Abstract
During 1990-2019, universal infant and childhood vaccination for hepatitis B resulted in a 99% decline in reported cases of acute hepatitis B among children, adolescents, and young adults aged <19 years in the United States; however, during 2010-2019, cases of acute hepatitis B plateaued or increased among adults aged ≥40 years. We conducted a topical review of surveillance strategies that will be critical to support the elimination of hepatitis B as a public health threat in the United States. In 2019, notifiable disease surveillance for acute hepatitis B showed continued transmission, especially among people who inject drugs and people with multiple sexual partners; rates were highest among people who were aged 30-59 years, non-Hispanic White, and living in rural areas. In contrast, newly reported cases of chronic hepatitis B (CHB) were highest among people who were aged 30-49 years, Asian or Pacific Islander, and living in urban areas. The National Health and Nutrition Examination Survey documented the highest CHB prevalence among non–US-born, non-Hispanic Asian people during 2013-2018; only one-third of people with CHB were aware of their infection. In the context of universal adult vaccination (2022) and screening (2023) recommendations for hepatitis B, better data are needed to support programmatic strategies to improve (1) vaccination rates among people with behaviors that put them at risk for transmission and (2) screening and linkage to care among non–US-born people. Surveillance for hepatitis B needs to be strengthened throughout the health care and public health systems.
Although asymptomatic in 85% of infants and 50% of older children and adults, hepatitis B virus (HBV) infection can cause an acute illness of variable severity characterized by fever, malaise, anorexia, nausea, vomiting, abdominal pain, and jaundice. 1 HBV spreads through exposure to blood and body fluids: vertically to infants from pregnant people who are hepatitis B surface antigen (HBsAg) positive 2 and horizontally (person to person), for example, among family members, 3 sexual partners, 4 people who inject drugs,4-6 and people in health care settings with inadequate infection control.7,8
Most people with acute hepatitis B resolve their infection 9 ; they have detectable hepatitis B core antibody (anti-HBc) and undetectable HBsAg, indicating past infection. 10 However, some develop chronic hepatitis B (CHB) 9 and remain positive for anti-HBc, HBsAg, and HBV DNA. 10 Complications from CHB include cirrhosis,11,12 hepatocellular carcinoma (HCC),12,13 and death. 14 These outcomes vary as a function of age at the time of initial infection. Without preventive measures, 90% of infants born to HBsAg-positive and hepatitis B e antigen–positive pregnant people develop HBV infection, as do 5% to 20% of infants born to HBsAg-positive and hepatitis B e antigen–negative pregnant people. 15 Among infected infants, up to 90% develop CHB, 15 yet <10% of people aged ≥30 years who acquire HBV infection develop CHB. 9 Perinatal, infant, and childhood vaccination programs in Alaska,16,17 Taiwan, 18 and Korea 19 have reduced the incidence of acute hepatitis B 16 and the prevalence of CHB18,19 and HCC17,19,20 among children, adolescents, and young adults.
In the United States, according to data from the National Notifiable Diseases Surveillance System (NNDSS), cases of acute hepatitis B declined 87%, from a high of 26 654 in 1985 to 3350 in 2010, after implementation of national vaccination strategies 21 (Figure 1). However, during 2010-2019, hepatitis B incidence remained stable at approximately 3000 cases per year in the United States and even increased in some areas, including Central Appalachian states. 22 Consistent with the World Health Organization 23 strategy to eliminate viral hepatitis, the US Department of Health and Human Services 24 published a strategic plan to eliminate it as a public health threat by 2030. Goals include reducing acute hepatitis B infections by 90%, increasing hepatitis B birth dose vaccination rates to 90%, increasing the proportion of people with hepatitis B infection who are aware of their infection to 90%, reducing hepatitis B–related deaths by 65%, and reducing disparities in awareness and deaths among people who are non-Hispanic Black and Asian and Pacific Islander. 24 This topical review provides an overview of surveillance systems that can be used to measure progress toward national elimination goals.
