Abstract
The United States has a goal to eliminate hepatitis C as a public health threat by 2030. To accomplish this goal, hepatitis C virus (HCV) care cascades (hereinafter, HCV cascades) can be used to measure progress toward HCV elimination and identify disparities in HCV testing and care. In this topical review of HCV cascades, we describe common definitions of cascade steps, review the application of HCV cascades in health care and public health settings, and discuss the strengths and limitations of data sources used. We use examples from the Massachusetts Department of Public Health as a case study to illustrate how multiple data sources can be leveraged to produce HCV cascades for public health purposes. HCV cascades in health care settings provide actionable data to improve health care quality and delivery of services in a single health system. In public health settings at jurisdictional and national levels, HCV cascades describe HCV diagnosis and treatment for populations, which can be challenging in the absence of a single data source containing complete, comprehensive, and timely data representing all steps of a cascade. Use of multiple data sources and strategies to improve interoperability of health care and public health data systems can advance the use of HCV cascades and speed progress toward HCV elimination.
Hepatitis C is a serious liver disease that can cause cirrhosis, liver cancer, and death. Direct-acting antiviral medications cure most people with hepatitis C in 8-12 weeks. 1 The US Department of Health and Human Services plans to eliminate hepatitis C as a public health threat by 2030. 2 Hepatitis C virus (HCV) care cascades (hereinafter, HCV cascades) can be used to measure progress toward elimination, evaluate programs and policies, provide population-level longitudinal surveillance, offer insights into clinical care, and identify disparities in HCV testing and care. The objective of this topical review is to describe HCV cascades and review the application of HCV cascades in health care and public health settings, including a description of data sources used. We provide a case study of HCV cascades in Massachusetts and explore health informatics solutions to optimize continuous monitoring of HCV cascades from health system, jurisdictional, and national perspectives.
For this topical review, we conducted a literature search in MEDLINE/PubMed on September 24, 2021, using the following search terms: “hepatitis C/ or (hepatitis-C or hepacvirus OR HCV).ti,ab. AND (care cascades or cure cascades or care continuum or cure continuum).ti,ab.” We placed no restrictions on language or time frame. We reviewed citations in the returned articles to identify additional relevant articles. This project was reviewed by the Centers for Disease Control and Prevention (CDC), was exempted from institutional review board oversight because it did not involve human subjects, and was conducted consistent with applicable federal law and CDC policy (eg, 45 CFR 46.102[e]).
HCV Cascade Components
An HCV cascade, also referred to as an HCV clearance, cure, or care cascade or continuum, characterizes steps from HCV screening to postcure monitoring and tracks the proportion of people who complete each step; dropoffs between cascade steps can highlight areas for improvement. HCV cascades vary in their purpose, data sources, and definitions, although standardized cascades have been proposed globally 3 and in the United States. 4
HCV cascades can be constructed from multiple data sources: public health surveillance data, including laboratory and case reporting 5 ; medical records 6 ; prevention, linkage, and care coordination program data 7 ; quality improvement data 8 ; population-based studies 9 ; health services claims data 10 ; and interviews or surveys of people living with or at risk for HCV. 11 The means by which data are collected and managed affect the quality, volume, completeness, and timeliness of data. For public health surveillance data, disease-reporting requirements vary by jurisdiction; states voluntarily submit case reports to CDC. Cases reported to the health department are typically prompted by laboratory reporting. Cascades should be interpreted in the context of their intended use, data sources, and data collection methods. HCV cascades include 1 or more components with multiple possible steps per component (Figure):
HCV screening (HCV antibody) and diagnosis (HCV RNA);
Referral, linkage to care, and evaluation for treatment;
HCV treatment and cure; and
Monitoring for hepatocellular carcinoma and reinfection.
