Abstract
This study recycling the industrial byproducts of furnace slag and fly ash to produce slag/ash based zeolite. A scanning electron microscopic/energy dispersive X-ray spectroscopic (SEM/EDS) analysis of this zeolite indicates a high SiO2 content of 53.94%, an Al2O3 content of 23.20%, a silicon-to-aluminum ratio of 2.049, a density of 2.88 g/cm3, and a water content of 0.13% while the zeolite appears as a porous crystalline structure. Results of weighing experiments revealed effective adsorption of liquid salad oil and highly volatile organic solvents after reusing the zeolite up to 6 times. When an adsorbed liquid pollutant was desorbed and the heating temperature exceeded 170 °C and 350 °C, the samples exhibited two exothermic reactions, respectively, the mean maximum enthalpy were 0.427 and 0.461 mW, and the mean per gram of accumulated heat were 80.92 and 45.64 J/g. For all samples, the mean maximum loss of mass rate was 9.26%. Analogously, for gaseous pollutants, the samples exhibited an exothermic reaction when the heating temperature exceeded 180 °C; the average maximum enthalpy was 0.395 mW, the average per gram of accumulated heat was 119.60 J/g, and the average maximum loss of mass rate was 4.79%. This slag/ash based zeolite has the advantages of low cost, high thermal stability, reusability, etc., and can be used as a new adsorbent material for indoor ventilation equipment.
Introduction
Accordingly, a WHO report noted that 100,000 people worldwide die every year from indoor air pollution–induced asthma, 35% of whom are children.1,2 Therefore, to address air pollution at its source, effective adsorption and filtering materials must be used in indoor ventilation and air conditioning equipment.3–5 Since the 1970s, countries worldwide have adopted zeolite, activated carbon, silica gel, and activated alumina as adsorption materials for indoor ventilation equipment to treat harmful indoor exhaust gases, such as volatile organic compounds (VOCs), carbon dioxide (CO2), toluene (benzene), acetone (acetone), and heavy metal pollutants.6,7 Activated carbon is primarily composed of wood, wood chips, or coal. After the carbonization of this material at a high temperature of 600 °C–800 °C, a porous hydrocarbon is formed, which has a large specific surface area and a porous structure that provides excellent adsorption capacity to its surface. However, activated carbon is a thermal hazard at temperatures higher than 500 °C, 3 which limits its applicability as an adsorbent material for industrial high-temperature exhaust gas.4,7 Moreover, because reused activated carbon is very inefficient, its waste causes serious secondary pollution. Although silica gel and activated alumina themselves do not have an exothermic effect, they can, in theory, be applied as industrial ventilation adsorbent materials. Furthermore, various zeolites have been widely adopted in the industry as pollution adsorption materials, and clinoptilolite, mordenite, analcime, and chabazite have also been commonly used. 8 Zeolite is primarily composed of crystalline aluminosilicates formed by silicon oxide and aluminum oxide in an alkaline environment under high water-vapor pressure. Natural zeolite is commonly found in the crevices and pores of igneous rock. Zeolite is a porous material with a large surface area, and it is widely used in adsorbents, catalyst conversion agents, and catalyst carriers. The structural formula of zeolite is M x /n[(AlO2) x (SiO2)], and it has low silicalite (Si/Al = 1–1.5), medium silicalite (Si/Al = 1.5–5.0), and high silicalite (Si/Al > 5) variants. Zeolite has a very good adsorption effect on industrial pollutants, such as VOCs.9–11 According to differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) results,7,8 the composition of zeolite—at all silicon-to-aluminum ratios—is such that it exhibits only one endothermic peak and no exothermic peak or exothermic effect. The endothermic reaction properties of the various types of zeolites, which differ by their silicon-to-aluminum ratios, are detailed as follows. For zeolites with a silicon-to-aluminum ratio of 2, the highest reported endothermic reaction value is 162 J/g, and the maximum thermal-mass-loss ratio proportion of TGA is reportedly 16%. These values indicate an extremely high efficiency in adsorbing moisture in the air. Zeolite will begin to undergo desorption after being heated to >100 °C, indicating that it has the ability to adsorb pollutants again after heating. These findings reveal zeolite's excellent reuse efficiency. 12
However, the current method for making zeolite requires considerable quantities of silicon dioxide (SiO2) and sodium silicoaluminate powder, which are mixed with other materials at different ratios before being added to sodium hydroxide (NaOH) and stirred to gelatinize the mixture into a dough. The dough must then be extruded and calcined at high temperature for more than 10 h to complete the process. The zeolite produced through this process is often highly expensive and does not sell well. 13 A report noted that in the coal and steel industry, only a small proportion of the by-product of furnace slag and fly ash is recycled and reused, and most of the slag and ash is still treated as waste, which harms the environment.14,15 The use of industrial by-products to make slag/ash based zeolite can replace activated carbon and other substances, as a new adsorbent material for indoor ventilation and ventilation equipment to purify indoor air quality.
