Abstract
Objectives:
Although the existing literature highlights the success of pharmacist-led hepatitis C virus (HCV) treatment, the impact of these models on time to treatment initiation has not been studied. This study compared outcomes for HCV treatment using direct-acting antivirals (DAAs) in 2 groups: pharmacist-led programs in 5 federally qualified health centers in Ohio and an external cohort from an out-of-state academic medical center. We assessed the time to HCV treatment initiation, rates of DAA initiation, and sustained virologic response (SVR) rates.
Methods:
We conducted a retrospective cohort analysis using electronic health records and pharmacy claims for patients with HCV referred to pharmacists from November 2021 through December 2023. We collected data on demographic characteristics, pre-entry characteristics, treatment initiation rates, time to initiation, and cure rates. The treatment initiation rate included all patients referred to a pharmacy. We defined time to initiation as the period from pharmacy referral to the date of medication pickup. We defined cure as achieving SVR 12 weeks posttreatment (SVR12) and analyzed SVR12 rates in an intent-to-treat and per-protocol manner. We compared time to initiation with an external cohort median of 300 days.
Results:
Of 477 referred patients, 317 (66.5%) initiated DAAs. The mean (SD) time to treatment initiation was 63.7 (74.2) days, with a median of 37 days. Among the 212 patients completing SVR12 laboratory tests, 194 achieved cure, yielding a per-protocol SVR12 of 91.5% and an intent-to-treat SVR12 of 61.2%. Patients without (vs with) health insurance and with (vs without) a history of substance abuse had longer treatment delays.
Conclusions:
This study demonstrates the value of integrating pharmacists into HCV treatment teams to meet public health needs.
Hepatitis C virus (HCV) affects approximately 50 million people worldwide, yet only 36% are aware of their infection. 1 In the United States, half of the 2.4 million people with HCV are aware of their status. 2 With only 5% of Medicaid enrollees receiving treatment each year, a need exists to increase screening, simplify delivery models, and reduce barriers for populations at high risk of HCV. 3 These strategies are essential to achieving the World Health Organization’s goal of eliminating HCV by 2030. 4
Untreated HCV infections are associated with costly outcomes including fibrosis, cirrhosis, and hepatocellular carcinoma. 5 In addition, extrahepatic manifestations of HCV, such as type 2 diabetes, depression, cardiovascular disease, and renal disease, contribute to disease burden and increase health care costs. 5
Direct-acting antivirals (DAAs) have revolutionized HCV treatment by simplifying treatment regimens.6,7 To eradicate HCV, national societies have recommended that HCV treatment be expanded to nonspecialist health care providers. 8 Treatment in primary care is possible; however, recent data indicate barriers to implementation. A survey of health care providers (hereinafter, providers) in Washington State elucidated their HCV treatment practices and hesitancies. 9 Of 547 providers, only 28% reported treating people with HCV. Common reasons for not treating were knowledge deficit (64%) and no organizational support (24%). Of these providers, only 52% indicated an interest in providing HCV treatment if given resources to support them. Providers who treated people with HCV reported they were most uncomfortable discussing health insurance coverage and cost support, drug interactions, and adverse effects of treatment. 9 Given pharmacists’ knowledge of medication use, monitoring, and health insurance adjudication, they are uniquely qualified to increase access to HCV treatment. Pharmacist-led HCV treatment cascades demonstrate favorable treatment outcomes.10-12 These models have proven to be cost-effective and feasible, particularly in safety-net clinics and populations at high risk of HCV, such as people who inject drugs (PWID).13-15
While the existing literature highlights the success of pharmacist-led HCV treatment, the effect of these models on time to treatment initiation (TTTI) has not been extensively studied. Ortiz and Rasch 16 found that TTTI improved after implementation of a pharmacist-led model; however, their approach still required referral to a specialist. 16 Currently, it is not well understood how pharmacist-led treatment models in primary care settings, implemented at the point of screening and diagnosis, affect TTTI.
