Abstract
Objective
Curative treatments for hepatitis C virus (HCV) infection are available, but access and barriers to treatment can delay initiation. We investigated the time to first negative RNA test result among people with HCV infection and examined differences by homeless status and whether people were tested at a correctional facility or substance use treatment facility.
Methods
We used surveillance data to identify New York City residents first reported with HCV infection during January 1, 2015–December 31, 2018, with ≥1 positive RNA test result during January 1, 2015–November 1, 2019. We used Kaplan–Meier survival analysis to determine the time from the first positive RNA test result to the first negative RNA test result, with right-censoring at date of death or November 1, 2019. We determined substance use treatment, incarceration, or homelessness by ordering facility name and address or from patient residential address.
Results
Of 13 952 people with an HCV RNA–positive test result first reported during 2015-2018, 6947 (49.8%) subsequently received an RNA-negative test result. Overall, 25% received an RNA-negative test result within 208 (95% CI, 200-216) days and 50% within 902 (95% CI, 841-966) days. Homelessness, incarceration, or substance use treatment was indicated for 4304 (30.9%) people, among whom 25% received an RNA-negative test result within 469 (95% CI, 427-520) days and <50% received an RNA-negative test result during the study period.
Conclusions
Efforts to connect people to treatment should occur soon after diagnosis, especially for people who could benefit from hepatitis C care coordination.
As of 2015, an estimated 116 000 (1.4%) New York City residents were living with chronic hepatitis C virus (HCV) infection, based on surveillance reports collected by the New York City Department of Health and Mental Hygiene (DOHMH). 1 If left untreated, chronic HCV infection can cause liver damage, liver cancer, and death. 2 However, starting in late 2013, new treatments for HCV infection became available, with cure rates >90% for almost all people after 8-12 weeks of therapy. 3 Initiating HCV treatment decreases the risk of developing or exacerbating health conditions and can reduce transmission to uninfected people. 4,5 These direct-acting antivirals (DAAs) make HCV elimination a viable option. However, since the release of DAAs, access to such treatment has not been uniform for all people with HCV infection. Health insurance–related restrictions, instituted because of the high cost of the medications, have included restricting medication to people with advanced liver disease, requiring abstinence from drugs and alcohol, allowing only health care providers in particular specialties to prescribe medications, requiring health care provider training before they are allowed to prescribe DAAs, and requiring prior authorization before medications can be provided. 6,7
These barriers can delay HCV treatment initiation 8 ; however, few studies have examined the timing of treatment initiation with DAAs after diagnosis on a population level. 9 In addition, barriers to treatment initiation can disproportionately affect populations that have high rates of HCV infection. Drug use, especially injection drug use, is a primary route of transmission for HCV, and people with a history of injection drug use have rates of HCV infection as high as 60%, usually dependent on the length of injection history. 10 -12 People who use drugs can face difficulties in accessing HCV treatment, from a lack of health insurance coverage, health insurance restrictions because of drug use, or health care provider reluctance to treat because of drug use, 6,13,14 despite evidence that treatment is as effective in these populations as in other groups. 15,16
Likewise, people who are or have been incarcerated also have high rates of HCV infection: detainees of New York City jails had a seropositivity rate of 20.6% in 2014, 17 and overall estimates of HCV prevalence among people who are incarcerated ranged from 10% to 40%. 12,17 -20 People who are incarcerated also have barriers to treatment access, both while incarcerated and after release; the high cost of medications has led some states and jurisdictions to limit treatment access for people who are incarcerated, although strategies to pay for HCV medications and ensure access to HCV medications are being developed. 19,21,22 In New York City, efforts to pay for HCV medications to treat patients in jails, despite challenges with variable lengths of stay, are underway. 23,24 However, upon release, barriers to linkage to and retention in health care services may remain. 19,25 -27
People who are experiencing or have experienced homelessness also have high rates of HCV infection; estimates range from 8% to 50%, 12,20,28 -30 often connected to a concurrent history of drug use and/or incarceration. 29 -31 In 2018, almost 60 000 people were in the New York City shelter system on a given day, and at least 3675 people were unsheltered. 32,33 People experiencing homelessness face many barriers to HCV care, including lack of health insurance or access to no-cost care, competing health and social priorities, difficulty remaining in care, and having a secure place to store medications while on treatment. 34
To assess the dynamics of HCV treatment access in New York City and how potential barriers can affect access to treatment, we evaluated the time to first RNA-negative test result, calculated as the time between specimen collection dates for a person’s first reported positive RNA test result and first reported negative RNA test result, among residents in New York City reported with a positive or negative RNA test result from 2015 through 2018. We measured the time to first RNA-negative test result overall and by year of first report, as well as separately among people for whom a history of incarceration, homelessness, and/or substance use treatment could be identified in surveillance data.