Slag/ash based zeolite
Sample production
This study analyzed the industrial by-product of steelmaking and coal burning from Taiwan Dragon Steel (Taichung, Taiwan) were selected as furnace slag as shown in Figure 1(a) and fly ash as shown in Figure 1(b). Using these two materials to replace other raw materials required to make zeolite, such as silicon dioxide and sodium aluminosilicate. First of all, this study slowly added a small amount of sodium hydroxide (NaOH) and stirred the mixture to gelatinize the dough into a high-temperature furnace. Then, according to the general zeolite production method, calcining temperature and holding time and other process conditions, it was made in the laboratory. This zeolite samples in the form of a solid strip is shown in Figure 1(c). Then, the composition and structure of the slag/ash-based zeolite were analyzed using an Energy Dispersive Spectrometer (EDS) and a scanning electron microscope (SEM).

Photos of the furnace slag and fly ash from industrial byproducts and slag/ash based zeolite in this study (a) furnace slag powder (b) fly ash powder (c) slag/ash based zeolite.
Furthermore, the furnace slag powder and fly ash powder are used to prepare the zeolite in a ratio of 1:1, and an appropriate amount of 75% NaOH was added during uniform mixing. After the dough was formed, it was formed into a strip by extrusion molding, and then dried it for 4 h at 100 °C and cut into shape. After drying, it would be calcined for 8 h at 600 °C to finally became slag/ash based zeolite. The preparation flow showed in Figure 2.
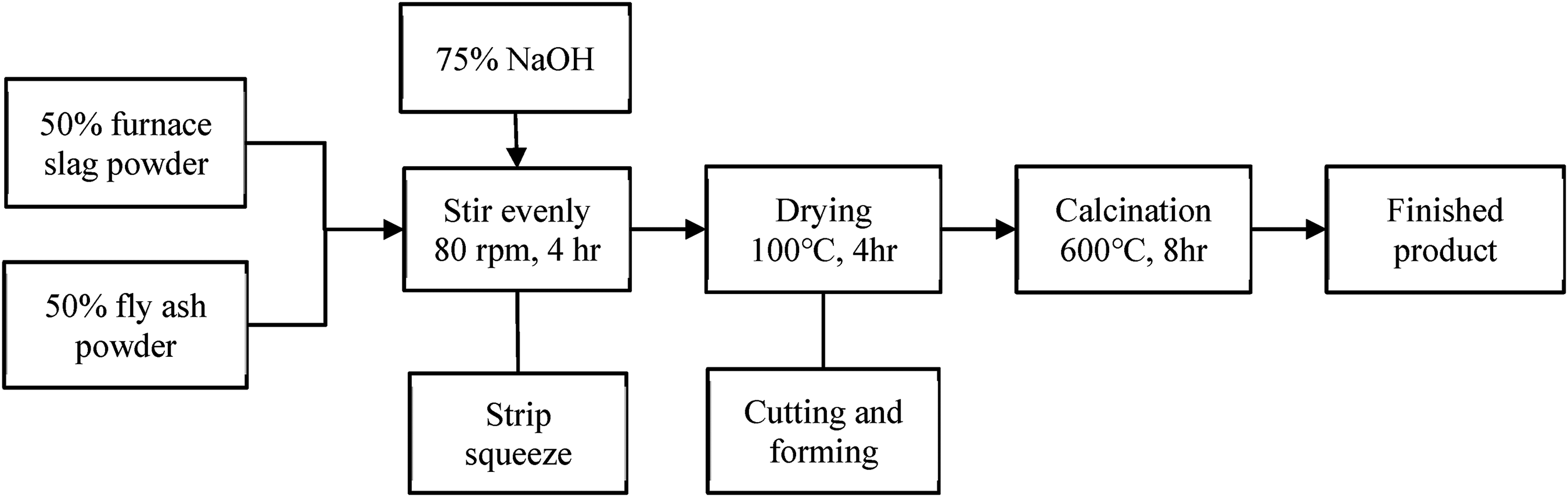
Chart of the experiment procedure for the slag/ash based zeolite.
EDS composition analysis and SEM observations
This study conducted SEM observations (magnifications of 5000 times and 10,000 times) of the slag/ash based zeolite in Figure 1 according to the ASTM C1723 test standard.16–19 The results presented in Figure 3 indicate that the slag/ash based zeolite was structurally identical to general zeolite; it had a porous crystalline structure with many regular pores.

Photos of scanning electron microscope of the slag/ash based zeolite in this study (a) 5000 times (b) 10,000 times.
At the same time, referring to the ASTM D3906 test standard,20–22 the EDS analysis was performed on the aforementioned slag/ash based zeolite in Figure 1. In the composition results, presented in Table 1, the density of the slag/ash based zeolite was 2.88 g/cm3, approximately equal to the 2.80 g/cm3 average density of earth's surface rocks and higher than the density of the concrete used in general construction. The slag/ash based zeolite was composed of 0.10% water, 53.94% SiO2, 23.20% Al2O3, and 0.41% Fe2O3.
Composition analysis of the slag/ash based zeolite in this study.

Description: The pink line in the photos above is the composition measurement range.
