Abstract
Objectives:
Military training centers and seagoing vessels are often environments at high risk for the spread of COVID-19 and other contagious diseases, because military trainees and personnel arrive after traveling from many parts of the country and live in congregate settings. We examined whether levels of SARS-CoV-2 genetic material in wastewater correlated with SARS-CoV-2 infections among military personnel living in communal barracks and vessels at US Coast Guard training centers in the United States.
Methods:
The Coast Guard developed and established 3 laboratories with wastewater testing capability at Coast Guard training centers from March 2021 through August 2022. We analyzed wastewater from barracks housing trainees and from 4 Coast Guard vessels for the presence of SARS-CoV-2 genes N and E and quantified the results relative to levels of a fecal indicator virus, pepper mild mottle virus. We compared quantified data with the timing of medically diagnosed COVID-19 infection among (1) military personnel who had presented with symptoms or had been discovered through contact tracing and had medical tests and (2) military personnel who had been discovered through routine surveillance by positive SARS-CoV-2 antigen or polymerase chain reaction test results.
Results:
Levels of viral genes in wastewater at Coast Guard locations were best correlated with diagnosed COVID-19 cases when wastewater testing was performed twice weekly with passive samplers deployed for the entire week; such testing detected ≥1 COVID-19 case 69.8% of the time and ≥3 cases 88.3% of the time. Wastewater assessment in vessels did not continue because of logistical constraints.
Conclusion:
Wastewater testing is an effective tool for measuring the presence and patterns of SARS-CoV-2 infections among military populations. Success with wastewater testing for SARS-CoV-2 infections suggests that other diseases may be assessed with similar approaches.
Keywords
Military training commands face several factors that contribute to a high risk for the spread of COVID-19: people arrive from various US regions and from around the globe and are housed in barracks that hold as many as 100 people in a single large room. Individuals then undergo training, which includes strenuous physical exertion in close proximity to others. Mitigation measures, including remote work, are not possible without degradation of the training. Furthermore, trainees are typically younger than trainers and support staff and are more likely to have asymptomatic COVID-19 and less risk of developing severe COVID-19. 1 Seagoing vessels provide a similarly dense living environment that increases the risk for outbreaks, and they have limited isolation space once an outbreak occurs; an outbreak on France’s aircraft carrier Charles de Gaulle in early 2020 resulted in 668 sailors with SARS-CoV-2 infection, 29 of whom required oxygen therapy. 2
Several US Coast Guard training centers initiated enhanced weekly surveillance of COVID-19 by polymerase chain reaction (PCR)–based nasopharyngeal testing beginning in August 2020 (Gray JP, Hall GJ, and Page EJ, unpublished data, March 2021). With limited COVID-19 testing supplies and with COVID-19 vaccines not yet available, the goal was rapid identification and transmission mitigation of COVID-19 cases through isolation and contact tracing. Weekly PCR testing was randomly performed on 20% of trainees at on-site laboratories established for public health purposes—a labor-intensive and costly process.
SARS-CoV-2 is shed in fecal matter within the small intestine, providing a biomarker for infection.3 -5 A single case in a building can be detected by shedding in wastewater, and this information may be used to deploy testing to identify asymptomatic people with SARS-CoV-2 infection.6 -8 In March 2021, the Coast Guard established wastewater testing for SARS-CoV-2. Wastewater testing operated concurrently with the enhanced PCR surveillance to determine the potential efficacy and suitability of wastewater testing to replace the expensive and labor-intensive randomized individual PCR testing as a measure of SARS-CoV-2 infection in the population. In this study, we report the earliest, longest-running US military implementation of wastewater surveillance for SARS-CoV-2, which was conducted at 3 US Coast Guard training installations. We also discuss successes and challenges with wastewater surveillance on military seagoing vessels, specifically Coast Guard cutters.
Methods
Coast Guard Installations and Cohort of COVID-19 Cases
We named the 3 Coast Guard training centers A, B, and C. Training center A has a single large barracks that houses >1000 military trainees. Most trainees arrive in 2 cohorts of 500 and remain at the center from August through May. Training centers B and C have smaller barracks that are capable of housing 50 to 200 trainees who arrive sporadically throughout the year and stay for various periods for numerous training courses. Training center B mostly has rooms shared by 2 trainees, whereas training center C has large rooms shared by as many as 100 trainees. Trainees share classrooms, living spaces, and dining spaces at all 3 locations. By mid-June 2021, the Coast Guard clinic had provided COVID-19 vaccines to about 99% of trainees. At training center B, the Coast Guard Clinic had fully vaccinated 58.5% of trainees by day 1 and 79.5% by day 117. However, subsequent cohorts of trainees arriving had varying vaccination rates. At training center C, vaccination status varied throughout the testing period and did not have a consistent pattern.