Methods
DOHMH Hepatitis C Surveillance System
DOHMH has had electronic laboratory reporting since 2006, and it is considered complete and comprehensive. 35 All New York City residents with a positive HCV test result (antibody, RNA, and genotype) are reportable to DOHMH. Beginning in 2014, negative RNA test results were reportable to DOHMH. 36 HCV test results are de-duplicated at the person level, such that each person with HCV infection has a complete history of reportable test results in the DOHMH surveillance system. De-duplication is automatic, but daily manual review is conducted for ambiguous matches. Information provided on laboratory reports includes patient name, sex, date of birth, residential address, ordering health care facility name and address, and ordering health care provider name and address. Demographic information, such as race/ethnicity and gender identity, and risk factor information (eg, injection drug use) are not typically available. Annual matching is conducted with the New York City Office of Vital Statistics death certificates to determine vital status for all people ever reported with a positive HCV test result.
Cohort Selection
We selected for analysis New York City residents with HCV infection (positive antibody or RNA test result) who were first reported to DOHMH during January 1, 2015–December 31, 2018, with at least 1 positive RNA test result during January 1, 2015–November 1, 2019. We excluded people if they had a negative RNA test result reported before or on the same day as their first positive RNA test result. We included people who were reported during 2015-2016 to allow for enough follow-up time for patients to receive a negative RNA test result within 6 months, 1 year, and 3 years. In addition, DAA medications became widely available in 2014, and hepatitis C treatment restrictions in New York State have diminished markedly since 2014. We hypothesized that people who were diagnosed earlier during the analytic period would take longer to receive an RNA-negative test result, and we assessed what proportion received an RNA-negative test result within 3 years. We included a later group (reported during 2017-2018) to determine if the time to RNA-negative test result decreased for this group. Fewer people were diagnosed with HCV infection during 2017-2018 than during 2015-2016, but access to treatment increased and the cost of medications decreased; as such, we anticipated that the time to an RNA-negative test result would shorten for this group.
Ascertainment of Risk History
Given the large number of people newly reported with chronic HCV infection in New York City each year (range, 4682 in 2018 to 7513 in 2014), 37 most reported cases are not investigated to determine characteristics that would put a person at increased risk of infection or that could increase barriers to health care and treatment access. However, some information can be inferred from the information provided in laboratory reports. Using the ordering health care facility name and address and ordering health care provider address from all laboratory tests, and all reported patient residential addresses, we identified known New York City–area jails, substance use treatment centers, and homeless shelters/supportive housing sites and used this information to infer whether people had a history of homelessness, or if they were tested at a correctional facility or substance use treatment facility. We also identified people as having experienced homelessness if a residential address was reported with variations of “homeless,” “undomiciled,” or “transient.”
Time to First RNA-Negative Test Result
We calculated the number of days from the blood draw date of the first positive RNA test result to the first negative RNA test result. For all people in the cohort, we calculated follow-up time from the date of the first positive RNA test result with right-censoring to the earliest of the date of the first negative RNA test result, date of death, or November 1, 2019. Using Kaplan–Meier survival analysis, we calculated the time to first RNA-negative test result for the cohort, by year of first report (2015-2018), and we compared people who did or did not have a known history of homelessness and/or testing at a correctional facility or substance use treatment facility. Using this approach, we assessed the probability of having an RNA-negative test result at 6 months, 12 months, and 3 years after the first RNA-positive test result and the time needed for 25% and 50% of each group to have an RNA-negative test result. We used the Peto–Peto–Prentice test to compare survival functions across groups after observing that the proportional hazard assumption failed and the survival curves crossed. We used a Cox proportional hazard model to test for significant differences in RNA-negative status after adjusting for sex and age at the time of diagnosis. We considered P < .05 to be significant. The model included an interaction term for sex and homelessness or testing at a correctional facility or substance use treatment facility. We performed all analyses using SAS version 9.4 (SAS Institute, Inc). The New York City DOHMH Institutional Review Board considered this analysis not to be human subjects research.
