Abstract
Tumoral calcinosis is a kind of tumorous calcified masses, which may progress in patients with chronic renal failure. Uremic tumoral calcinosis in the brachioradialis muscle presented like acute infection of the arteriovenous fistula has not been previously reported. A 49-year-old man came to our hospital with a 9-year history of hemodialysis and secondary hyperparathyroidism. He had a huge painful mass in the left forearm at admission. The treatment was debridement and cytoreductive surgery accompanied by parathyroidectomy and medical management of calcium phosphate imbalance. After the comprehensive treatment, the patient recovered from his previously affected forearm. This is an enlightening case of intramuscular tumoral calcinosis mass that presented mimicking the infection of the arteriovenous fistula. Nephrologists and surgeons should pay attention to early diagnosis and proper management of the primary disease to master the indication and opportunity of resection.
Keywords
Background
Tumoral calcinosis (TC) is a kind of tumorous calcified mass frequently associated with a periarticular distribution, first described in 1943 by Inclan. It was classified into three pathogenically distinct subtypes including two primary TC with normal or elevated phosphate levels and one secondary to chronic renal failure, hyperparathyroidism, scleroderma, vitamin D intoxication, and so on. 1 In the case of uremia, sometimes accompanied by hyperparathyroidism, elevated serum calcium-phosphate products are usually caused by the calcium and phosphorus metabolism disorder. Precipitation of calcium-phosphate salts occurs when the solubility product accumulated is generally greater than 70, which leads to the occurrence of tumoral calcinosis. 2 Secondary TC arises mainly in acral sites, including upper and lower extremities. Almost all of those reported occurred near the joints, 3 but rarely in the muscles. We report a huge recurrent lesion of TC in the brachioradialis muscle with the presentation mimicking a regional infection of the arteriovenous fistula in a patient suffering end-stage renal failure, focusing on disease progression, diagnosis, and treatment outcomes. To the best of our knowledge, there was no such case previously reported.
Case presentation
Chief complaints
A 49-year-old man on long-term hemodialysis (HD) was admitted for pain in his brachioradialis region of the left forearm.
History of present illness
A muscle swelling near the fistula of the left forearm has existed for 1 year. The swelling of the left brachioradialis worsened and local skin temperature elevated for about 1 week (Figure 1).

Physical examination revealing swelling in the brachioradialis region nearby the puncture sites of the fistula, with elevated local skin temperature.
History of past illness
The patient was receiving hemodialysis for the treatment of end-stage renal failure for about 9 years. The arteriovenous fistula was obtained with native vessels of the forearm (radial-cephalic). He was diagnosed with secondary hyperparathyroidism for 1 year. The BUN level was 18.35 (normal, 2.9–8.2 mmol/L) and the creatinine level was 789 (normal, 59–104 µmol/L) at that time. The parathyroid hormone levels was 1865 (normal, 15–65 pg/mL). It was considered secondary to his severe renal failure. Within 1 year before his admission, the patient was undergoing the HD three times per week. The HD lasted about 4 h. The blood pump speed was 300 mL/min and the dialysis pump speed was 500 mL/min. The weekly Kt/V level was kept above 1.2. The latest one before admission was 4.1. Six months before admission, the BUN level of the patient was 14.22 (normal, 2.9–8.2 mmol/L) and the creatinine level was 691 (normal, 59–104 µmol/L). Not long before admission, he was in the waiting period of parathyroidectomy on the advice of his doctor.
Physical examination upon admission
Physical examination revealed muscle swelling near the fistula of the left forearm. The swelling mass was reddish, with elevated skin temperature, and local tenderness.
Laboratory findings
Laboratory studies showed a reduction in leukocyte count of 3.2 (10E9/L) an elevated C-reactive protein (CRP) level of 60.2 (normal, 0–8 mg/L). The BUN level was 29.83 (normal, 2.9–8.2 mmol/L). The serum creatinine level was 1040 (normal, 59–104 µmol/L), and parathyroid hormone levels was 1723 (normal, 15–65 pg/mL). The serum calcium level was 9.68 (normal, 8.12–10.16 mg/DL) and serum phosphorus was 8.68 (normal, 2.70–4.50 mg/DL), with a calculated calcium-phosphate solubility product of 84 (Table 1). The strange thing is that blood cultures and tissue biopsy were unrevealing. Chest X-ray showed no signs of a lung infection. The tumor markers were within the normal range. Technetium-99m methoxyisobutylisonitrile-single-photon emission computed tomography/computed tomography (99mTc-MIBI SPECT/CT) revealed bilateral parathyroid hyperplasia (Figure 2).

