Abstract
Objectives:
Mpox surveillance was integral during the 2022 outbreak response. We evaluated implementation of mpox surveillance in Tennessee during an outbreak response and made recommendations for surveillance during emerging infectious disease outbreaks.
Methods:
To understand surveillance implementation, system processes, and areas for improvement, we conducted 8 semistructured focus groups and 7 interviews with 36 health care, laboratory, and health department representatives during September 9-20, 2022. We categorized and analyzed session transcription and notes. We analyzed completeness and timeliness of surveillance data, including 349 orthopoxvirus-positive laboratory reports from commercial, public health, and health system laboratories during July 1–August 31, 2022.
Results:
Participants described an evolving system and noted that existing informatics platforms inefficiently supported iterations of reporting requirements. Clear communication, standardization of terminology, and shared, adaptable, and user-friendly informatics platforms were prioritized for future emerging infectious disease surveillance systems. Laboratory-reported epidemiologic information was often incomplete; only 55% (191 of 349) of reports included patient address and telephone number. The median time from symptom onset to specimen collection was 5 days (IQR, 3-6 d), from specimen collection to laboratory reporting was 3 days (IQR, 1-4 d), from laboratory reporting to patient interview was 1 day (IQR, 1-3 d), and from symptom onset to patient interview was 9 days (IQR, 7-12 d).
Conclusions:
Future emerging infectious disease responses would benefit from standardized surveillance approaches that facilitate rapid implementation. Closer collaboration among informatics, laboratory, and clinical partners across jurisdictions and agencies in determining system priorities and designing workflow processes could improve flexibility of the surveillance platform and completeness and timeliness of laboratory reporting. Improved timeliness will facilitate public health response and intervention, thereby mitigating morbidity.
Keywords
For decades, mpox, which is caused by the monkeypox virus (MPXV), was predominantly a zoonotic disease endemic in some African countries. However, in spring 2022, nonendemic countries, including the United States, began reporting mpox cases predominantly among men who have sex with men. 1 The first case of mpox in Tennessee was detected in July 2022, shortly before the World Health Organization declared the outbreak a public health emergency of international concern. 2
During the COVID-19 pandemic in the United States, state, tribal, local, territorial (STLT), and national surveillance programs adapted to new response methods for data collection and data processing, providing an experience that could improve future surveillance system implementation. 3 Two years after the onset of the COVID-19 pandemic, US public health agencies responded to the multicountry outbreak of mpox with rapidly implemented surveillance systems at STLT and national levels. 4 Like other health departments, the Tennessee Department of Health (TDH) developed and implemented an mpox surveillance system in May 2022 to detect cases, ensure useful data collection, and facilitate reporting to the Centers for Disease Control and Prevention (CDC).
We evaluated implementation of Tennessee’s mpox surveillance system to understand successes and opportunities for improvement, with a goal of guiding future approaches to surveillance for acute emerging infectious disease outbreaks. Our objectives were to (1) describe the system and its implementation, (2) determine whether laboratory reporting provided acceptable case identification and sufficient epidemiologic information to guide public health response, (3) assess timeliness of information gathered, and (4) describe whether available resources (eg, personnel, technology) were efficiently used in data management processes and information exchange between TDH and CDC.
Methods
We based the evaluation process on previously described methods for evaluating public health surveillance system attributes of acceptability, data quality (completeness), timeliness, simplicity, and flexibility. 5 Taking a mixed-methods approach, we incorporated information provided by representatives of key collaborating groups and surveillance system data. Using purposeful sampling, we recruited representatives of collaborating groups that were determined a priori to include health care organizations serving populations at increased risk for mpox, laboratories testing clinical specimens for mpox, local health departments, and informatics departments within TDH and CDC. We invited representatives to participate in focus groups. If only 1 representative of the collaborating group could join at a given time, we conducted an in-depth interview instead. We spoke with a total of 36 representatives in 8 focus group discussions (29 representatives) and 7 key informant interviews during September 9-20, 2022 (Table 1). The roles of representatives related to mpox surveillance activities included patient diagnosis and treatment; laboratory administration, testing, and reporting to TDH; interviewing patients for public health investigations; and data entry, management, and reporting to TDH outbreak response leadership and CDC.