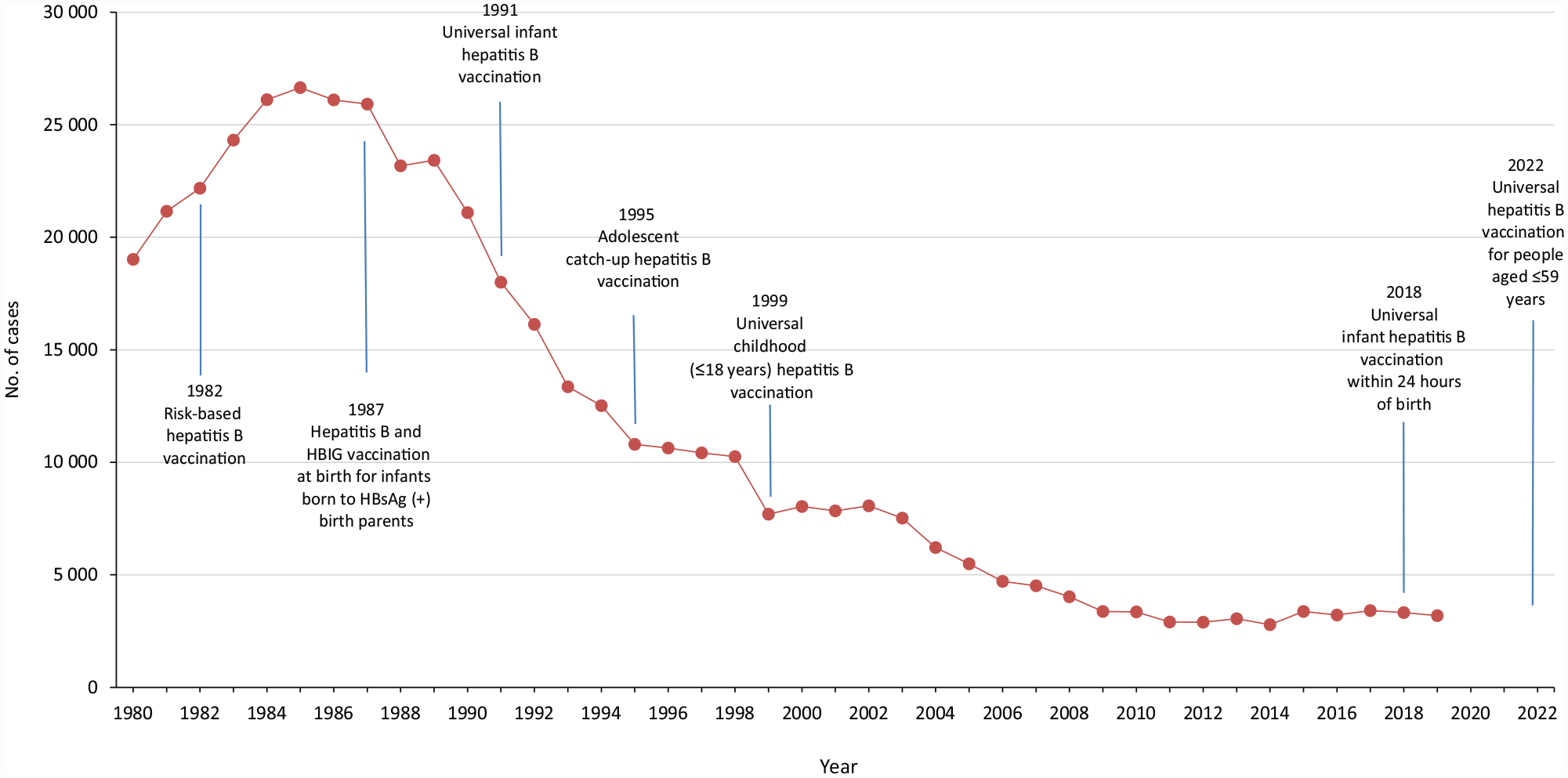
Reported cases of acute hepatitis B and key hepatitis B vaccine recommendations from the Centers for Disease Control and Prevention (CDC), United States, 1980-2022. Data sources: CDC,21,25-29 Schillie et al, 30 and Weng et al. 31 Abbreviations: HBIG, hepatitis B immune globulin; HBsAg, hepatitis B surface antigen.
Vaccination, Screening, and Testing Strategies in the United States, 1982-2023
The first hepatitis B vaccine, licensed in the United States in 1982, 26 was recommended for health care workers with risk of exposure to blood, staff and clients of institutions for people with developmental disabilities, men who have sex with men, people who inject drugs, and other people at increased risk for HBV infection 25 (eTable 1 in Supplemental Material). Hepatitis B vaccine and immune globulin were recommended for infants born to HBsAg-positive pregnant people 25 (eTable 2 in Supplemental Material).
Nonetheless, the incidence of hepatitis B continued to rise in adults after 1982. In sentinel surveillance data from 1982 to 1988, primary risk behaviors for acute hepatitis B included injection drug use and heterosexual and male-to-male sexual activity. 32
In 1984, hepatitis B immune globulin within 12 hours of birth and hepatitis B vaccine within 7 days of birth were first recommended for infants born to HBsAg-positive pregnant people 33 ; in 1988, the Advisory Committee on Immunization Practices (ACIP) first issued comprehensive recommendations for universal screening for HBsAg in all pregnant people during the first trimester. 34
In 1991, ACIP recommended universal infant hepatitis B vaccine. 27 By 1999, this recommendation was expanded to universal hepatitis B vaccine for all children, adolescents, and young adults (aged <19 y). 29 In 2018, ACIP recommended universal hepatitis B vaccine within 24 hours of birth for all medically stable infants weighing ≥2000 g and adopted the 2016 American Association for the Study of Liver Diseases recommendation to test for HBV DNA among HBsAg-positive pregnant people and to offer antiviral therapy for HBV DNA levels ≥200 000 IU/mL.30,35
In 2022, ACIP recommended hepatitis B vaccination for all adults aged 19-59 years and for adults aged ≥60 years with risk factors. 31
Centers for Disease Control and Prevention (CDC) guidelines, first published in 2008 36 and updated in 2017, 37 recommend hepatitis B testing for the following populations: people born in regions of high or intermediate HBV endemicity, people who have ever injected drugs, men who have sex with men, people with HIV or hepatitis C, and others (people needing immunosuppressive therapy; people with elevated alanine transaminase or aspartate transaminase of unknown