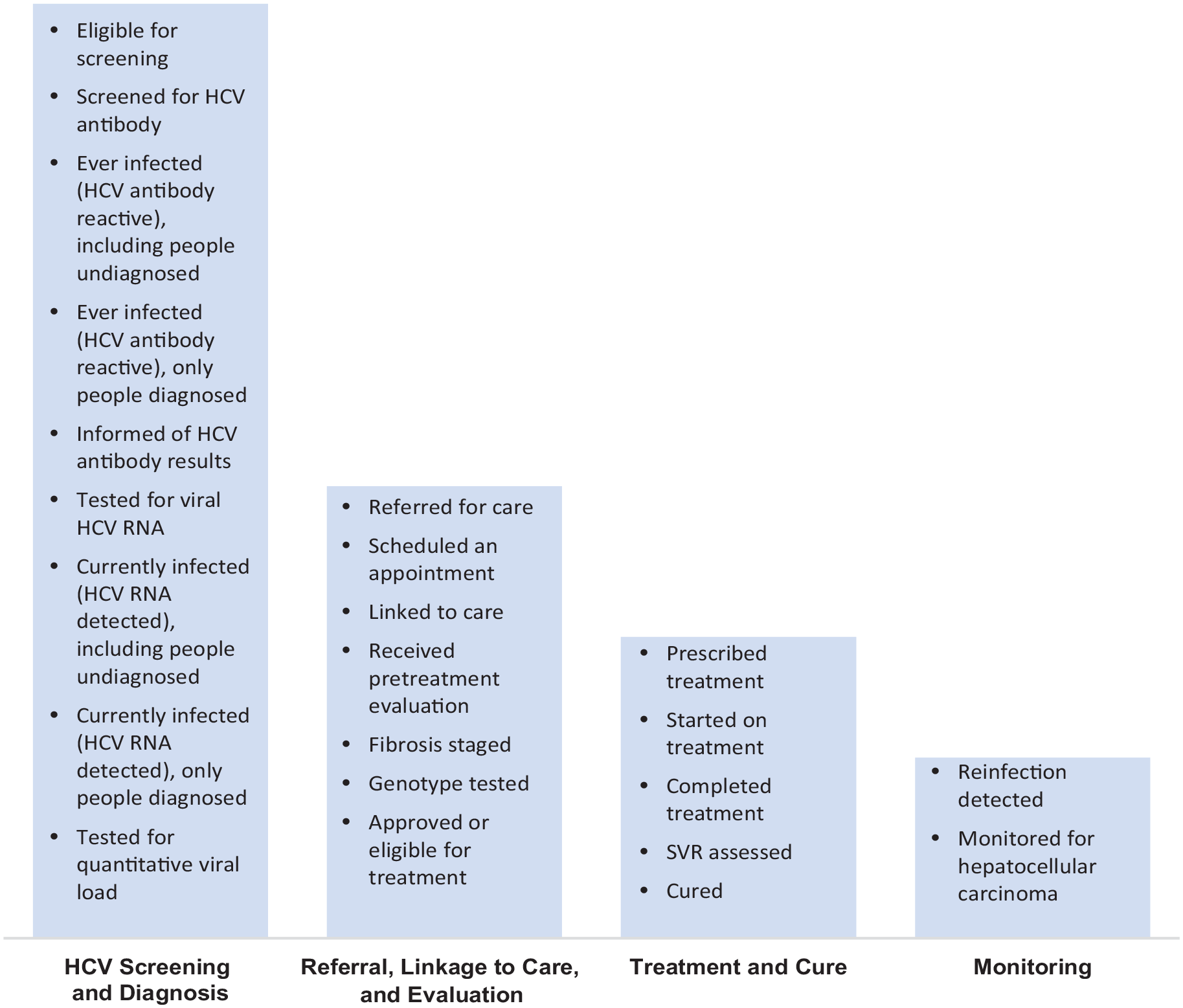
Common hepatitis C virus (HCV) care cascade components and steps. HCV care cascades include multiple steps across 4 main components. The steps within each component are selected depending on the purpose of the cascade and the data sources available. Definitions for each step can vary between cascades, which limits the ability to compare across cascades developed for different purposes. Abbreviation: SVR, sustained virologic response.
HCV cascades often begin with the number of people who screen positive for HCV antibody during a specified time frame, which is commonly available through surveillance or clinical records. Some cascades include the number of people screened or undiagnosed,12-15 which can help identify HCV-screening gaps. In addition to HCV antibody, the number tested for and confirmed to have detectable HCV RNA should be documented to distinguish active viremia from past exposure.
