Abstract
Objective:
Inpatient vaccination is an opportunity to increase vaccine uptake among patients at high risk for severe COVID-19 illness. We designed and implemented a hospital-based COVID-19 vaccination program with the aim of increasing documentation of vaccine eligibility and COVID-19 vaccination to eligible inpatients before discharge.
Methods:
We integrated a templated note into the electronic medical records and trained health care personnel to screen inpatients and document COVID-19 vaccine eligibility at the Atlanta Veterans Affairs Medical Center. Vaccination staff deployed to inpatient wards administered the vaccine to eligible and consenting patients at the bedside. We calculated the number of inpatients whose vaccine eligibility was assessed and documented during a 4-week period after health care personnel were trained. We used the Wald χ2 test to compare the proportion of eligible patients who were vaccinated before discharge 4 weeks before (March 29–April 23, 2021) and 4 weeks after (May 3-28, 2021) the training period.
Results:
During the 4 weeks before the training period, COVID-19 vaccine eligibility was not routinely assessed and documented. Of 793 inpatients discharged during the 4 weeks after the training period, 470 (59%) had COVID-19 vaccine eligibility documented. Of 86 patients who were eligible for vaccination, 61 (71%) received COVID-19 vaccination before discharge. COVID-19 vaccination rates during hospitalization increased significantly from 16 of 769 inpatients (2%) during the 4 weeks before training to 61 of 793 inpatients (8%) during the 4 weeks after training (P < .001).
Conclusion:
An inpatient vaccination program that integrated COVID-19 vaccination into discharge planning increased vaccine screening and uptake. Future studies are needed to identify barriers to vaccination and strategies to increase vaccine uptake among those who are hesitant.
Vaccination against COVID-19 has been shown to reduce hospitalizations and death among older adults (aged ≥65 y). 1 As of May 2023, 70% of people in the United States were fully vaccinated, but fewer than 20% had received the bivalent booster dose. 2 Inpatient vaccination before hospital discharge is an opportunity to increase vaccine uptake among patients at high risk of severe COVID-19–related illness and death.
The rationale for an inpatient vaccination program is that an inpatient program prioritizes vaccinating patients who are at high risk of severe illness because hospitalized patients often include older adults and those with chronic comorbidities. This rationale is the same rationale used for integration of influenza and pneumococcal vaccinations into hospital-based settings, with data showing that recently discharged older adults (aged >65 y) account for a substantial proportion of subsequent influenza-related hospitalizations and deaths. 3 Hospital-based vaccination against influenza and pneumococcus has been recommended by the Centers for Disease Control and Prevention since the 1980s,4,5 and multiple studies have shown that hospital-based interventions increase influenza or pneumococcal vaccine uptake.6 -8 Thus, prior experiences with these inpatient vaccination programs suggest that similar strategies could be applied to increase COVID-19 vaccination uptake.
Few studies have described how to operationalize an inpatient COVID-19 vaccination program or have reported results of vaccine uptake with such a program.9,10 We implemented an inpatient COVID-19 vaccination program at a Veterans Affairs (VA) medical center during April 2021. Our aims were to (1) describe our strategies to train health care personnel and implement a sustainable inpatient COVID-19 vaccination program, (2) document the proportion of discharged patients with vaccine eligibility, and (3) compare the number and proportion of patients vaccinated at discharge during the 4 weeks before versus the 4 weeks after training of health care personnel.
Methods
Study Design and Patient Population
We trained health care personnel in April 2021 to review and document COVID-19 vaccine eligibility and interest among adult patients hospitalized in medical, surgical, and psychiatric wards at the Atlanta VA Medical Center (AVMC). Health care personnel on primary treatment teams evaluated patients for eligibility, using a templated note integrated into the electronic medical record (EMR) to document COVID-19 vaccine eligibility during discharge planning (Figure 1). The AVMC outpatient vaccination center is located in the hospital, and personnel from the outpatient vaccination center administered COVID-19 vaccines at discharge every day except Sundays and after hours.

Example of COVID-19 vaccine screening on a discharge note, which was integrated into the Atlanta Veterans Affairs Medical Center’s electronic medical record at implementation of an inpatient COVID-19 vaccination program, 2021.