etiology; donors of blood, plasma, organs, tissues, or semen; people with end-stage renal disease, including those on dialysis; all pregnant people; infants born to HBsAg-positive pregnant people; household, needle-sharing, or sexual contacts of those who are HBsAg positive; people who are the source of blood or body fluids for exposures that might require postexposure prophylaxis; and inmates of correctional facilities) (eTable 3 in Supplemental Material). However, risk-based testing recommendations have not translated into adequate testing rates. In population-based studies of insured people and refugees who are Asian and Pacific Islander, testing rates are estimated at 30% to 60%.38-40 Among people in the United States with CHB, 50% were estimated to be aware of their infection in an analysis of data from the January 2017–March 2020 National Health and Nutrition Examination Survey (NHANES). 41 Ogawa et al 42 estimated that only 18.6% of privately insured people with CHB had been diagnosed during January 2007–December 2014 based on a comparison of expected rates from NHANES and observed rates from US Truven Health MarketScan data. As a result, CDC now recommends onetime screening of all adults for hepatitis B with a triple panel (HBsAg, total anti-HBc, and hepatitis B surface antibody). 43
Surveillance Strategies and Findings
Vaccine Coverage Surveillance
National surveillance for vaccine coverage is accomplished primarily through the National Immunization Survey (NIS), 44 which measures immunization rates among children aged 19-35 months and adolescents aged 13-17 years; the National Health Interview Survey (NHIS), 45 which collects data on vaccination status for adults aged ≥18 and ≥50 years, health care workers, pregnant women, and children; and other surveys. 46 According to NIS, for children born during 2017-2018, the completion rate for the hepatitis B vaccine birth dose was 78.4% and that of the 3-dose series at age 24 months was 91.9% 47 ; in 2020, the completion rate among children aged 13-17 years was 92.3%. 48 According to NHIS, in 2018 hepatitis B immunization rates were 40.3% among adults aged 19-49 years, 19.1% among those aged ≥50 years, and 33.0% among people with chronic liver disease. 49 Low response rates are a limitation in NIS and NHIS.47-49 Hepatitis B vaccination rates among people who inject drugs, 50 refugees, 51 and men who have sex with men52,53 are suboptimal, and coverage data for these risk groups are not available through NHIS. 49 Additional sources of vaccine coverage data are immunization information systems, 54 which contain records of immunizations administered within a geopolitical region.
Perinatal Hepatitis B Prevention Program