For analysis of quantified data, we used COVID-19 cases that were diagnosed by on-base medical personnel among (1) trainees who had presented with symptoms or had been discovered through contact tracing and had medical tests and (2) trainees who had been discovered through routine surveillance by SARS-CoV-2 antigen or PCR testing (GeneXpert; Cepheid). The institutional review board of the US Coast Guard Academy (IORG0007428) concluded on August 19, 2020, that wastewater testing was public health surveillance and, therefore, not human subjects research and was exempt from further review in accordance with 45 CFR part 46.
Processing of Wastewater Samples and RNA Extraction
To collect wastewater samples, we connected a tampon (OB Ultra Absorbency; Edgewell Personal Care) to a line and placed it in the wastewater stream outside each barracks for 24 to 96 hours 9 at identified access points. We placed retrieved wastewater samples in sealed bags that were spiked with an RNA control (bovine respiratory syncytial virus [BRSV] vaccine; Inforce 3 [Zoetis Inc]) if samples were to be stored for >24 hours before analysis. To extract the sample, we cut a corner of the plastic bag, poured the liquid into a 50-mL conical centrifuge tube, and adjusted the volume to about 48 mL with distilled water. We then concentrated samples by using 600 mL of Nanotrap Microbiome A Particles (Ceres Nanosciences Inc). 10 We used the QIAmp Viral RNA Mini Kit (Qiagen) according to the manufacturer’s protocol to extract RNA. For seagoing vessels, Coast Guard personnel aboard the vessels obtained concentrated human waste sewage (blackwater) samples directly from the storage tank or while vessels pumped and disposed wastewater ashore. We diluted 5 mL of blackwater with distilled water to a volume of about 48 mL, spiked the samples with BRSV process control, and purified samples using Nanotrap Microbiome A Particles.
Assessment of Viral Genes by PCR
We used real-time quantitative reverse transcription (RT)–PCR to analyze RNA. We used the primers N1 and N2, designed by the Centers for Disease Control and Prevention, 11 to assess viral gene N, and we used a primer designed by the Germany Centre for Infection Research to assess the viral gene E. 12 Haramoto et al 13 at the University of Yamanashi designed the pepper mild mottle virus (PMMoV) primers and probe. The Veterinary and Agrochemical Research Center (Uccle, Belgium) designed the BRSV primers and probe. 14 We purchased primers and probes from Integrated DNA Technologies.
We performed 1-step real-time RT-PCR according to the manufacturer’s protocol (TaqPath 1-Step RT-qPCR Master Mix; Thermo Fisher Scientific). For the N1 primer set, we included BRSV or N2 in the multiplex RT-PCR assay and used a thermocycler (CFX96; Bio-Rad) with the following conditions: 50 °C for 10 minutes, 95 °C for 10 minutes, and 45 cycles at 95 °C for 10 seconds and at 60 °C for 30 seconds. For the E primer set, we included PMMoV in the multiplex RT-PCR assay and used the following conditions: 50 °C for 10 minutes, 95 °C for 10 minutes, and 44 cycles at 95 °C for 15 seconds and at 58 °C for 30 seconds. We determined values for the quantification cycle by setting the threshold value to 200 (arbitrary fluorescence units).
We used BRSV primers to validate sample stability in samples stored for >24 hours. During each validation run, we used a positive control for viral genes N and E. We compared expression levels of the viral genes with expression levels of PMMoV to account for sample dilution by extraneous wastewater sources. We used the delta-delta cycle threshold method with the assumption of 100% efficiency. 15 We analyzed each sample daily for the presence of 2 or 3 viral genes in singlicate.
