Abstract
Endoscopic variceal ligation (EVL) with vasoconstrictors has been recommended for acute esophageal variceal bleeding. However, the optimal duration of vasoconstrictors after EVL is controversial. This systematic review and meta-analysis was conducted to explore the efficacy of short-course vasoconstrictors (≤3 days) versus standard combination (3–5 days). A comprehensive literature review was conducted using the PubMed, Embase, and Cochrane library databases with subsequent meta-analysis. The primary outcomes were 5-day rebleeding, mortality, and treatment failure rates. A risk ratio (RR) with 95% confidence interval is used for outcome comparison. Nine randomized studies with 838 patients were included. The initial hemostasis (96.8% vs 97.4%, p = 0.919), 5-day rebleeding (11.2% vs 8.3%, RR = 1.05, 95% CI = 0.62–1.76), mortality (0 vs 1.3%, RR = 0.48, 95% CI = 0.10–2.39), and treatment failure (7.4% vs 5.9%, RR = 1.10, 95% CI = 0.48–2.49) were similar in both groups. Subgroup analysis suggested EVL alone had no significant difference of 5-day re-bleeding (15.9% vs 7.1%, RR = 2.25, 95% CI = 0.87–5.77), mortality (0 vs 0.7%, RR = 0.71, 95% CI = 0.08–6.03), treatment failure (9.6% vs 6.7%, RR = 1.43, 95% CI = 0.54–3.75) compared to standard combination. Clinical heterogeneity was found for the rebleeding rate for the subgroup during sensitivity analysis. EVL with short-course vasoconstrictors is highly efficacious for esophageal variceal bleeding. Further studies are required to determine the genuine need of subsequent vasoconstrictor after successful EVL.
Introduction
Acute esophageal variceal bleeding is one of the major complications of portal hypertension, being notorious for an appreciably high morbidity and mortality. 1 Although its prognosis has improved in recent years,2,3 multi-disciplinary care is still required to optimize the outcome. Vasoconstrictors are generally used as a first-line therapy for acute variceal hemorrhage, 1 and then followed by endoscopic therapy.
Endoscopic injection sclerotherapy (EIS) was the first endoscopic modality that was found to be useful in acute hemostasis of variceal hemorrhage. 4 Despite its efficacy, studies have demonstrated that the rebleeding rate is high with EIS alone,5,6 and a combination of vasoconstrictors for up to 5 days had been recommended.1,7 Endoscopic variceal ligation (EVL) has replaced EIS as the endoscopic therapy of choice in the recent two decades owing to fewer rebleeding and complications.8,9 Because EVL is highly effective in arresting acute esophageal variceal bleeding, the optimal duration of adjuvant vasoconstrictors after EVL has been an issue of controversy.10–12
An earlier systematic review and meta-analysis by Bañares et al. 13 demonstrated the benefit of combination therapy, however, most studies in this review were published before 2000 with an adaptation of EIS as the primary endoscopic treatment. Recently, several studies suggested short-course (≤3 days) of vasoconstrictors had similar outcomes compared to standard combination (3–5 days) after successful EVL for acute variceal bleeding,11,12,14–18 and pooled evidence is required to make a more confident recommendation in terms of the duration of vasoconstrictors. Though Yan et al. 19 reported another meta-analysis that was in favor of short-course vasoconstrictors after EVL, the conclusion may be undermined as some randomized trials were not included.12,14
We hypothesized that, with the superior efficacy of EVL, the need for subsequent vasoconstrictors may be reduced and a short-course of vasoconstrictors should be sufficient. Hence, we conducted an updated systematic review and meta-analysis to compare the outcomes between short-course vasoconstrictors and standard combination therapy in patients with acute esophageal variceal bleeding.