The Perinatal Hepatitis B Prevention Program (PHBPP) has funded perinatal hepatitis B coordinators in multiple state, local, and territorial jurisdictions and freely associated states 55 since 1990. 56 Coordinators use enhanced case management to ensure that infants born to HBsAg-positive pregnant people are identified and receive vaccination and postvaccine serologic testing. 56
For 9950 infants born to HBsAg-positive pregnant people enrolled in PHBPP during 2018, 9617 (97%) received the hepatitis B vaccine and hepatitis B immune globulin birth dose; 8609 (87%) completed the 3-dose hepatitis B vaccine series by age 12 months; and 6828 (69%) had postvaccine serologic testing. 57 Twenty-three infants born in 2018 were HBsAg positive after postvaccine serologic testing, 57 an 89% decline from a peak of 113 in 2001. 58
One limitation to PHBPP data is that about half the infants estimated to be perinatally exposed annually are identified58,59; another limitation is that some pregnant people arrive at delivery with HBsAg status unknown.60-62 Modeling predicted a baseline perinatal hepatitis B infection rate of 3.8%, as compared with PHBPP-measured rates of 0.8% to 0.9%, for an estimated 952 infants who developed CHB in 2009. 63
National Notifiable Diseases Surveillance System
Notifiable disease surveillance in the United States is conducted for acute hepatitis B, 64 newly reported cases of CHB, 65 and perinatal hepatitis B. 66 According to data from NNDSS, 21 during 1990-2019, cases of acute hepatitis B fell 99% (from 2180 to 13) among people aged 0-19 years and 97% (from 7261 to 218) among those aged 20-29 years, but case reports plateaued or increased during 2010-2019 among adults aged ≥40 years (Figure 2). In 2019, 3192 cases of acute hepatitis B were reported, for a national incidence rate of 1.0 per 100 000 population, and 17 infants were reported with perinatal hepatitis B. 67 The incidence of acute hepatitis B was highest among people who were aged 30-59 years, male, non-Hispanic White, and living in rural areas (Table). The most common risk factors for cases with complete information were among people reporting use of injection drugs (631 of 1780, 35%) and multiple sexual partners (241 of 1042, 23%). 67 For newly identified cases of CHB in 2019, the rate was highest among people who were aged 30-49 years, male, Asian and Pacific Islander, and living in urban areas. 67
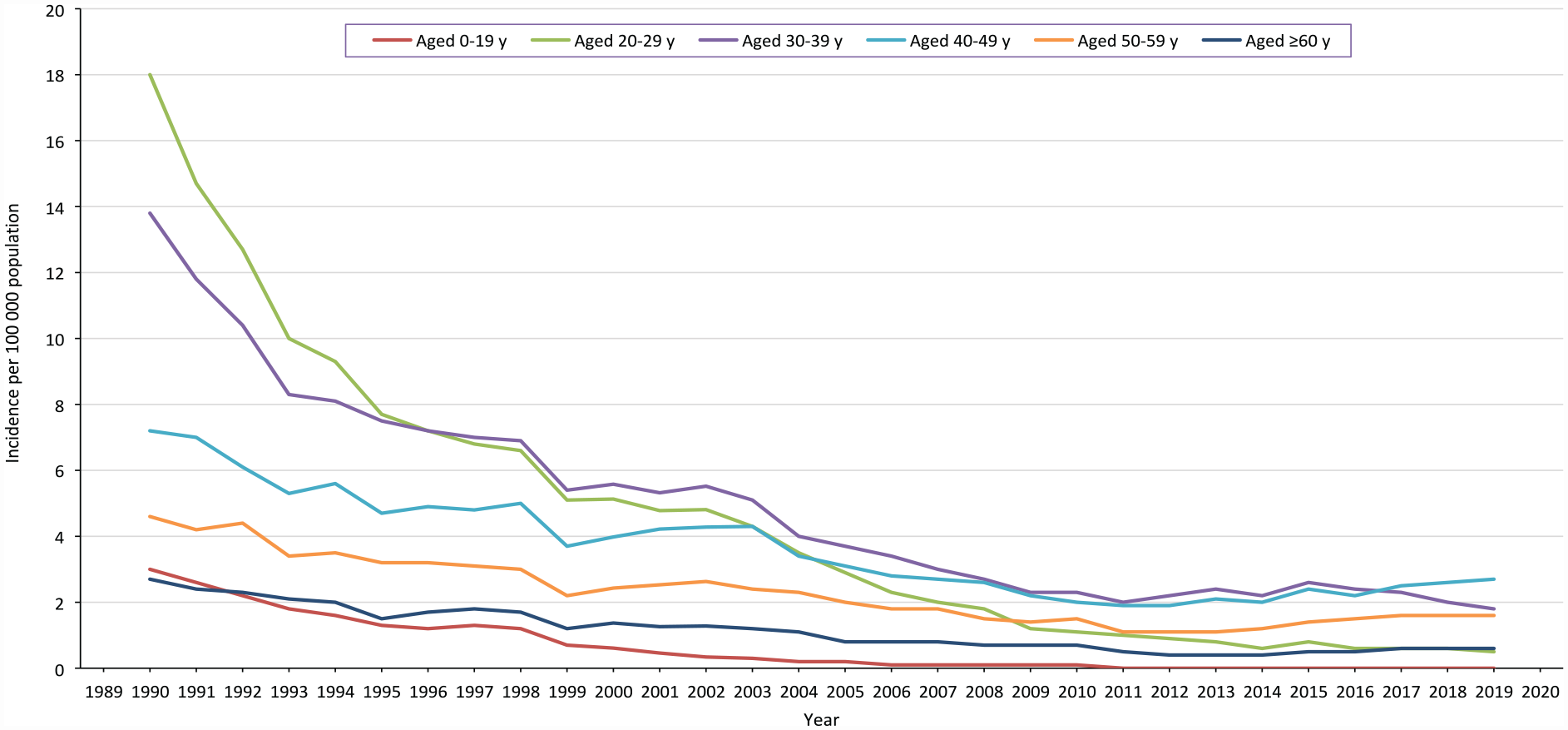
Age-specific incidence of acute hepatitis B, United States, 1990-2019. Data source: National Notifiable Diseases Surveillance System. 21
Characteristics of people with newly reported cases of acute and chronic hepatitis B, United States, 2019 a
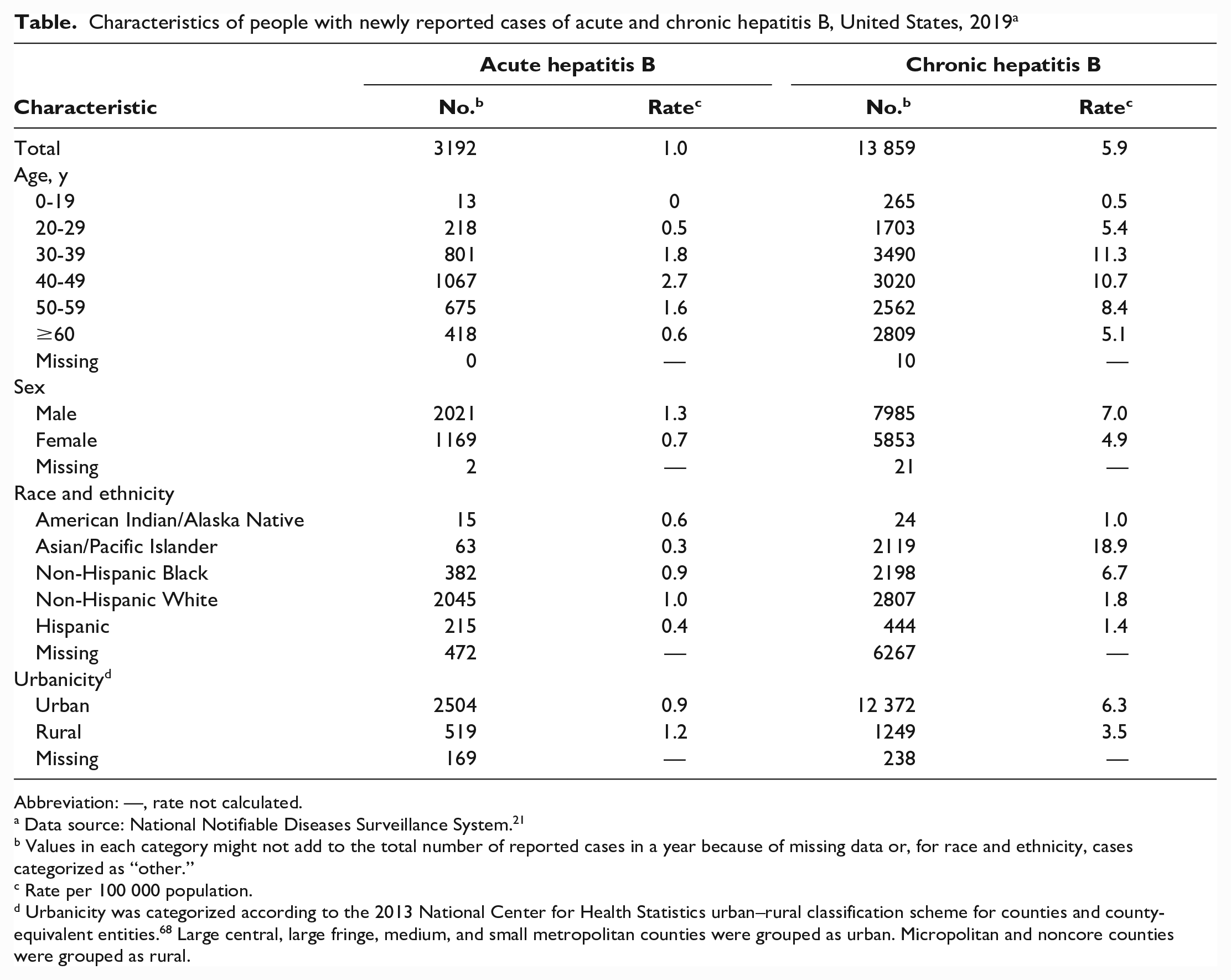
Abbreviation: —, rate not calculated.