TTTI is a crucial factor for patients, health systems, and public health. Shortening this time frame can reduce the prevalence and severity of HCV-related extrahepatic manifestations and result in cost savings for health care systems. Faster treatment initiation may reduce loss to follow-up and increase treatment completion in challenging populations such as PWID, one of the largest HCV reservoirs in the United States.17,18 Timely treatment initiation is also a preventive measure. Modeling data demonstrate that 1 person with HCV who injects drugs can transmit HCV to 20 others within 3 years of initial infection. 19 Eliminating HCV in the population of PWID can, thus, have a substantial effect on curbing HCV transmission.1,4
This study occurred in 5 federally qualified health centers (FQHCs) in Ohio. Patients of FQHCs typically are medically underserved, have multiple chronic diseases, and have problems accessing health care.20,21 We evaluated TTTI for pharmacist-led treatment models.
Methods
Design and Setting
We conducted a retrospective cohort analysis using electronic health record data from 5 FQHCs in Ohio using a pharmacist-led treatment model. This model was independently implemented at each FQHC prior to the study.
Although workflows varied, a consistent process was followed across all FQHCs. After a primary care provider referred a patient to the program, pharmacists managed patients with HCV under a collaborative practice agreement (CPA). Under CPA, pharmacists can prescribe and manage drug therapy, including ordering any necessary laboratory tests to initiate and monitor DAA treatment. Pharmacists in the program (1) assessed appropriateness of the referral, (2) evaluated patient history and treatment candidacy, (3) selected and prescribed DAAs, (4) provided patient education, (5) facilitated medication access, (6) monitored adherence and tolerability, and (7) assessed for sustained virologic response (SVR) at least 12 weeks posttreatment (SVR12).
Prior authorization was required for all DAA prescriptions for health insurance coverage, with varying restrictions based on health insurance type. Type of health insurance dictated which pharmacies could fill DAA prescriptions, with some requiring a specialty pharmacy.
Study Outcomes
The primary outcome was TTTI, measured in days from the date of the referral to the pharmacist to the date the patient received medication. This metric was defined for the overall study cohort and stratified by patient characteristics, including fibrosis staging, health insurance status, history of substance use disorder, and the dispensing pharmacy type (in-house, community retail, mail order, or patient assistance program). We compared TTTI with an external cohort from an out-of-state academic medical center for benchmarking purposes. 22 Secondary outcomes included treatment initiation rates and SVR12 rates in both intention-to-treat and per-protocol analyses.
Data Collection and Analysis Plan
We collected data from electronic health and pharmacy dispensing records and managed the data using Microsoft Excel. We compiled information from each site for analysis. We reviewed all referrals for HCV treatment to the pharmacists from November 2021 through December 2023 for inclusion. We collected data on demographic characteristics, dates, and laboratory results from the initial referral date until SVR assessment, treatment discontinuation, or loss to follow-up as of June 30, 2024.
The calculation of treatment initiation rate encompassed all patients referred to the pharmacist program. The TTTI analysis and intention-to-treat SVR12 analysis group included patients who had picked up their DAA medication by December 31, 2023. The per-protocol SVR12 analysis included patients who had picked up their DAA medication by December 31, 2023, and SVR12 laboratory results.
Patients were excluded from the TTTI and SVR12 analyses if they were outside the pharmacist’s scope under the CPA, including decompensated cirrhosis, HIV, hepatitis B virus, previous DAA treatment experience, or pregnancy. Additional exclusions included patients who did not complete pretreatment laboratory work or did not pick up their DAA medication to begin therapy.
We imported data into SPSS version 25.0 (IBM Corp). We summarized baseline demographic characteristics for the study cohort using frequencies and percentages. We summarized outcome data for time milestones using means and SDs, and we summarized SVR12 laboratory data using frequencies and percentages. We calculated 95% CIs for the primary and secondary outcomes. We performed exploratory analyses to determine factors associated with differences in the primary outcome variable. We used analysis of variance tests with Tukey honestly significant difference post hoc tests for factors with >2 subcategories, and we used the Welch Student t test for dichotomous factors. All tests were 2-sided, with significance set at P < .05. No data were missing for the primary outcome. Missing data for secondary analyses (n = 19 for genotype, n = 2 for fibrosis stage) were treated as missing at random without imputation.