Materials and methods
Study design and search strategy
In this study, we included studies that compared short-term outcomes by either standard course (EVL plus 3–5 days of vasoconstrictors) or short-course (EVL plus 0–3 days of vasoconstrictors) combination therapy for acute esophageal variceal bleeding. Two authors (JHY and GHL) independently underwent comprehensive literature searches of three online database resources: PubMed, Embase, and Cochrane library in February 2021. The search queries and keywords were “(variceal bleeding) AND (endoscopic variceal ligation) AND (vasoconstrictor) OR (vasoactive agents) OR (vasoactive drugs) OR vasopressin OR octreotide OR terlipressin OR somatostatin OR vapreotide)” in all the database.
After excluding duplicated articles, the authors (DSP and WLW) proceeded manual reference review of all remaining studies to retrieve relevant articles. All records identified through database searches were reviewed via their abstracts, and full-text articles as necessary, for eligibility. The exclusion criteria were: (1) EVL was not performed as endoscopy treatment, (2) using vasoconstrictors alone without early endoscopy in the control group, (3) gastric variceal bleeding, (4) lack of outcomes within 5 days and during hospitalization, (5) studies in which EIS was used as alternative endoscopic treatment.
Data extraction and assessment of outcome and validity
The following data were extracted: name of the first author, year of publication, country of origin, number and characteristics of participants, study design, treatment modality, dosage and administration of vasoconstrictors, as well as the primary and secondary outcomes. The primary outcome of these studies included: (1) 5-day rebleeding, defined as any recurrent bleeding episode after initial hemostasis, (2) 5-day mortality, and (3) 5-day treatment failure, defined as the presence of persistent bleeding, rebleeding, or mortality after successful endoscopic hemostasis. The secondary outcome was the 6-week mortality, adverse event, hospital stay, and transfusion requirement. All data were extracted as originally stated or following appropriate calculations. For studies with missing or unavailable data, we attempted to contact the corresponding author to request additional information.
The included randomized clinical trials were independently reviewed by two authors (RYH and CWL) using Cochrane Collaboration’s risk of bias assessment tool. Any discrepancy found in the review process was resolved after discussion with the corresponding author (GHL).
Statistical analysis
Statistical analyses were performed by Comprehensive Meta Analysis version 3.3.070 (Biostat, Englewood, NJ, USA, 2014). In this study, RR was used generally for discrete variables, with the corresponding 95% CI were used to compare the outcomes between EVL alone and standard combination groups. Because the outcome contained a zero value in either or both groups, continuity correction was used to facilitate meta-analysis. 20 The pooled effect sizes were considered statistically significant if (1) p-value <0.05, or (2) the range of 95% confidence interval spare 1 for RR. In this study, a random effects model was chosen for pooled effect estimates because of limited study numbers which may underpower statistical and clinical heterogeneity. Of note, because some included studies compared EVL alone versus standard combination therapy, and the other studies compared combination therapy with different duration of vasoconstrictors, a mixed-effect model was used for the forest plots in the primary outcomes.
Statistic heterogeneity among studies was assessed by I square (I2) statistics, in which a higher value generally suggested significant heterogeneity. The presence of significant statistical heterogeneity was defined as p < 0.1 by a chi-square test or I square statistics >50%. Publication bias was evaluated by the funnel plot, in which the natural logarithm of the risk ratio was plotted against inverse standard error for each study, as well as Egger’s test; in which p < 0.1 in a two-tailed test was considered positive. In this study, we used the 5-day rebleeding to examine the publication bias because it was reported among all included studies. Sensitivity analysis was performed by excluding one study at each time to examine the robustness. Subgroup analysis by different study design, and other clinically important variables was also conducted as necessary.
Results
Search results and studies included
After excluding duplicated articles, we had 660 potentially relevant records based on the search strategy and additional manual search. During a subsequent review, 19 studies that met inclusion criteria were further evaluated for eligibility, and 9 full-text articles from 1995 to 2019 were included in this meta-analysis.11,12,14–18,21,22 The review process is illustrated by a flow chart of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (Figure 1 and Supplemental Appendix 1). 23

Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow chart.