Data source: National Notifiable Diseases Surveillance System. 21
Values in each category might not add to the total number of reported cases in a year because of missing data or, for race and ethnicity, cases categorized as “other.”
Rate per 100 000 population.
Urbanicity was categorized according to the 2013 National Center for Health Statistics urban–rural classification scheme for counties and county-equivalent entities. 68 Large central, large fringe, medium, and small metropolitan counties were grouped as urban. Micropolitan and noncore counties were grouped as rural.
Limitations to NNDSS data include underreporting by a factor of about 6.5 new infections of hepatitis B for every reported case in the United States 69 and missing risk factor data in 37.1% of acute hepatitis B cases in 2019. 67 Cases of CHB are also underreported through NNDSS. HBV testing rates are less than optimal among patients in medical care and groups at high risk for hepatitis B,38-42 and CHB is usually asymptomatic. In addition, CHB surveillance through NNDSS is predominantly conducted through laboratory reporting; as such, data on race and ethnicity and country of birth are often unknown.
National Health and Nutrition Examination Survey
Unlike NNDSS, which records newly diagnosed cases of CHB, NHANES measures CHB prevalence.41,70-75 NHANES is a probability cluster survey of the noninstitutionalized civilian US household population. 70 Consenting participants have their serum tested for anti-HBc, indicating past or present HBV infection. 70 Those who test positive for anti-HBc are tested for HBsAg; those who test positive for anti-HBc and HBsAg are considered to have CHB. 70 In the 1988-2018 NHANES cohorts,70,71 prevalence estimates for past or present HBV infection decreased 68% (1.9% to 0.6%) among people aged 6-19 years and 48% (5.8% to 3.0%) among adults aged 20-49 years (Figure 3). Populations with the highest prevalence of past or present HBV infection in recent NHANES cohorts include non–US-born people of Asian or African descent,70,71 men who have sex with men, 70 people who have past or current hepatitis C virus (HCV) infection or HIV, 70 and people who inject drugs.74,75 During 2001-2006 and 2013-2018, the prevalence of past or present HBV infection among US-born people who inject drugs increased from 35.3% (95% CI, 24.8%-47.6%) to 58.4% (95% CI, 39.5%-75.1%); concurrently, that among all US-born people was 3.5% in 2001-2006, which leveled off to 2.5% in 2007-2012 and rose to 2.6% in 2013-2018. 75
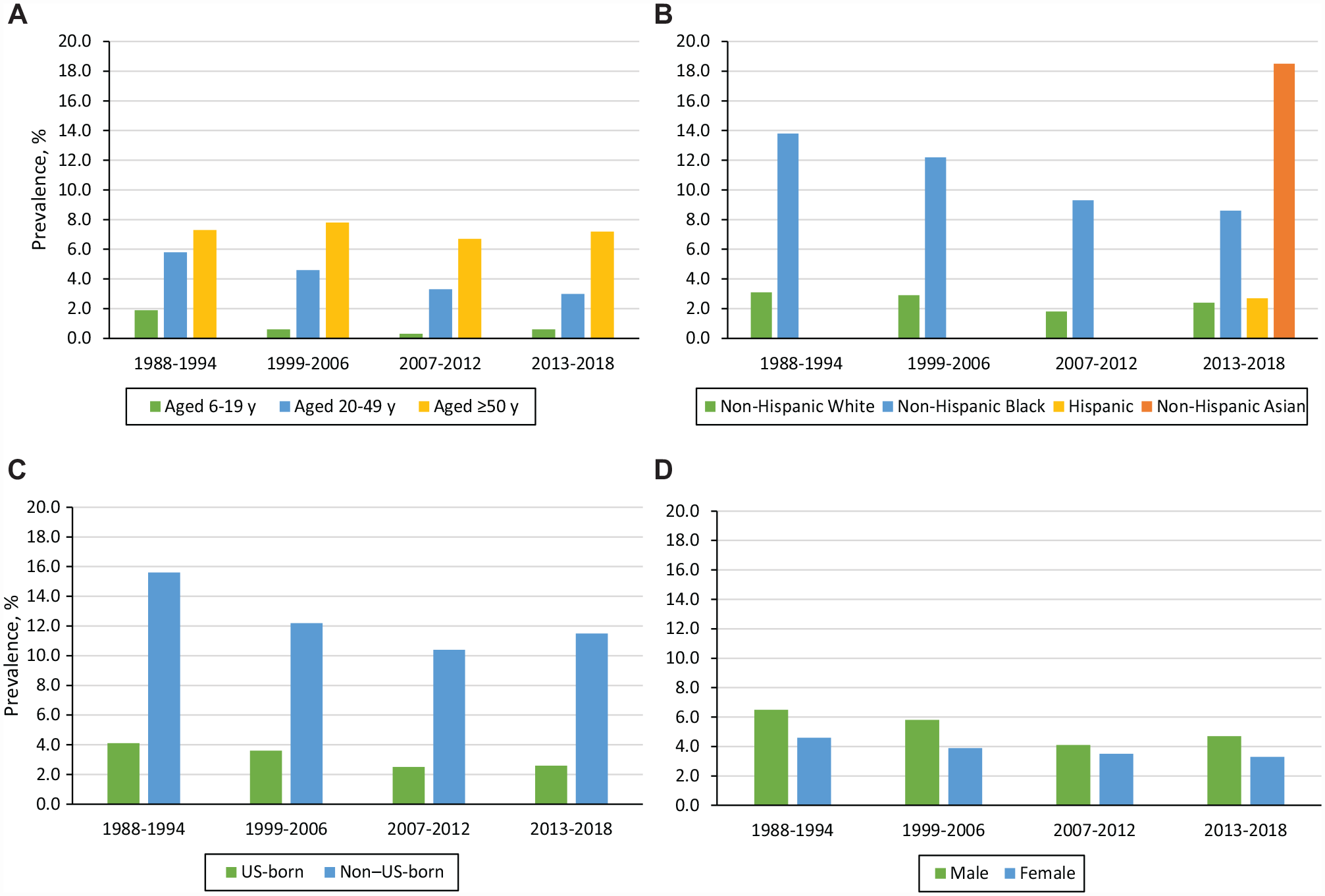
Prevalence (%) of past or present (anti-HBc positivity) HBV infection among NHANES cohorts, stratified by (A) age group, (B) race and ethnicity, (C) place of birth, and (D) sex, United States, 1988-2018. Data were not available for the years 1988-1994, 1999-2006, and 2007-2012 for Hispanic and non-Hispanic Asian people. No data were collected during 1995-1998. Data source: Roberts et al.70,71 Abbreviations: anti-HBc, hepatitis B core antibody; HBV, hepatitis B virus; NHANES, National Health and Nutrition Examination Survey.
In the 2013-2018 NHANES cohort, the prevalence of CHB was 0.3% (95% CI, 0.2%-0.4%) for an estimated prevalence of 880 000 (95% CI, 580 000-1 170 000) people with CHB in the United States. 70 Non–US-born residents, who accounted for 69.1% of those with CHB, were 9.1 times more likely than US-born people to have CHB 70 (Figure 4). Non–US-born non-Hispanic Black/African American and Asian people had a CHB prevalence of 2.5% (95% CI, 1.3%-4.7%) and 3.2% (95% CI, 2.6%-4.1%), respectively. 70
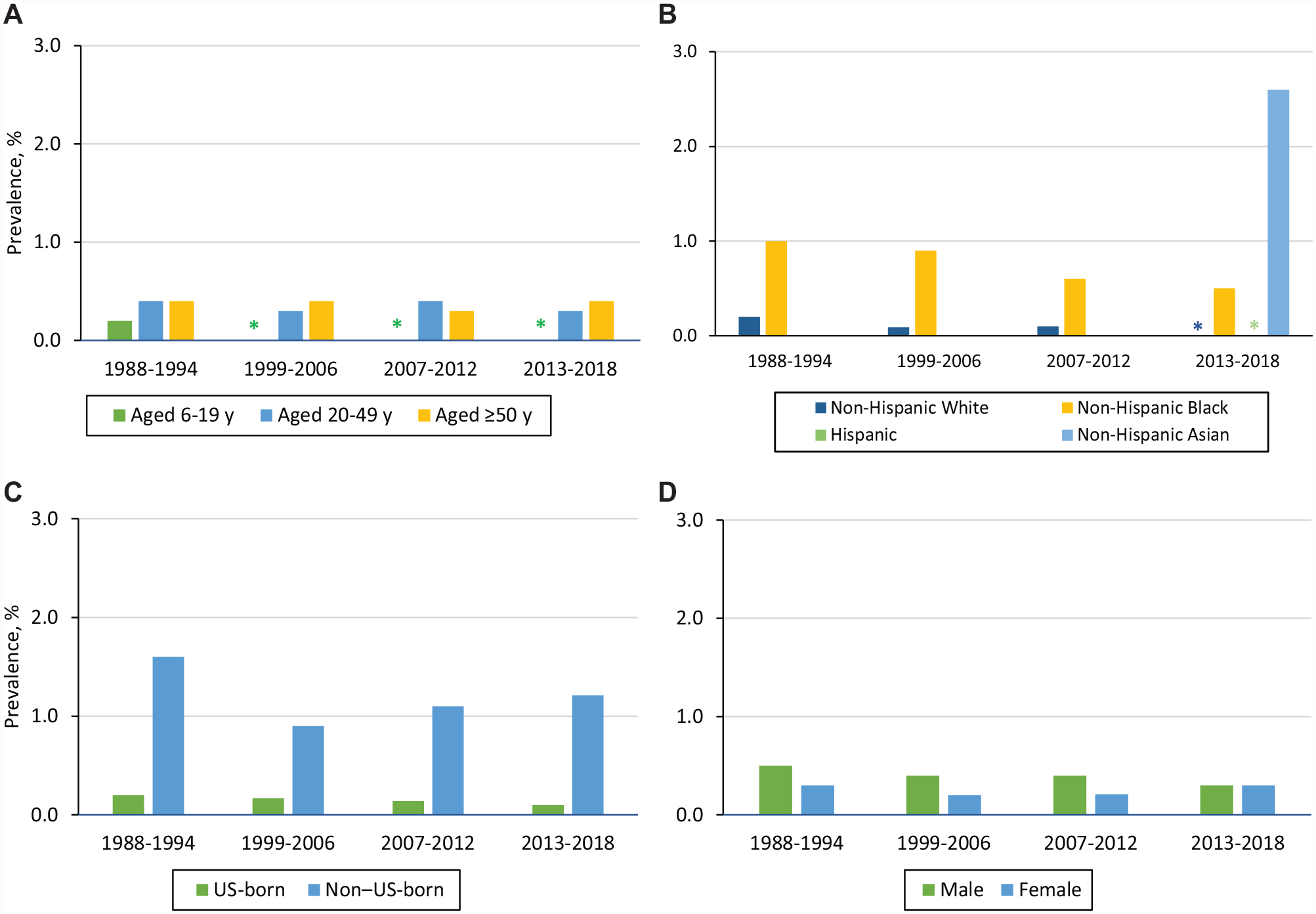
