Abstract
The purpose of this study was to determine the preventive effect of ultrashort wave diathermy on immobilization-induced myogenic contracture and to explore its underlying mechanisms. Forty-two rabbits were randomly assigned into control (Group C), immobilization (Group I, which was further divided into one week, Group I-1; two weeks, Group I-2; and four weeks, Group I-4, subgroups by the length of immobilization) and ultrashort wave prevention (Group U, which was further divided into one week, Group U-1; two weeks, Group U-2; and four weeks, Group U-4, by time of treatment) groups. Intervention effects were assessed by evaluating rectus femoris cross-sectional area (CSA), knee range of motion, and the protein levels for myogenic differentiation (MyoD) and muscle atrophy F-box (MAFbx-1) in the rectus femoris. Compared with those of Group C, in Groups I and U, total contracture, myogenic contracture, MyoD and MAFbx-1 levels were significantly elevated, and CSA was significantly smaller (p < 0.05). Compared with those of Group I at each time point, MyoD levels were significantly elevated, MAFbx-1 levels were significantly lower, CSA was significantly larger, and myogenic contracture was significantly alleviated in Group U (p < 0.05). In the early stages of contracture, ultrashort wave diathermy reduces muscle atrophy and delays the process of myogenic contracture during joint immobilization; the mechanism of this may be explained as increased expression of MyoD triggered by suppression of the MAFbx-1-mediated ubiquitin-proteasome pathway.
Introduction
Joint immobilization is used to treat musculoskeletal disorders, for example, to repair ligaments and to allow fractures to heal. However, prolonged immobilization can also result in joint contracture, which reduces the quality of life in patients. 1 Physical therapy and medications, which are considered as the conventional methods for the treatment of joint contracture, can achieve effective therapeutic effects to an extent, but can't completely reverse joint contracture. 2 Furthermore, in the later stages of joint contracture, even with surgical management, the prognosis for restoring total range of motion (ROM) is unsatisfactory. 3 Therefore, a more efficacious therapy to prevent and treat joint contracture is urgently needed.
Joint contracture is caused by both myogenic (e.g. muscles, tendons) and arthrogenic (e.g. bones, cartilages) factors; joint contracture as a result of short-term (less than four weeks) immobilization is mainly caused by myogenic factors.2–5 Joint contracture is almost always associated with muscle atrophy, whose pathological characteristics are the decrease of muscle tissue mass, the shortening of muscle fiber length and a decrease in muscle cross-sectional area (CSA). 6 Muscle atrophy promotes the expression of certain muscle-specific genes, such as MuRF-1 for muscle ring finger protein-1, Atrogin-1 for muscle atrophy F-box protein (MAFbx-1) and myogenic differentiation (MyoD).7–9 MAFbx-1 is a muscle-specific F-box protein that is highly expressed in the process of muscle atrophy and is the rate-limiting enzyme in the process of protein degradation. 7 The gene is up-regulated in many muscle atrophy models, such as disuse muscular atrophy and denervated atrophy models, which affects the balance between protein synthesis and degradation and is a sign of muscle atrophy. 7 MyoD, a member of the myogenic regulatory factor family, also plays an important role in guiding muscle satellite cell specialization and skeletal muscle regeneration.8,10 Recent studies have shown that MyoD levels may affect proteolysis and muscle atrophy through the ubiquitin-proteasome pathway. 11
Ultrashort wave diathermy is a high-frequency electric field that produces thermal and non-thermal effects and acts on deep tissue and bone. 12 Ultrashort wave diathermy is used to treat inflammation of the nervous system, joint and soft tissue, which has been proved to accelerate blood circulation, reduce inflammation, improve joint stiffness and relieve pain.3,12,13 Zhu et al. found that ultrashort wave diathermy promotes the regeneration of injured peripheral nerves by dilating nerve vessels, improving blood circulation and inhibiting inflammation. 13 Another study found that ultrashort wave diathermy has a significant effect on the improvement of knee osteoarthritis in rabbits. 14 Zhang et al. showed that ultrashort wave diathermy enhances the effect of stretching on joint capsule fibrosis in a rabbit knee model. 3 Up till the present moment, there are few studies on ultrashort wave alone in the prevention and treatment of joint contracture. Moreover, it is still unclear whether ultrashort wave diathermy during the plaster cast period could ameliorate joint contracture by preventing muscle atrophy.