Testing at Training Center A
We collected wastewater from 2 untreated wastewater collection points adjacent to training center A from March 9, 2021 (day 1), through August 15, 2022 (day 525). From late summer 2021 through day 428, to measure SARS-CoV-2 infection, Coast Guard medical personnel conducted weekly antigen tests for the entire training center A population and conducted clinical PCR tests for trainees with COVID-19 symptoms or used PCR tests to confirm positive antigen results in trainees during routine surveillance. Coast Guard personnel immediately isolated trainees diagnosed with COVID-19 in a different barracks outside the wastewater stream of the tested barracks from day 1 through day 475, which allowed us to confirm that diagnosed COVID-19 cases did not contribute to the raw wastewater. After day 475, military trainees diagnosed with COVID-19 continued to shed into the wastewater. This design provided a natural experiment whereby wastewater levels could be correlated with subsequently diagnosed cases of COVID-19.
Testing at Training Centers B and C
At training center B, we collected wastewater from June 21, 2021 (day 1), through September 14, 2022 (day 451). Each of the 3 barracks at training center B had 2 wastewater sampling points. We collected samples once weekly beginning on day 4 and twice weekly beginning on day 18. Trainees arrived and departed for training as staggered cohorts for multiple weeks, increasing the likelihood of individuals from throughout the United States bringing infections to the base. Unlike at training center A, Coast Guard medical personnel did not conduct routine weekly SARS-CoV-2 antigen testing of trainees at training center B. Instead, trainees received antigen testing when they arrived and at sporadic situation-specific intervals thereafter. At training center C, we collected wastewater from March 10 (day 1) through September 9, 2022 (day 183). Similar to training center B, trainees received routine SARS-CoV-2 antigen testing when they arrived at training center C. However, medical personnel did not conduct subsequent routine testing of asymptomatic trainees.
Testing on Seagoing Vessels
Sampling wastewater from seagoing vessels is more difficult than sampling from maintenance access points ashore: samples taken while the vessel is at sea must be stored until the vessel returns to port, and blackwater access points can be difficult to reach and are sometimes pressurized. Coast Guard personnel made modifications to 4 vessels (all Coast Guard cutters) to allow for wastewater sampling while at dock, which involved adding a valve that allowed sampling of blackwater as it was pumped from the bilge to dockside septic containers. We required multiple samples to be collected and mixed because storage tanks did not agitate the blackwater and they may have contained many days of accumulated wastewater.
Results
Training Center A
Initially, wastewater testing underrepresented diagnosed cases of COVID-19. After day 120, passive samplers were deployed for 48 to 72 hours; a new sampler was deployed upon retrieval of the previous one, with more frequent deployment of passive samplers when new cohorts arrived. From July through August 10, 2021, no cases of COVID-19 were clinically diagnosed, and no SARS-CoV-2 was detected in the wastewater. A new cohort of trainees arrived in mid-August 2021; thereafter, most trainees were tested weekly for COVID-19 with antigen tests. From mid-August through mid-October 2021, 9 cases of COVID-19 were clinically diagnosed. SARS-CoV-2 gene fragments were detected in wastewater on days 165, 172, 179, and 221 at 1 sampling site (Figure 1A). In each of these instances, a COVID-19 case was clinically diagnosed within 4 days before or after detection in wastewater. Before the winter holiday in December 2021, a sustained spike in wastewater levels corresponded with 6 diagnosed cases.