Prevalence (%) of chronic hepatitis B (HBsAg and anti-HBc positivity) among NHANES cohorts, stratified by (A) age, (B) race and ethnicity, (C) place of birth, and (D) sex, United States, 1988-2018. HBsAg prevalence was suppressed for the years 1999-2006, 2007-2023, and 2013-2018 for age 6-19 years; for the years 2013-2018 for non-Hispanic White people; and for the years 2013-2018 for Hispanic people. Data were not available for the years 1988-1994, 1999-2006, and 2007-2012 for Hispanic people and for non-Hispanic Asian people. No data were collected during 1995-1998. Data source: Roberts et al.70,71 Abbreviations: anti-HBc, hepatitis B core antibody; CHB, chronic hepatitis B; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; NHANES, National Health and Nutrition Examination Survey.
The primary limitation to NHANES is that it excludes people experiencing homelessness or incarceration, active-duty military, residents of long-term care facilities, and American Indian/Alaska Native people living on reservations.70,75 Survey nonresponse and missing or unknown responses for questions on drug use and sexual history are additional limitations.70,75 Because of concerns that NHANES underestimates CHB prevalence, Lim et al 76 combined NHANES data with estimated CHB prevalence data among groups underrepresented in NHANES to estimate the US prevalence at 1.59 million people (range, 1.25-2.49 million). Wong et al 77 combined the prevalence of CHB among US-born NHANES participants with the estimated prevalence among non–US-born people to estimate the US prevalence at 1.89 million people (range, 1.49-2.40 million).
Hepatocellular Carcinoma
Rates of HCC among people with a diagnosis of HBV infection can be ascertained from Surveillance Epidemiology and End Results–Medicare (SEER-Medicare).78-80 From 2001 to 2013, the prevalence of HCC per 100 000 people with HBV infection increased from 1.3 to 1.8, representing 8% of all cases of HCC during this time frame. 78 Among people with HBV infection and HCC, 22.4% were non-Hispanic White, 5.1% non-Hispanic Black, 53.3% non-Hispanic Asian, 2.3% Hispanic, and 16.8% other or unknown racial or ethnic group. 78 During 2000-2014 among adults aged ≥68 years, the median survival time was 10.3 months after diagnosis. 79 Limitations of SEER-Medicare include a lack of generalizability to adults aged <65 years and likely underascertainment of HBV infection status based on medical claims data.78-80 SEER-Medicare data do not include place of birth. 81
Liver Transplantation
Surveillance for transplant indications, including CHB and HCC, and transplant outcomes is conducted through the United Network for Organ Sharing/Organ Procurement and Transplantation Network registry in the United States.82,83 During 2005-2015, of the 3046 people on a wait list for liver transplantation because of a primary or secondary diagnosis of hepatitis B, 80.8% were male, 39.0% had HCC, and the median age was 54.1 years. Furthermore, 32.9% were non-Hispanic White, 10.9% non-Hispanic Black, 5.8% Hispanic, and 50.4% non-Hispanic Asian. 82 The 12-month survival rate after transplantation was 91.0% and that on the wait list was 93.8%; 52.5% of people received a liver transplantation within 12 months. 82 The registry is limited primarily by accuracy of diagnostic code input by transplant coordinators 83 and does not collect data on place of birth for liver recipients. 84
Mortality Surveillance
Mortality surveillance is conducted through analysis of multiple cause-of-death data, based on the death certificates of US residents. 67 In 2019, the age-adjusted hepatitis B–related mortality rate was 0.42 (95% CI, 0.40-0.44) per 100 000 population, and the highest age-adjusted mortality rate per 100 000 people was recorded among Asian and Pacific Islander people (2.10; 95% CI, 1.90-2.29). 67