The steps between diagnosis and treatment include referral to a health care provider, linkage to care, and treatment evaluation. Ideally, a cascade should document the steps closer to linkage than to referral because this step better identifies opportunities for clinical or public health intervention. However, the actual steps vary based on data sources available. Public health surveillance systems are often unable to directly measure linkage to care. In Philadelphia, repeat HCV RNA testing in public health surveillance data was used as a linkage-to-care proxy. 5 These referral, linkage, and treatment evaluation steps are more easily measured in health systems than in public health surveillance systems. Some examples include referrals recorded by treatment coordinators at a clinical site, 16 referral orders extracted from electronic health records (EHRs), 17 reminders to make an appointment with an HCV treatment provider, 18 or recorded attendance at a clinic appointment. 8
Prior to initiating treatment, patients might be screened for treatment eligibility, related to health insurer–imposed treatment restrictions based on sobriety, fibrosis, or prescriber type. Fibrosis staging and genotype testing have been included in cascades, both as proxies of linkage to care10,19 and as independent cascade steps as part of evaluation for treatment. 20 With evolving, simplified treatment guidelines, 1 newer pangenotypic treatment options, and policy reforms that eliminate treatment restrictions, 21 steps such as genotype testing 22 become increasingly less relevant.
The next HCV cascade component measures the number of people who are treated and cured during a specified time frame. Certain steps can be measured through EHR or prescription databases. 23 Some programs track treatment initiation and completion through case management data. 24 While these detailed HCV cascades can be useful to guide clinical quality improvement interventions, they are not feasible to implement at a population level. Subtle variations in definitions of treatment (eg, prescribed, initiated, or completed treatment) make comparisons across published cascades challenging. Many HCV cascades include a separate step for HCV cure, defined clinically as sustained virologic response at 12 weeks after completion of therapy (SVR-12). 1 Because not all people return for testing after 12 weeks, SVR-12 might underestimate the number of people who are cured, in contrast with definitions that rely on a single reported negative HCV RNA test result. 4
After HCV cure, people with cirrhosis require monitoring for hepatocellular carcinoma. People with ongoing HCV exposure, particularly people who inject drugs, require access to harm reduction services and repeat HCV testing to identify reinfection. Few HCV cascades include reinfection as an additional step.25,26 Defining reinfection and distinguishing reinfection from treatment failure or virologic relapse is challenging without intensive investigation, but the importance of tracking reinfections is recognized, 27 and efforts to systematically define and detect reinfections are emerging. 28
Applications of HCV Cascades
We describe HCV cascades from 3 perspectives: health systems, public health jurisdictions, and national. Cascades from each perspective are constructed using various data sources and systems, are used for various purposes, and have unique advantages and challenges.
Health Systems Perspective
Most published HCV cascades have been constructed in health systems settings and reflect the characteristics of the patient population or facilities in that system. Health care providers, health insurers, and administrators use health systems HCV cascades to evaluate outcomes of screening and treatment programs,16,29 guide quality improvement efforts,8,30 and identify disparities in HCV treatment.31,32 Clinical data from EHR systems are invaluable for constructing health systems–level HCV cascades and contain demographic, clinical, and treatment data that are often not available to public health departments. Health systems–based HCV cascades can provide near–real-time understanding of HCV testing and treatment occurring within their patient population, providing actionable data to inform changes in practice. The Cherokee Nation Health Services constructed an HCV cascade from EHR data and an HCV treatment database to document improvements in HCV testing, treatment, and cure related to an HCV elimination program. 33 The Veterans Health Administration leveraged the Corporate Data Warehouse, a patient-level database with demographic, laboratory, treatment, and clinical encounter information, combined with an HCV Clinical Case Registry to construct a cascade of HCV care for veterans across the United States.6,34 The Veterans Health Administration used the HCV cascade to monitor the impact of interventions to improve HCV testing and treatment. 29
Production of HCV cascades for use by health systems can be challenging and may rely on manual processes that make it difficult to scale up and maintain. Automated processes to extract EHR data and develop cascades are needed, such as those implemented by health researchers from urban medical centers in New York City and Atlanta.35,36 Use of EHR data to generate HCV cascades may also rely on defining and maintaining laboratory (eg, Current Procedural Terminology codes) and prescription code sets, which may be labor intensive and infeasible for many health systems. Standardized, open-source codes from EHR systems would increase accessibility for health systems with fewer staff resources. Testing, care, and treatment that occur outside a health system are difficult to capture in an EHR; therefore, cascades generated from a health system’s EHR can underestimate progression at each step. 36
Public Health Jurisdictions’ Perspectives
State and local health departments develop population-based cascades to measure progress in achieving jurisdiction elimination goals,37,38 identify disparities by subpopulations, 9 and monitor engagement in HCV treatment. 5 The primary data source is often HCV surveillance data, which historically rely on laboratory results and clinician-submitted case reports. CDC has produced programmatic guidance for state and local health departments to develop population-based HCV cascades from laboratory data. 4 Ideally, timely, complete, person-level laboratory data would be received electronically and managed in a longitudinal database to optimize utility. In addition, identifying people who are cured in a laboratory-based cascade relies on receiving negative HCV RNA test results, which are not reportable in all jurisdictions.