Demographic and occupational characteristics of focus group and interview participants for evaluation of mpox surveillance system (N = 36), Tennessee, United States, 2022
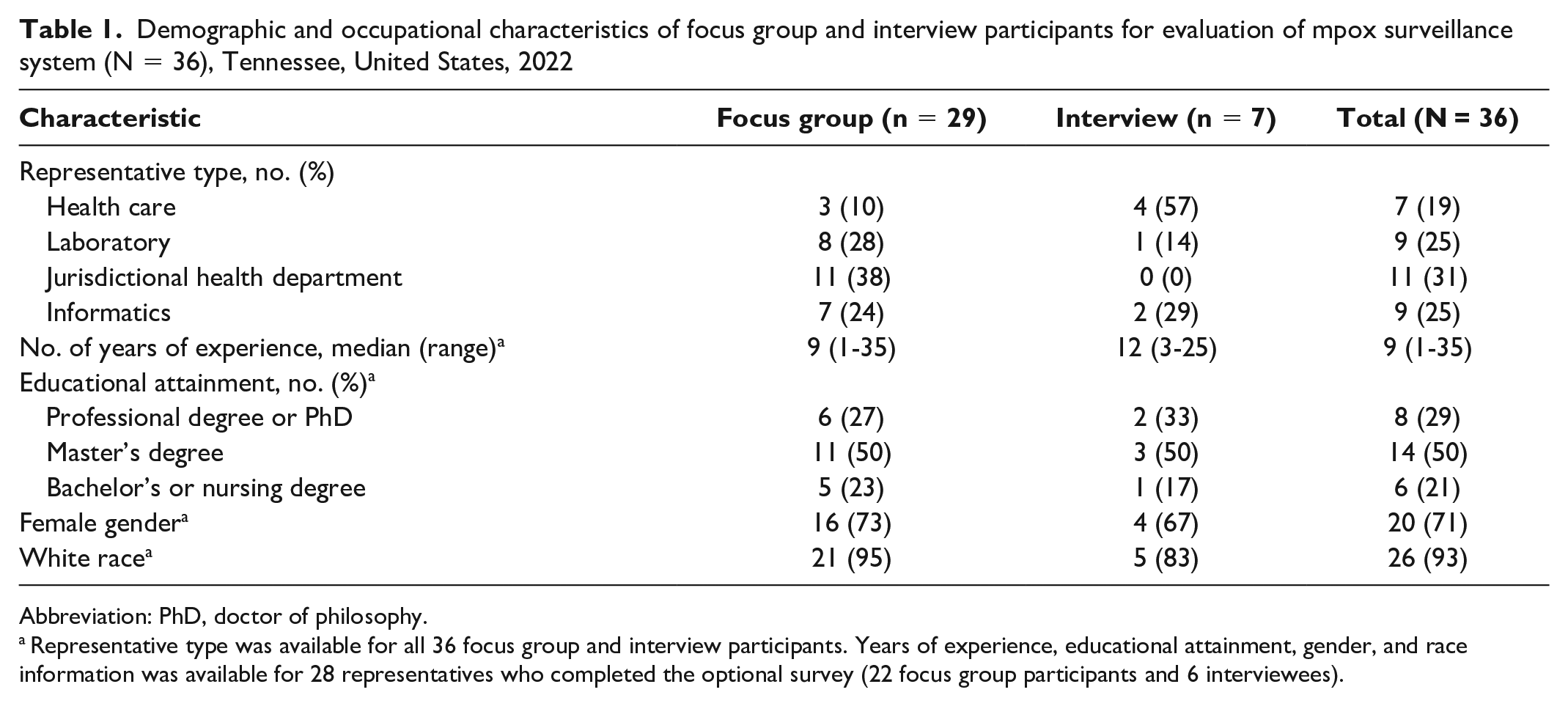
Abbreviation: PhD, doctor of philosophy.
Representative type was available for all 36 focus group and interview participants. Years of experience, educational attainment, gender, and race information was available for 28 representatives who completed the optional survey (22 focus group participants and 6 interviewees).