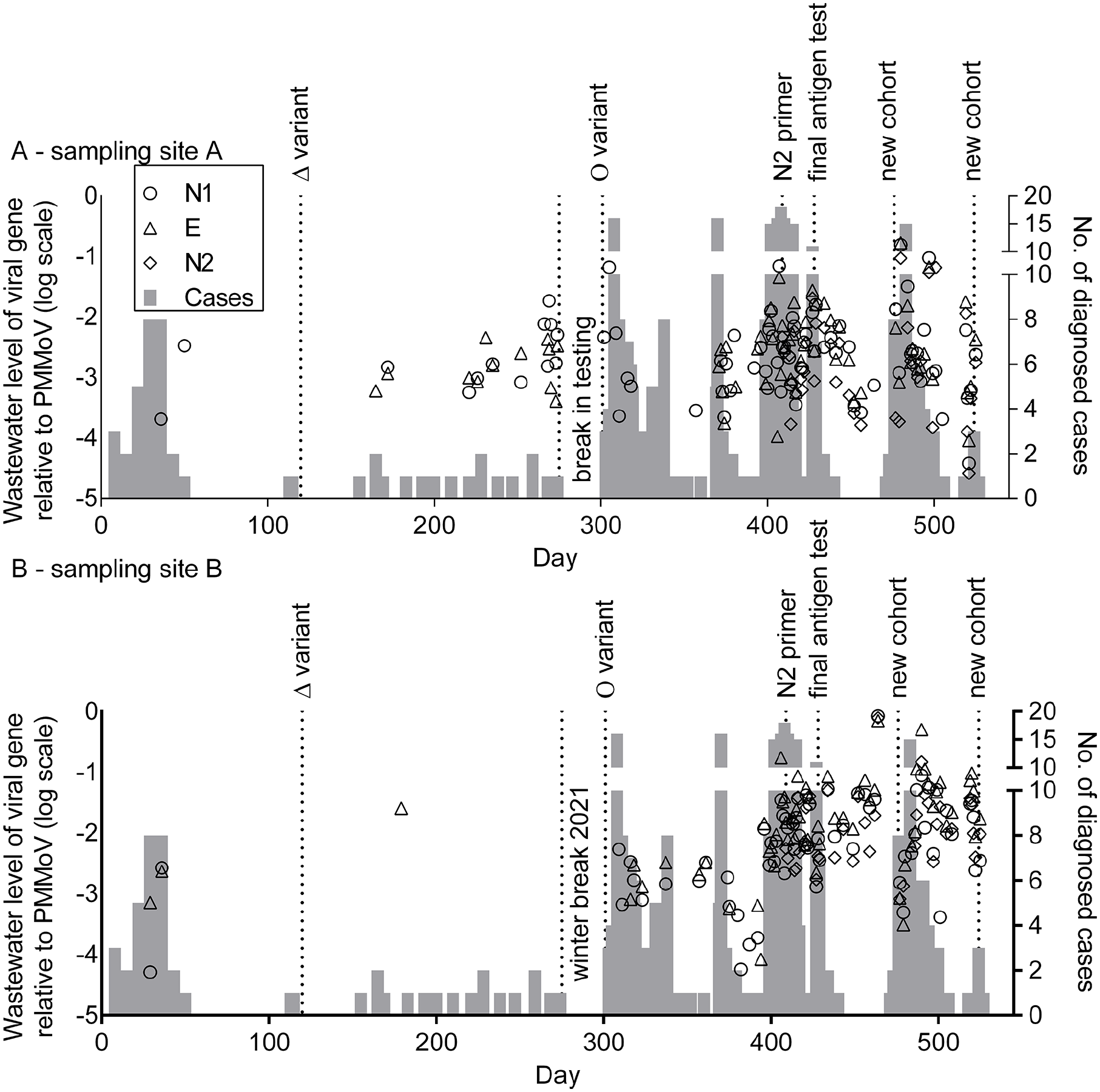
SARS-CoV-2 levels relative to a fecal biomarker from samples collected at 2 untreated wastewater collection points (A and B) and diagnosed COVID-19 cases among military trainees at US Coast Guard training center A, March 9, 2021 (day 1), through August 15, 2022 (day 525). Training center A consisted of a single barracks, and wastewater samples were collected from 2 distinct portions of the same building for 525 days. Symbols show the presence of SARS-CoV-2 RNA N and E as detected by primer pairs (N1, E, N2) in the wastewater relative to the human fecal matter indicator pepper mild mottle virus (PMMoV). Primers N1 and E were used throughout, and N2 primers were added on day 409. Cases indicate military trainees with clinically diagnosed COVID-19 for the entire barracks; diagnosed cases were not separated between the sampling locations because of freedom of choice of restroom facilities by military trainees in the barracks. Wastewater sampling was temporarily halted during winter break and near day 335 because of a weather-related event preventing wastewater access. Abbreviations: Δ, Delta; Ο, Omicron.
In early 2022, a surge in levels of viral genes in wastewater correlated with >20 diagnosed cases of COVID-19. During the remainder of 2022, the number of COVID-19 cases increased, consistent with the emergence of the Omicron variant. 16 A break in wastewater sampling occurred from day 328 through day 336 because of winter weather, during which time 15 people were diagnosed with COVID-19. Outside of this break, diagnosed cases of COVID-19 remained correlated with high levels of viral genes detected in wastewater (Figure 1). Weekly SARS-CoV-2 antigen tests of military personnel were halted on day 428. After day 428, viral genes were more frequently detected in the absence of diagnosed cases. Overall, wastewater detection of viral genes correlated with ≥1 diagnosed COVID-19 case 69.8% of the time, ≥2 diagnosed cases 78.3% of the time, and ≥3 diagnosed cases 88.3% of the time, peaking at 97.5% for ≥6 cases.
