocytes on the day after ovum aspiration. The proportion of good embryos was defined as the number of good embryos divided by the number of total available embryos on the third day after fertilization.
Questionnaire
Before retrieving oocytes, patients completed the questionnaire, including the questions about regular physical activity, usage of sunscreen, and the mean duration of sunlight exposure every day.
Statistical analysis
Continuous variables, including age, BMI, and serum concentration of vitamin D, are expressed as mean ± standard deviation, or median (interquartile ranges) as appropriate, whereas categorical variables are presented as number and percentage. Clinical and cycle characteristics by ovarian reserve were assessed using a t-test and Mann-Whitney U-test for continuous predictors and a chi-square test for categorical predictors. The effect of each variable on FF vitamin D level was estimated using univariable regression analysis. Multivariable linear regression analysis was used to assess the association between DOR and FF vitamin D adjusting for basal FSH and the level of serum vitamin D. Multicollinearity in the regression analysis was evaluated by the variance inflation factor and condition index. The normal distribution of the residuals in the regression model was analyzed by the Kernel density plot and Shapiro-Wilk test, Homoscedasticity was evaluated by plotting the residuals versus fitted values. Nonconstancy of variance of the residuals was complemented using regression analysis with robust standard error. Count outcome variables, including the number of retrieved oocytes, available embryos on day 3, and good embryos on day 3, were regressed on FF vitamin D level using Poisson regression and negative binomial regression, as appropriate. We analyzed proportion outcomes such as fertilization rates on day 1 or good embryos/ available embryos on day 3 with fractional response regression. All statistical analyses were conducted using Stata 15.1 (StataCorp LLC, College Station, Texas, USA). Statistical significance was determined using 2-sided P values of 0.05 or less.
Results
Table 1 provides the clinical and cycle characteristics. Women with DOR tended to be older (37.9 ± 4.5 vs. 35.6 ± 3.4 years, P = 0.08) and to have higher basal FSH (7.8 ± 1.2 vs. 4.5 ± 2.3 mIU/ml, P = 0.01), as compared to women with normal ovarian reserve. They were also more likely to have lower BMI (21.3 ± 2.3 kg/m2) than women with normal ovarian reserve (23.6 ± 4.8 kg/m2, P = 0.03). However, the proportion of sunscreen use, sunlight exposure, regular exercise, and primary infertility were not statistically different according to ovarian reserve status. Also, serum concentration of vitamin D was comparable between the two groups. As expected, the number of retrieved oocytes or available embryos on day 3 was lower in women with DOR than in those with normal ovarian reserve. However, the proportion of good embryos on day 3 was not different in the two groups.
Baseline characteristics of study participants.

Values are presented as mean ± SD, median (interquartile range) or number (%).
DOR, decreased ovarian reserve; BMI, body mass index; FSH, follicle stimulating hormone; E2, estradiol.
P by t-test or chi-square test.
P* by Mann-Whitney U test or Fisher's exact test.
Both ovarian reserve status and serum vitamin D were significant predictors for FF vitamin D (Table 2). Women with DOR had 7.88 ng/ml higher FF vitamin D concentration than women with normal ovarian reserve. The estimated marginal mean of FF vitamin D was still higher in women with DOR than those with normal ovarian reserve (24.1 ± 2.1 vs. 18.8 ± 1.4 ng/ml, P = 0.048), holding basal FSH and the level of serum vitamin D constant (Table 3). However, scaled FF vitamin D did not demonstrate any significant associations with cycle outcomes, including fertilization rate and the number and proportion of good embryos on day 3 (Table 4).
Univariable regression analysis for follicular fluid 25-(OH) vitamin D3.

FSH, follicle stimulating hormone; E2, estradiol.
p value by regression analysis.
Multivariable analysis for follicular fluid 25-(OH) vitamin D3.

Unadjusted data are presented as mean ± standard deviation and adjusted data are presented as mean
Data† are adjusted for basal follicle stimulating hormone and serum 25-(OH) vitamin D3.
p value by regression analysis with robust standard errors.
Regression analysis of follicular fluid 25-(OH) vitamin D3 /10 on cycle outcomes.