Economic analysis
Scholars Ruen-ngam and others had made a preliminary assessment on the production cost of synthetic zeolite X. After considering the production materials and electricity costs, the production cost of 1 kg of synthetic zeolite X was approximately 96 EUR (109.74 USD), while the commercial price of zeolite X was approximately 243.42 EUR (278.26 USD) per 10 g. 23 When calculating the aforementioned commercial price, the agency company mainly considered four basic cost categories, including technology development cost, production operation cost, replacement of manufacturing equipment and components, and transportation operation cost. Therefore, the commercial price of zeolite X was much higher than the production cost of scholars in the laboratory. 24
Since the raw material furnace slag and fly ash of zeolite in this paper were both industrial by-products, the raw material cost required to make 1 kg of slag/ash based zeolite was approximately USD 2.65, and the cost of electricity and the replacement of manufacturing equipment and components was approximately USD 25.2, totaling about 27.85$. After the slag/ash based zeolite adsorbs pollutants, it could be reused after the desorption procedures shown in Figure 4(c)–(e), and the cost of the aforementioned 1 kg was only about USD 3.36. Therefore, the production cost of the slag/ash based zeolite in this paper was much lower than the aforementioned synthetic zeolite X, and after a low-cost desorption procedure, it could be reused and had high economic value.

Photos of the weighing experiment for adsorption of liquid pollutants of slag/ash based zeolite (a) weighed sample (b) soaked in edible peanut oil for 2 min (c) rinsed with clean water and household detergent (d) boiled in water for 5 min (e) placed into an above 400 °C oven for 10 h (f) placed in a sealed bag for thermal analysis.
Experimental results and discussion
According to research, the main sources of pollutants that cause poor indoor air quality in US residences were cooking, smoking, burning incense and candles, and operating office equipment and other appliances, and by resuspension from settled dust. 1 In the cooking process, fried food was the main source of pollutants in indoor air. 25 If the household range hood could effectively absorb the pollutants produced by fried food, it will prevent these pollutants from scattering into the indoor air, resulting in poor indoor air quality. Therefore, in order to explore whether the slag/ash based zeolite in this paper could be used as an adsorption material inside a household range hood. Firstly, household salad oil, which was commonly used in fried food, was used as a liquid pollutant, and an electronic balance was used to carry out a weighing experiment method to measure the weight data before and after the adsorption of liquid pollutants by the slag/ash based zeolite. A proper desorption method was employed. The weighing experiment was repeated, and the sample after each desorption was subjected to thermal analysis experiments to determine (1) the number of times the sample could be reused and (2) the appropriate desorption procedure for the slag/ash based zeolite after the liquid pollutant had been adsorbed. The later sections of this paper detail the experiments.
In another experiment, bromine propane (C3H7Br), a highly volatile organic solvent commonly used in industry, was used as a gaseous pollutant. Self-assembled adsorption experimental equipment was used, and the repeated weighing and desorption procedures for the adsorbed pollutants, per the prior experiment for household salad oil, were executed. The samples after each desorption were also subjected to thermal analysis experiments to determine how well the slag/ash based zeolite adsorbs gaseous pollutants, how many times the zeolite can be reused, and what the appropriate desorption method is. The following sections of this study detail the experimental results.
Liquid pollutants
Weighing experiment method
The slag/ash based zeolite in this study was divided into several groups, and several samples from each group were weighed on an electronic balance, as depicted in Figure 4(a). The samples were then soaked in household salad oil for 2 min. and then taken out to drain, as depicted in Figure 4(b). In the subsequent cleaning procedure, the samples were washed with used water and household cleaner and then placed in boiling water for 5 min, as depicted in Figure 4(c) and (d). For subsequent desorption, the samples were placed in a 400 °C oven for 10 h, as depicted in Figure 4(e). After desorption, one of the groups (labeled Sample 1) was removed, weighed, and then placed in a sealed bag as preparation for subsequent thermal analysis experiments, as depicted in Figure 4(f).
The remaining five groups of samples were subjected to a similar procedure. They were soaked in household salad oil for 2 min, drained, rinsed repeatedly with water and household detergent, and then boiled in water for 5 min. All samples were then placed into a 400 °C oven for 10 h for desorption. After desorption, one of the groups (labeled Sample 2) was removed, weighed, and then placed in a sealed bag as preparation for subsequent thermal analysis experiments—identical to those applied to Sample 1. The experiment was repeated several times, and the experimental processes are depicted in the flowchart in Figure 5.

Flowchart of the weighing experiment for adsorption of liquid pollutants of slag/ash based zeolite.
The masses at the start and end of the experiments are presented in Figure 6. Six samples, labeled Samples 1 to 6, were analyzed; they were subjected to one to six iterations of the adsorption and desorption process, respectively. Sample 6 had approximately equal masses before and after the experiment. This indicates that under the aforementioned desorption procedure, the slag/ash based zeolite was useless by the fifth use. To understand the thermal stability of the zeolite samples after each desorption, this study will perform the relevant thermal analysis experiments on the 6 groups of slag/ash based zeolite.

Results of the weighing experiment for adsorption of liquid pollutants of slag/ash based zeolite.
Thermal analysis experiment after desorption
Samples 1 to 6, with the masses of 4.58, 4.98, 4.60, 4.78, 4.71, and 5.02 mg, respectively, were each subjected to thermal analysis experiments DSC—at a heating rate of 4 °C per minute from 30 °C to 500 °C—was used to obtain the sample heat release curves and maximum enthalpy values (Figure 7(a), Table 2). The results in Figure 7(a) indicate that the six samples had their first exothermic reaction when the temperature exceeded 170 °C, where the maximum enthalpy values were 0.54, 0.52, 0.38, 0.40, 0.37, and 0.35 mW for Samples 1 to 6, respectively (mean: 0.427 mW). The per gram of accumulated heat values were 112.66, 107.84, 76.91, 72.14, 60.26 and 55.72 J/g for Samples 1 to 6, respectively (mean: 80.92 J/g).