Training Centers B and C
Fewer diagnosed cases of COVID-19 were identified at training center B (Figure 2). Elevated viral levels in the wastewater suggested that asymptomatic cases of COVID-19 occurred in all 3 barracks. After viral genes became undetectable in wastewater at day 275, viral genes reemerged at about day 375, consistent with the increased pervasiveness of the Omicron variant in the United States. At training center C, near day 125, all trainees at 1 barracks were tested for COVID-19 after an exceptionally large wastewater spike; nearly 70% of trainees tested positive for COVID-19 by antigen testing (Figure 3C).
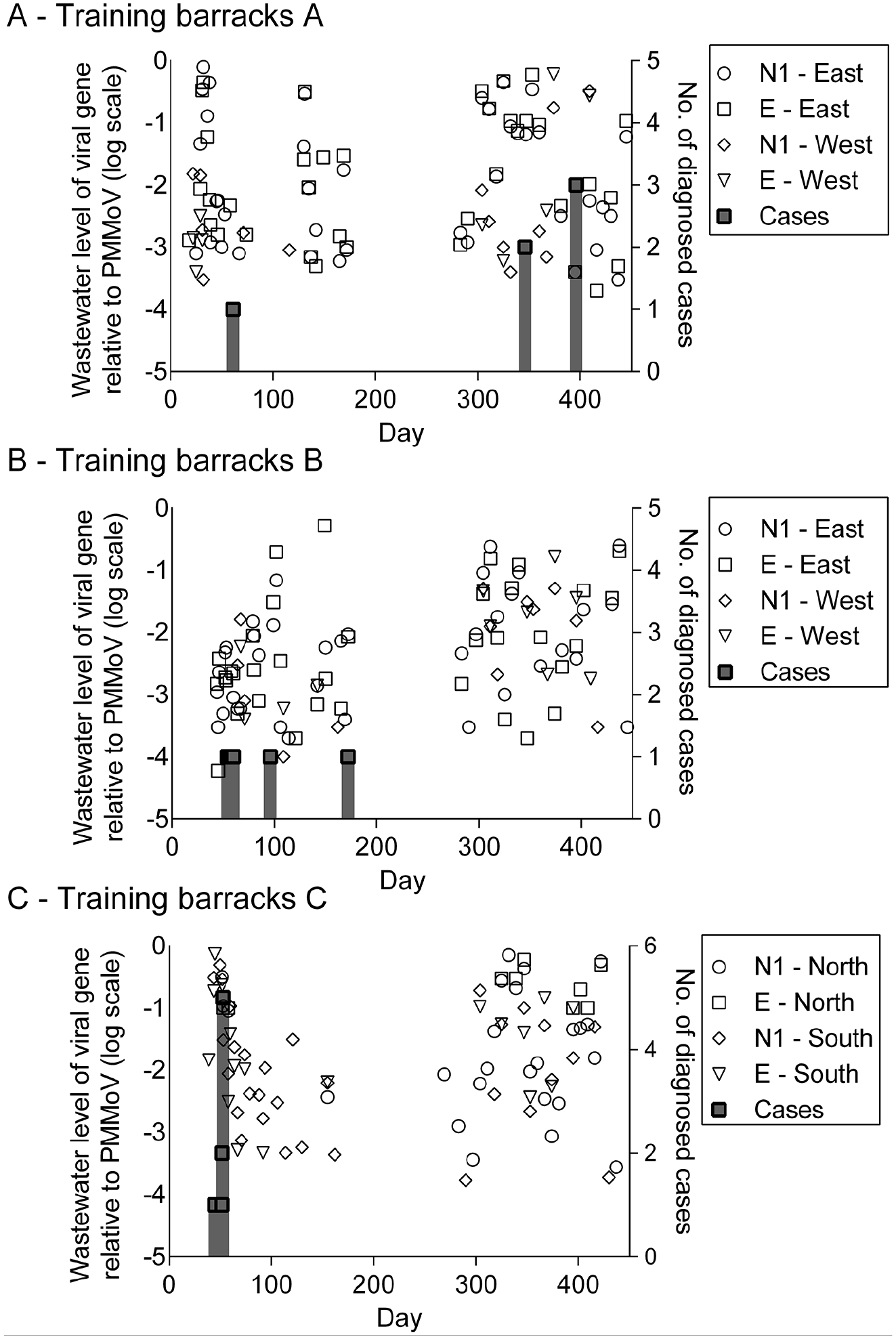
SARS-CoV-2 levels relative to a fecal biomarker from samples collected at 3 barracks (A-C) and diagnosed COVID-19 cases among military trainees at US Coast Guard training center B, June 21, 2021 (day 1), through September 14, 2022 (day 451). Each barracks had 2 sampling sites, East/West or North/South as indicated. Symbols show the presence of SARS-CoV-2 RNA detected by the indicated polymerase chain reaction primers (primer set N1, primer set E) in the wastewater relative to the human fecal matter indicator pepper mild mottle virus (PMMoV). Cases indicate people with clinically diagnosed COVID-19; such people were moved to an isolation building with a separate watershed. After initial SARS-CoV-2 antigen testing of trainees at the time of arrival, subsequent antigen tests were not routinely performed.