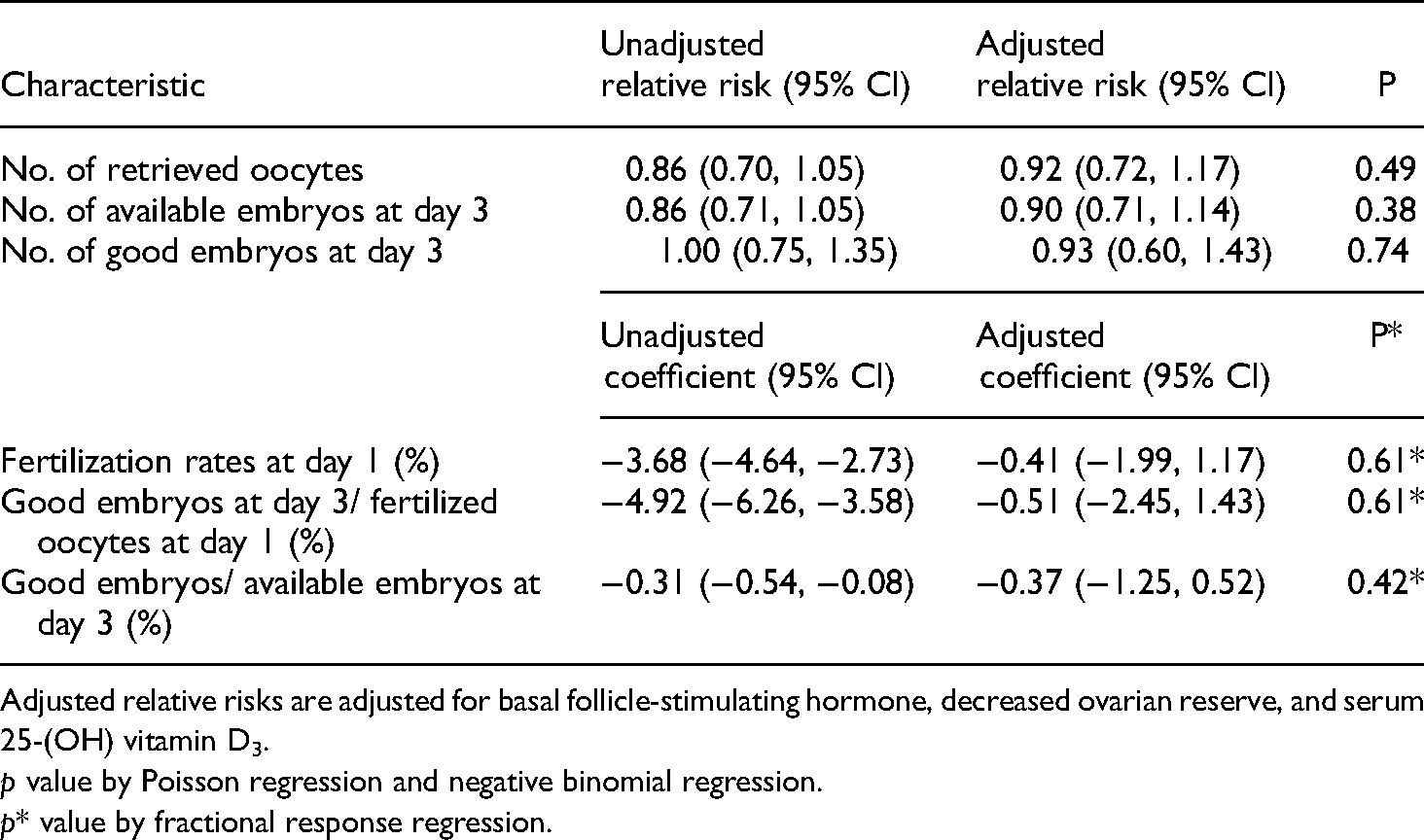
Adjusted relative risks are adjusted for basal follicle-stimulating hormone, decreased ovarian reserve, and serum 25-(OH) vitamin D3.
p value by Poisson regression and negative binomial regression.
p* value by fractional response regression.
Discussion
In the present study, women with DOR demonstrated significantly higher vitamin D levels in FF than those with normal ovarian reserve. To our knowledge, this is the first study to evaluate FF vitamin D levels according to baseline serum AMH concentration in infertile women.
AMH is one of the most reliable ovarian reserve biomarkers and has commonly been used to predict ovarian response in ART practice.22,23 Previous studies have investigated the association between serum vitamin D levels and AMH levels in infertile women.24–28 However, previous studies observed conflicting results because serum vitamin D might not represent the actual status of the follicular microenvironment. This study noted no significant differences in serum vitamin D levels between women with DOR and those with normal ovarian reserve. However, FF vitamin D level was significantly associated with ovarian reserve, and women with DOR had higher FF vitamin D concentration by 7.88 ng/ml compared to those with normal ovarian reserve. Additionally, this difference was persistent after adjustment for serum vitamin D, which is correlated with FF vitamin D level. Recently, Bednarska-Czerwińska et al. reported a change-point problem in the relationship between AMH and vitamin D; however, all study participants had a serum level of AMH ≥ 0.7 ng/ml, and were likely to have a normal ovarian reserve. 29 Increased FF vitamin D level in women with DOR in our study is partly in accordance with previous reports linking low FF vitamin D levels and polycystic ovary syndrome (PCOS).30,31 Moreover, vitamin D receptor gene expression decreased in granulosa cells in PCOS patients, 30 and women with low FF vitamin D concentrations were more likely to have more follicles in COS. 32 These findings suggest that FF vitamin D level is different according to the status of ovarian reserve; however, further studies for a clear explanation of this association are needed.