Thermal analysis experimental results (graph) after each desorption of liquid pollutants from slag/ash based zeolite (a) heat release (b) mass loss.
Thermal analysis experimental results (form) after each desorption of liquid pollutants from slag/ash based zeolite.

A second exothermic reaction occurred when the temperature exceeded 350 °C, where the maximum enthalpy values were 0.36, 0.73, 0.46, 0.41, 0.4 and 0.41 mW for Samples 1 to 6, respectively (mean: 0.461 mW). The per gram of accumulated heat values were 30.64, 98.14, 43.31, 36.46, 33.16, and 32.15 J/g for Samples 1 to 6, respectively (mean: 45.64 J/g). Table 2 presents the results for the two exothermic reactions. TGA was also applied to the desorbed Samples 1 to 6 with a heating rate of 4 °C per minute from 30 °C to 900 °C. The thermogravimetric loss results are presented in Figure 7(b) and Table 2. The maximum loss of mass values were 10.32%, 10.08%, 9.39%, 8.88%, 8.74%, and 8.16% for Samples 1 to 6, respectively (mean: 9.26%).
When the heating temperature of the six samples exceeded 480 °C, the associated curves, presented in Figure 7(a), were all close to horizontal, and the samples exhibited no thermal reactive behavior. In addition, as detailed in Table 2, Sample 1 and Sample 6 had, respectively, the exothermic peak temperatures of 226.36 °C and 282.13 °C, maximum enthalpy values of 0.54 and 0.35 mW, per gram of accumulated heat values of 112.66 and 55.72 J/g, and maximum loss of mass rate values of 10.32% and 8.16%. In general, as desorption time increased, the safety of the thermal reaction increased, and the maximum enthalpy, per gram of accumulated heat, and maximum loss of mass decreased. This was because the internal pores of the slag/ash based zeolite were very small, which meant that most household salad oil contaminants were adsorbed on the surface without penetrating into the pores of the slag/ash based zeolite. Therefore, the salad oil adsorbed by the slag/ash based zeolite had almost been completely desorbed after the zeolite was subjected to the cleaning and desorption procedures. Therefore, our experimental procedures and conditions resulted in the slag/ash based zeolite effectively desorbing liquid contaminants (at least that of salad oil). Furthermore, as noted in Figure 7(a) and Table 2, although the desorbed samples still had a two-stage exothermic reaction when the heating temperature exceeded 170 °C and 350 °C, the mean maximum enthalpy value was <0.5 mW, and the per gram of accumulated heat was also <100 J/g, which indicates an unclear exothermic reaction. Therefore, the slag/ash based zeolite, which adsorbed liquid oil, retained excellent thermal stability at the sixth use.
According to Figure 7(b) and Table 2, the samples’ post-desorption maximum thermal loss of mass (and, by implication, adsorption capacity) decreased with each reuse. However, Table 2 indicates that the difference in the maximum thermal losses of mass at the first and sixth use was merely 2%. Therefore, the slag/ash based zeolite effectively adsorbs liquid oil pollutants at the sixth use, and possibly more, as long as the appropriate desorption process is applied. Our slag/ash based zeolite is thus economical by virtue of its reusability.
Gaseous pollutants
Weighing experiment method
The reusability of the slag/ash based zeolite was analyzed for gaseous pollutants. The same experimental method as that for liquid pollutants was used, with the exception of adsorption experimental equipment, as illustrated in Figure 8, being used for the gaseous-pollutant experiments.
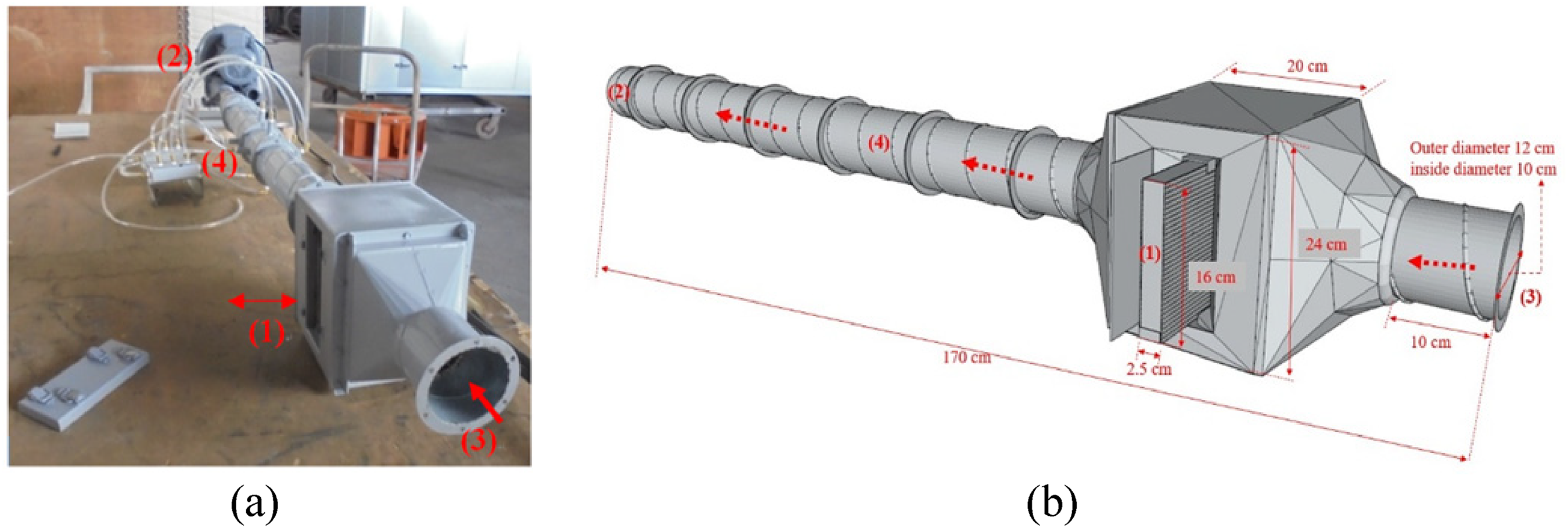
Photos of the adsorption experimental equipment installed by the research team (a) physical photo (b) 3D structure diagram.