Baseline characteristics of included studies
The enrolled 9 studies were all prospective, randomized controlled trials, and their baseline characteristics are summarized in Table 1 and Supplemental Table 1. Five studies compared the outcomes with EVL alone versus standard combination therapy, and the other four studies compared a shorter duration (the “limited combination”) to standard combination therapy.
Study designs and characteristics of the included studies.
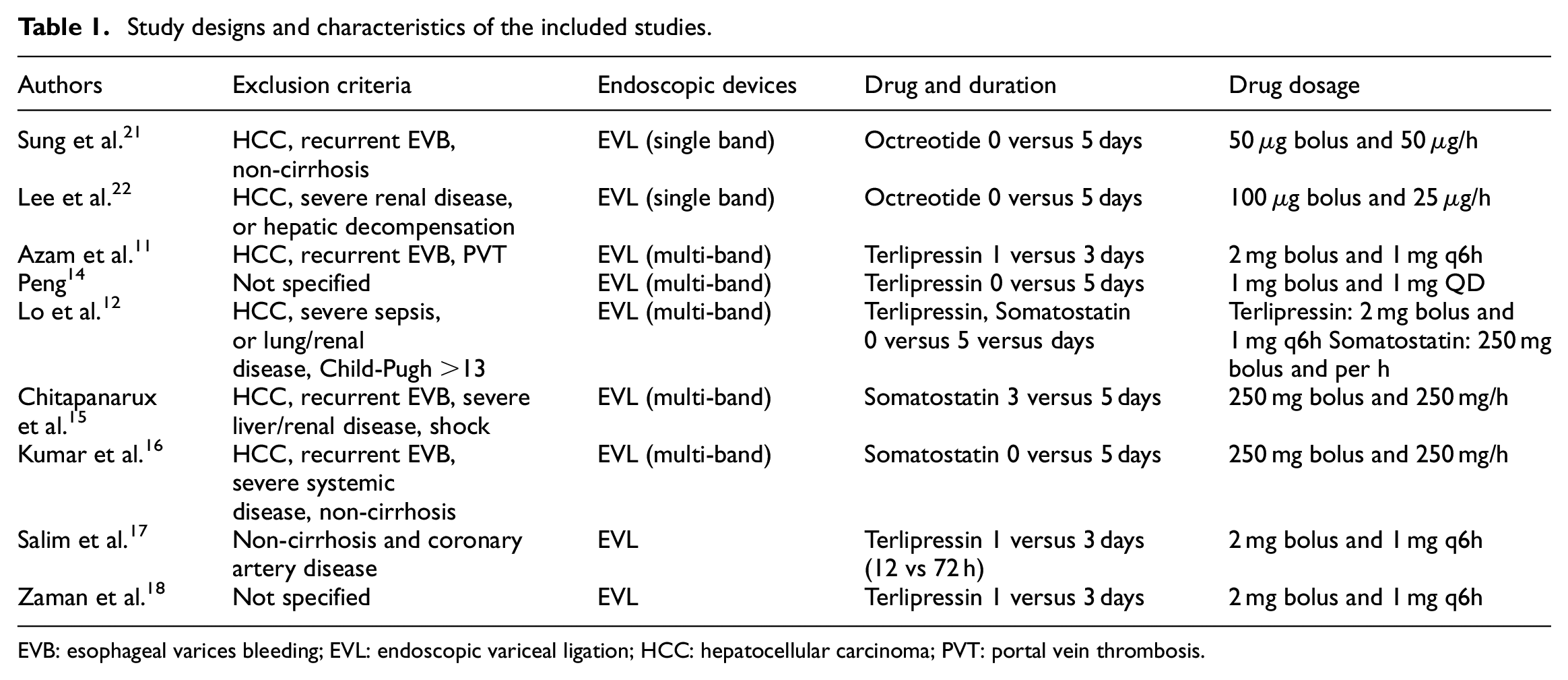
EVB: esophageal varices bleeding; EVL: endoscopic variceal ligation; HCC: hepatocellular carcinoma; PVT: portal vein thrombosis.