This study was conducted in accordance with the Declaration of Helsinki. The institutional review board (IRB) at Northeast Ohio Medical University was the central IRB, whose review was accepted by all participating institutions’ IRBs (ref. 24-004). The central IRB determined that this research involved minimal risk and approved a waiver for informed consent. We used the Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines for cohort studies. 23
Results
From November 2021 through December 2023, a total of 477 patients were referred to pharmacists for HCV treatment under the CPA at 5 FQHCs (Figure). Of these, 386 (80.9%) were established for HCV treatment with the pharmacists and completed pretreatment laboratory tests. Subsequently, 333 (69.8%) were prescribed DAAs. Patients out of the pharmacist’s scope as defined by the CPA were referred elsewhere. These included patients with decompensated cirrhosis (n = 26), multiple exclusions (n = 12), hepatitis B virus (n = 3), previous treatment (n = 3), and pregnancy (n = 3). Finally, 317 patients picked up their medications by December 31, 2023, resulting in a treatment initiation rate of 66.5%. Of these 317 patients (Table 1), 212 (66.9%) returned for SVR12 laboratory results. Among those tested, HCV was undetectable in 194 patients, yielding a per-protocol SVR12 rate of 91.5% and an intention-to-treat SVR12 rate of 61.2%.
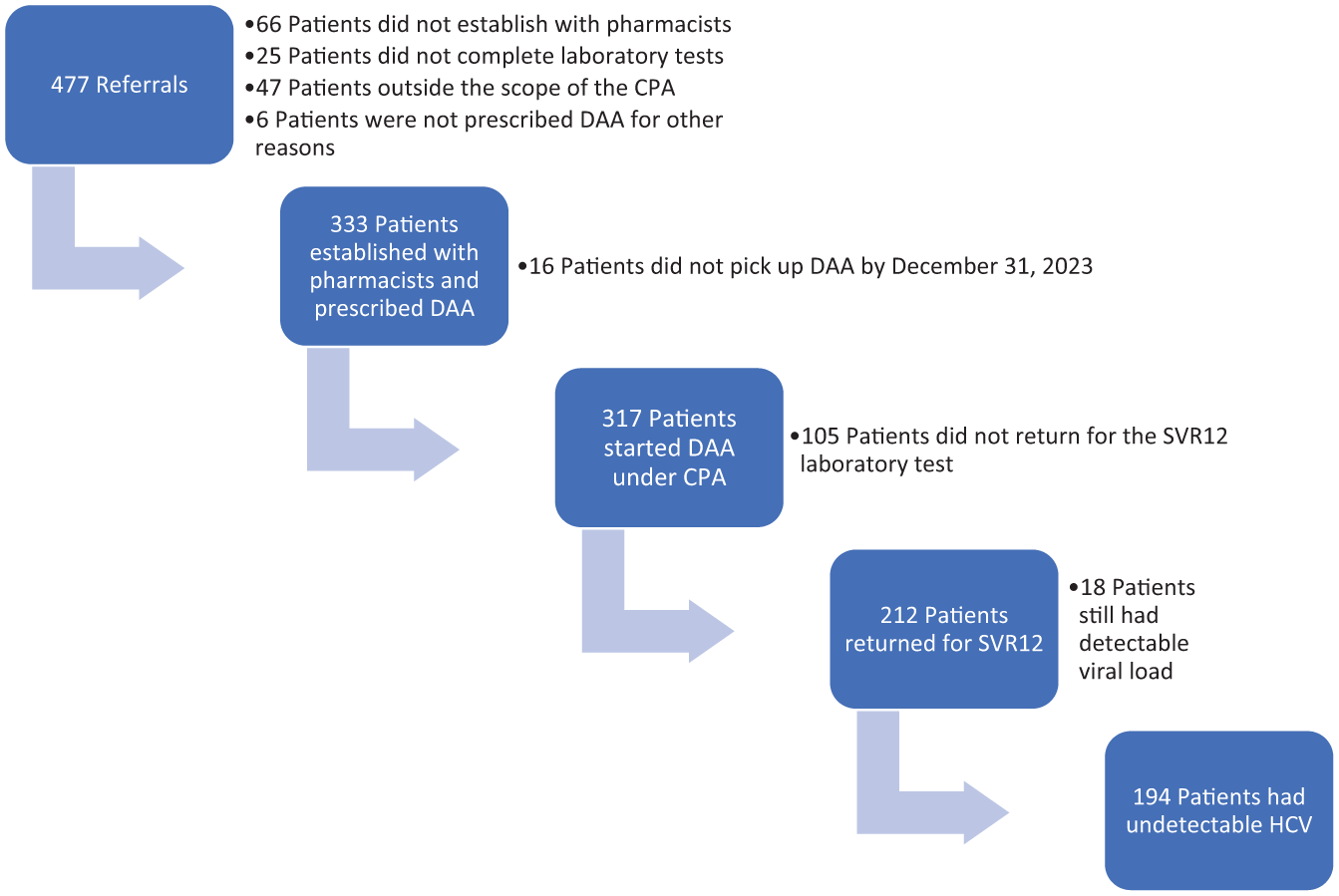