Among 17 483 decedents with hepatitis B during 2010-2019, 10 823 (61.9%) were US-born, 6285 (35.9%) were non–US-born, and 375 (2.1%) had unknown birthplace information. 85 When compared with non–US-born decedents with hepatitis B, US-born decedents with hepatitis B were more likely to be non-Hispanic Black (24.7% vs 9.1%) or non-Hispanic White (67.1% vs 10.8%) and less likely to be non-Hispanic Asian and Pacific Islander (1.6% vs 68.0%) (all P < .001). 85 Hepatitis B was listed as the underlying cause of death for 30% of US-born and non–US-born decedents with hepatitis B. 85 When hepatitis B was listed as a contributing cause of death, US-born decedents were significantly less likely than non–US-born decedents to have liver cancer (20.6% vs 53.7%) and significantly more likely to have hepatitis C, other viral hepatitis, alcohol-related liver disease, non–alcohol-related liver disease, HIV infection, injuries or trauma (including overdose), and various disorders (circulatory, respiratory, mental, or behavioral) listed as the underlying cause of death (all P < .001). 85
Among all decedents with hepatitis B, coinfections were reported with HCV (n = 4297, 24.6%), HIV (n = 961, 5.5%), HCV and HIV (n = 295, 1.7%), and hepatitis delta virus (n = 6, 0.03%); coinfection with HCV, HIV, or hepatitis delta virus was more common among US-born decedents with hepatitis B (39.4%; 95% CI, 38.5%-40.3%) than non–US-born decedents with hepatitis B (8.8%; 95% CI, 8.1%-9.5%). 85 National death rates per 100 000 population declined from 0.58 during 2000-2009 to 0.47 during 2010-2019. 85 Death rates increased significantly in only 2 states: West Virginia and Kentucky. 85
Mortality data underestimate CHB as a cause of death in the United States. In a well-characterized cohort of people with CHB, only 19% of CHB decedents and 40% of people who died of liver disease had hepatitis B reported on their death certificates. 86
Unfinished Business
With high rates of infant and childhood vaccination, the United States has reduced the incidence of acute hepatitis B among children and adults aged <30 years. However, unvaccinated adults with risk behaviors have elevated rates of acute hepatitis B in NNDSS data 67 and an elevated prevalence of past or present HBV infection in NHANES,70,74,75 while non–US-born people who acquired HBV infection vertically or during early childhood have an elevated prevalence of CHB.41,70 People of Asian race have disproportionately elevated rates of the most serious consequences of CHB, including mortality,67,85 HCC, 78 and liver transplantation. 82 In NHANES, the highest prevalence of CHB is among non–US-born Asian people. 70 To guide hepatitis B elimination activities and national public health policy, better data are needed on acute hepatitis B infection and CHB prevalence stratified by race and ethnicity, place of birth, and risk behaviors such as injection drug use.
The most urgent surveillance and data needs for US-born adults with risk behaviors for hepatitis B are to characterize incident and prevalent infections sufficiently to identify places where screening and vaccination services can be delivered to people at risk for hepatitis B, including correctional settings, substance use treatment facilities, syringe service programs, and sexually transmitted infection clinics.