These studies contained a total of 838 patients (76.6% men) with the weighted-average age of 50.8 years old, and alcoholic cirrhosis ranged from 11% to 55% of cases; in addition, a substantial portion of the population was Child class B (39%–54%) or class C (24%–55%). Typical patients included were cirrhosis with the first attack of acute esophageal variceal bleeding; in other words, those who were non-cirrhotic, having hepatocellular carcinoma or portal vein thrombosis, or who had previous variceal bleeding, were generally excluded in these studies.11,12,15–17,21,22
After the index bleeding episodes, endoscopic treatments were performed within 6–24 h; and five studies after 2000 stated the use of multi-band ligator.11,12,14–16 The rate of active spurting during the index endoscopy was similar in both groups (27.1% in short-course vs 28.6% in standard combination group, p = 0.437). Vasoconstrictors, including terlipressin, octreotide, and somatostatin, were prescribed either before,11,15,18,21 or after emergent endoscopy.12,14,16,22 Their dosages adhered to the recommendation of current international guidelines,1,7 except for the study by Peng 14 that terlipressin was prescribed at 1 mg QD. In studies without vasoconstrictors in the control group; proton pump inhibitor, 12 propranolol, 14 and placebo 16 were prescribed as concomitant medical therapy.
Primary outcomes: 5-day rebleeding, mortality, and treatment failure rates