Results
Descriptive Statistics of the Cohort
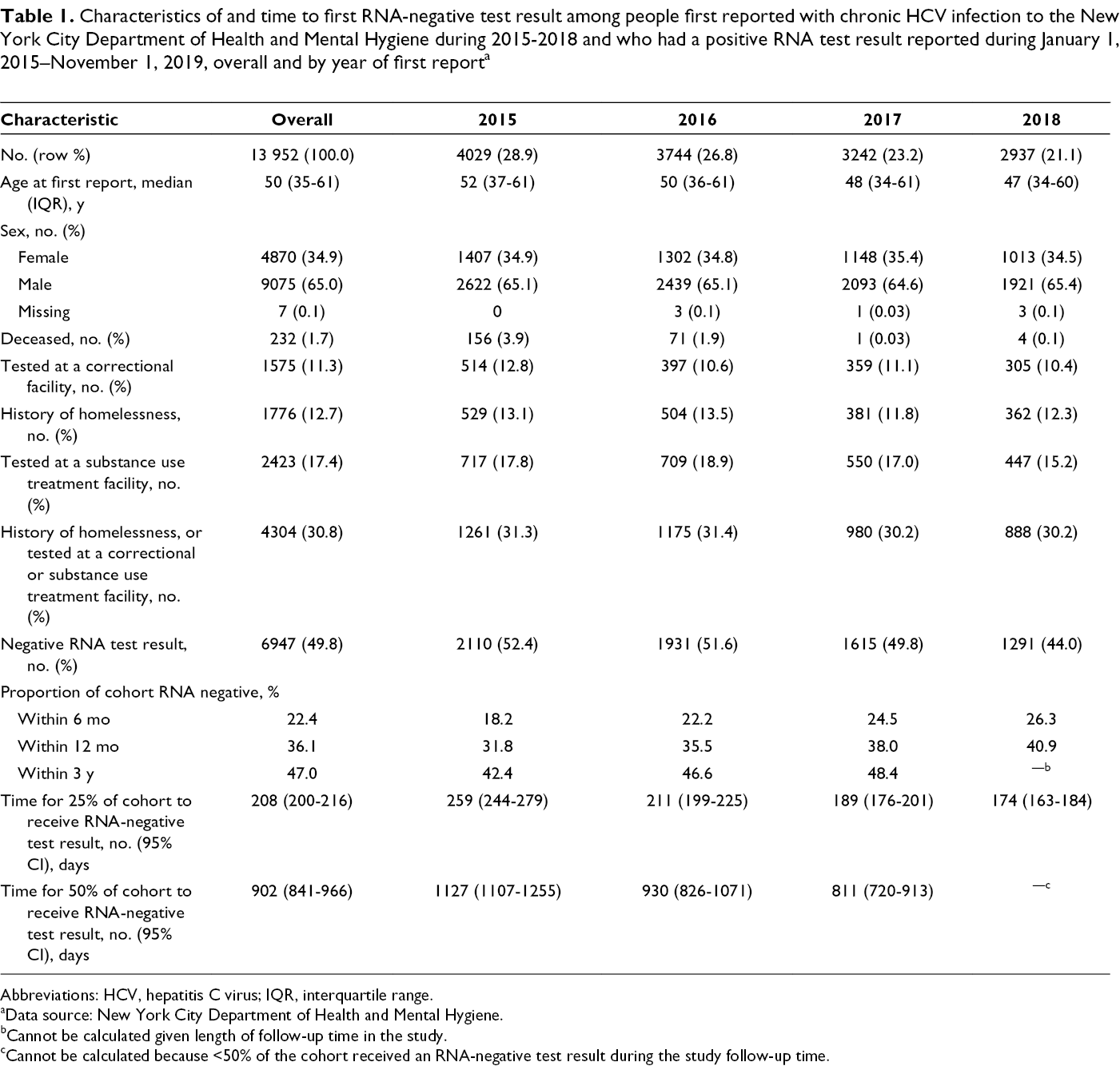
We identified 13 952 people with HCV infection during 2015-2018 with a positive RNA test performed during January 1, 2015–November 1, 2019. The median age at first report was 50 (interquartile range: 35-61), and 65.0% were male (Table 1). The number of people reported each year ranged from 4029 in 2015 to 2937 in 2018. Overall, 1575 (11.3%) people were tested at a correctional facility, 1776 (12.7%) had a history of homelessness, and 2423 (17.4%) were tested at a substance use treatment facility; these proportions did not vary substantially by year of first report. A small proportion of people (1.5%, n = 214) had all 3 risk factors (history of testing at a correctional facility, history of homelessness, and testing at a substance use treatment facility), and 30.9% (n = 4304) had a history of at least 1 of these risk factors. Half of the cohort (n = 6947, 49.8%) had a negative RNA test result after having a positive RNA test result, ranging from 52.4% in 2015 to 44.0% in 2018.