Once we completed training of health care personnel, we measured the number and proportion of discharged patients with vaccine eligibility documented by using the templated note and the number of patients who received COVID-19 vaccines. We compared the number of patients who received COVID-19 vaccines at discharge during the 4 weeks before training (March 29–April 23, 2021) and the 4 weeks after training (May 3-28, 2021). Hospital personnel did not routinely assess or document COVID-19 vaccine eligibility before the training period. Of note, all Georgia adults became eligible for COVID-19 vaccines on March 25, 2021. 11
According to criteria set forth by the VA’s Office of Research and Development, this project was considered a quality improvement project that did not require local research office approval and was, therefore, not reviewed by an institutional review board.
Standard Operating Procedure
During discharge planning, primary treatment teams used the EMR note to assess and document the eligibility of inpatients for vaccination with either the Janssen (Ad.26.COV2.S) COVID-19 vaccine or the Moderna (mRNA-1273) COVID-19 vaccine (Figure 2). Primary treatment teams determined eligibility if (1) the patient did not have active COVID-19 infection during the current admission, (2) the patient was not up to date on COVID-19 vaccination, and (3) the patient had verbally consented to receiving a COVID-19 vaccine during admission. If patients did not meet 1 or more of these criteria, the primary treatment teams indicated on the EMR note that the patient did not qualify for COVID-19 vaccination. Primary treatment teams could also select “other” on the EMR note to account for other relative contraindications or reasons for deferring vaccination.
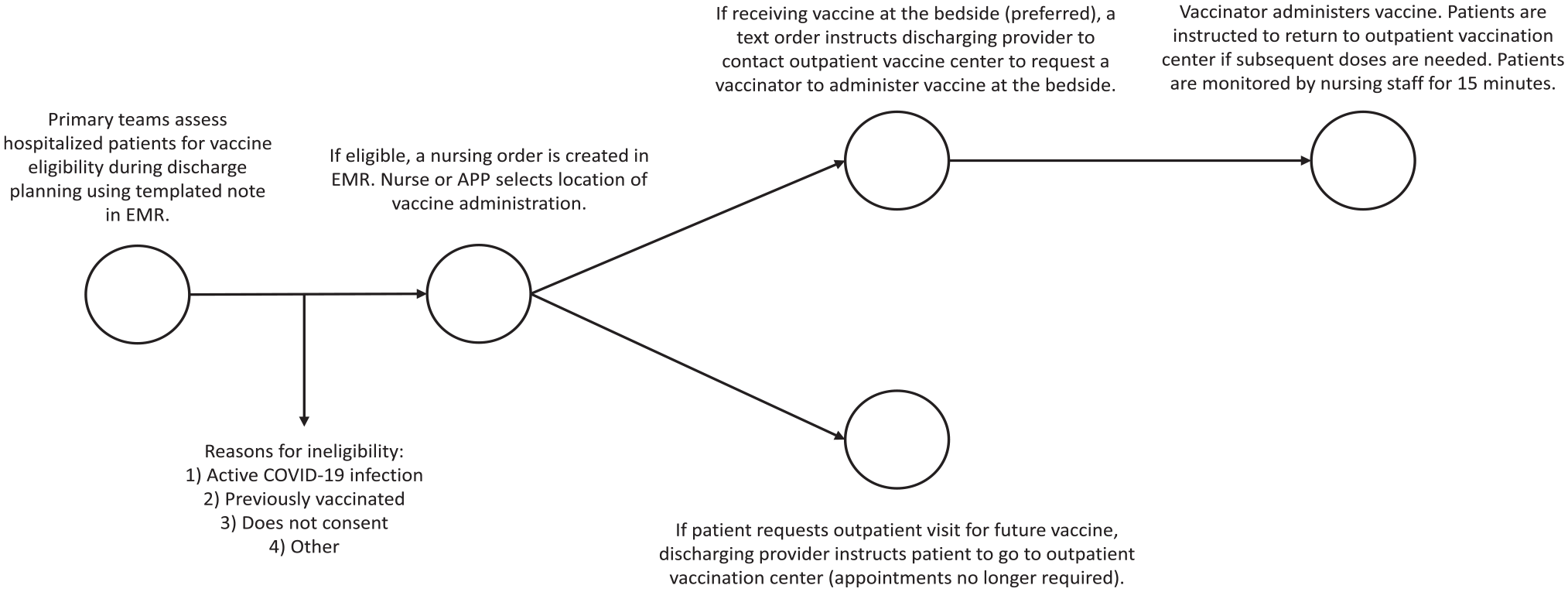
Workflow of an inpatient COVID-19 vaccination program as part of discharge planning for inpatients at the Atlanta Veterans Affairs Medical Center, 2021. Abbreviations: APP, advanced practice provider; EMR, electronic medical record.