State and local public health agencies have an interest in evaluating HCV-related policies and regulations, including uptake of screening guidelines and the impact of treatment restrictions. Health insurance claims, such as all-payer claims or state Medicaid program data, can be used to monitor components of the HCV cascade. 13 Many health departments invest in hepatitis C prevention, care, and treatment access programs; cascades constructed from program data are a useful tool to evaluate investments made in these publicly supported programs.
Data quality and completeness can be a challenge when using surveillance systems, because information such as risk history, race, and ethnicity needed to generate population-specific cascades is often missing. Surveillance systems may receive only reports related to initial case identification and may not receive negative test results needed to evaluate cascade progression. Enacting policies to enable reporting of negative test results would improve utility. While interoperability between EHRs and health department surveillance systems is improving, the extent to which longitudinal reporting mechanisms are available is unclear. The technical and analytic capacity of health departments is highly variable, and tools such as code sets, training to use administrative claims, and capacity to analyze and visualize administrative data would be beneficial.
National Perspectives
Federal agencies, national health care and advocacy organizations, and policy makers can use a national HCV cascade to measure progress toward HCV elimination goals, identify disparities in elimination progress, and direct public health resources efficiently. National HCV screening and treatment databases have been used to construct national, population-based HCV cascades in the country of Georgia 39 but are not available in the United States. Data approaches to constructing national HCV cascades in the United States have included modeling,14,40 literature reviews, 41 extrapolation from cohort and population-based studies, 42 administrative claims databases, 10 and systematic reviews and meta-analyses. 43 The National Health and Nutrition Examination Survey (NHANES) collects population-based data through interviews and laboratory testing. NHANES is central to estimating national HCV prevalence and can estimate the proportion of people who are unaware of their infection42-44; however, NHANES misses certain populations with a disproportionate prevalence of hepatitis C in its sampling frame, including people who are incarcerated or experiencing unsheltered homelessness (homeless shelters are included in the sampling frame).
Moving forward, new data sources and approaches are needed to construct comprehensive HCV cascades. Health informatics strategies such as privacy-preserving record linkages could be used to merge large, national datasets at the person level. 45 These combined datasets could be used to construct national HCV cascades that include data on testing and treatment. A research team in Australia has developed a national cohort to monitor blood-borne viruses, including HCV, by conducting automated, anonymous, patient-level linkage across health clinics, testing sites, and laboratories. 46 An important limitation to using large health care databases for producing HCV cascades is an inherent selection bias toward including people with better health care access. People who inject drugs are at increased risk for HCV infection 47 and are more likely than people who do not inject drugs to lack health insurance, have negative experiences with health care providers, and experience barriers to accessing health care (eg, transportation difficulties or inflexible work schedules).48,49
Case Study From the Massachusetts Department of Public Health (MDPH)
MDPH uses HCV cascades to (1) monitor progress toward hepatitis C elimination, (2) evaluate and monitor implementation of screening and treatment policies and guidelines, (3) monitor health department–funded screening and linkage programs, and (4) support clinical and operational quality improvement. MDPH leverages multiple data sources to generate various cascades, because no single data source provides complete, comprehensive, and timely data. Each cascade serves a distinct purpose. The strengths and limitations of these data sources are described hereinafter (Table).