An investigator (C.M.T.) facilitated each session and used a semistructured interview guide that inquired about representatives’ experiences with surveillance activities during the mpox response, including their role, what went well, what could be improved, and recommendations for future systems. Representatives of health care organizations were asked about experiences at the patient–provider interface. The investigator used Microsoft Teams videoconferencing transcription software (Microsoft Corp) and written notes to record responses; 1 focus group was conducted in person. At the end of each session, we invited participants to complete an optional demographic survey facilitated by REDCap Electronic Data Capture tools hosted at TDH.6,7 Drawing from rapid qualitative methods for health services implementation research,8,9 an author (C.M.T.) reviewed and cross-checked notes with the transcriptions immediately after each session and categorized findings by interview guide topic and respondent type into a matrix using Excel (Microsoft Corp). After the final session, an author (C.M.T.) analyzed matrix content with a quasi-deductive approach in which evaluation objectives determined result sections.
All probable and confirmed mpox cases, defined as an individual with laboratory-confirmed Orthopoxvirus species or MPXV infection, 10 identified during July 1–August 31, 2022, were included in the analysis. Surveillance data included laboratory reports of all clinical specimens that tested positive for Orthopoxvirus species and case investigation information. Variables included dates (initial symptom onset, specimen collection, laboratory report, and patient interview) and reporting laboratory. Additional laboratory report variables included patient information (name, date of birth, address, and telephone number), demographic information (race and ethnicity), and specimen information (specimen type, collection method, and anatomic site of collection). 11 One commercial laboratory provided a list of all positive test results for Orthopoxvirus species among Tennessee residents as of August 31, 2022, to facilitate evaluation of surveillance system data completeness.
We used descriptive analyses to compare results from a commercial laboratory with cases identified by the surveillance system and to determine the proportion of laboratory reports with complete patient, demographic, and specimen information. We described the time between symptom onset, specimen collection, laboratory report, and patient interview and the total time between symptom onset and patient interview. When a case was linked with multiple laboratory reports, we used the report with the earliest report date.
We calculated exact odds ratios (ORs) with 95% CIs to compare the proportion of health system and commercial laboratory reports that included complete recommended patient, demographic, and specimen information with reports from the TDH Laboratory Services (TDHLS). We also used a Wilcoxon test, with P < .05 considered significant, to compare the distribution of days between specimen collection and result reporting between commercial laboratories and TDHLS, excluding health system laboratories that were not internally testing for mpox. We generated statistical analyses using SAS version 9.4 (SAS Institute, Inc). This activity was reviewed and approved by CDC and conducted consistent with applicable federal law and CDC policy (45 CFR part 46.102[l][2]; 21 CFR part 56; 42 USC Sect 241[d]; 5 USC Sect 552a; 44 USC Sect 3501 et seq).
Results
Mpox Surveillance System Description and Implementation
TDH began preparing for the 2022 mpox outbreak on May 18, 2022. By May 20, 2022, a surveillance system had been implemented and communicated to health care providers and public health staff across the state. The system was continuously refined and improved as the outbreak progressed. The system (Figure) primarily used mandatory laboratory reporting of clinical specimens that tested positive for either Orthopoxvirus species or MPXV (ie, probable and confirmed mpox cases), and clinicians or other public health agencies could also report cases. Individual, community, and clinician disease awareness and decision-making determined testing practices. TDHLS and commercial laboratories tested specimens and reported results electronically to TDH’s integrated disease surveillance system, the National Electronic Disease Surveillance System Base System (NBS). On receipt of a positive laboratory test result, the local health department initiated case investigation and contact tracing and subsequently offered symptom monitoring through a text messaging application or email and postexposure vaccine prophylaxis for contacts who were exposed. TDH shared information with CDC through 2 pathways: (1) results within 2 hours of completed TDHLS testing through the Laboratory Response Network and (2) daily information about identified cases through the Data Collection and Integration for Public Health Event Response (DCIPHER).