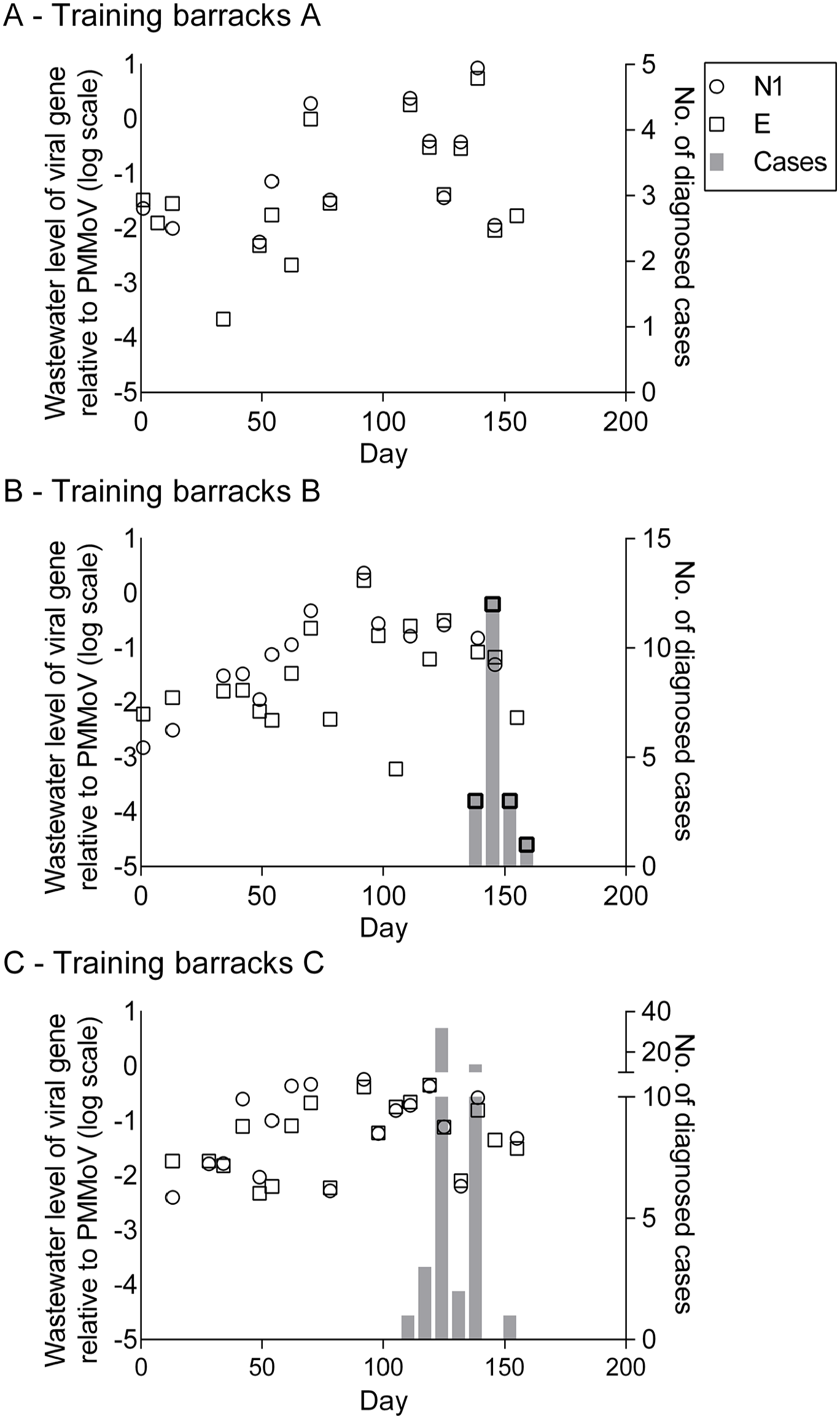
SARS-CoV-2 levels relative to a fecal biomarker from samples collected at 3 separate barracks (A-C) and diagnosed COVID-19 cases among military trainees at US Coast Guard training center C, March 10, 2022 (day 1), through September 9, 2022 (day 183). Each building had 1 sampling site. Symbols show the presence of SARS-CoV-2 RNA detected by the indicated polymerase chain reaction primers (N1, E) in the wastewater relative to the human fecal matter indicator pepper mild mottle virus (PMMoV). Cases indicate people with clinically diagnosed COVID-19; such people were moved to an isolation building with a separate watershed. After initial SARS-CoV-2 antigen testing of trainees at the time of arrival, subsequent antigen tests were not routinely performed.
Seagoing Vessels
SARS-CoV-2 was detected on 2 separate Coast Guard cutters. On the first cutter, a positive result on a SARS-CoV-2 antigen test resulted in confirmation of COVID-19 and isolation onboard for 1 military personnel. When the vessel returned to port, 4 samples were obtained while the blackwater storage tank was emptied. One of the 4 samples was positive, showing a value of 2.64 × 10−5 for the N1 primer set and 1.96 × 10−5 for the E primer set, and PMMoV levels were significantly higher than those from the barracks. There was a long interval between diagnosis and testing and the discharging of blackwater several times before the sample was taken. The outcome shows that SARS-CoV-2 can be detected in blackwater from vessels.