However, the role of FF vitamin D is unknown and several hypotheses have been proposed. The promoter region of the AMH gene is highly similar to the vitamin D response elements, and it has been suggested that vitamin D concentration may be associated with AMH expression. 33 Anifandis et al. reported that the level of FF glucose decreased in women with high FF vitamin D concentration and suggested the association between FF vitamin D and insulin action. 11
Several studies have investigated the association between FF vitamin D levels and ART outcomes, but the results were inconsistent. Ciepiela et al. reported that the level of FF vitamin D was significantly lower in women with successful outcomes in ART practice. 12 Similarly, Anifandis et al. demonstrated that FF vitamin D levels were negatively correlated to the quality of embryos and clinical pregnancy rate. 11 On the contrary, in other studies, women with higher FF vitamin D levels were more likely to be pregnant after ART,14,34,35 have better embryo quality,34,36 or no significant differences were observed between FF vitamin D levels and pregnancy rate. 13 These discrepancies might have resulted from the fact that the ovarian reserve status was never considered in previous studies. As described before, the ovarian reserve was associated with FF vitamin D levels in our study, and the ovarian reserve has also been reported to be correlated with ART outcomes. Therefore, the ovarian reserve should have been incorporated as a confounder in the analyses in prior studies that defined FF vitamin D as an exposure and ART results as outcomes. Therefore, we analyzed cycle outcomes according to FF vitamin D with adjustment for the ovarian reserve status, and we did not find any significant associations of FF vitamin D with fertilization rate and the number and proportion of good embryos at day 3. Moreover, vitamin D supplementation might not be needed for a good IVF outcome. Because serum vitamin D and FF vitamin D are associated in the present study and excess vitamin D can cause nausea, weakness, and nephrolithiasis, vitamin D supplementation is not recommended in premenopausal women. 37 Therefore, routine vitamin D supplementation should be considered when the benefits outweigh the risks in infertile women.
Contrary to expectations, sunscreen usage, sunlight exposure, and regular exercise did not show any associations with FF vitamin D levels. This result might be due to the relatively high sunscreen usage (80.9%) in our study population. Frequent use of sunscreen might conceal the beneficial effect of sunlight exposure on vitamin D levels. Furthermore, the intake of nutritional supplements, including vitamin D needs to be considered. However, we did not include nutritional supplements in the analyses because the content of vitamin D in supplements was not assessed in the questionnaires.
One of the shortcomings of this study is the small sample size. Because only ten study participants were pregnant, the clinical pregnancy rate could not be analyzed with multivariable analysis. Moreover, three patients who planned fertility preservation were included, and ET was not performed on these patients. Therefore, the findings in our study need to be interpreted with caution and the margin of error might have increased due to the small sample size. However, the power of the study reached over 70% with multivariable regression analysis, and we decided to report our findings as a pilot study. Second, we could not assess the outcomes of individual oocytes because the embryos or oocytes in the present study were not cultured separately. Third, we could not adjust for the pituitary suppression method because most women with DOR used GnRH antagonists in the present study. Finally, vitamin D supplementation was not considered a covariate in the present analysis.
The strength of the present study is the consideration of confounders in analyses. For example, the association of DOR and FF vitamin D or the association between cycle outcomes and FF vitamin D were analyzed with adjustment for serum vitamin D. Serum vitamin D can affect both FF vitamin D and cycle outcomes; therefore, serum vitamin D should be adjusted in the analysis of the association between FF vitamin D and cycle outcomes. Another strong point in the present study is that FF was collected only from a dominant follicle of each ovary, although most previous studies collected FF from several follicles.
We observed significantly higher FF vitamin D levels in women with DOR. However, FF vitamin D levels did not demonstrate any significant associations with fertilization rate and the number and proportion of good embryos at day 3. A further prospective study on a larger scale is needed to investigate the effect of FF vitamin D levels on the clinical pregnancy rate and live birth rate with consideration of the ovarian reserve and other possible confounders.
Footnotes
Acknowledgments
The Biospecimens and data used in this study were provided by the Biobank of Seoul National University Hospital, a member of Korea Biobank Network (KBN4_A03).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Seoul National University Hospital research fund, (grant number 04-2016-0350).
Ethics approval and consent
This study was approved by the institutional review board of Seoul National University Hospital (IRB No. 1706-052-858).