The samples were first placed in a square removable cassette (length: 16 cm, thickness: 2.5 cm; position (1) in Figure 8) and then inserted into the equipment, as depicted in Figure 9(a) and (b). By adjusting the rotation speed of the exhaust fan (position (2) in Figure 8), the wind speed and air volume in the duct can be reduced from 13.5 m/s and 26.0 m3/min to 9 m/s and 16.5 m3/min, and 4.5 m/s and 8.5 m3/min wind speed and air volume to increase the contact time of harmful substances with the zeolite samples, as illustrated in Figure 9(c). Subsequently, highly volatile organic solvent bromopropane was poured into the container, as depicted in Figure 9(d), so vaporized bromopropane gas could enter the cassette from position (3) in Figure 8. After 30 min, the cassette was removed for sample weighing; the weight was then used to compute the adsorption ratio (cassette mass before adsorption to cassette mass after adsorption).

Photos of the weighing experiment for adsorption of gas pollutants of slag/ash based zeolite (a) putting a sample into the cassette (b) inserting the cassette into the equipment (c) measuring the wind speed and volume in the duct (d) pouring in highly volatile organic solvent bromopropane.
After adsorbing the pollutant, the slag/ash based zeolite was placed into a high-temperature furnace to desorb the pollutant at 800 °C for 1 h. The desorbed slag/ash based zeolite was then placed into the square cassette again. The aforementioned adsorption process, weighing experiments, and post-desorption thermal analysis experiments were repeated. The results were as follows.
Divide the weight difference value of the aforementioned sample before and after each adsorption by the weight before adsorption to obtain the ratio of each adsorption, as shown in Figure 10. It can be seen from Figure 10 that when the wind speed and air volume of the duct are 13.5 m/s and 26.0 m3/min, the adsorbed percentage decreases with repeated use (8.05%, 7.77%, 4.58%, 4.03, %, 3.48% and 2.79% for the first to sixth use, respectively); when the duct wind speed and air volume are reduced to 9 m/s and 16.5 m3/min, the adsorbed percentage after the first to the sixth use are 10.31%, 9.18%, 6.03%, respectively, 4.07%, 3.93%, and 2.94%; when the wind speed and air volume of the duct are reduced to 4.5 m/s and 8.5 m3/min, the adsorbed percentage after the first to sixth use are 13.42%, 11.90%, 8.82%, 6.59%, 5.01% and 3.74%. It can be seen that when the wind speed and air volume are maintained at 4.5 (m/s) and 8.5 (m3/min), the adsorption effect of the zeolite samples is the best. Moreover, after the zeolite samples adsorbed gaseous hazardous substances, all of the adsorbed percentage were only 4% or less when the number of repeated uses reached the sixth time. The foregoing results show that with a desorption process at a high temperature of 800 °C and continuous heating for 1 h, after the slag/ash based zeolite adsorbs gaseous harmful substances, it can be used repeatedly for about 6 times. In addition, in order to understand the thermal stability of the zeolite samples after each desorption, this study will perform the relevant thermal analysis experiments on the 6 groups of desorbed slag/ash based zeolite, and the results will be described in the following section.

Results of the weighing experiment for adsorption of gas pollutants of slag/ash based zeolite.
Thermal analysis experiment after desorption
Samples 1 to 6, with the masses of 4.97, 4.66, 4.85, 4.88, 4.95, and 4.65 mg, respectively, were each subjected to thermal analysis experiments DSC—at a heating rate of 4 °C per minute and from 30 °C to 450 °C—was used to obtain the sample heat release curves and maximum enthalpy values (Figure 11(a), Table 3). The results in Figure 11(a) indicate that the six samples had their first exothermic reaction when the temperature exceeded 180 °C, where the maximum enthalpy values were 0.47, 0.47, 0.50, 0.35, 0.33, and 0.25 mW for Samples 1 to 6, respectively (mean: 0.395 mW). The per gram of accumulated heat values were 165.06, 161.72, 148.98, 91.81, 86.82, and 63.22 J/g for Samples 1 to 6, respectively (mean: 119.60 J/g). Table 3 details the results of this exothermic reaction. TGA was then applied to the desorbed Samples 1 to 6 with heating from 30 °C to 800 °C at a rate of 4 °C per min. The thermal-mass-loss results are presented in Figure 11(b) and Table 3. The maximum loss of mass values were 5.97%, 5.74%, 5.63%, 4.53%, 3.53%, and 3.35% for Samples 1 to 6, respectively (mean: 4.79%).