The primary purpose of this study was to determine whether ultrashort wave diathermy could prevent muscle atrophy and joint contracture by investigating the following using a rabbit knee model: 1. improvement in knee ROM; 2. histopathological characterization of skeletal muscle atrophy; and 3. changes in MyoD and MAFbx-1 protein levels in the rectus femoris.
Materials and methods
Animals
The experimental protocol was carried out in accordance with the guide of the Humane Care and Use of Laboratory Animals issued by the Ministry of Science and Technology of the People's Republic of China in 2006 and was approved by the Institutional Animal Care and Use Committee of Anhui Medical University on 10 October 2019 (NO. LLSC20190761). Forty-two male New Zealand white rabbits (aged 3–4 months, mass 2-2.5 kg, Anhui, Hefei, China) were used. The animals were housed individually in iron cages with controlled environmental conditions and free access to standard food and water.
Groups
Groups: The rabbits were randomly assigned to control (Group C, n = 6), immobilization (Groups I-1, I-2 and I-4 for one-week, two-week and four-week immobilizations, respectively; n = 6 each) and ultrashort wave prevention (Group U-1, U-2 and U-4 for one-week, two-week and four-week treatments, respectively; n = 6 each) groups.
Intervention measures
Prior to our former experiment, in order to prove the effect of ultrashort wave on tissues inside the plaster, a neon lamp was placed inside the plaster, and then the ultrashort wave was turned on. We found that ultrashort wave diathermy treatment could light the neon lamp in the plaster model (Figure 1), which indicated that ultrashort wave diathermy treatment may play an effect on the tissue inside plaster.

Ultrashort wave diathermy could light the neon lamp encased in plaster.
In the former experiment, the rabbits in the control group were not subjected to either immobilization or treatment. In Groups I group, the left knee joints of the rabbits were immobilized with tubular plasters, as previously described by Zhou et al. 15 The rabbits in the immobilization group were first anesthetized with an injection (to the ear margin) of sodium pentobarbital (30 mg/kg), and the fully extended unilateral knee joint was immobilized from the left groin to the proximal toe (Figure 2(a)). Rabbits in Group U were treated with ultrashort waves (DL-C-BII, 2013NO226049, Shantou Medical Equipment Factory) for the periods during which the plaster was worn. During treatment, the rabbits were placed on insulated wooden boards, and towels were placed on the front and back of the left lower limb. The electrode pairing method was selected; after the device was preheated, the mode of the therapeutic apparatus was converted to microcalorimetry (a frequency of 27.12 MHz, a maximum export power of 200 W and a current intensity controlled at approximately 80 mA). Treatments were applied six times a week (Monday through Saturday), and each treatment lasted 15 min (Figure 2(b)).

(A) A rabbit model of knee extension contracture. (B) The rabbits in Group U received ultrashort wave diathermy treatment.
ROM measurements
The rabbits were sacrificed with an overdose of sodium pentobarbital. The left hind limb of the rabbit was disarticulated at the hip, and knee joint ROM was measured using a mechanical arthrometer and the method described by Zhang et al. 3 : two experimenters fixed the hind limbs of rabbits to a joint mobility meter and used a standard torque of 0.077 Nm to measure the flexion angle before (Figure 3(a)) and after myotomy (Figure 3(b)), three times each. The mean of the six measurements is reported. Generally speaking, a smaller flexion angle of knee joint represents a greater loss of motion or severe contracture. The loss of the knee joint flexion angle is called total contracture, and after myotomy, the loss ROM is called arthrogenic contracture. According to previous studies, myogenic contracture = total contracture − arthrogenic contracture.3,6

Mechanical arthrometer used to measure knee (A) flexion angle before myotomy and (B) flexion angle after myotomy.
Tissue preparation and staining
After ROM was measured, the rectus femoris was excised from the left knee. The muscle samples were fixed with 4% paraformaldehyde solution at 4 °C for 12 h. The samples were cut into five-µm sections and stained with hematoxylin and eosin. Then, the sections were photographed using a microscope (400× magnification), and CSA was measured using Image J. Six randomly selected fields of a section were analyzed. After the areas in each field were measured, the average CSA was calculated. 16 The remaining muscle samples were stored at −80 °C for the protein experiments.