If patients met the eligibility criteria, the EMR note automated a vaccine order by text to the discharge nurse. The vaccine order was a standing order so that individual physicians did not have to order each vaccine. Personnel from the outpatient vaccination center administered the COVID-19 vaccine to patients before discharge (recommended) or scheduled a COVID-19 vaccination appointment with patients at a future date after discharge per patient preference or if the vaccination center was closed (ie, if the patient was discharged after hours or on Sunday).
If the treatment team selected in the EMR note to have the patient receive the COVID-19 vaccine as an inpatient, an automated text order instructed the discharge nurse to contact the outpatient vaccination center to request a vaccinator to administer the vaccine at the bedside. If the patient received the Moderna vaccine, the vaccinator scheduled a return visit for the patient’s second dose. After vaccination, nursing staff monitored patients for adverse reactions for 15 minutes and documented vaccine administration and monitoring in a separate templated note. If the patient requested an outpatient visit after discharge to receive vaccination, the discharging nurse instructed the ward clerk to make an appointment in the outpatient vaccination center for the patient. Later in the pandemic and after the study period, the outpatient vaccination center welcomed walk-ins; thus, the outpatient vaccination center no longer required vaccine appointments. In addition, since the beginning of our inpatient COVID-19 vaccination program, we have updated the hospital policy to offer COVID-19 boosters and have aligned our eligibility criteria to reflect current vaccine guidance.
Until May 6, 2021, primary treatment teams had the option to select “ambulatory” or “nonambulatory” instead of “inpatient” or “outpatient” locations for vaccination. During that time, if the patient was nonambulatory, a staff member from the outpatient vaccination center would administer the vaccine to the patient at the bedside; however, discharge staff instructed ambulatory patients to go to the outpatient vaccination center after discharge to receive their vaccination. Because some patients left the hospital after discharge without going to the vaccination center, we changed the workflow to recommend bedside administration of vaccines unless the patient declined or the outpatient vaccination center was closed. We avoided asking nurses to escort patients to the outpatient vaccination center because they already had competing tasks and priorities.
Training Health Care Personnel
A physician (E.J.C.) and pharmacist (T.A.G.) in the infectious diseases division mainly led the inpatient COVID-19 vaccination program. Either the physician or pharmacist met with nurses, advanced practice providers, and physicians on each of the participating wards during the training period, which took place in April 2021. The infectious diseases physician or pharmacist trained hospital providers and staff in small-group sessions on how to assess patients for vaccine eligibility and interest, how to use the integrated template in the EMR, and the importance of inpatient vaccination to increase COVID-19 vaccine uptake. The advanced practice providers and physicians primarily performed vaccine eligibility assessments and documentation. Infectious diseases leadership regularly updated the standard operating procedures, and department leaders circulated emails with detailed instructions to all personnel.
Data Collection
Using data extracted from the EMR, we collected data on all adult inpatients on medical, surgical, and psychiatric wards who were discharged during the 4 weeks before training (March 29–April 23, 2021; preintervention) and after training (May 3-28, 2021; postintervention). We established a 1-week period (April 24–May 2, 2021) after initial training during which no data were collected to allow adoption of the intervention, and we then measured data from discharged patients during the subsequent 4 weeks. We collected data on the patients’ medical team (direct-care hospital medicine, academic hospital medicine, surgical, psychiatric), eligibility status, reasons for ineligibility, whether patients received the vaccination as inpatient or whether staff referred the patient to the outpatient vaccination center, and whether patients received COVID-19 vaccination before discharge. We had data on vaccine eligibility, reasons for ineligibility, the primary treatment team, and the location of vaccination only for the 4 weeks postintervention because staff did not collect these variables preintervention.