Care cascade of patients referred to pharmacist-run hepatitis C clinics (n = 317) at 5 federally qualified health centers in Ohio, 2021-2023. Abbreviations: CPA, collaborative practice agreement; DAA, direct-acting antiviral; HCV, hepatitis C virus; SVR12, sustained virologic response 12 weeks posttreatment.
Demographic and entry characteristics of patients who received direct-acting antiviral therapy at pharmacist-run hepatitis C programs (n = 317) in 5 federally qualified health centers in Ohio, 2021-2023

Abbreviation: HCV, hepatitis C virus.
For the 317 patients who initiated treatment, the mean (SD) [95% CI] TTTI was 63.7 (74.2) [55.5-71.9] days, with a median of 37.0 days. The mean time from referral to initial visit was 31.7 days, and the mean time from initial visit to receipt of treatment was 32.0 days.
The TTTI did not differ significantly based on age, sex, ethnicity, fibrosis staging, or HCV genotype. Patients without (vs with) health insurance who were using manufacturer patient assistance programs and patients with (vs without) a history of substance abuse had longer treatment delays (Table 2).
Factors associated with a difference in time to treatment initiation with direct-acting antivirals in patients who were treated at pharmacist-run hepatitis C programs (n = 317) at 5 federally qualified health centers in Ohio, 2021-2023
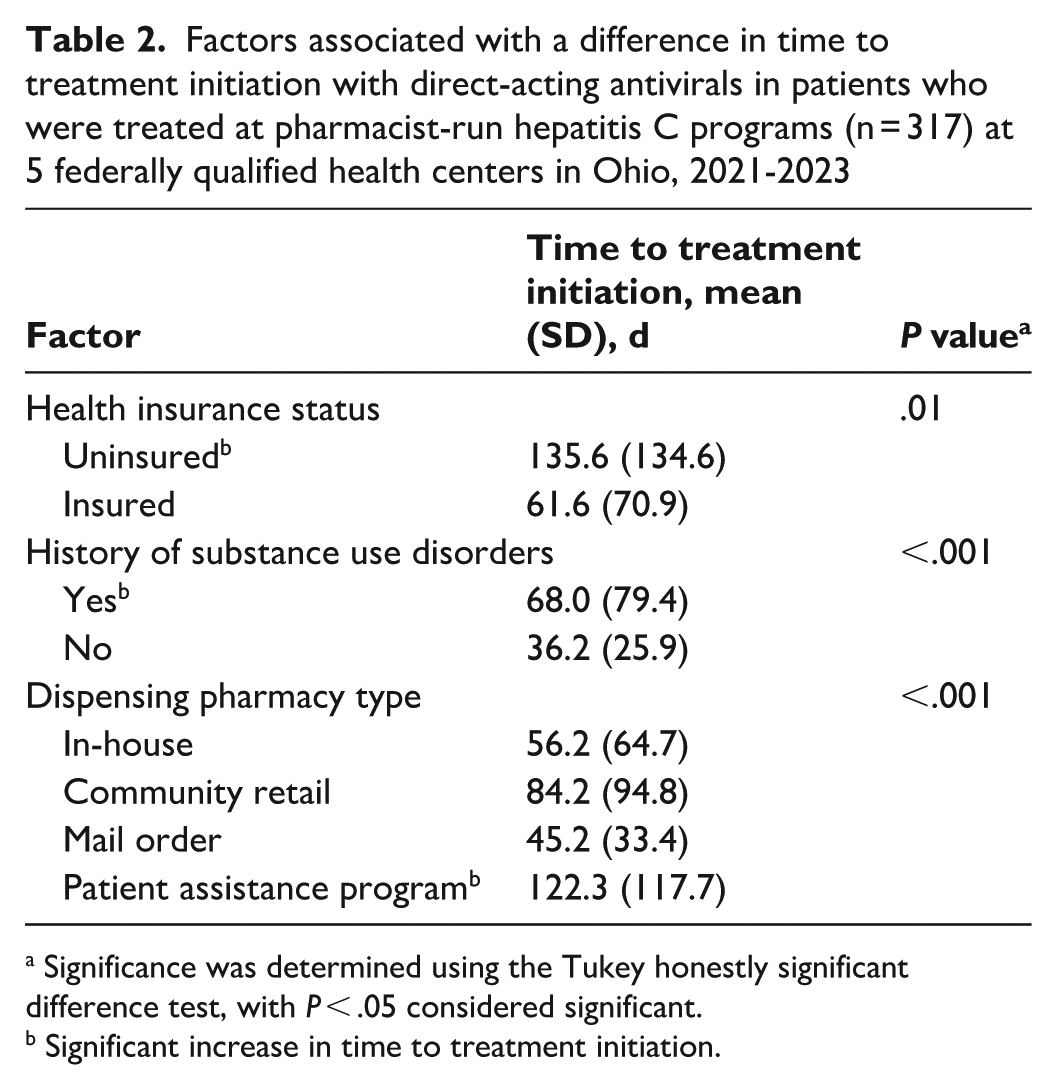
Significance was determined using the Tukey honestly significant difference test, with P < .05 considered significant.
Significant increase in time to treatment initiation.
Discussion
Previous studies have documented TTTI in patients with HCV. A retrospective cohort analysis by Kwo et al 22 examined treatment initiation rates, TTTI, and SVR12 rates with DAAs in a cohort of 8611 patients from a large health system in Indiana. The analysis reported a treatment initiation rate of 9.9%, a median TTTI of 300 days (range, 25-675 days), and a per-protocol SVR12 rate of 84.7%. In contrast, our study found a treatment initiation rate of 66.5%, a median TTTI of 37 days, and a per-protocol SVR12 rate of 91.5%. Kwo et al did not evaluate the type of provider prescribing HCV treatment. Some differences exist between the methods used in our study and the methods for calculating TTTI in the external cohort. Kwo et al defined TTTI as the period from the first clinical encounter for HCV diagnosis to the first medication order of a DAA agent, whereas our study measured the time from referral to the date of DAA pickup. We chose this metric because our data source did not reliably capture the time from initial HCV diagnosis to the time of referral for HCV treatment, and accounting for the pickup date captures delays between prescribing and medication pickup such as those caused by prior authorizations and primary nonadherence. The external cohort study reported the median TTTI. To facilitate a comparison, we reported our median TTTI. However, our multivariate analysis used mean TTTI, because this metric provides more information about the dataset.