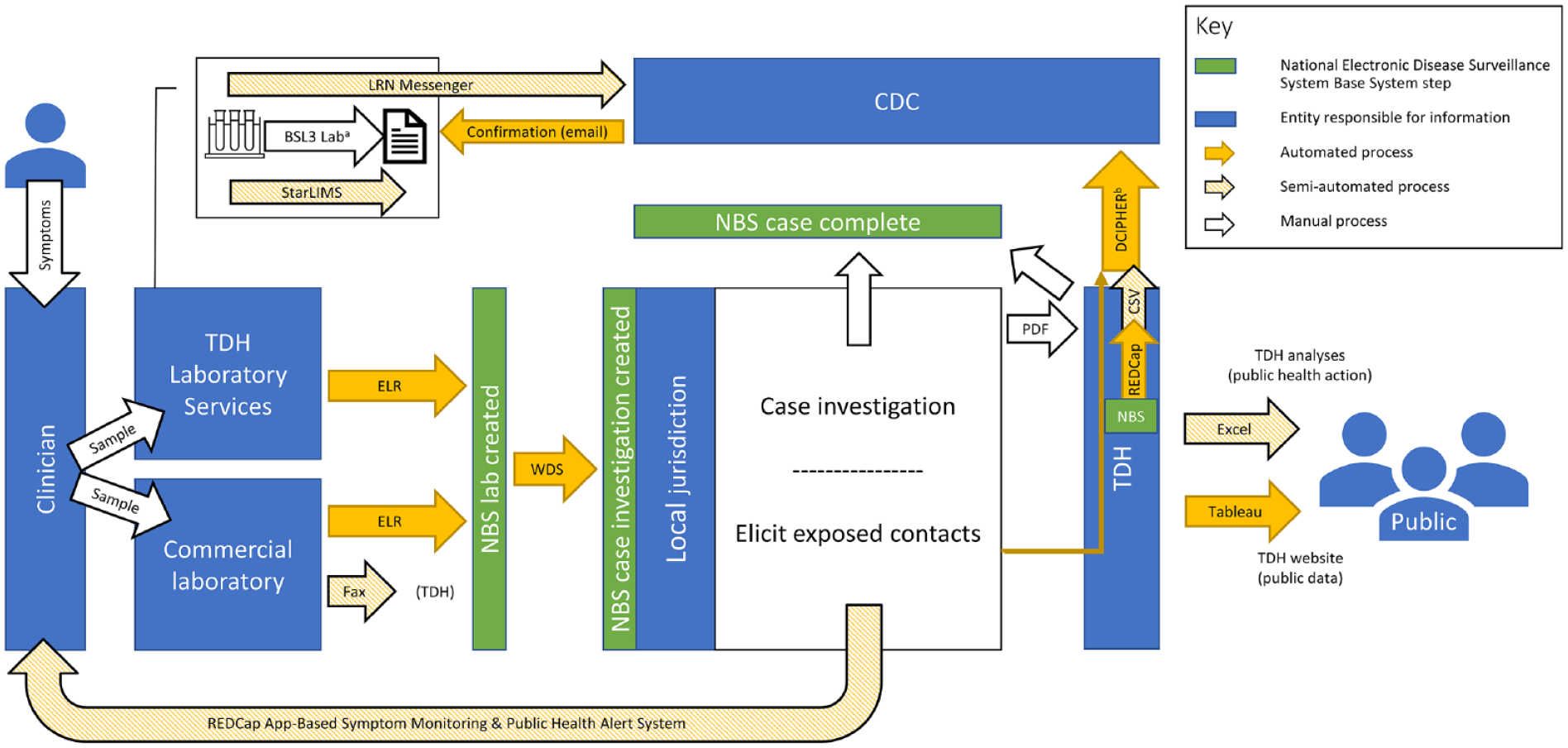
Mpox surveillance system processes, Tennessee, 2022.