Statistical Analysis
We collected data using Microsoft Excel version 16.57, and we used SAS version 9.4 (SAS Institute Inc) for data analysis. We used descriptive statistics to calculate the proportion of patients whose vaccine eligibility was documented, the proportion of patients who were eligible for COVID-19 vaccines, and the proportion of patients who received COVID-19 vaccines. We calculated unadjusted odds ratios (ORs) to estimate the association of exposure to the inpatient COVID-19 vaccination program (postintervention period) with vaccination before discharge. We used the Wald χ2 test to compare the number of inpatients who received vaccination before discharge pre- and postintervention. We also assessed the association between location of vaccination (inpatient vs postdischarge vaccination appointment or at the outpatient vaccination center) with completion of vaccination. We considered P < .05 to be significant.
Results
In total, 769 patients were discharged from participating wards during the 4 weeks before the intervention, and 793 inpatients were discharged during the 4 weeks after the intervention.
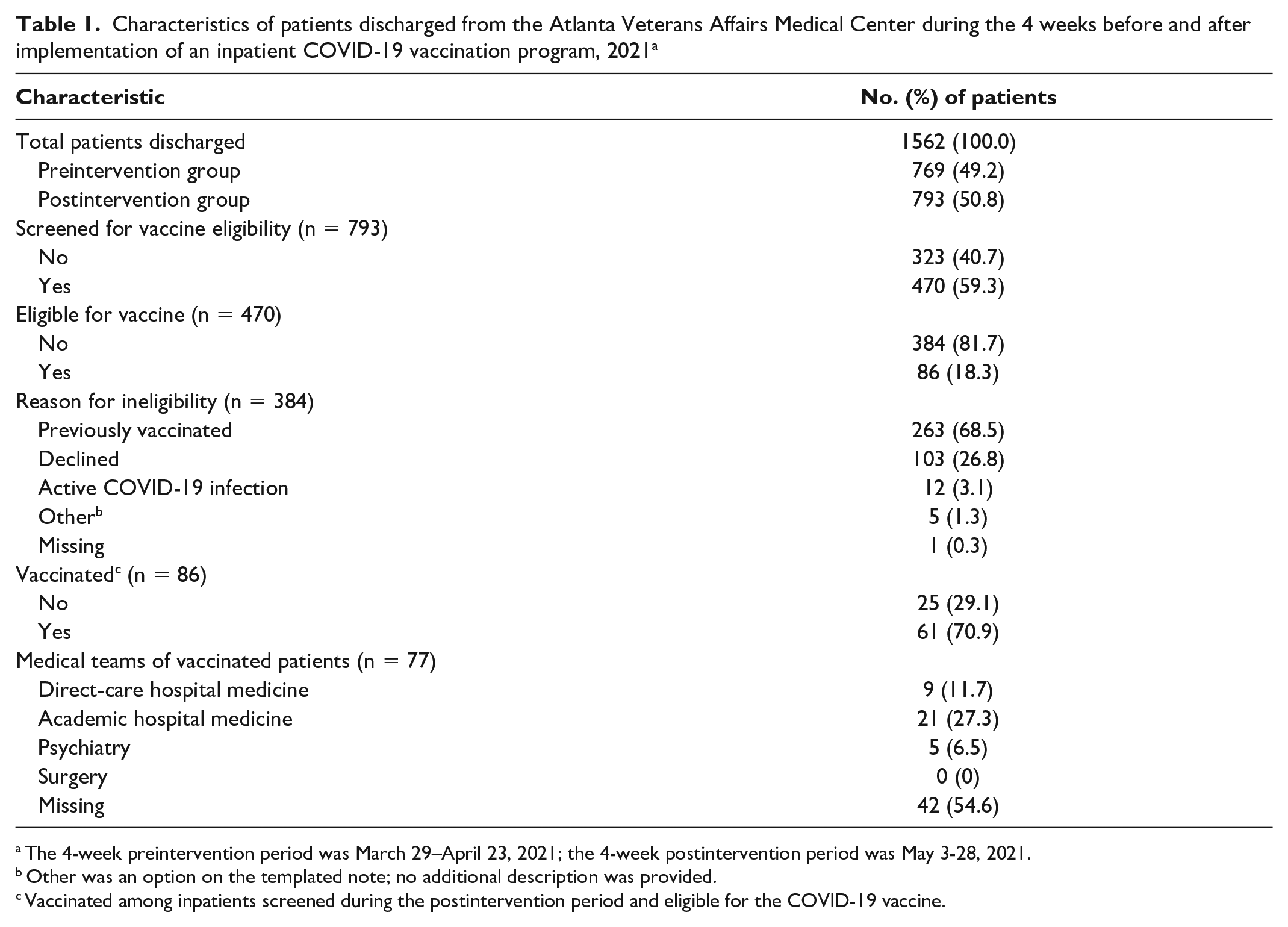
Of the 793 patients discharged after the intervention, 470 (59.3%) were screened for COVID-19 vaccine eligibility (Table 1). When we analyzed the proportion of patients by week, the rate was sustained across the 4-week postintervention period (Table 2).