Characteristics of and time to first RNA-negative test result among people first reported with chronic HCV infection to the New York City Department of Health and Mental Hygiene during 2015-2018 and who had a positive RNA test result reported during January 1, 2015–November 1, 2019, overall and by year of first report a
Abbreviations: HCV, hepatitis C virus; IQR, interquartile range.
aData source: New York City Department of Health and Mental Hygiene.
bCannot be calculated given length of follow-up time in the study.
cCannot be calculated because <50% of the cohort received an RNA-negative test result during the study follow-up time.
Time to RNA-Negative Test Result
When we used a Kaplan–Meier approach, we found that 22.4% of people received an RNA-negative test result within 6 months, 36.1% within 12 months, and 47.0% within 3 years (Table 1; Figure 1A). The length of follow-up was shorter for people with later years of first report than for people with earlier years of first report, but we found steady increases in the percentage of people who received an RNA-negative test result within 6 months and 12 months with later years of first report; 18.2% of people first reported with a positive RNA test result in 2015 had a negative RNA test result within 6 months and 31.8% within 12 months compared with 26.3% and 40.9%, respectively, of people first reported with a positive HCV test result in 2018 (Figure 1B).
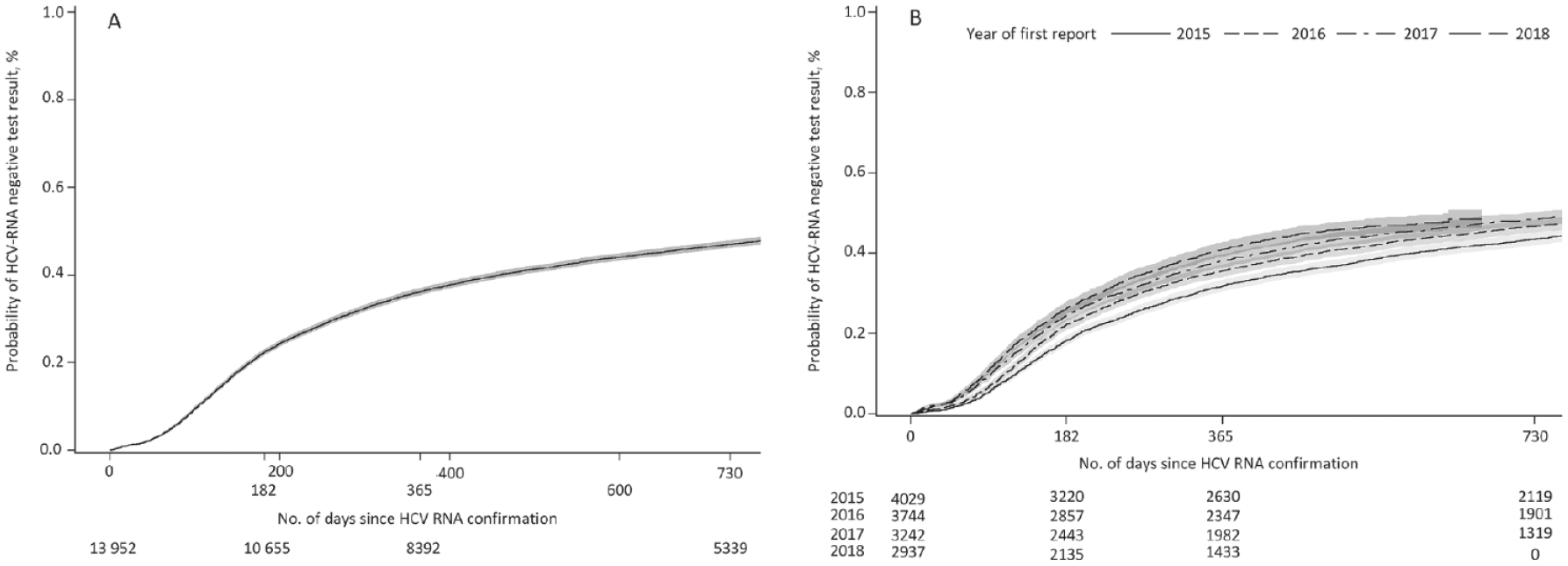
Time to first RNA-negative test result from RNA confirmation of hepatitis C virus (HCV) infection in New York City, with counts of the number of people remaining RNA positive (A) overall and (