Focus group participants described mpox surveillance system implementation as dynamic and evolving to meet response needs and priorities at local, state, and national levels as the outbreak progressed. Multiple iterations of case investigation, data management, and reporting processes occurred as the national response and CDC guidance changed over time. The evolving national response included changes to requested case information, length of case report forms, and the process for case notification to CDC. For example, early in the outbreak, health department staff created fields in NBS to store information from the initial case report form. When CDC released an abbreviated case report form, TDH staff modified NBS to accommodate the change. Similarly, CDC initially requested case notification verbally by telephone, but this process was later replaced with daily transmission of line lists through DCIPHER. Participants noted successfully building on COVID-19 experiences to rapidly adapt to and meet current response needs and that building on prior experiences relied heavily on institutional knowledge of surveillance and informatics needs for a new condition. For example, through COVID-19, TDH informatics staff became adept at modifying NBS pages throughout an event and learned to anticipate reporting requests from CDC. This learned experience resulted in fewer negative effects to the end-user experience for local and regional health department staff for mpox as compared with COVID-19. Participants suggested that more flexible informatics platforms and clearer communication between public health entities early in the response would improve the efficiency of implementation processes.
Acceptability and Completeness of Laboratory Reporting
The surveillance system performed well in reporting cases with positive laboratory test results. One commercial laboratory provided information about all tests detecting Orthopoxvirus species in Tennessee residents, which included 91 specimens collected from 64 individuals during July 11–August 31, 2022. When compared with surveillance system data, all 64 individuals had been identified as cases. However, representatives of health care organizations noted that patient health care–seeking behaviors and clinician testing practices limited the surveillance system’s ability to detect all MPXV infections because of barriers to testing, including lack of disease awareness, stigmatization, uncertain or burdensome testing processes, and unknown test costs.