Characteristics of patients discharged from the Atlanta Veterans Affairs Medical Center during the 4 weeks before and after implementation of an inpatient COVID-19 vaccination program, 2021 a
The 4-week preintervention period was March 29–April 23, 2021; the 4-week postintervention period was May 3-28, 2021.
Other was an option on the templated note; no additional description was provided.
Vaccinated among inpatients screened during the postintervention period and eligible for the COVID-19 vaccine.
COVID-19 vaccine eligibility and vaccination before discharge during the 4 weeks after implementation of an inpatient COVID-19 vaccination program at the Atlanta Veterans Affairs Medical Center, reported by week, 2021a
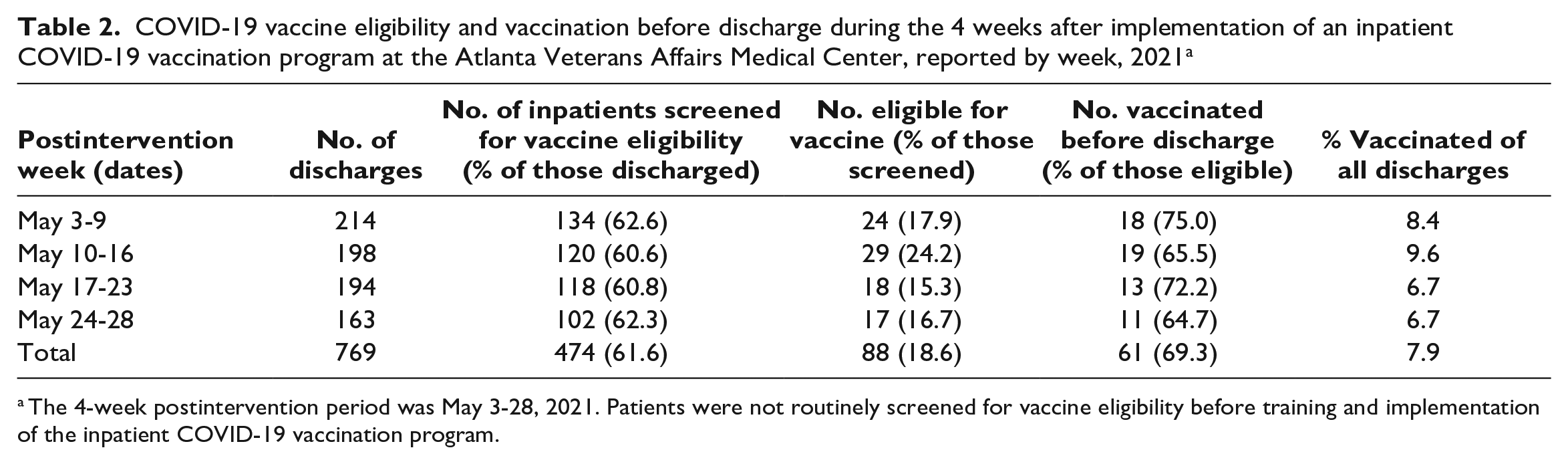
The 4-week postintervention period was May 3-28, 2021. Patients were not routinely screened for vaccine eligibility before training and implementation of the inpatient COVID-19 vaccination program.
Of 470 patients who were screened for COVID-19 vaccine eligibility, 86 (18.3%) were eligible. Among 384 patients who were ineligible for COVID-19 vaccination, the most common reasons for ineligibility were previous vaccination (n = 263, 68.5%), patient refusal (n = 103, 26.8%), and active COVID-19 infection (n = 12, 3.1%). Of 86 eligible patients, 61 (70.9%) received a COVID-19 vaccine before discharge.