Twenty-five percent of people in the cohort had a negative RNA test result within 208 (95% CI, 200-216) days, or approximately 7 months, after their first positive RNA test result. Fifty percent of people in the cohort had an RNA-negative test result within 902 (95% CI, 841-966) days, or approximately 2.5 years, after their first positive RNA test result (Table 1; Figure 1A). When examining the proportion of people who had a negative RNA test result by year of first report, a similar pattern held, although the time to RNA-negative test result for 25% and 50% of the cohort decreased with increasing year of first report, and the incremental time required for 25% and 50% of the cohort to receive an RNA-negative test result also decreased with increasing year of first report (Table 1; Figure 1B).
Time to RNA-Negative Test Result by Risk History
Among people with a known history of homelessness, or who were tested at a correctional facility or substance use treatment facility, 11.8% had an RNA-negative test result within 6 months compared with 27.2% of people with no known history of any of these factors; after 3 years, the percentage of people with a known history of risk factors who had an RNA-negative test result was 32.1% and 53.7%, respectively (Table 2; Figure 2A). Twenty-five percent of people with a known history of homelessness or who were tested at a correctional facility or substance use treatment facility initiated treatment within 469 (95% CI, 427-520) days, or approximately 1.3 years, after their first positive RNA test result, compared with 170 (95% CI, 164-175) days, or approximately 6 months, for people with no known history of these risk factors (P < .001). When we examined each factor separately, only 7.3% of people tested at a correctional facility, 11.9% of people with a history of homelessness, and 14.3% of