We demonstrated a shorter TTTI compared with the external cohort, despite the significant proportion of patients with substance use disorder (81.7%) and Medicaid insurance coverage (78.6%). This finding underscores the effect of pharmacist-led treatment in primary care on this outcome. Our findings are further supported by data on Medicaid enrollees from 2019-2020 as reported by the Centers for Disease Control and Prevention, which showed that most patients with HCV (77%) had not initiated treatment within 1 year of diagnosis. 24 Additional evidence indicates that the median TTTI among Medicaid enrollees was approximately 216 days from 2014 to 2018. 25 Furthermore, data from a nationwide cohort study examining Medicaid claims for DAA initiation during 2017-2019 demonstrated that 20% of patients initiated treatment with DAAs within 6 months of diagnosis. 26 In our study, 62% of patients referred started treatment within 6 months. When examining the totality of evidence on TTTI for HCV with DAAs, a gap in care is present, which was improved in our study population. While we show promising results for pharmacist-led HCV treatment helping patients access medication sooner, current delays are still unacceptable given the risk of loss to follow-up. Therefore, efforts to further decrease delays and bridge gaps in care should be continued.
Although some sites had in-house pharmacies that could dispense DAAs, they did not demonstrate a shorter TTTI, implicating other causes of delay in the care process. We observed delays in initiating DAA treatment between the time the patient was referred to the pharmacist and the initial pharmacist visit and between the initial pharmacist visit and prescribing of medication. The delay between the patient being referred to the pharmacist and the initial pharmacist visit was likely due to many initial pharmacist visits being scheduled on separate days from the initial visit with the medical provider. Implementation of provider–pharmacist co-visits or pharmacist availability at the time of the medical provider visit should be considered to eliminate this delay. The delay seen between the pharmacist visit and prescribing was likely a result of laboratory tests needing to be completed prior to prescribing. Point-of-care HCV testing has been linked to significantly decreased TTTI. 27 Using point-of-care HCV testing to determine HCV status would allow treatment laboratory results to be ordered and collected on the same day. This practice would allow many patients to be prescribed treatment at the initial visit and could open the door to telehealth opportunities, further decreasing TTTI.
Swift initiation of treatment with DAAs is crucial to HCV care. While progression of HCV to cirrhosis or hepatocellular carcinoma may take 20 to 40 years, the time to development of extrahepatic manifestations from HCV is less defined. 28 Prompt HCV treatment therefore offers numerous benefits, including a decrease in extrahepatic morbidity. Our study predominantly included patients with low baseline fibrosis—an ideal population for primary care providers and pharmacists to treat, potentially preventing the need for high-cost specialist care by averting the development of complications.
Rapid treatment initiation also serves as a public health measure because of the high transmission rates of HCV, particularly among PWID. Our study population had a high prevalence of substance use disorder. Treating these patients presents unique challenges, because they often face barriers to care and require interdisciplinary teams. Notably, their transient nature necessitates prompt treatment to prevent loss to follow-up within the HCV care cascade. 29 Our data indicate that pharmacists were sometimes unable to establish ongoing care with patients after the initial referral, resulting in missed opportunities for treatment. In addition, a number of patients in our study did not return for laboratory work, highlighting the difficulties faced by providers in clinical settings that provide care for medically underserved populations when they have limited opportunities to engage with patients. These findings underscore the importance of prescribing HCV treatment while patients are actively engaged in care. Same-day, point-of-care test-and-treat models would be a potential solution to mitigate loss to follow-up, particularly in a transient patient population. Treatment of individuals with a history of substance use disorder does present a possibility of reinfection. However, patients with substance use disorders should still be offered treatment because treatment decreases the prevalence of HCV.30,31
Achieving SVR12 is the primary goal of HCV antiviral therapy. Manufacturer-funded studies using DAAs report SVR12 rates as high as 98%.32-35 However, in practice, DAA effectiveness may be lower because of greater patient diversity and decreased adherence. Real-world SVR12 rates with DAAs are as high as 95%. 36 An analysis of a US Department of Veterans Affairs population reported SVR12 rates of 90% to 94% depending on genotype. 37 Our study represents a medically underserved population in the United States with a large proportion of Medicaid enrollees and a high prevalence of substance use disorder. Our per-protocol SVR12 rate of 91.5% is in line with real-world studies. We did not collect data on reasons for treatment failure. The transient nature of the population studied may have led to some patients not returning for posttreatment testing.