During May 3-6, 2021, when patients were noted to be ambulatory or nonambulatory, among vaccine-eligible patients, 12 were nonambulatory and all 12 were vaccinated at the bedside before discharge. In contrast, among 5 patients who were eligible but ambulatory, only 3 patients attended their scheduled appointment at the outpatient vaccination center. After we adjusted the protocol so that vaccination staff administered vaccines at bedside to patients who were eligible for COVID-19 vaccination, 46 of 59 patients (78.0%) who were eligible received vaccination at bedside.
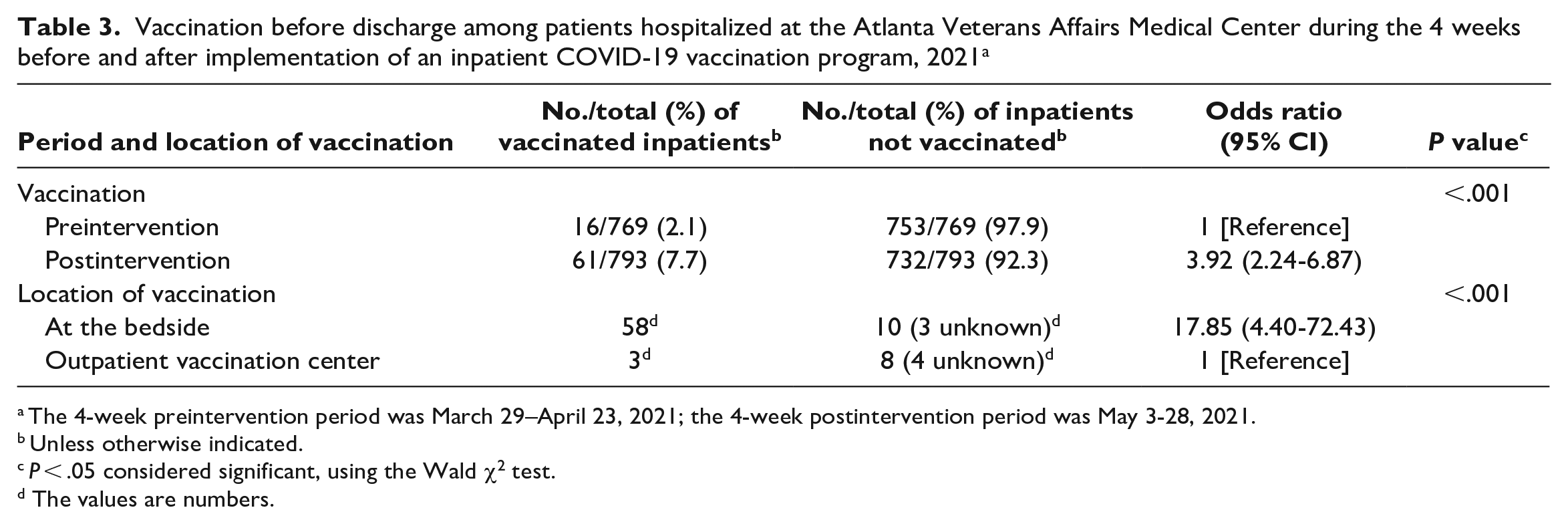
COVID-19 vaccination rates during hospitalization increased from 16 of 769 (2.1%) before the intervention to 61 of 793 (7.7%) after the intervention (P < .001) (Table 3). The unadjusted odds of COVID-19 vaccination among patients postintervention was nearly 4 times the odds of vaccination among patients preintervention (OR = 3.92; 95% CI, 2.24-6.87). Among COVID-19 vaccine–eligible and consenting patients, the unadjusted odds of COVID-19 vaccination among patients who were assigned to be vaccinated at the bedside were nearly 18 times the odds of COVID-19 vaccination among patients who were instructed to go to the outpatient vaccination center after discharge or who were scheduled for a postdischarge COVID-19 vaccination appointment (OR = 17.85; 95% CI, 4.40-72.43).
Vaccination before discharge among patients hospitalized at the Atlanta Veterans Affairs Medical Center during the 4 weeks before and after implementation of an inpatient COVID-19 vaccination program, 2021 a
The 4-week preintervention period was March 29–April 23, 2021; the 4-week postintervention period was May 3-28, 2021.
Unless otherwise indicated.