The numbers of patients with major outcomes in each study were summarized in Table 2. The rate of initial hemostasis was reported in all articles, with the range from 82% to 100%, and the pooled estimate was about the same between the short-course or standard combination groups (96.8% vs 97.4%, p = 0.919). Patients who did not receive vasoconstrictors also have similar initial hemostasis to their counterparts (98.5% vs 98.6%, p = 0.963).
Intervention and numbers of patients with major outcomes among the included studies.
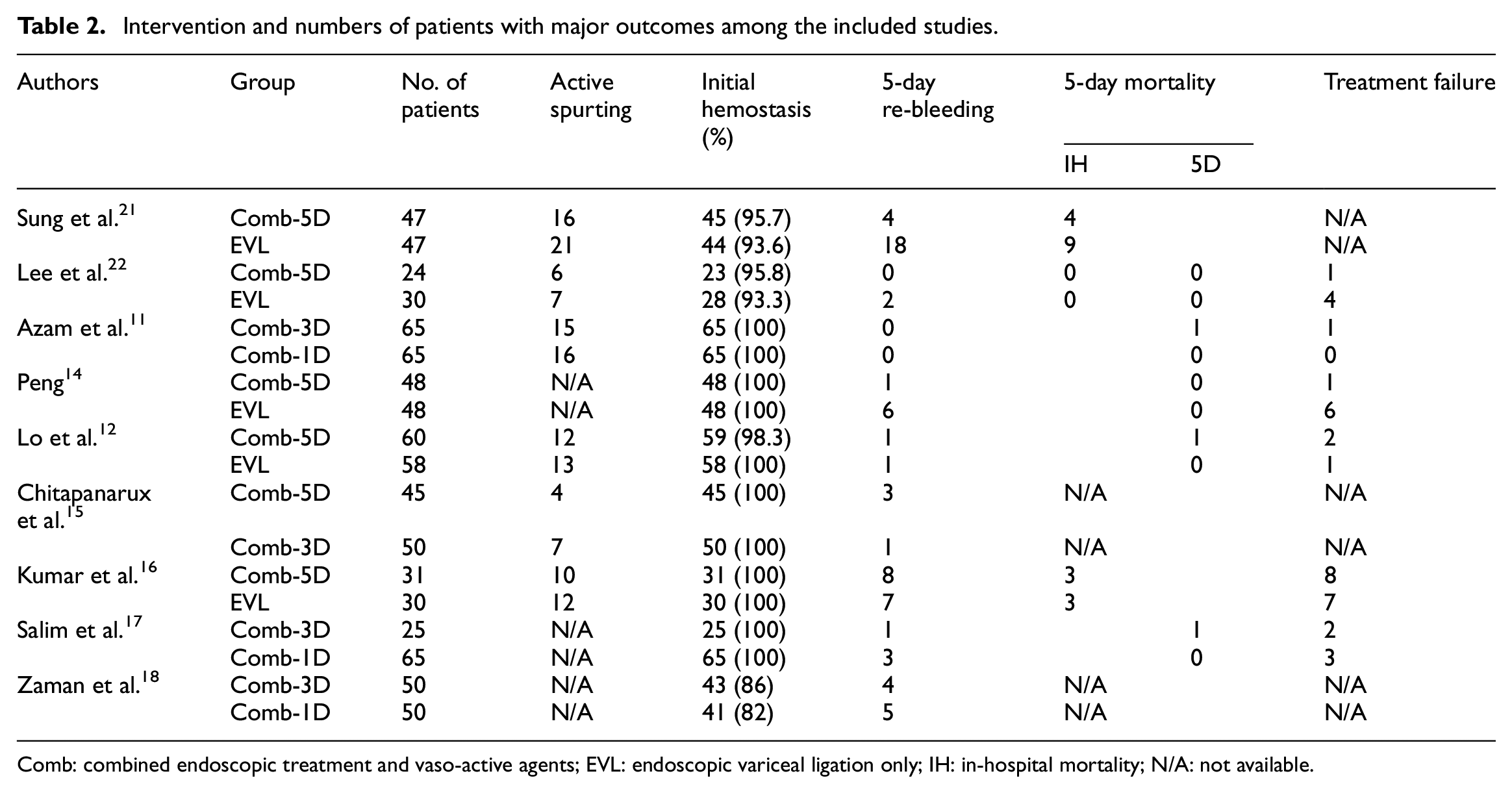
Comb: combined endoscopic treatment and vaso-active agents; EVL: endoscopic variceal ligation only; IH: in-hospital mortality; N/A: not available.
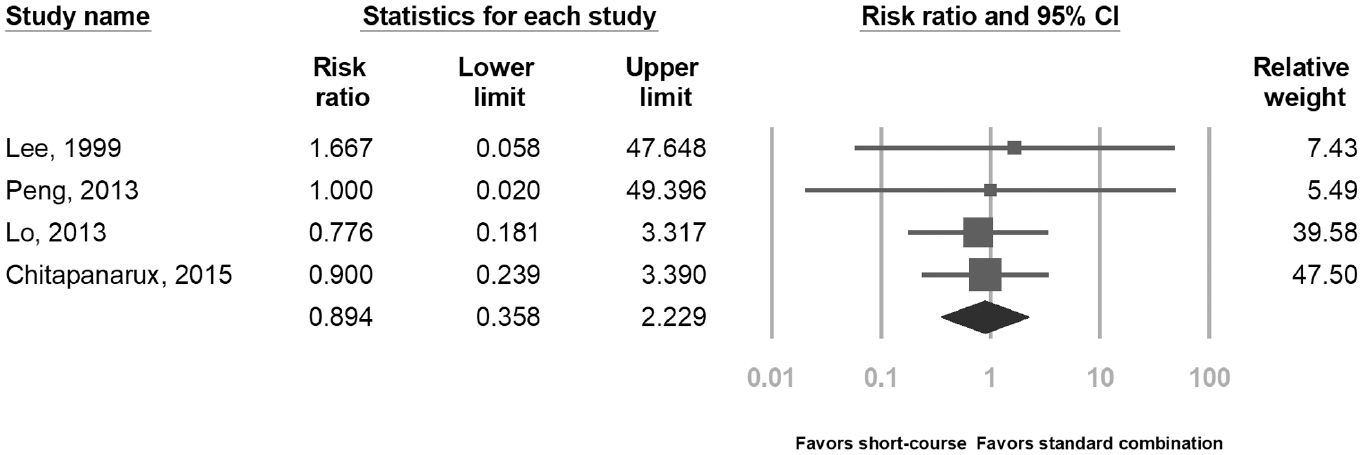
Five-day rebleeding was available in all the included studies, and most of them showed an insignificant risk ratio during the meta-analysis except for Sung et al., 21 which favored combination therapy. Pooled estimates of 5-day rebleeding rate were shown in Figure 2. The rebleeding rate was similar between short-course and standard combination groups (11.2% vs 8.3%, RR = 1.05, 95% CI = 0.62–1.76), and limit-days versus standard combination groups (6.9% vs 10.2%, RR = 0.75, 95% CI = 0.40–1.40). Although EVL alone had higher rebleeding rate versus standard combination groups (15.9% vs 7.1%, RR = 2.25, 95% CI = 0.87–5.77), it did not reach statistical significance.