Laboratory reports (N = 349) for 180 cases frequently lacked patient contact and demographic information necessary for guiding public health efforts. All reports included patient name, date of birth, and current sex, but only 66% (230 of 349) included addresses and 55% (192 of 349) included telephone numbers. Fifty-four percent (187 of 349) included race or ethnicity information. Regarding specimen information, 72% (253 of 349) indicated the specimen type, 42% (146 of 349) indicated the collection method, and 20% (70 of 349) indicated the anatomic site. A laboratory report was considered fully complete if all patient, demographic, and specimen information was included. Fewer than 1% of commercial laboratory reports were complete, whereas 19% of TDHLS and 17% of health system reports were complete (Table 2). Commercial laboratory reports were 10 times less likely than TDHLS reports to include race or ethnicity information (OR = 0.10; 95% CI, 0.05-0.17), but inclusion of patient information was similar (OR = 0.79; 95% CI, 0.50-1.25).
Completeness of recommended patient, demographic, and specimen information in laboratory reports for Orthopoxvirus species–positive specimens (N = 349 a ), by laboratory type, Tennessee, July–August 2022
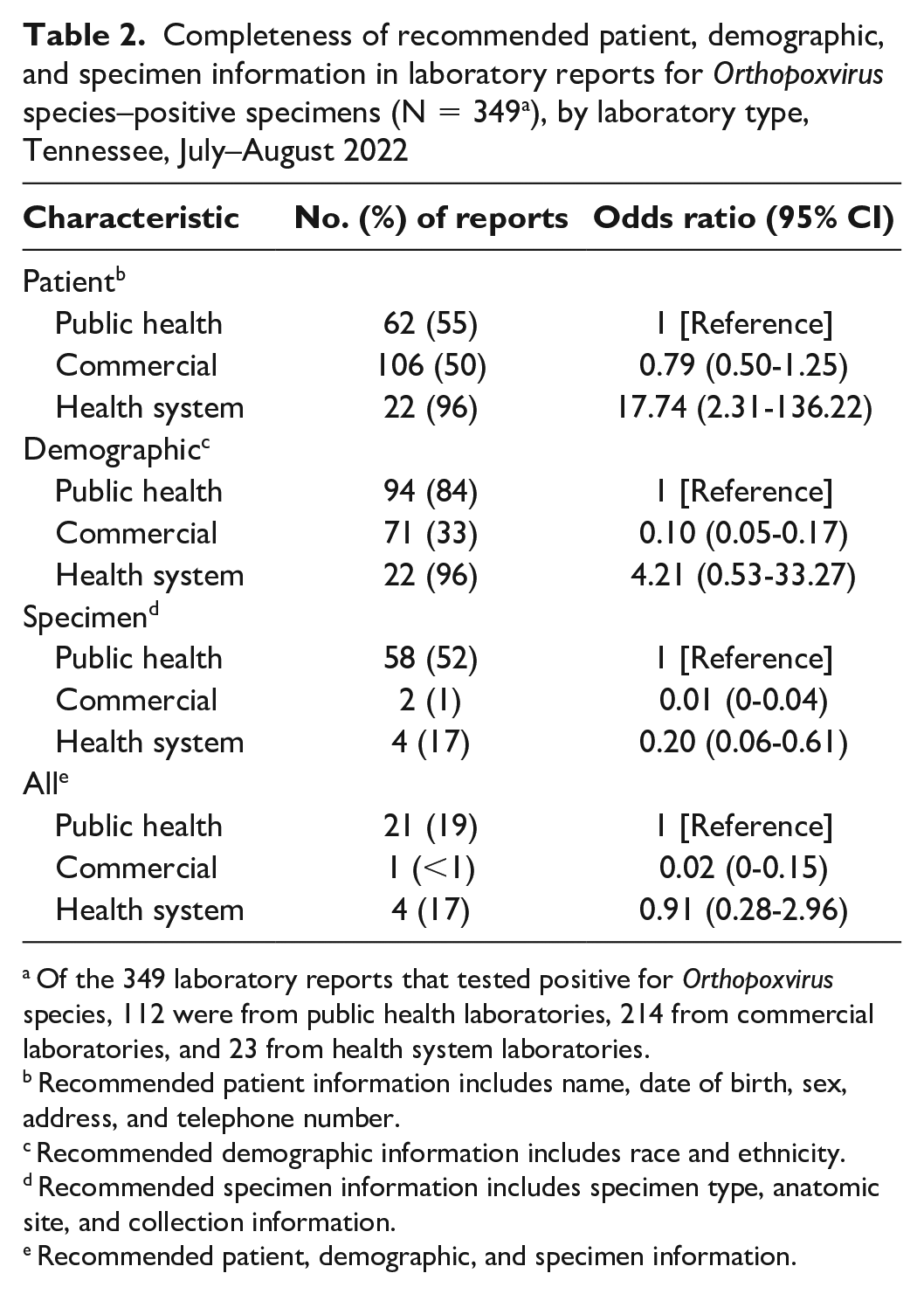
Of the 349 laboratory reports that tested positive for Orthopoxvirus species, 112 were from public health laboratories, 214 from commercial laboratories, and 23 from health system laboratories.
Recommended patient information includes name, date of birth, sex, address, and telephone number.
Recommended demographic information includes race and ethnicity.
Recommended specimen information includes specimen type, anatomic site, and collection information.
Recommended patient, demographic, and specimen information.
When asked what aspects of mpox surveillance system implementation had not gone well, a commercial laboratory representative relayed challenges in collecting demographic information, noting, “I would see more issues with missing demographics for monkeypox th[a]n the other reportable tests.” In addition to missing data on demographic characteristics, they noted that sometimes false patient information (eg, addresses) would be given and explained that, from the laboratory perspective, little can be done to fill in the missing or inaccurate information because this information is provided by the clinician or patient.
Timeliness of Information
In total, 118 of 203 cases (58%) had date information available for symptom onset, specimen collection, laboratory report, and patient interview. The median time between symptom onset and specimen collection was 5 days (IQR, 3-6 d). Laboratory reporting occurred a median of 3 days (IQR, 1-4 d) after specimen collection. Patient interviews occurred a median of 1 day (IQR, 1-3 d) after laboratory reporting. In total, the median time between symptom onset and patient interview was 9 days (IQR, 7-12 d). Commercial laboratories (n = 214 reports from 119 cases) took longer to report positive specimen test results than TDHLS (n = 112 reports from 60 cases), with a median of 4 days (IQR, 3-5 d) versus 1 day (IQR, 1-3 d) between specimen collection and laboratory reporting (P < .001).