Data sources, strengths, and limitations of the Massachusetts hepatitis C virus (HCV) care cascade
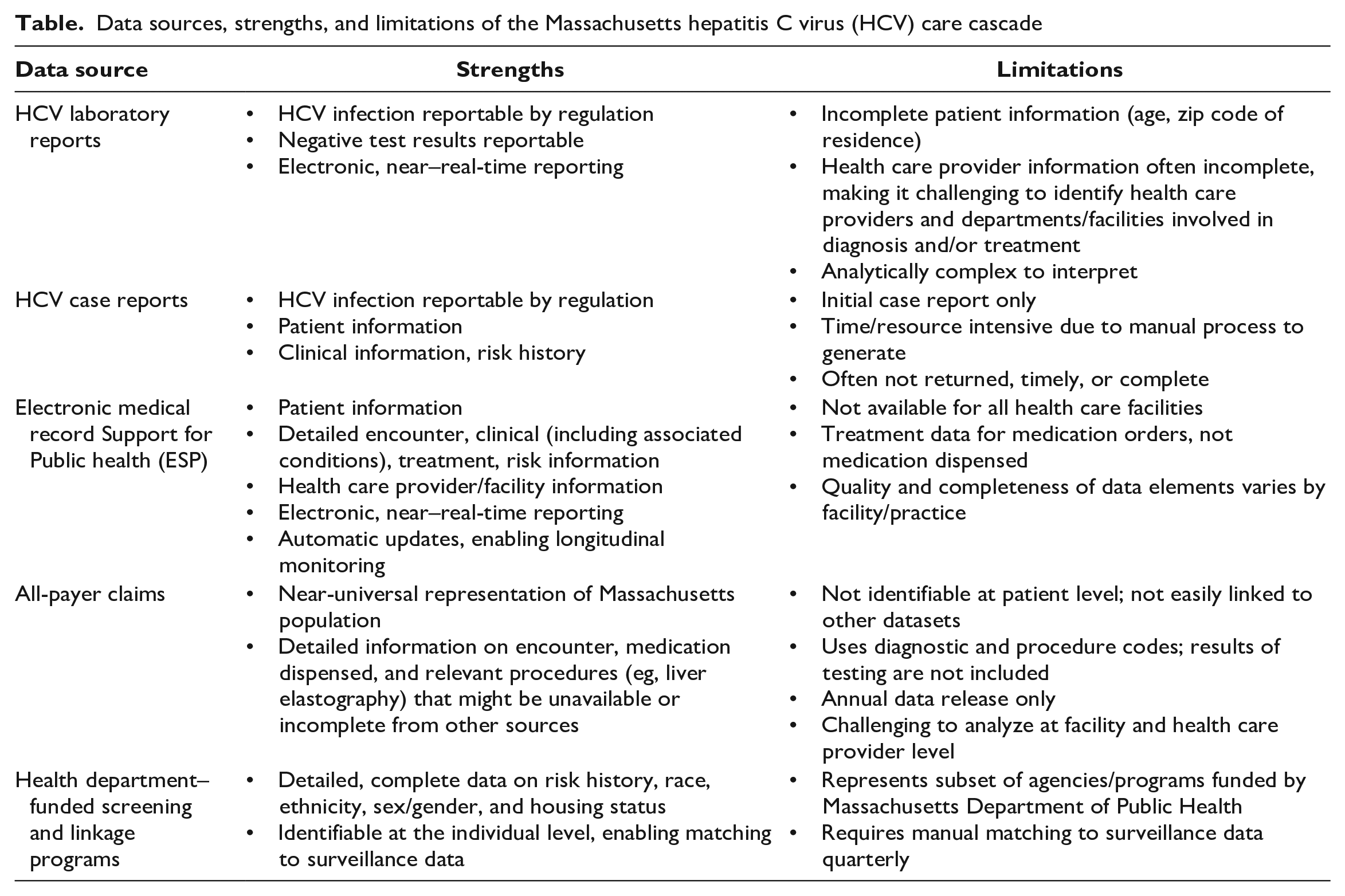
To monitor hepatitis C elimination, MDPH mainly uses a cascade constructed from HCV surveillance data. 37 All laboratories conducting HCV testing on Massachusetts residents are required to report evidence of HCV infection to MDPH as well as all negative antibody and HCV RNA test results. Laboratory reports contain limited patient information (eg, patient age, zip code of residence). MDPH mails a single-page case report form to the health care provider to collect patient demographic, clinical, and risk history information. Clinician-completed case report forms are often incomplete and pertain only to the initial case report, therefore lacking treatment information.