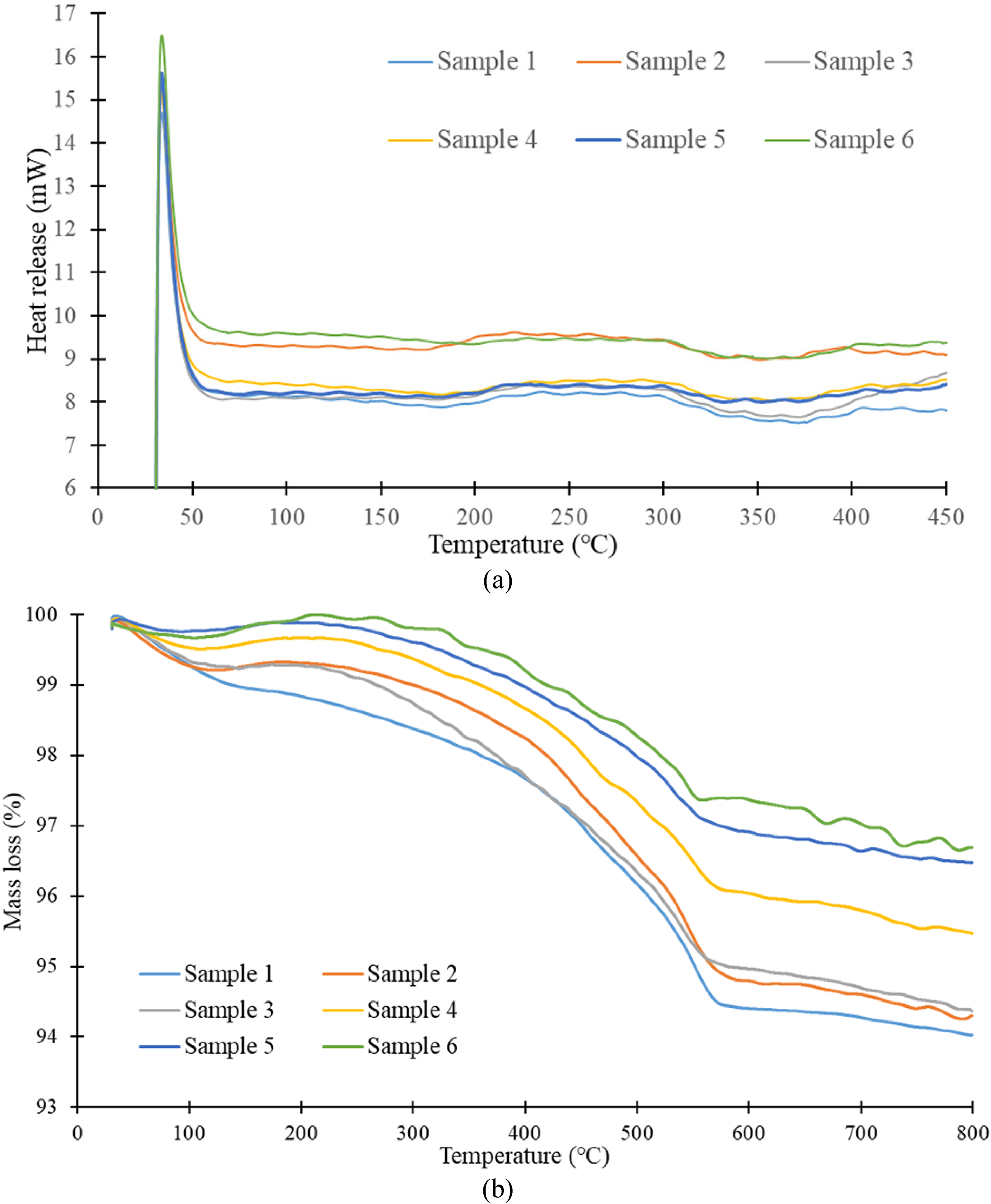
Thermal analysis experimental results (graph) after each desorption of gas pollutants from slag/ash based zeolite (a) heat release (b) mass loss.
Thermal analysis experimental results (form) after each desorption of gas pollutants from slag/ash based zeolite.

When the temperature of the six samples exceeded 400 °C, the associated curves, presented in Figure 11(a), were all close to horizontal, and the samples exhibited no tendency towards an exothermic reaction. In addition, as detailed in Table 3, Samples 1 and 6 had exothermic peak temperatures of 277.21 °C and 284.91 °C, respectively, maximum heat enthalpy values of 0.47 and 0.25 mW, respectively, per gram of accumulated heat values of 165.06 and 63.22 J/g, respectively, and maximum loss of mass rates of 5.97% and 3.35%, respectively. In general, as desorption time increased, the safety of the thermal reaction increased, and the maximum enthalpy, per gram of accumulated heat, and maximum loss of mass decreased. Therefore, the slag/ash based zeolite can be used to remove gaseous pollutants, in general, and highly volatile organic solvents, in particular, after desorption through heating at 800 °C for 1 h. Furthermore, as shown in Figure 11(a) and Table 3, the samples had a post-desorption exothermic reaction only when the temperature exceeded 180 °C; the reaction had a mean maximum enthalpy value of <0.5 mW and a mean per gram of accumulated heat of <120 J/g, which indicates an unclear exothermic reaction. Therefore, the slag/ash based zeolite, which adsorbed gaseous pollutants, retained excellent thermal stability at the sixth use.