Pooled risk ratio of 5-day re-bleeding rate. Overall heterogeneity: I2 = 37%, τ2 = 0.309, p = 0.122; EVL alone versus standard combination: I2 = 43%, τ2 = 0.448, p = 0.134; limited combination versus standard combination: I2 = 0, τ2 = 0, p = 0.84.
Five-day mortality rate was present as death within the first 5 days11,12,14,17,22 and in-hospital mortality.16,21,22 With meta-analysis, there was neither significant difference in 5-day (0% vs 1.3%, RR = 0.48, 95% CI = 0.10–2.39, Figure 3) nor in-hospital mortality (11.2% vs 6.8%, RR = 1.69, 95% CI = 0.70–4.04, Supplemental Figure 1) between both groups. Likewise, 5-day treatment failure rates were also similar between short-course and standard combination groups (7.4% vs 5.9%, RR = 1.10, 95% CI = 0.48–2.49, Figure 4). The subgroup analysis for 5-day mortality (0 vs 0.7%, RR = 0.71, 95% CI = 0.08–6.03) and treatment failure (9.6% vs 6.7%, RR = 1.43, 95% CI = 0.54–3.75) based on the study design (EVL alone or limited combination compared to standard combination) implied no difference between groups.

Pooled risk ratio of 5-day mortality rate. Overall heterogeneity: I2 = 0%, τ2 = 0, p = 0.954; EVL alone versus standard combination: I2 = 0, τ2 = 0, p = 0.965; limited combination versus standard combination: I2 = 0, τ2 = 0, p = 0.694.

Pooled risk ratio of 5-day treatment failure rate. Overall heterogeneity: I2 = 0%, τ2 = 0, p = 0.434; EVL alone versus standard combination: I2 = 24.7%, τ2 = 262, p = 0.263; limited combination versus standard combination: I2 = 0, τ2 = 0, p = 0.935.
Secondary outcomes: 6-week mortality, adverse events, hospital stay and transfusion requirement
The 6-week mortality was documented in four studies.12,14,15,22 With meta-analysis, it was similar between short-course and standard combination groups (4.3% vs 4.4%, RR = 0.89, 95% CI = 0.35–2.29, Figure 5). Adverse events of both groups were reported in three studies. In the study by Lee et al., 22 aspiration pneumonia (two in EVL alone and three in standard combination group) and one spontaneous bacterial peritonitis in the standard combination group were observed during the period of hospitalization, and Azam et al. 11 reported one myocardial infarction and stroke in each group, respectively. On the other hand, the study by Lo et al. 12 found significantly more chest pain and abdominal pain in the standard combination group (22/60 vs 0/60, p < 0.001).
Pooled risk ratio of mortality within 6 weeks. Heterogeneity: I2 = 0%, τ2 = 0, p = 0.982.