Our study highlights the effect that lack of health insurance has on TTTI in patients with HCV. 38 Uninsured patients had longer delays in initiating treatment than those with health insurance, mainly because of delays in receiving DAA therapy from the manufacturers’ patient assistance programs. As of August 2024, the White House proposed a national HCV elimination plan in its 2024-2034 budget. 39 For this initiative to be effective, budget allocations should target the uninsured population. Furthermore, revisions to the manufacturers’ patient assistance programs are needed to enhance accessibility to DAA therapy for uninsured individuals.
Prior authorizations present another barrier to prompt HCV treatment. Some state-funded health insurers have removed prior authorization requirements completely, allowing for rapid initiation after prescribing. 40 Prior authorization requirements should be removed by all payors for HCV medications, because it will decrease TTTI and increase treatment initiation. In the current practice environment, pharmacists are well positioned to navigate the prior authorization process. Pharmacists have clinical training to submit prior authorizations with minimal denials and can quickly determine prior authorization status via pharmacy dispensing software. This methodology minimizes the administrative time required to process and determine prior authorization status.
Strengths and Limitations
Our study had several strengths. First, we addressed a gap in the literature of the effect of pharmacist-led HCV treatment models on TTTI. While previous research demonstrated the effectiveness of these models in increasing treatment initiation and completion rates, our study examined how implementing these models in primary care can shorten TTTI—a key factor for improving patient outcomes and reducing disease transmission. Second, our study provides real-world evidence of the effectiveness and feasibility of pharmacist-led HCV treatment models. Third, the inclusion of patients at high risk of contracting HCV; medically underserved populations, such as people with substance use disorder; and people with barriers to accessing HCV treatment, such as uninsured patients, demonstrates the feasibility and effect of pharmacist-led treatment models in addressing the health care disparities encountered by these groups. Given the ongoing shortage of specialists, this model can be a sustainable solution for expanding HCV care and improving access to HCV treatment in resource-limited environments.
Our study also had several limitations. First, the retrospective design limited control of potential confounders, and the study was geographically restricted to Ohio, which may have limited the broader applicability of our results. Health insurer treatment criteria differ by state, potentially affecting treatment initiation timelines across regions. Guideline and insurer treatment criteria have broadened since data on TTTI were previously reported in Kwo et al. 22 This change potentially increased access to HCV treatment, which could have overstated the effect of the pharmacist-led HCV treatment model. Ohio has removed some restrictions to DAA coverage but during the study period received a grade of “C” from the Hepatitis C: State of Medicaid Access reports, indicating that barriers to DAA coverage persist. 40 Finally, we did not collect data on specific DAA regimens or reasons for treatment failure, and the lack of a direct comparator group may have influenced interpretation of the results.
Conclusion
This study provides insight into the HCV care cascade in a medically underserved population with a high prevalence of substance use disorder. It also highlights the effect of the integration of pharmacists into hepatitis care teams on multiple outcomes. While our average TTTI of 63.7 days represents an improvement from previously described national trends, more interventions are needed for more rapid and widespread HCV care. Same-day point-of-care test-and-treat models without the need for prior authorization are an ideal goal, aligning with the care models used for HIV. Continued removal of state-based Medicaid restrictions on DAA use are also important to decrease waiting periods to start treatment. Finally, a pressing need exists for more nonspecialists to screen and treat patients with HCV, as current transmission rates exceed cure rates. The combination of these public health interventions has the potential to result in rapid TTTI, and they should be implemented to progress toward elimination of HCV. Pharmacists play an integral role in this endeavor, and their role should continue to be expanded in outpatient settings as members of the HCV care team.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