According to Figure 11(b) and Table 3, the samples’ post-desorption maximum thermal loss of mass (and, by implication, adsorption capacity) decreased with each reuse. However, from Table 3, the difference in the maximum thermal losses of mass at the first and sixth uses was merely 2%. Therefore, the slag/ash based zeolite effectively adsorbs gaseous pollutants at the sixth use, and possibly more, as long as the appropriate desorption process is applied.
Summary
The experimental results indicate the reusability of the slag/ash based zeolite (effective until at least the sixth use) for removing organic solvents—such as bromopropane—and other harmful substances, with the application of proper desorption procedures. According to previous studies,26–29 the adsorption efficiency of zeolite for different harmful substances is related to its silicon-to-aluminum ratio and specific surface area, where a high (>5) silicon-to-aluminum ratio is more suitable for high-molecular-mass harmful industrial waste gases. The spontaneous ignition temperature of activated carbon is 450 °C, and the ignition temperature of bromopropane is 490 °C. If high temperatures are used to remove pollutants, activated carbon will also be pyrolyzed. By contrast, slag/ash based zeolite of this study can maintain its integrity up to 900 °C, making it reusable, and also can be used in industrial ventilation equipment. Specifically, the slag/ash based zeolite is fabricated using a by-product from the iron and steel industry, and its silicon-to-aluminum ratio is low at only 2.049. It may be necessary to increase this ratio to improve adsorption performance for removing industrial waste gases with high molecular mass. Therefore, in our laboratory, the method of reference5,27 used the method of adding silica (85.1% silica, specifically) as a raw material. The ratio of furnace slag to fly ash to silica is 4.5:4.5:1, which results in a silicon-to-aluminum ratio for the slag/ash based zeolite of 5.93, with SiO2 and Al2O3 compositions of 66.81% and 11.26%, respectively; the EDS composition analysis results are presented in Figure 12(a). In addition, this study further applied the ratio of slag to fly ash to silica is 1:1:2 mixture to adjust the compositions of SiO2 and Al2O3 to 98.24% and 1.77%, respectively, and the silicon-to-aluminum ration to as high as 55.50; the EDS composition analysis results are presented in Figure 12(b). Thus, silica raw materials can be used in the iron and steel industry to adjust the furnace slag and fly ash by-product to the appropriate type. These adjustments improved the slag/ash based zeolite's applicability, albeit at the expense of increased production cost, specifically by improving the zeolite's adsorption capacity for the harmful substance in question.
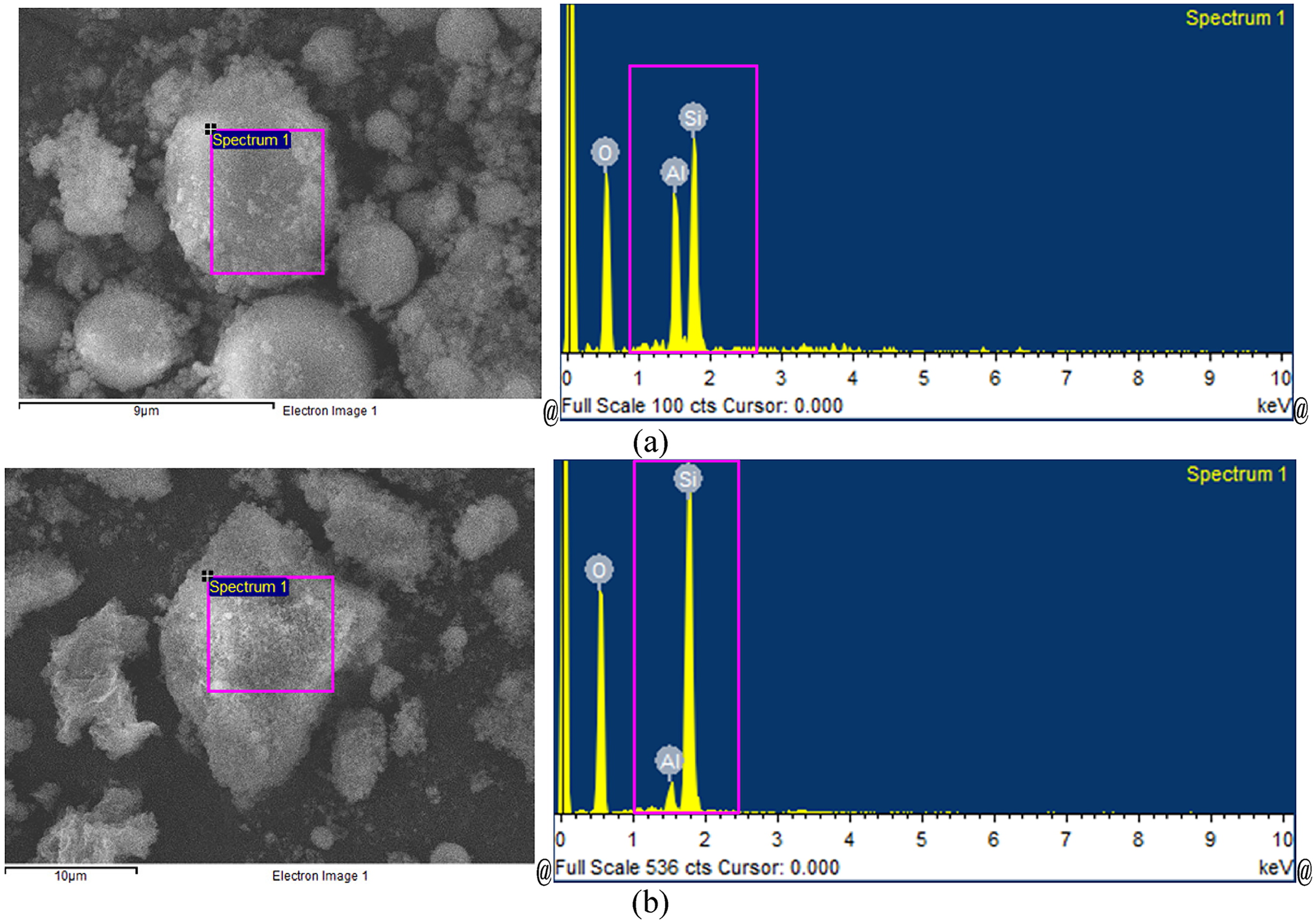
EDS analysis results (graph) of slag/ash based zeolite at different ratios of added silica (a) the ratio of furnace slag to fly ash to silica is 4.5:4.5:1 (b) the ratio of furnace slag to fly ash to silica is 1:1:2. Description: Fe and Si both represent SiO2; Al represents Al2O3 in the photos above.
Conclusions and recommendations
In the traditional method for producing zeolite, a large quantity of raw materials, such as silica and sodium aluminosilicate, are required. This makes zeolite much more expensive to produce than its counterparts, such as activated carbon, which greatly limits its commercial use. Therefore, activated carbon is still the current adsorbent material of choice for buildings and factories. However, under high-temperature operating environments, activated carbon poses a fire hazard. Activated carbon is also difficult to reuse and recycle after it has adsorbed pollutants, which usually causes secondary damage to the environment. Therefore, to produce zeolite more cheaply, this study proposed a slag/ash based zeolite fabricated using furnace slag and fly ash from the iron and steel industry, and relevant research conclusions are as follows:
It exhibited a density of 2.88 g/cm3 and a water content of 0.10%. Through EDS composition analysis, the slag/ash based zeolite was composed of 53.94% SiO2, 23.20% Al2O3, and 0.41% Fe2O3. After conversion, the silicon-to-aluminum ratio was 2.049. SEM observations revealed that the zeolite variant had a porous crystalline structure. The slag/ash based zeolite to repeated adsorption and desorption experiments using the weighing method and involving salad oil, gaseous bromopropane, and other harmful substances. Thermal analysis experiments revealed that the slag/ash based zeolite could be reused at least 6 times, and possibly more.
Since this study uses industrial byproducts such as furnace slag and fly ash to make zeolite samples, some property of the metal elements may be leached when adsorbing liquid pollutants, which will endanger human, animal and plant health.30,31 And scholars Feng et al. conducted a leaching test which evidenced a noticeable mobility of toxic elements from fly ash including arsenic, selenium, lead, chromium and cadmium under acidic and alkaline conditions.
32
In view of this, after the slag/ash based zeolite in this paper adsorbed liquid pollutants such as household salad oil, although the pollutants adsorbed on the surface of the zeolite could be burned off by using a high-temperature oven (as shown in Figure 4(e)), during the cleaning process. (as shown in Figure 4(c) and (d)) heavy metals or toxic substances may be leached out. Therefore, the waste liquid generated in the aforementioned cleaning process must be properly treated before it can be discharged to the outside environment to avoid environmental pollution and harm to human health. According to previous studies,33,34 there are many types of substances that harm human health and affect indoor air quality. Slag/ash based zeolite can be adjusted to better perform in removing a given harmful substance. Specifically, the silica-to-aluminum ratio, surface area, and contact area with the harmful substance can be adjusted by adding silica and other raw materials. Furthermore, this study uses the method of the rotating speed of the fan will be adjusted to reduce the wind speed in the adsorption experiment air duct in Figure 8 to maintain 3 different wind speeds and air volumes. In the future, if it is possible to reduce the wind speed and air volume in the air duct again, or increase Figure 9(a) of the thickness of the cassette will increase the contact time of gaseous hazardous substances with the zeolite samples and increase the adsorption capacity of the zeolite samples.
The proposed slag/ash based zeolite is cheap to produce, highly resistant to fire, and reusable, thus making it an excellent novel adsorption material. Recommendations for the main points of future experiments are as follows:
The slag/ash based zeolite could be used as a substrate, after coating it with silver sulfate and other substances.35–38 This would provide the zeolite a sterilizing effect and would make it applicable to medical and biological fields. Indoor air can be purified and disinfected if the slag/ash based zeolite is used as an adsorbent material in indoor ventilation equipment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
During the research time, this study received a kind subsidiary budget from the Ministry of Science and Technology (project number MOST 110-2221-E-274-002-). Besides, the colleagues from TBM corporation and Golden Flag ventilator engineering Co., Ltd. also providing precious information and experimental assistant. This study would like to express our sincerely gratitude.