Protein extraction and western blot
The protein levels of MyoD and MAFbx-1 were determined with western blotting analysis. Proteins from muscle specimens were extracted using RIPA lysis buffer (Wanleibio Co, China), and the protein concentrations were measured using a BCA protein assay kit (Wanleibio Co, China). Later, the equivalent protein was separated on 12% polyacrylamide electrophoresis gel and transferred onto polyvinylidene fluoride (Millipore) membranes. The membranes were blocked with 5% skim milk dissolved and TBST (tris-buffered saline and Tween 20) for 0.5 h at 20–25 °C. Subsequently, the membranes were incubated with rabbit anti-MyoD antibody (1:5000; Beyotime), rabbit anti-MAFbx-1 antibody (1:2000; Beyotime) and rabbit anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibody (1:8000; Beyotime) at 4 °C for 12 h. The following day, the membranes were washed in TBST five times for 3 min and incubated with peroxidase-conjugated affinity-purification goat anti-rabbit IgG-HRP (1: 10,000; Beyotime), as the secondary antibody, for 1 h at 20–25 °C. The membranes were evaluated with a chemiluminescence system, and bands were quantified using Image J to analyze pixel density. The protein levels of MyoD and MAFbx-1 were normalized with those of GAPDH.
Statistical analyses
All data were analyzed with SPSS 21.0 and presented as the mean ± SD. Distribution normality was assessed using the Shapiro–Wilk test. An analysis of variance (ANOVA) was performed, and Tukey post hoc tests were used to examine differences between groups for total and myogenic contracture, rectus femoris CSA as well as MyoD and MAFbx-1 levels in the rectus femoris. A p-value less than 0.05 was deemed significant.
Results
During the immobilization period, one rabbit in Group U-2 died from the anesthesia and one rabbit in Group I-2 died from diarrhea. All other rabbits survived until the end of the experiments.
Range of motion
Knee joint contracture is illustrated in Figure 4(a) and (b) and Table 1: Total contracture and myogenic contracture were significantly increased in Group I and Group U compared with those of Group C (p < 0.001). Total contracture in rabbits in Group U was significantly reduced compared with that in rabbits in Group I for corresponding immobilization duration less than four weeks; (p < 0.05), while there was no significant difference between Group I-4 and Group U-4. For corresponding immobilization durations, myogenic contracture was significantly reduced in Group U compared with that in Group I (p < 0.05). The results of the one-factor ANOVA and the Tukey–Kramer test showed that total contracture and myogenic contracture in Group I and Group U significantly increased with prolonged immobilization (p < 0.05).

(A) Total contracture and (B) myogenic contracture of knee in each group. ap < 0.05 vs. Group C, bp < 0.05 vs. Group I-1, cp < 0.05 vs. Group I-2, a’p < 0.05 vs. Group I-0, b’p < 0.05 vs. Group U-1, c’p < 0.05 vs. Group U-2, ***p < 0.001, **p < 0.01 and nsp = 0.115.
Total contracture and the myogenic contracture and CSA of rectus femoris.

Morphological changes of the rectus femoris
The CSA values were significantly smaller in Group I and Group U compared with Group C (p < 0.05). For corresponding immobilization durations, CSA values were significantly larger in Group U compared with those in Group I (p < 0.05). CSA values in both Group I and Group U significantly decreased with prolonged immobilization (p < 0.05) (Figure 5 and Table 1).

(A) Hematoxylin and eosin staining (400× magnification; 100 µm scale bar) of rectus femoris specimens from group C, group I and group U. (B) Rectus femoris cross-sectional area (CSA); ap < 0.05 vs. Group C, bp < 0.05 vs. Group I-1, cp < 0.05 vs. Group I-2, a’p < 0.05 vs. Group I-0, b’p < 0.05 vs. Group U-1, c'p < 0.05 vs. Group U-2 and *p < 0.05.
Expressions of MyoD and MAFbx-1 protein
The relative protein expression levels for MyoD and MAFbx-1 were normalized to mean GAPDH expression levels (Figure 6(a)). Results showed that MyoD and MAFbx-1 levels were significantly higher in Group U and Group I compared with those in the control group (p < 0.05).