The length of hospital stay was also reported in two studies.12,22 Lee et al. suggested longer days of hospitalization in the group with EVL alone (16 ± 11 vs 11 ± 4 days, p = 0.028), whereas Lo et al. 12 did not found any difference between both groups (8.8 ± 3.8 vs 9.4 ± 2.3 days, p = 0.45). In addition, the duration of intensive care unit stay was similar in both groups.16,22 Finally, blood transfusion requirements were reported as mean and standard difference in three studies,12,15,22 the pooled estimate showed similar need of transfusion in both groups (standardized mean difference = 0.15, 95% CI = −0.37 to 0.68).
Sensitivity analysis and publication bias
The sensitivity analysis for 5-day mortality and treatment failure rates revealed that the pooled estimates had no significant changes for overall studies or the two subgroups with different design (EVL alone vs standard combination, and limited combination vs standard combination). However, the 5-day rebleeding rate for EVL alone versus standard combination exhibited significantly more rebleeding (8.9% vs 2.6%, RR = 3.30, 95% CI = 1.54–7.60) when Kumar et al. 16 was excluded. Furthermore, the statistic heterogeneity by I2 became zero when either Kumar or Sung’s study 21 was excluded, indicating the source of heterogeneity. By removing the latter study, the 5-day rebleeding rate become much more similar in both groups (5.7% vs 2.3%, RR = 1.24, 95% CI = 0.61–2.51). Publication bias was also assessed by the pooled estimate of 5-day re-bleeding and is displayed in Figure 6. Neither obvious bias on the funnel plot nor significant Egger’s test (one-tailed p = 0.643) was observed.

Funnel plot for risk ratios of 5-day rebleeding.
Risk of bias assessment
Risk of bias in each randomized clinical trial was evaluated by the Cochrane Collaboration’s risk of bias assessment tool, and the results were summarized in Supplemental Figure 2. Among these studies, three had a double-blind design11,15,16 and one was blind to examiner only 12 ; in addition, three studies did not report randomization and allocation concealment procedure.14,21,22 Studies that were considered a high risk of bias in reporting were attributed to the lack of a 5-day mortality rate.14,15,17,18 However, excluding these studies did not significantly change the primary outcomes.
Discussion
For acute esophageal variceal bleeding, vasoconstrictors plus endoscopic treatment, and prophylactic antibiotics have been the standard of care.1,7,24 Vasoconstrictors are generally recommended to be used for up to 5 days owing to the high risk of re-bleeding within the first 5 days. 25 Past studies showed that vasoconstrictors may have an additional benefit in patients received EIS by alleviating the paradoxical portal hypertension.26,27 EVL, on the other hand, is not associated with rebounding portal hypertension 28 and thus with superior efficacy for hemostasis.8,9 Coincidently, Kumar et al. 16 also demonstrated that hepatic venous pressure gradient was similar at baseline and post-treatment period after EVL. Hence, whether the use of vasoconstrictors can be reduced is a critical clinical question that was pointed out in a recent international guideline. 24
Our study demonstrated that a short course of vasoconstrictors had similar hemostasis, 5-day re-bleeding, mortality, and treatment failure compared to standard combination therapy, after successful EVL. Furthermore, the meta-analysis of published clinical trials suggested EVL alone might be as effective as standard combination therapy, though the latter conclusion needs to be made with caution because of clinical heterogeneity among studies and small overall case numbers.
However, these findings have some important clinical implications. For instance, the outcomes with EVL alone of earlier studies (e.g. Sung et al. 21 ) may be suboptimal owing to the lack of some modalities of modern care such as multi-band ligator and prophylactic antibiotics. As a result, the role of vasoconstrictors may be more important to maintain hemostasis and prevent early rebleeding. While this study was well-designed and the pioneer among randomized trials in comparison of EVL alone and combination therapy, it was the only positive trial for standard combination therapy and the 5-day rebleeding rate without vasoconstrictor (38%) was much higher than other subsequent studies. Hence, we believe further clinical trials to compare the necessity of vasoconstrictors after EVL should be valuable, and more evidence can guide the optimal approach in the future.