people tested at a substance use treatment facility had an RNA-negative test result after 6 months, all of which were substantially lower percentages than the percentage of people without a history of each risk factor who had an RNA-negative test result (Table 2; Figure 2B–D). The time required for 25% of the cohort to receive a negative RNA test result among people with a history of homelessness or testing at a correctional facility or substance use treatment facility was 748 (95% CI, 676-847) days (2.0 years), 403 (95% CI, 361-464) days (1.1 years), and 356 (95% CI, 320-380) days (1.0 years), respectively, which was significantly (P < .001) longer than among people who did not have a history of these risk factors. When also examined by year of first report, patterns in time to first RNA-negative test result for people with a history of 1 of these risk factors were similar to stratifying by year of first report among the overall cohort.
Time to first RNA-negative test result among people first reported with chronic HCV infection to the New York City Department of Health and Mental Hygiene during 2015-2018 and who had a positive RNA test result reported during January 1, 2015–November 1, 2019 (N = 13 952), among people with and without a known history of homelessness, and/or tested at a correctional facility or substance use treatment facility (N = 13 952) a
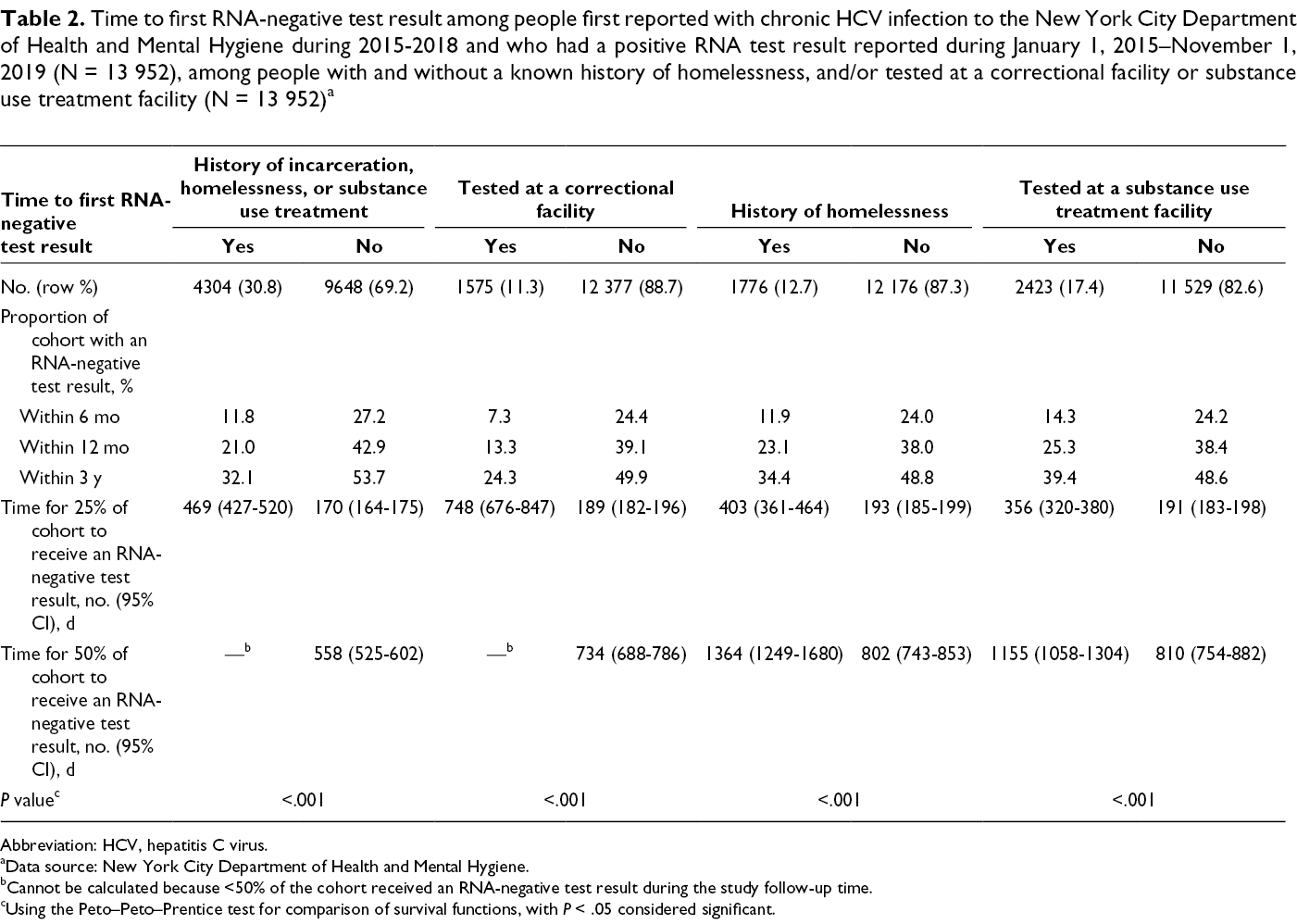
Abbreviation: HCV, hepatitis C virus.
aData source: New York City Department of Health and Mental Hygiene.
bCannot be calculated because <50% of the cohort received an RNA-negative test result during the study follow-up time.
cUsing the Peto–Peto–Prentice test for comparison of survival functions, with P < .05 considered significant.