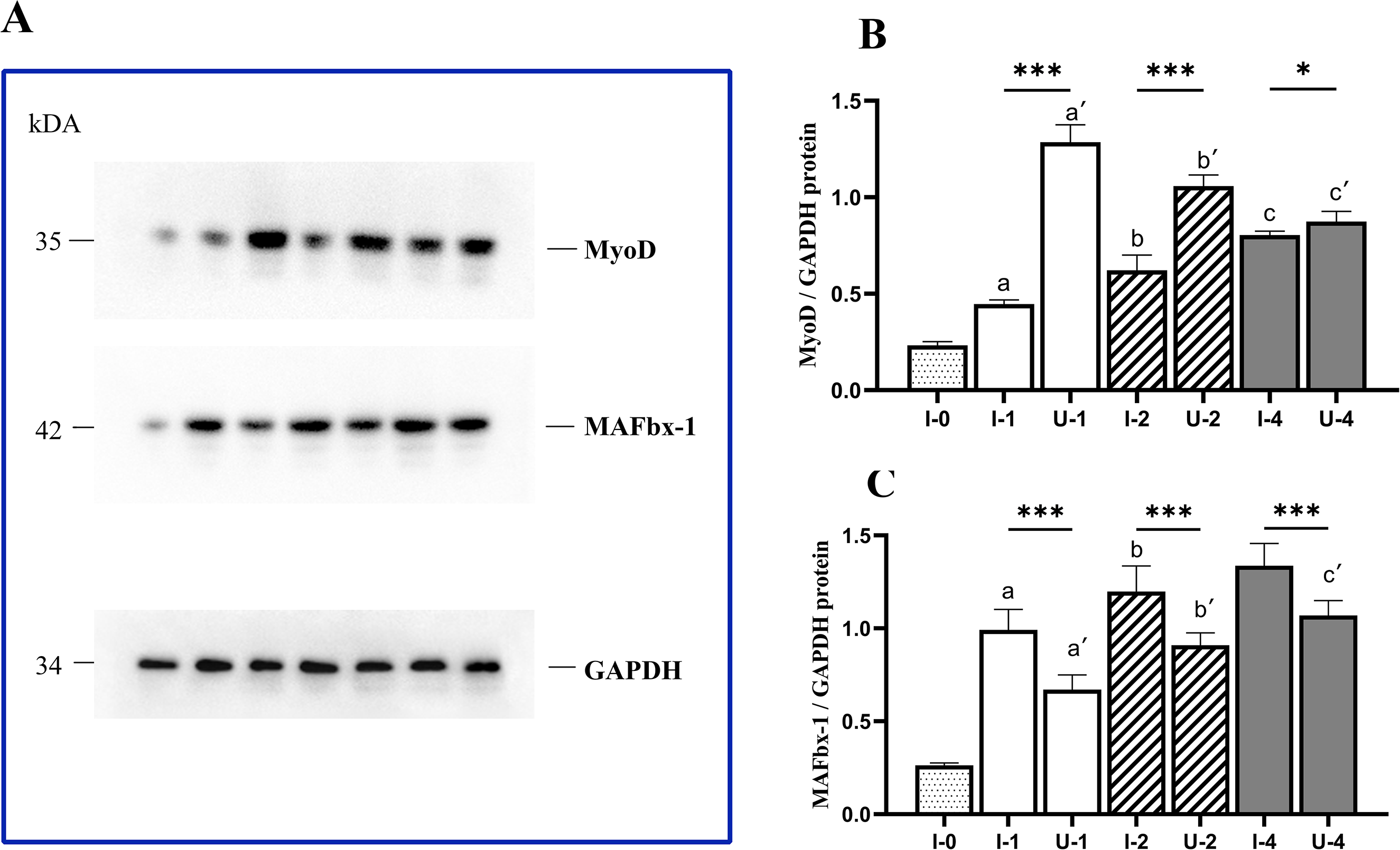
Protein levels of myogenic differentiation (MyoD) and muscle atrophy F-box (MAFbx-1) relative to glyceraldehyde 3-phosphate dehydrogenase (GAPDH). (A) Photographs of MyoD, MAFbx-1 and GAPDH protein-band intensities. (B) MyoD protein levels relative to GAPDH. (C) MAFbx-1 protein levels relative to GAPDH. ap < 0.05 vs. Group C(I-0), bp < 0.05 vs. Group I-1, cp < 0.05 vs. Group I-2, a’p < 0.05 vs. Group I-0, b’p < 0.05 vs. Group U-1, c’p < 0.05 vs. Group U-2, ***p < 0.001, **p < 0.01 and *p < 0.05.