During focus groups, representatives of jurisdictional health departments commented that the longer time required to receive results from the commercial laboratories than from TDHLS constrained case investigations. The long interval between the date of symptom onset and patient interview reduced the ability to identify contacts of the patient at risk for mpox to assess for disease transmission and offer postexposure prophylaxis (ie, Jynneos vaccine) within the window of greatest efficacy.
Simplicity and Efficiency of Information Exchange
Laboratory and informatics representatives reported on inefficiencies and relayed a need to improve automation of processes before another outbreak response. For example, TDHLS technicians manually created 2 entries for each tested specimen; 1 in TDH’s Laboratory Information Management System to report results to TDH epidemiologists and 1 in the Laboratory Response Network Messenger, which communicates directly with CDC. Informatics personnel at TDH created a platform at NBS to capture information from laboratory reports and case report forms. However, at this time during the outbreak response, processes and protocols to validate and receive automatic data transmissions for mpox were still being developed by CDC. Because of the rising incidence of disease and the need for an immediate solution to provide timely and complete data, TDH developed a mechanism of sharing information stored in NBS through CDC’s required DCIPHER platform using a REDCap application programming interface.
When asked how to improve surveillance systems for future emerging infectious diseases, focus group participants frequently provided ideas for improving clarity and standardizing information exchange among departments and agencies. Laboratory representatives recommended standardization of terminology and testing protocols. Jurisdictional health department personnel requested clear communication about standardized data collection, including which questions on case report forms should be prioritized and efficient processes for reporting information from patient interviews. Informatics representatives recommended standardizing data collection early to avoid discrepancies between data collected by health departments across the country. Informatics representatives also recommended improving the flexibility of current surveillance system software so that existing surveillance processes can be easily expanded to respond to emerging threats. One representative recommended developing “a more flexible and rapidly adapting system that everyone can share.”
Discussion
In Tennessee, implementation of mpox surveillance during an outbreak response was largely successful because of institutional knowledge and experience, with staff building on lessons learned during the COVID-19 response. Focus group participants attributed the development of processes for data capture and storage to experience gained during the COVID-19 response. However, some lessons learned during the COVID-19 response could have been better applied. For example, participants noted that early uncertainty about commercial laboratory test costs and testing locations for mpox might have led to a decrease in the number of tests conducted, thereby affecting the completeness of surveillance. Substantial room for improvement exists in the public health infrastructure and operations for future surveillance of emerging infectious diseases, including addressing the limitations of laboratory-based surveillance; increasing the completeness of information received by public health; shortening the duration of information gathering; increasing the flexibility of informatics platforms; and improving the standardization, communication, and efficiency of reporting between public health agencies at state and federal levels.
Laboratory reporting is often relied on as the basis of public health surveillance out of convenience, but many factors can influence testing decisions and timeliness. Interview and focus group participants acknowledged these limitations, providing reasons why patients might not seek evaluation by a clinician or for clinicians not to test for MPXV (eg, feeling stigmatized, being unaware of mpox presentation, or not knowing the availability or cost of testing). Improving community and clinician engagement to increase awareness and emphasize the importance of appropriate testing for emerging infectious diseases might help to address these concerns and reduce delays between symptom onset and diagnostic testing. Health care representatives indicated the importance of partnering with community health workers and organizations to increase disease awareness in the community. Clinicians noted that education of health care staff might be more effective when presented in a multimodal and easily digestible approach (eg, an infographic on social media) than through paragraphs of text.
In addition to testing practice limitations, the usefulness of laboratory reporting is limited by frequently missing patient contact, demographic, and specimen information in laboratory reports. In Tennessee’s mpox surveillance system, laboratory reporting was followed by standardized patient interviews; for these interviews to take place, public health personnel needed patient contact information. Unfortunately, this information was often missing from laboratory reports, possibly in part because health care facilities do not always provide this information when specimens are submitted. Missing information can delay patient interviews by requiring public health personnel to search alternative sources for contact information. Delayed patient interviews could further delay identifying people who have been exposed to the disease and hinder efforts to interrupt disease transmission, such as timely provision of postexposure vaccine prophylaxis. Increasing communication between laboratory and health care facilities can improve the completeness of laboratory report information and allow public health surveillance data to be used quickly. For example, conversations that include laboratory, health care facility, and local public health partners when determining what patient information will be included with specimen submission and result reporting could be prompted when a health care facility introduces a new diagnostic test for a reportable public health condition.