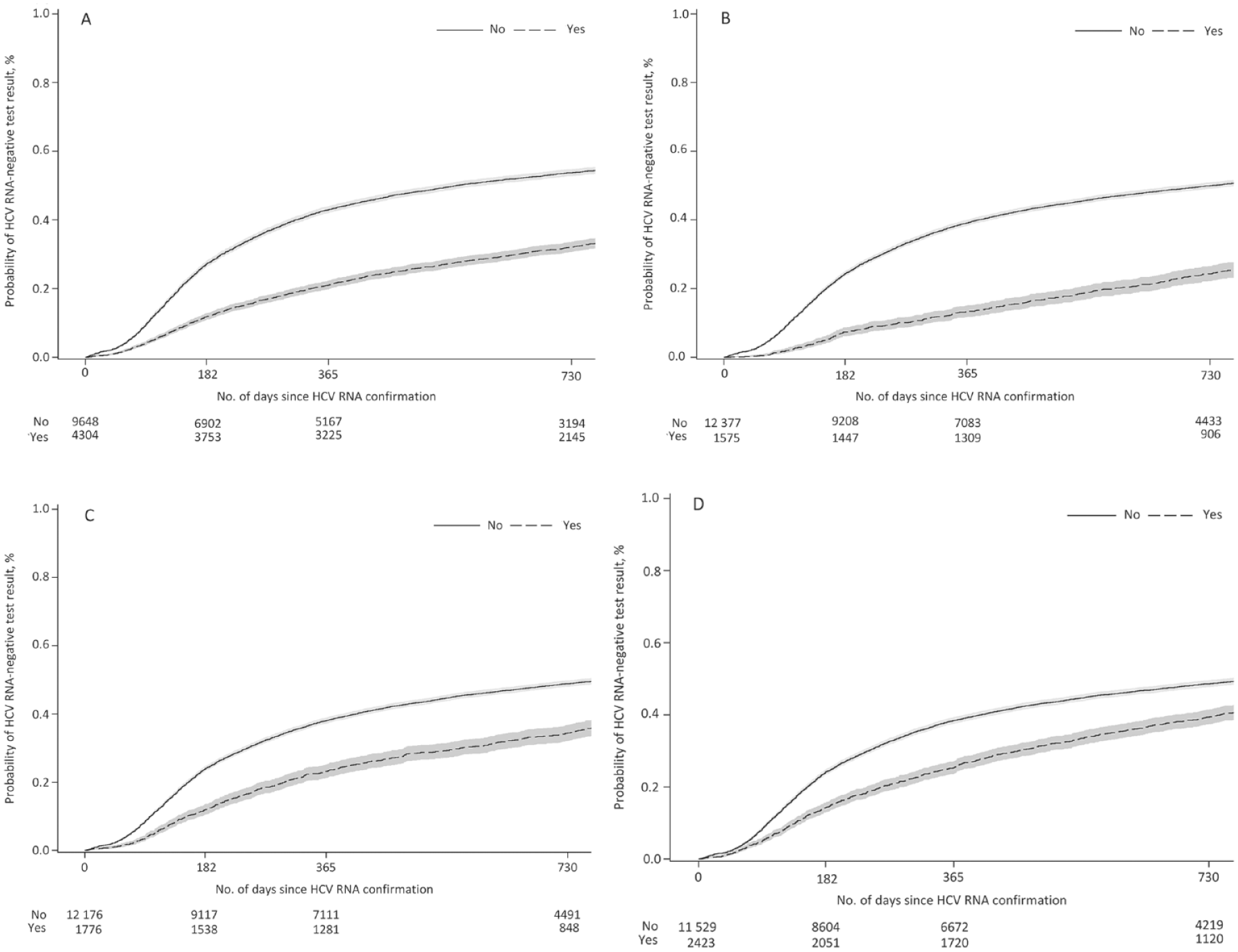
Time to RNA-negative test result from RNA confirmation of hepatitis C virus (HCV) infection in New York City, with counts of the number of people remaining RNA positive, among people reported to the New York City Department of Health and Mental Hygiene, 2015-2018, who (
The time to first RNA test result was still longer for people with a history of homelessness or testing at a correctional facility or substance use treatment facility after adjusting for age and sex. The adjusted hazard ratio (aHR) among females who tested at a correctional facility was 2.75 (95% CI, 2.19-3.44) and 2.05 (95% CI, 1.85-2.27) for males. The aHR was lower among people with a history of homelessness (females: aHR = 1.70 [95% CI, 1.43-2.01]; males: aHR = 1.33 [95% CI, 1.22-1.40] or who tested at a substance use treatment facility (females: aHR = 1.59 [95% CI, 1.40-1.81]; males: aHR = 1.11 [95% CI, 1.03-1.20]) than among people without any known risk factors.