The 99 mTc-MIBI SPECT/CT revealing bilateral tertiary hyperparathyroidism: (a) low-density nodules with tracer agent aggregation in the posterior and lateral of the right lobe of the thyroid (arrow) and (b) low-density nodules with tracer agent aggregation in the upward side and lateral of the left lobe of the thyroid (arrow).
Clinical course
The nephrologists first diagnosed it as infection of the arteriovenous fistula. Repeated puncture was considered as the cause of infection. It never happened in his 9 years of hemodialysis. The patient developed a high fever at 38.7°C on the day of admission. He began to feel drowsy with the blood pressure dropped. Considering the risk of septic shock at that moment, parathyroidectomy was put on hold while antibiotic treatment was initiated. The patient was treated with Vancomycin (Eli Lilly, Japan; 1000 mg every 12 h). But the Blood cultures and tissue biopsy were unrevealing. Antibiotic treatment was not effective to release the local distending pain. The fever lasted for the next three days. The brachioradialis mass increased in size, and thus, the pain also increased. Plain radiographs showed extensive soft tissue calcification without bone and joint involvement (Figure 3). Magnetic resonance imaging (MRI) revealed low signal intensity on TI sequence with septa inside forming a honeycomb appearance, which was known as “chicken wire” described in the previous literatures. 4 It also displayed a large irregular lesion with multiple cavities of mixed-signal density on a T2-weighted sequence. It was blended with muscle without a clear boundary, which was considered an intermuscular abscess (Figure 4).

Plain radiographs showing extensive soft tissue calcification without the involvement of the radius, ulna, or the elbow.
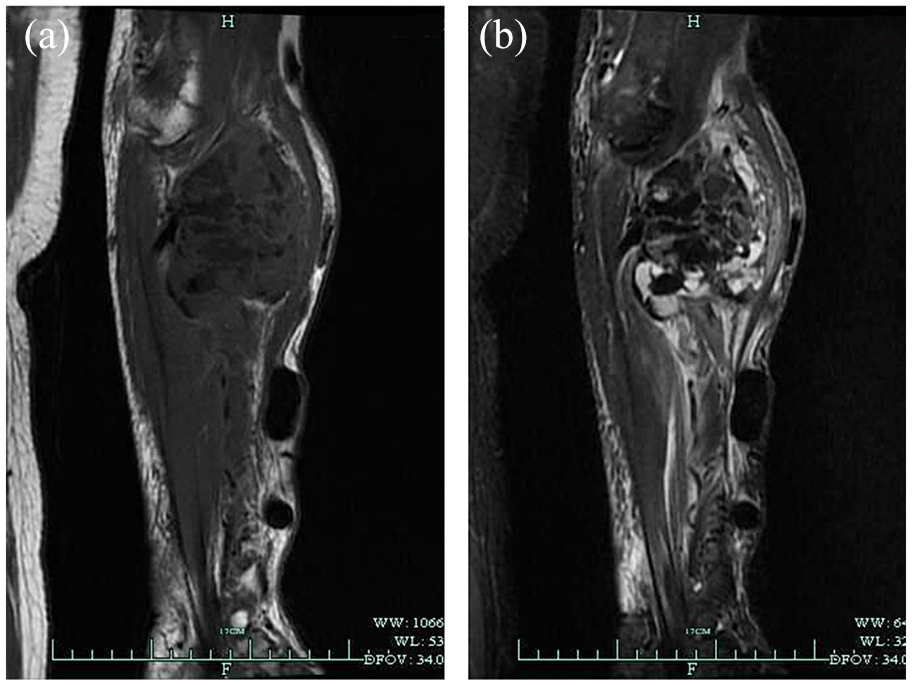
(a) MRI displaying a large irregular lesion with multiple cavities of low signal density on T1-weighted sequence and (b) multiple cavities of mixed-signal density on T2-weighted sequence.