Laboratory and case report data in this cascade are supplemented by EHR data provided through MDPH’s Electronic medical record Support for Public Health (ESP) platform,50,51 which covers approximately 64% of Massachusetts residents. ESP uses algorithms composed of diagnosis codes, symptoms, laboratory results, and prescriptions to identify reportable HCV cases. Data reported to MDPH include patient demographic characteristics, laboratory results, encounters, clinical information, medications, risk history, and information about ordering and treating health care providers. Data are updated as new information is recorded in the EHR. Cascade development using surveillance data is time and labor intensive because of the complexity and volume of data analyzed.
MDPH has generated HCV cascades using the Massachusetts All Payer Claims Database to evaluate state policies and national guidelines. 52 This database includes health claims data for nearly all Massachusetts residents during 2016-2020. Individuals screened for HCV using a combination of diagnostic and procedure codes are followed through treatment based on a combination of laboratory tests performed and prescriptions dispensed. Claims data include limited demographic data, thus restricting the ability to generate cascades for subpopulations. The complexity of analyzing health care provider and facility information hinders the ability to use the cascade to inform facility- or provider-specific quality improvement activities.
Lastly, MDPH generates HCV cascades to monitor its funded programs and support quality improvement. MDPH contracts with 40 clinical and nonclinical agencies across the state to provide integrated HIV, HCV, and sexually transmitted infection testing and linkage to treatment services in various settings, including syringe exchange programs, substance use treatment facilities, and correctional facilities. All testing is performed by the Massachusetts State Public Health Laboratory. Approximately one-fifth of all HCV cases reported annually come from these funded agencies. The granular data available (race, ethnicity, age, risk history, sex/gender, current housing status, incarceration history, and linkage to treatment) provide an essential tool to advance health equity. MDPH produces agency-specific HCV cascades and provides technical assistance to highlight opportunities for operational changes, such as refining procedures, adjusting workflows, or strengthening collaboration with referring agencies. MDPH is developing capacity to leverage ESP infrastructure to automate HCV cascade reports for participating facilities, which would provide a near–real-time tool for clinical and operational quality improvement.
HCV Cascades and Health Disparities
From all 3 perspectives, it is important to consider how HCV cascades can be developed for populations that are disproportionately affected by hepatitis C. HCV cascades have been developed for people who use drugs, 53 people living with HIV,54,55 people experiencing homelessness, 56 people who are recently incarcerated,57,58 and American Indian and Alaska Native people. 33 Researchers in New York City constructed an HCV cascade that encompassed all 12 jail facilities in the city by extracting data from the Correctional Health Services’ EHR. 58 The researchers used the findings to identify opportunities to increase HCV testing and treatment for a large number of HCV-infected individuals. Examining HCV cascades for certain populations will help to monitor whether progress toward HCV elimination is being achieved equitably. Health informatics strategies to produce national HCV cascades should consider how to incorporate demographic and risk history information to stratify HCV cascades by specific populations.
Public Health Implications
Automating cascade development will be critical for improving timeliness, sustainability, and scalability of HCV cascades. For health systems, HCV cascades can be improved through efforts to ensure open-source terminology and value sets for definitions of cascade steps. Public health departments need sufficient resources and staffing to develop and maintain data systems for longitudinal, patient-level surveillance of hepatitis C. Ideally, these data systems should be able to receive and deduplicate electronic laboratory results 59 and supplement with longitudinal clinical data from EHRs. 60 Health departments can use CDC’s program guidance on laboratory-based HCV cascades to improve consistency of HCV cascades across jurisdictions. 4 Mandated reporting of all HCV RNA test results, including negative HCV RNA test results, is essential for identifying people who have cured or cleared HCV infection. Informatics solutions to improve data interoperability across multiple health systems and public health surveillance systems are emerging and will be important to explore for future development of population-level cascades. 61 Integrating additional data sources, including health insurance claims, commercial laboratory, prescription databases, and vital statistics, can further improve data completeness.
These efforts complement CDC’s Data Modernization Initiative objective to increase data linkages across diverse datasets. 62 Strengthening collaborations between health systems and public health is critical for improving HCV cascade quality and using HCV cascade data to drive public health action. Moving forward, HCV cascades will continue to be an important tool to evaluate progress toward HCV elimination in the United States.
Footnotes
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Monica Morrison and Liisa Randall are employed by the Commonwealth of Massachusetts, Department of Public Health, which is a recipient of Integrated Viral Hepatitis Surveillance and Prevention Funding for Health Departments (CDC-RFA-PS21-2103).