Discussion
For about 20% of our cohort, the time to an RNA-negative test result occurred within 6 months of an RNA-positive test result. This percentage is similar to rates of spontaneous clearance, 38 which occurs within 6 months of becoming infected with HCV; however, most people are not tested so soon after infection. As such, having an RNA-negative test result within 6 months of an RNA-positive test result likely represents how quickly people can access care and initiate treatment or clear infection naturally. If treatment information were reportable, we could accurately classify treatment initiation versus natural clearance among people who receive an RNA-negative test result within 6 months of report and among the general analytic population. The first 25% of the cohort received an RNA-negative test result within approximately 7 months, but almost 2 years passed before the next 25% of the cohort received an RNA-negative test result. These findings suggest that it is increasingly difficult to connect people to appropriate care and treatment and that delays in treatment initiation can increase the chance that someone is lost to follow-up and never treated. Some proportion of people with HCV infection are likely able to initiate treatment relatively quickly because of a combination of advantages in resources, life situations, health insurance, and health care providers who facilitate access to treatment. Efforts should be focused on people with a history of homelessness or on improving access to patient care navigators, substance use treatment, and housing support for people tested at correctional facilities and substance use treatment facilities. 39 -42
When facilitating timely treatment initiation, it is important to consider the administrative barriers to treatment access instituted by health insurance companies. We found that the time to first RNA-negative test result steadily decreased by year of first report; the time required for 25% of the cohort to have an RNA-negative test result was about 8.5 months among people first reported in 2015, approximately 7 months among people reported in 2016, and <6 months among people first reported in 2018. The proportion of the population that had an RNA-negative test result within 6 months of first report similarly increased each year. During this time, health insurance restrictions were lessened in New York State. In 2016, most public and private health insurers in the state removed treatment restrictions based on liver disease stage or drug or alcohol use. 7,43 In addition, New York State Medicaid removed health care provider training and specialty requirements in August 2018. 44 The lessening of these restrictions likely contributed to the improved access to and timeliness of having an RNA-negative test result seen in our analysis. 45
Despite some progress, access to care and treatment is still not equitable. People tested at a correctional facility had the longest times to an RNA-negative test result, likely related to a short length of stay at the correctional facility, making them ineligible for treatment in jail and leading to difficulties in maintaining continuous and coordinated care when interacting with the criminal justice system. 19 In addition, some incarcerated people were sentenced to prison, and their negative RNA test results were not reported to NYC DOHMH. Efforts are being made locally, nationally, and internationally to increase access to hepatitis C treatment among people who are incarcerated. 46 -49 Although people with a history of homelessness or who tested at a correctional facility or substance use treatment facility had similar relative improvements in timeliness to first RNA-negative test result to what we observed overall, additional efforts are still required to ensure that more people in these groups are able to access care and initiate treatment with few delays. 42,50 On-site treatment or telemedicine at substance use treatment facilities is one such approach, 51 -54 as is providing HCV treatment at homeless shelters that offer medical care. 55
Limitations
This analysis had several limitations. First, when assessing time to first RNA-negative test result by year of first report, the length of follow-up time differed for people first reported early in the study period (eg, 2015) and those reported later in the study period (eg, 2018), which limited our ability to assess the percentage of people with an RNA-negative test result within 3 years for each group. Second, treatment information is not reportable to NYC DOHMH; therefore, we could not differentiate between treatment initiation and natural clearance, which is important for people who received an RNA-negative test result within 6 months of diagnosis. Third, patient-level data are lacking because our surveillance data rely on laboratory reports, which have limited information on demographic characteristics, risk factors, and treatment. Enhanced surveillance investigations to obtain this information were not routinely conducted during this period. Information on risk history is likely underascertained in this cohort. Fourth, history of homelessness could be identified only using information on testing facility/health care provider and patient addresses, meaning many people could have experienced homelessness without it being noted in these data fields. Finally, deaths were unknown if they occurred outside New York City, and outmigration from New York City is not accounted for in the survival analysis. Death certificate matching for people first reported in 2018 was incomplete at the time of analysis, potentially leading to some missed dates of death.
Conclusion
This analysis provides novel insights into timing of an RNA-negative test result at the population level. Our observation that the proportion of the population with an RNA-negative test result decreased after receiving an RNA-positive test result suggests that efforts to connect people to treatment should occur soon after diagnosis, especially for people who could benefit from hepatitis C care coordination. To eliminate HCV infection, hepatitis C navigation should be provided to people who use drugs, are incarcerated, have a history of experiencing homelessness, and/or have delayed treatment and linkage to care.
Footnotes
Acknowledgments
The authors acknowledge the Bureau of Communicable Disease’s Reportable Disease and Data Analysis Unit, especially Eric Peterson for his analytic work on classifying homelessness; the Viral Hepatitis Program for assistance with data processing and cleaning; and Joseph Kennedy from the Office of Vital Statistics for matching New York City death certificates to determine vital status for people reported with hepatitis C virus.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a New York City tax levy.