Surveillance system timeliness is essential for guiding public health actions and patient follow-up during an acute infectious disease response. For mpox, timeliness is essential for providing valuable epidemiologic information to public health agencies and preventing additional MPXV infections. Postexposure vaccine prophylaxis is recommended within 4 days but is permitted up to 14 days after exposure. 12 In Tennessee, health department personnel identified contacts of patients with mpox during patient interviews and then contacted them to offer postexposure vaccination and education about preventive measures. Although patient interviews tended to occur within 1 day of the health department receiving a report identifying a patient with mpox, long durations between specimen collection and laboratory reporting can reduce the ability of people who were exposed to mpox to receive postexposure vaccine prophylaxis within the recommended time frame. Reasons for this long duration are multifactorial and related to health care–seeking behavior, testing practices, specimen processing at the health care facility, transportation to the commercial laboratory, and testing procedures. For example, 1 study found that transportation factors (eg, geographic location) might be associated with delays. 13 Public health agencies could focus on reducing these delays by engaging with and incentivizing commercial laboratories to identify and address barriers to timely specimen processing and result reporting.
Current informatics platforms and reporting processes are not efficient or user-friendly for rapid implementation of disease surveillance or sharing information between public health agencies. Substantial institutional expertise was necessary to overcome these barriers and manually develop software options to capture necessary data to guide public health actions and accommodate CDC’s case notification requirements for mpox surveillance. Our findings indicate a need for more flexible informatics systems, in advance of urgent responses, that can be easily and quickly tailored for various diseases. In addition, information sharing among agencies could be improved through data standardization, maximizing automation of data flow (eg, through application programming interfaces), and reducing redundancies in required CDC case notification procedures. Other researchers have also raised the need for an integrated and standardized information system that is interoperable among agencies and meets both routine surveillance and outbreak management needs. 14 CDC’s Data Modernization Initiative, 15 an effort to unify public health data nationally, is a step toward improving interoperability and standardization throughout US public health agencies. For success, collaboration among software developers and public health agencies is essential to identify and develop solutions for inefficiencies. Ideally, addressing inefficiencies would occur before the next major outbreak response and include representatives from various levels of public health meeting with software developers to determine system priorities and design workflow processes.
Limitations
This study had several limitations. First, the description of our surveillance system may not be generalizable to other jurisdictions because it represents 1 state health department in the United States. Aspects of our experience will differ from those in other states and jurisdictions with different public health laboratory capabilities, case burden, and informatics platforms. However, our recommendations might be applicable to all jurisdictions in varying degrees. Second, our quantitative findings comparing commercial laboratory reporting with cases captured by the surveillance system were limited because we included only 1 commercial laboratory in the analysis. Third, our qualitative findings were limited by our purposeful sampling approach, which might have excluded additional insights from others involved with mpox surveillance efforts in Tennessee.
Conclusion
Lessons learned from implementation of mpox surveillance in Tennessee highlight the need to improve future surveillance approaches for acute emerging infectious disease outbreaks. Improving commercial laboratory quality and timeliness, informatics platform flexibility, and data standardization can streamline future responses and avoid reinventing key aspects of acute outbreak response. These improvements will require engaging and partnering with commercial laboratories, informatics companies, and other STLT and federal public health agencies to understand and address the systemic barriers to improving these aspects of public health infrastructure. Taking these steps as part of ongoing outbreak preparedness could be most helpful for the next response to an emerging infectious disease.
Footnotes
Acknowledgements
The authors acknowledge the valuable contribution to this work from people who participated in focus groups and interviews, and Anna Blackstock, PhD, Division of Scientific Education and Professional Development, Center for Surveillance, Epidemiology, and Laboratory Services, CDC, for review of the statistical methods.
Authors’ Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC) or the Tennessee Department of Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