On the second cutter, in June 2021, wastewater detection showed levels of SARS-CoV-2 of 1.21 × 10−4 for gene N1 and 3.34 × 10−4 for gene E. A subsequent test about 2 weeks later yielded a positive result only for gene N1, with a value of 2.42 × 10−4. The detection of SARS-CoV-2 in wastewater triggered active case finding among the crew, which yielded no cases of COVID-19 but did provide reassurance to begin the subsequent deployment.
Discussion
This study described the wastewater testing program conducted at 3 US Coast Guard training centers from March 2021 through August 2022, the end of the Coast Guard’s emergency phase of COVID-19 management. The original goal was to prevent uncontrolled viral transmission among fellow trainees, staff, and instructors and disruption of the unique training mission at each center. Leadership received timely information on wastewater levels of SARS-CoV-2 and interpretations of the estimated number of infections, as well as recommendations, such as risk mitigation strategies, additional testing, the need for face masks and social distancing, and other changes depending on the phase of the pandemic and the mission of the training center.
SARS-CoV-2 genetic material was least likely to be detected in the wastewater if military trainees with SARS-CoV-2 infection were isolated >48 hours before a 24-hour sampling period. We increased the deployment time of the passive sampler from 24 hours to 48 to 72 hours to reduce the risk of false-negative test results. Individual cases of COVID-19, largely among military trainees who were vaccinated, shed SARS-CoV-2 above the lower limit of detection for our assay in dormitories housing >1000 people 69.8% of the time. When ≥3 military trainees were clinically diagnosed with COVID-19 within a 7-day period, SARS-CoV-2 was detected in wastewater 88.3% of the time.