P < .05 considered significant, using the Wald χ2 test.
The values are numbers.
Discussion
With the design and implementation of our inpatient COVID-19 vaccination program, we demonstrated that a brief training period and templated note integrated into the EMR led to sustained, high rates of vaccine eligibility screening and documentation. We also showed that our inpatient COVID-19 vaccination program increased COVID-19 vaccine uptake among patients before discharge. Our findings are important because they show that hospitalizations are an alternate setting for offering vaccination that may reduce logistical barriers to vaccine access, allow opportunities to deliver evidence-based messaging about vaccines, and prioritize vaccination of patients aged ≥65 years with comorbidities who are at high risk of COVID-19–related hospitalization and death.
The strengths of our inpatient COVID-19 vaccination program that made it successful were the small-group training sessions for health care providers and staff, the integration of the program into the discharge-planning process, the templated note in the EMR, and the nurse-driven protocol. These elements are not unique to the AVMC or VA health care systems, and, theoretically, our inpatient COVID-19 vaccination program could be implemented in other health care systems, especially with federal support.
Assessing vaccination status, addressing vaccine misinformation, and offering vaccination services during the hospital discharge process are aligned with best practices recommended by the Centers for Disease Control and Prevention. 12 As previously shown, integrating influenza and pneumococcal vaccination into discharge planning is not only feasible but also effective in increasing vaccine uptake.3,6 -8 In contrast, few studies have shown how to operationalize hospital-wide inpatient COVID-19 vaccination programs. One study showed that intentional discussions between rehabilitation physicians and patients on biweekly ward rounds led to increased vaccination rates among patients in a rehabilitation ward during a 3-week period. 13 However, this intervention was limited to a single rehabilitation unit, and the intervention was not a systems-level change that could be applied to a hospital system.
Two articles in particular highlight challenges and strategies in implementing inpatient COVID-19 vaccination programs: a program at New York–Presbyterian (NYP) Hospital and another at the Priscilla Chan and Mark Zuckerberg San Francisco General Hospital and Trauma Center (ZSFG).9,10 Both hospitals integrated vaccine order sets into admission order sets, with the aim of avoiding patient discharge delays. Similarly, we integrated our vaccine screening and order set into the EMR; however, we integrated the inpatient COVID-19 vaccination program into discharge planning. One advantage to integration into discharge planning orders rather than admission orders is that patients may be temporarily ineligible for the COVID-19 vaccine at the time of admission due to acute, critical illness, and patients may be hesitant to accept COVID-19 vaccination with their symptoms. In our study, a disadvantage to waiting until discharge was that patients who were discharged after hours or on Sundays were unlikely to follow up with their scheduled COVID-19 vaccination appointment. Thus, integrating vaccine order sets earlier in the hospitalization may allow more flexibility in timing of vaccine administration.
Both the NYP and ZSFG vaccination programs began with centralized models of care, in which a limited number of designated vaccination staff were trained and deployed to patients’ bedsides to administer vaccines.9,10 Eventually, both programs switched to decentralized models, where patients’ primary nurses delivered the vaccine. In our program, we used a centralized model in which the patient’s primary treatment team screened for eligibility and requested vaccination, but a designated vaccinator from the outpatient vaccination center was deployed to administer the vaccines. This model allowed efficient training of a few staff members to rapidly start the program, rather than training every nurse in the hospital. With gained experience over time, decentralizing vaccine administration to primary nurses may reduce the additional resources needed to hire dedicated vaccination staff. Decentralizing the inpatient COVID-19 vaccination program will also be necessary if the outpatient vaccination center is closed and outpatient vaccinations are shifted to clinic settings.
One concern is that decentralized models create additional tasks for primary nurses who have multiple competing priorities in caring for inpatients. 9 Both NYP and ZSFG, as well as our program, found that administration of vaccines on the ward at the patient bedside was the most effective means of vaccinating inpatients. Another theme that was clear in both hospitals and our own program was the need to leverage multidisciplinary teams, which included physicians, inpatient nurses, dedicated vaccination staff, pharmacists, and information technology leadership.9,10
Another logistical challenge that was addressed by centralizing the inpatient COVID-19 vaccination program was avoiding vaccine waste. Because of COVID-19 vaccine stability and limited supply, especially earlier in the COVID-19 pandemic, ensuring minimal vaccine waste was a priority. Having a separate supply of COVID-19 vaccines stored in the inpatient pharmacy would risk wasting unused doses if few inpatients were eligible for vaccination, whereas using a central supply of COVID-19 vaccines obtained from the outpatient vaccination center limited waste and allowed for more convenient tracking of the single vaccine supply.