The nephrologists suspected that the repeated puncture of hemodialysis was the path of infection in the forearm. So ligation of the internal fistula was performed in case of sepsis spread. The patient was then treated with surgical debulking for symptomatic relief and drainage of the abcess. During the first operation, the muscle appeared intact on the surface (Figure 5(a)), and milk white liquid blended with blood were found inside the muscle. (Figure 5(b)). It was partly cleared and vacuum sealing drainage was performed. The lateral antebrachial cutaneous nerve was released and protected. Consistent with preoperative biopsy culture, there was no pathogenic bacteria found in the bacteriological results intraoperatively. One week after the operation, the patient was pain-free, and his temperature and inflammatory index returned to normal values. Postoperative pathological examination confirmed the diagnosis of tumoral calcinosis, showing up as deposits of calcified material blended with giant cells and inflammatory cells as well as fibrous tissue septa (Figure 6). Parathyroidectomy was suggested, although the patient refused, being unaware of its importance. So the patient was required to receive long-term medication of phosphate-binding antacids. Lanthanum carbonate (Shire, America, 1500 mg every day) was given during the hospitalization and after he was discharged and the dose was adjusted according to the phosphate level. After the patient was discharged, he continued with the previous HD regime using the central venous catheter. And the weekly Kt/V level was 3.6 the last time of HD before his second admission.

(a) During the first operation, the muscles appeared intact on the surface, and the lateral antebrachial cutaneous nerve is inside the lesion (arrow), (b) during the first operation, calcifications resembling the consistency of toothpaste blended with bloody fluid are found interspersed throughout the muscle, and (c) during the second operation, the mass contains chalky calcification and milk-white fluid, that blended with the muscle.

Postoperative pathological examination confirming deposits of calcific material and giant cell reaction: (a) the lesions comprise several calcospherites and granular calcific debris (H&E stain,; magnification, ×50) and (b) giant cells and scattered inflammatory cells aggregation within the lesion (H&E stain; magnification, ×200).
Six months after the first resection, the calcified mass recurred with progressive swelling and worsening of pain. In the meanwhile, he experienced persistent pain due to the tertiary hyperparathyroidism. During this second hospital course, he accepted the parathyroidectomy procedure, and the excision of the tumoral calcinosis lesion was successful. The mass contained chalky calcification and milk-white fluid, that blended with the muscle (Figure 5(c)). It was removed as much as possible while the deep branch of the radial nerve and the integrity of the brachioradialis were protected. The patient was asked to take Dihydroxycholecalciferol (Qingdao Chia Tai Haier Pharmaceutical Co Ltd. China, 1 µg four times every day), and Calcium Carbonate and Vitamin D3 Tablets (Wyeth pharmaceuticals, America, 900 mg of Calcium Carbonate and 4.6875 µg of Vitamin D3 four times every day) after surgery. Hemodialysis frequency was increased to once every other day. During the next year, serum calcium and phosphate levels were controlled and the Ca x P product was found within the normal range. The serum calcium level was 7.76 (normal, 8.12–10.16 mg/DL) and serum phosphorus was 4.71 (normal, 2.70–4.50 mg/DL) at 1 year after surgery. The parathyroid hormone level reduced to 22.9 (normal, 15–65 pg/mL) at the same time (Table 1). The patient did not experience any recurrence until now. Long-term follow-up is underway as well.
Discussion
When the diagnosis of TC is considered while having a condition known that promotes ectopic calcifications, it is categorized as secondary TC. 1 End-stage renal disease is one of the most common causes of secondary TC. Hyperparathyroidism secondary to end-stage renal failure is supposed to accelerate the progress of calcium-phosphate precipitation. 5 Nevertheless, parathyroidectomy did not stop the progression of TC completely. Thakur has reviewed six studies with 24 patients with secondary TC. He found that TC resolved in all patients who underwent excisional biopsy. Only 27% had TC regression after subtotal or total parathyroidectomy. 6 Other studies proposed a high propensity of recurrence even after surgical removal. 7 A multidisciplinary approach are required for effective treatment.