In terms of the duration of vasoconstrictors after EVL, our study was in line with the previous systematic review 13 that the use of vasoconstrictors up to 3 days shows similar clinical outcomes compared to longer duration. However, the current review is different from the prior one in several aspects. Firstly, our study only includes studies with EVL as the primary treatment, whereas one study with EVL and EIS 29 was enrolled in the previous meta-analysis. Secondly, it did not contain two clinical trials in our study,12,14 and thus the results are more confident in the present meta-analysis.
The potential advantages with a short course of vasoconstrictors afterward may reduce adverse events, hospital stay time, and financial costs. Although vasoconstrictors generally have a high safety profile and they are also well-tolerated, the association with side effects such as coronary ischemia and abdominal cramp is still a concern. In this systematic review, only the study by Lo et al. 12 reported significantly more side effects in the standard group without major morbidities. Thus, in patients with risk factors of ischemic heart disease or stroke, it might be reasonable to limit vasoconstrictors use once hemostasis was achieved by EVL. On the other hand, two studies addressed the length of hospital stays and the results failed to show significant benefit,12,22 and there was no trial in this review that compared the cost with or without vasoconstrictors. Still, more studies are necessary to evaluate the cost-effectiveness in the real-world setting.
This study had some limitations. Firstly, the number of studies and patients comparing EVL alone and standard combination therapy was not large. Secondly, most included patients were Child-Pugh class B and C, which might not be applied in patients with better liver reserve. Thirdly, the implication from different duration and types of vasoconstrictors for all the short-course groups could not be evaluated. However, a recent randomized trial suggested the efficacy should probably be similar among different vasoconstrictors. 30 Lastly, most of our included trials were not double-blind and thus there might be some concerns of bias. However, our sensitivity analysis demonstrated that excluding these studies would not change the major outcomes of the meta-analysis.
In conclusion, our study suggests that EVL with short-course vasoconstrictors is highly efficacious for esophageal variceal bleeding. Further studies are required to determine the genuine need for subsequent vasoconstrictor after successful EVL.
Supplemental Material
sj-tif-1-sci-10.1177_00368504211031711 – Supplemental material for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis
Supplemental material, sj-tif-1-sci-10.1177_00368504211031711 for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis by Jen-Hao Yeh, Gin-Ho Lo, Ru-Yi Huang, Chih-Wen Lin, Wen-Lun Wang and Daw-Shyong Perng in Science Progress
Supplemental Material
sj-tif-2-sci-10.1177_00368504211031711 – Supplemental material for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis
Supplemental material, sj-tif-2-sci-10.1177_00368504211031711 for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis by Jen-Hao Yeh, Gin-Ho Lo, Ru-Yi Huang, Chih-Wen Lin, Wen-Lun Wang and Daw-Shyong Perng in Science Progress
Supplemental Material
sj-rtf-3-sci-10.1177_00368504211031711 – Supplemental material for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis
Supplemental material, sj-rtf-3-sci-10.1177_00368504211031711 for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis by Jen-Hao Yeh, Gin-Ho Lo, Ru-Yi Huang, Chih-Wen Lin, Wen-Lun Wang and Daw-Shyong Perng in Science Progress
Supplemental Material
sj-rtf-4-sci-10.1177_00368504211031711 – Supplemental material for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis
Supplemental material, sj-rtf-4-sci-10.1177_00368504211031711 for Short-course vasoconstrictors are adequate for esophageal variceal bleeding after endoscopic variceal ligation: A systematic review and meta-analysis by Jen-Hao Yeh, Gin-Ho Lo, Ru-Yi Huang, Chih-Wen Lin, Wen-Lun Wang and Daw-Shyong Perng in Science Progress
Footnotes
Acknowledgements
We would like to thank the help for English polishment by Harris Chen.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the EDAHP110009 project of E-Da Hospital granted to Dr. Jen-Hao Yeh. The funder had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