Although we had relatively high uptake of screening for COVID-19 vaccine eligibility, about 40% of patients were not screened for eligibility, with the lowest rates among patients who were seen by surgery and psychiatry treatment teams and the highest rates among patients seen by academic hospital medicine treatment teams. These rates may reflect discrepancies in knowledge, confidence, and attitudes toward the inpatient COVID-19 vaccination program by different hospital services; however, we did not formally evaluate reasons for discrepancies. Although the implementation gap in screening was addressed through additional education and reminders, understanding barriers to screening will be important to improve uptake of the inpatient COVID-19 vaccination program.
Of note, although the proportion of inpatients vaccinated before discharge increased during the 4 weeks postintervention compared with the 4 weeks preintervention, the overall proportion (7.9%) remained low. The low rate may in part be explained by the high vaccination rates in this patient population, with more than two-thirds of assessed patients reporting prior vaccination as the reason for ineligibility. Our rates are comparable to vaccination rates at ZSFG, which reported a vaccination rate of 6.7% among inpatients during its study period. 10 The article on the NYP program did not report overall vaccination rates; however, the NYP and ZSFG programs noted a downward trend in inpatient COVID-19 vaccination rates as community COVID-19 vaccination rates increased and the inpatient eligibility pool decreased. With the availability of booster vaccines, it would be interesting to reassess whether our inpatient COVID-19 vaccination program increases completion rates of the COVID-19 vaccine series.
Several challenges earlier in the COVID-19 pandemic evolved and may be less relevant today, including changes in COVID-19 vaccine eligibility criteria, recommendations on preferred COVID-19 vaccine brand, and safe COVID-19 vaccine storage. We initially had more widespread use of the Johnson & Johnson vaccine because of its single-dose administration, which was ideal for inpatients who may have had barriers to returning for a second dose. We are now only offering Moderna vaccines for our hospitalized patients. In addition, a diminishing number of patients who are interested and agreeable to receiving a COVID-19 vaccination remain unvaccinated. Thus, an increasing proportion of unvaccinated patients who are hospitalized may express COVID-19 vaccine hesitancy. Although our program focused on the feasibility of assessing and documenting COVID-19 vaccine eligibility, future work to address vaccine hesitancy through techniques such as motivational interviewing will be important to promote COVID-19 vaccine uptake among those who declined, who accounted for nearly 30% of our ineligible pool.
Limitations
Our study had several limitations. First, our inpatient COVID-19 vaccination program was implemented at a single center, specifically at an urban VA hospital, which represents a mostly older, male population with baseline high rates of COVID-19 vaccination even prior to our program. As such, our results may not be generalizable to more rural health care settings or to younger, majority female patient populations. Second, the intervention took place in April and May 2021, and much of what we know about COVID-19 vaccine eligibility, vaccine efficacy, COVID-19 variants, and the climate of vaccine hesitancy has changed since then. However, our protocols have evolved to reflect current vaccine guidance, including booster vaccines, and the features of our program that made it successful were not unique to our hospital and could be modeled at other facilities.
Conclusions
Our inpatient COVID-19 vaccination program demonstrated that a brief training period and integrated note template in the EMR are feasible, resource-efficient ways to integrate COVID-19 vaccination into discharge planning and increase vaccine uptake. Future longitudinal studies of inpatient COVID-19 vaccination programs at multiple sites may help elucidate whether integrating COVID-19 vaccination into discharge planning could help address attitudinal barriers and increase rates of booster vaccinations. Qualitative studies to assess perspectives of inpatient COVID-19 vaccination are needed to better understand barriers to screening and vaccination in this setting, and future inpatient COVID-19 vaccination programs should focus on how to increase COVID-19 vaccine uptake among those who are hesitant. Hospital settings remain an important opportunity to address COVID-19 vaccine misinformation and offer vaccination to patients at high risk for severe COVID-19 illness.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.W.F. is supported by the National Institute of Allergy and Infectious Diseases (T32AI157855).