Differences in MyoD protein levels
The protein levels for MyoD (Figure 6(b)) were significantly higher in Group U than those in Group I for corresponding immobilization durations (p < 0.05). The results of one-factor ANOVA and Tukey–Kramer test showed that MyoD expression in Group I significantly increased with the prolonged immobilization (p < 0.05).
Differences in MAFbx-1 protein levels
Figure 6(c) shows that the MAFbx-1 protein level was significantly lower in Group U than that in Group I (p < 0.05). The average protein levels for MAFbx-1 in Group I and Group U significantly increased with prolonged immobilization (p < 0.05).
Discussion
Joint contracture is a common clinical disease. Physiotherapy (such as physical factor rehabilitation, stretching and joint mobilization) is considered as the most prevalent treatment method for joint contracture.2,5 However, after the formation of joint contracture, these interventions may not produce satisfying outcomes all the time. Therefore, the prevention of joint contracture is particularly important, but there are few previous studies in this area. As a common physical factor rehabilitation, ultrashort wave diathermy has deep penetrating ability allowing therapy in the deep tissue. 12 Therefore, the purpose of this study was to explore the preventive effect of ultrashort wave diathermy on immobilization-induced myogenic contracture and to explore its underlying mechanisms.
In this study, with longer durations of immobilization, knee joint ROM was further limited (in Group I and Group U). Because the total contracture in Group U-4 was lower than that in Group I-4, these differences were small and did not reach statistical significance. It seems that ultrashort wave diathermy had little effect on the prevention of joint contracture. However, myogenic contracture in Group U was significantly reduced compared with that in Group I at corresponding time points. Rectus femoris CSA in Group U was significantly higher than that in Group I at corresponding time points (p < 0.05). This suggests that ultrashort wave diathermy may improve myogenic contracture by reducing muscle atrophy.5 Ultrashort wave diathermy can dilate local tissue blood vessels, enhance blood lymphatic circulation, increase vascular wall permeability, improve local tissue metabolism and then reduce myogenic contracture by reducing the viscoelasticity of the tissue and increasing the extensibility of the tissue.12,17 We also observed that total contracture in Group U was significantly lower than that in the corresponding immobilization group for one- and two-week-long immobilization. This is because early joint contracture is primarily myogenic.3,4
Muscle atrophy is the result of a change in the balance between protein synthesis and degradation. When the protein degradation is greater than protein synthesis, the mass of muscle tissue decreases. MAFbx-1 is one of two muscle-specific ubiquitin ligases selectively induced during skeletal muscle atrophy and promotes protein degradation through the ubiquitin-proteasome pathway. 18 Studies have found MAFbx-1 expression to be up-regulated, both at the mRNA and protein level in muscle atrophy models, such as the denervated muscular atrophy model7,17 and the disuse muscular atrophy model. 19 MAFbx-1 is a ubiquitin-protein ligase specifically for protein substrate recognition that transfers ubiquitin from an E2 ubiquitin-conjugating enzyme to the target substrate and degrades the protein substrate through covalent modification. 20 MAFbx-1 is commonly up-regulated and plays an important role in muscle atrophy, and hence considered as an important biomarker of muscle atrophy. 21 This study found that MAFbx-1 protein levels were significantly increased with prolonged immobilization, which suggested that fixation can result in muscle atrophy around the joint, and the results were consistent with the previous study on muscle atrophy conducted by Okamoto and Machida. 22 We also found that MAFbx-1 protein levels in Group I were significantly higher than those in Group U, which suggested that ultrashort wave diathermy decreases skeletal muscle protein degradation through inhibition of MAFbx-1 protein expression levels.