Most secondary TC appears around the joints, presenting single or multiple lesions.3,7 Swelling pain, nerve compression, decreased joint mobility, and skin ulcers are common in large tumoral calcinosis masses.8–10 The masses may blend with the adjacent muscles, rendering diagnosis difficult. Numerous causes may lead to metabolic and dystrophic calcification similar with TC, such as sclerosis, synovial sarcoma, synovial osteochondromatosis, calcific tendonitis, calcific bursitis, and tumors like extraskeletal chondrosarcoma, osteosarcoma, and others.11–13 The characteristic manifestation of TC on MRI is described as “chicken wire.” 4 The specific presentation was not mentioned in any other diseases referred above. In addition, the existence and progression of primary disease, especially the long-term end-stage renal failure with high-level calcium-phosphate product are the important identifying factors.2,10,12
In this case, the huge lesion was located in the brachioradialis and did not have a well-defined margin. During the first operation, multiple cystic spaces that contain milky mixed with reddish-brown fluid were observed in the mass, indicating there may be mild hemorrhage in it. The primary diseases of long-term end-stage renal failure and hyperparathyroidism may have been the original cause. It was proposed that repeated traumas might initiate a successive reaction to the development of TC in juxta-articular soft tissues and skin. 14 Another study reported that calcium ions might leak into tissues surrounding vessels and lead to calcium deposition during intravenous administration of calcium gluconate or calcium chloride. 15 Unlike these types of iatrogenic calcinosis, the lesion, in this case, was inside the muscle, close to, although not confined to the arteriovenous fistula region. Moreover, there was no calcinosis on the surface of the muscle. Therefore, we believe that it was not exogenous. Repeated minor trauma of punctures may be the cause of blood aggregation in the local region. The presentation of it was quite similar with local infection. However there was no direct evidence of infection.
In the authors’ experience, recurrence of secondary TC tends to occur unless the original cause of the elevated calcium-phosphate product is addressed. 6 Thus, the lesion was not controlled before parathyroidectomy in this case. Even if surgical intervention is the only palliative, to those masses impinged on nerves and vessels, causing symptoms, or secondary infections, surgical resection is necessary, as radiotherapy or systemic steroid treatment has proved ineffective. 11 However, the whole brachioradialis muscle could not be removed in this case. Thus, cytoreductive surgery was performed to relieve pain. Medication for calcium-phosphate imbalance, including bisphosphonates, calcium channel blockers, low-dose anti-vitamin K, probenecid, vinpocetine, cinacalcet, as well as the optimum dialysis and parathyroidectomy co-assist in the treatment of uremic tumoral calcinosis. In the end, renal transplantation is the ultimate cure for this problem. 16
Conclusion
We diagnosed the first case of a huge TC inside the brachioradialis muscle, mimicking acute infection of the arteriovenous fistula, and presented its progression and treatment course. The condition of this patient has rarely been reported in previous literatures. Increased awareness of this rare disease by both the nephrologists and surgeons may help in the early diagnosis and proper management of the primary disease to master the indication and opportunity of resection.
Supplemental Material
sj-pdf-1-sci-10.1177_00368504211018560 – Supplemental material for Intramuscular tumoral calcinosis near the arteriovenous fistula mimicking acute infection
Supplemental material, sj-pdf-1-sci-10.1177_00368504211018560 for Intramuscular tumoral calcinosis near the arteriovenous fistula mimicking acute infection by Qianjun Jin, Haiying Zhou and Hui Lu in Science Progress
Footnotes
Acknowledgements
I’d like to show my sincere gratitude to my colleague Dr. HL who gave me support in every step of this study. Also, I really appreciate the funding program for sponsoring our research. Besides, I want to thank my husband, Lawyer Hui Cai, for helping me with the figures and ethical principles.
Author contributions
JQJ and ZHY conceived of the study. LH and ZHY participated in the design of the study. JQJ drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data collection and Data analysis of this work were supported by Zhejiang Traditional Chinese Medicine Research Program (grant number 2016ZA124, 2017ZB057).
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Availability of data and materials section
The dataset supporting the conclusions of this article is included within the article.
Supplemental material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