The use of a commercial-grade tampon for passive sampling detected the fecal indicator virus PMMoV 100% of the time, consistent with previous studies.9,17 -19 Signal strength and the ratio of SARS-CoV-2 to PMMoV per infected person were inversely correlated to the number of trainees living in a barracks because of dilution of wastewater by trainees without SARS-CoV-2 infection. Training center A houses >1000 military trainees and had our strongest detected ratio of SARS-CoV-2 to PMMoV (0.784). Training center C houses 100 to 150 military trainees and had the largest ratio of viral gene to fecal control (11.6) in summer 2021. The ratio of viral gene to fecal control is also likely to be influenced by the number and strength of COVID-19 infections, the COVID-19 vaccination rate, and individual variability in viral shedding of SARS-CoV-2. 20
Routine shipboard wastewater testing by the French Armed Forces Biomedical Research Institute of a French aircraft carrier found a spike in SARS-CoV-2 levels in blackwater after each arrival of new personnel and during port calls. 21 We also developed a method to identify SARS-CoV-2 genetic material in ship wastewater but encountered logistical challenges, including storage of the samples and adapting the ship’s wastewater system hardware to allow sampling. When vessels are in port, many military personnel do not remain on the vessels, and workers or visitors may shed into the vessel’s wastewater. Substantial alterations to the normal shipboard routine would be required to make wastewater testing an alternative to regular screening by the SARS-CoV-2 antigen or PCR tests. Vessels with a longer deployment time and few port calls, such as submarines or Coast Guard polar icebreakers, may be more amenable to wastewater testing for screening purposes. New technologies designed for locations without clinical laboratories may also be adaptable to shipboard testing. 22
Limitations
This study had some limitations. First, for logistical reasons, a single sample was isolated from each location, and a single PCR was performed; it is unclear if additional samples or replicate PCRs would have increased the sensitivity of detection in the wastewater. At training center A, the higher number of military trainees reduced the rate of detection if <3 people were infected. Second, policies at training centers B and C precluded testing of asymptomatic vaccinated trainees, which prevented identification of people with SARS-CoV-2 infection several times at that location. Training center B did not perform weekly SARS-CoV-2 antigen testing, increasing the likelihood of asymptomatic vaccinated individuals shedding into the wastewater. The policies also precluded us from assessing the correlation between diagnosed cases and wastewater levels at these 2 locations. Third, shipboard testing proved impractical, preventing us from studying wastewater on seagoing vessels in detail. Fourth, we were not able to conduct tests on ships with longer deployments because of their placement on the West Coast.
Conclusions
Before wastewater testing was halted at all US Coast Guard locations in August 2022, as the Coast Guard concluded its emergency phase of COVID-19 management, wastewater testing was shown to be a useful epidemiologic tool for the Coast Guard, especially early in the pandemic, as also shown at the Air Force Academy. 23 Wastewater testing is not invasive and requires fewer resources than mass screening while still helping to identify people with asymptomatic SARS-CoV-2 infection. Wastewater testing results were some of the only forward-looking data available to senior leaders, unlike diagnosed case rates, which are retrospective with respect to infection rates. The work contributed to the US Coast Guard’s ability to bring science to bear on public health emergencies. The Coast Guard continues to assess lessons learned to develop policies that will better position the service for the next pandemic, which may include wastewater testing for other pathogens. In other settings, this surveillance method has been expanded to infectious diseases of significance, such as poliovirus, respiratory syncytial virus, and influenza.24 -27
Footnotes
Acknowledgements
The authors thank the many collaborators with this work and, in particular, the outstanding support of reservist US Coast Guard personnel, including Erin L. Barttelt, BS, Jolene Bruard, BA, Richard Burmeister, BS, William Cabreja, BS, Conrad Carruthers, BA, Chad R. Cruset, Erin Degenstein, MS, Samuel Encarnacion, Joseph Guajardo, AA, Jeremy Guerrero, Stephanie Jocis, MS, Yevginy Kislov, BS, Anthony Lebaron, Lorenzo Medina, BS, Michael Persun, MS, Meghan Peterson, BS, Rebecca Prendergast, MS, Eric Propst, BA, Tara Schendorf, MS, Caitlin Springer, BS, Shawn Tuthill, BS, Christopher Varga, BS, Jordan Wagstaff, and Paul Woodhead. The authors thank Kendra Maas, PhD, and Lisa Nigro, PhD, of the Microbial Analysis, Resources, and Services Center of the University of Connecticut for training and support in adopting the testing methodology. The authors thank Michael Raneri, BS, of MACOM Technology Solutions for his careful review of the work.
Previous Posting
Parts of this article were posted as a preprint on medRxiv.org on February 6, 2022. doi:10.1101/2022.02.05.22269021.
Disclaimer
The views expressed herein are those of the authors and do not necessarily reflect those of the US Coast Guard, US Department of Homeland Security, or US government.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