MyoD belongs to the family of myogenic regulatory factors, which plays an important role in guiding myogenic stem cell proliferation and differentiation, and is responsible for the regeneration of postnatal adult skeletal muscle.8,23 Studies have proven that there is increased expression of MyoD in muscle atrophy models, such as those for disuse muscle atrophy and denervated atrophy.10,24 As compared to Group I at each time point, MyoD protein levels were significantly elevated in Group U. This indicates that ultrashort wave diathermy can up-regulate the expression of MyoD and promote muscle development and muscle fiber maturation. The results of our study were in accordance with previous studies (Wilborn et al. and Lima et al.). Wilborn et al. 25 found that exercise can effectively increase MyoD mRNA levels and improve muscle atrophy, as demonstrated in this experiment. Lima et al. 26 found that electrical stimulation can improve skeletal muscle atrophy by increasing the expression of MyoD.
It is well known that MyoD is degraded by the ubiquitin-proteasome pathway, though the specific pathway is unknown. In an in vitro experiment, the increased expression of MAFbx-1 in atrophied muscles selectively inhibited the expression of MyoD. After suppressing MAFbx-1, the expression of MyoD increased and the amount of muscle atrophy reduced, which indicated that MyoD was the target of MAFbx-1. 27 Furthermore, an in vivo experiment indicated that the increased expression of MyoD is probably due to the inhibition of E3 ubiquitin ligase and the activation of the Akt/mTOR/FoxO3α signaling pathway, which promotes the expression of MyoD and enhances myoblast differentiation.28,29 In our experiment, the expression of MAFbx-1 and MyoD changed significantly, and joint contracture was alleviated to some extent.
The present research provides a basis for the use of ultrashort wave diathermy to prevent knee joint contracture and muscle atrophy. We found that ultrashort wave diathermy for 15 min a day improves muscle atrophy, delays the progression of myogenic contracture and changes the protein levels of MyoD and MAFbx-1 in rectus femoris muscles immobilized with a plaster cast. This study shows that ultrashort wave diathermy is a credible choice for the prevention of joint contracture caused by immobilization. However, we also observed that the ROM did not return to normal. This suggests that additional mechanisms are likely at play in the development of joint contracture, and multiple therapeutic strategies are needed for the prevention of joint contracture.
There were some important limitations to our study. First, the longest immobilization time was four weeks; therefore, the preventive effect of ultrashort wave diathermy on myogenic contracture and muscular atrophy is unknown after that. However, the main purpose of this study was to explore the preventive effects of ultrashort wave diathermy on myogenic contracture, and myogenic contracture progresses slowly after two weeks and reaches a balance at four weeks. 2 Second, the expression of MAFbx-1 and MyoD is measured in this study, but the expression of MyoD protein after inhibition or knockout of the MAFbx-1 gene was not analyzed. Further studies are required to confirm our findings and explore the detailed mechanisms underlying the preventive effect of ultrashort wave diathermy on immobilization-induced myogenic contracture. Finally, ubiquitin ligases related to muscle atrophy, including MAFbx-1 and MuRF-1, were not used to measure the expression of MuRF-1. This is a possible direction for future research.
We found that, in the early stages of knee joint contracture in a rabbit model, ultrashort wave diathermy could ameliorate muscle atrophy and delay the process of myogenic contracture. This may be because ultrashort wave diathermy therapy inhibits the MAFbx-1-mediated ubiquitin-proteasome pathway and up-regulates MyoD expression.
Footnotes
Authors’ note
Qi-Yu Xu and Quan-Bing Zhang contributed equally. QX, QZ and AL performed the experiments, wrote the manuscript, as well as the analysis and interpretation of data. FW collected and analyzed the data. YZ designed the study. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors declare that the research was conducted in the absence of any commercial or financial relationships that could be a potential conflict of interest.
Ethics approval
The Institutional Animal Care and Use Committee of Anhui Medical University approved the experimental procedures, NO. LLSC20190761. We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this report is consistent with those guidelines. The datasets used and/or analyzed during the current study are availablefrom the corresponding author. All data generated or analyzed during this study are included in this published article. The manuscript, including related data, figures and tables have not been previously published and are not under consideration elsewhere.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was jointly funded projects of Anhui Key Research and Development Program (201904a07020067; recipient: Yun Zhou) and Anhui Medical University Research Foundation (2018xkj050; recipient: Quan-Bing Zhang).
Correction (May 2023):
This article has been discovered a descriptive error in the Discussion section, specifically in the last two sentences of the second paragraph and last two sentences has been removed since its original publication.