The most critical surveillance and data needs for non–US-born people are data to identify where these people receive care so that screening and treatment services can be evaluated and improved in those settings. Electronic health record and claims data, in combination with the universal screening recommendation for adults, offer promise for improving access to care for these groups. Integration of hepatitis B surveillance registries with electronic health record or claims data could support active case finding, evaluation of access to care, and enhanced linkage to care. Active outreach to immigrants and refugees, coupled with culturally and linguistically appropriate care, will continue to be important to increase awareness of current infection. Further use of electronic health records, 87 claims data, 88 and integrated clinical databases 89 will be needed to evaluate and improve the treatment of people diagnosed with CHB.
To evaluate progress toward elimination goals, the Viral Hepatitis National Strategic Plan calls for improved surveillance at the national, state, tribal, local, and territorial health department levels; improved sharing of clinical viral hepatitis data; and routine analysis and dissemination of surveillance findings to inform public health action. 24
In 2021, CDC funded 59 state and local jurisdictions for surveillance and prevention of viral hepatitis. 90 One objective is to improve data completeness and quality, including risk factor data for viral hepatitis in NNDSS. This information is critical for jurisdiction activities to gear testing and treatment resources toward people at highest risk for hepatitis B and evaluate the new universal hepatitis B vaccine 31 and screening recommendations 43 for adults. Improved surveillance data will also enable jurisdictions to formulate plans for elimination of hepatitis B.
Public Health Implications
In the context of elimination, the small number of perinatal, acute, and chronic cases of hepatitis B in infants, children, and young adults should be scrutinized to identify missed opportunities for prevention. While universal adult vaccination for hepatitis B has now been recommended for adults through age 59 years, focused testing and vaccination programs will continue to be important among people at risk for acute hepatitis B (eg, people who inject drugs, people with multiple sexual partners). While universal screening of adults is now recommended, focused programs to screen and treat non–US-born people will remain a critical intervention, especially for immigrants and refugees from Asian and African countries.
Stronger, more integrated surveillance is needed to measure the impact of new screening, testing, and vaccination recommendations. Gaps in surveillance data for perinatal hepatitis B, acute hepatitis B, and prevalence of CHB are particularly important to address at every level of the primary care and public health systems in the United States if the promise of hepatitis B elimination is to be realized.
Supplemental Material
sj-docx-1-phr-10.1177_00333549231175548 – Supplemental material for Progress and Unfinished Business: Hepatitis B in the United States, 1980-2019
Supplemental material, sj-docx-1-phr-10.1177_00333549231175548 for Progress and Unfinished Business: Hepatitis B in the United States, 1980-2019 by Danae Bixler, Henry Roberts, Lakshmi Panagiotakopoulos, Noele P. Nelson, Philip R. Spradling and Eyasu H. Teshale in Public Health Reports
Footnotes
Acknowledgements
The authors acknowledge Erin Conners, PhD; Neil Gupta, MD; Kathleen Ly, MPH; and Mark Weng, MD, of the CDC Division of Viral Hepatitis for helpful discussions, editorial comments, and guidance on the content of this review.
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online and at ![]() . The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th Edition.
. The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th Edition.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
