Abstract
During 2014-2019, the Utah Department of Health (UDOH) enhanced its surveillance program for acute hepatitis C virus (HCV) infections by mandating electronic reporting of negative HCV test results in 2015 and liver function test results in 2016. UDOH also engaged with blood and plasma donation centers beginning in 2014 and syringe exchange programs in 2018 to encourage manual reporting of negative HCV test results from facilities without electronic reporting capabilities. UDOH hepatitis surveillance staff also provided training for case investigations in 2017. The number of cases detected increased 14-fold, from 9 during 2012 to 127 during 2019. In 2019, of 127 cases, 55% (n = 70) were detected through negative HCV test results reported electronically before positive test results (ie, recent seroconversions), 25% (n = 32) through positive HCV test results and elevated liver function test results, 18% (n = 23) through manually reported negative HCV test results, and 2% (n = 2) through positive HCV test results and clinical evidence. Challenges to surveillance included accessing patients for investigations and engaging donation centers in reporting negative test results. Utah’s experience demonstrates practical considerations for improving surveillance of acute HCV infections.
Keywords
Cases of acute hepatitis C virus (HCV) infection are often underascertained and underreported.1,2 People newly infected with HCV may have jaundice and clinical symptoms, such as abdominal pain, malaise, fever, headache, nausea, vomiting, and diarrhea. 3 However, because most patients are asymptomatic, sensitive reporting protocols are needed to identify and treat new cases and control the spread of illness. HCV is transmitted primarily through parenteral exposures to blood, such as injection drug use. 4 HCV also has been transmitted through needlestick injuries, poor injection safety practices in health care settings, and receipt of donated blood, plasma, and organs. 4
Although up to 50% of HCV infections resolve spontaneously, without treatment, the remainder progress to chronic infections that can result in fibrosis, cirrhosis, or liver cancer. 4 In 2020, universal HCV infection screening in the United States was recommended for all adults aged ≥18 years and for pregnant people during each pregnancy. One-time testing is recommended for people with HIV and people who ever injected drugs, ever received hemodialysis, received blood transfusions or organ transplants before 1992, or had other known exposures. Routine periodic testing is recommended for people with ongoing risk factors, such as currently injecting drugs and sharing needles or receiving maintenance hemodialysis. Any person requesting an HCV test should receive one. 5
In 2015, the voluntary report of a Utah patient’s HCV testing history to the Centers for Disease Control and Prevention (CDC) by a blood donation service helped to detect an opioid diversion (ie, use by a health professional of opioid medication intended for patients) occurring in 2 Utah health care facilities during 2011-2014. During the 3-year period, a nurse who was HCV-positive administered medications to patients after self-administering some of the dose, exposing about 7200 patients and infecting an estimated 16 people. 6 Had reporting of negative HCV test results or liver function test (LFT) results been mandatory during those years, the outbreak might have been detected sooner.
In 2012, the Council of State and Territorial Epidemiologists (CSTE) updated the definition of an acute case of HCV infection by requiring ≥1 clinical symptom and evidence of jaundice or dark urine, or serum alanine aminotransferase (ALT) levels >400 IU/L, to also include an alternative ascertainment method of a positive test result ≤6 months after a negative test result (ie, evidence of recent seroconversion). 7 In 2016, a further updated CSTE case definition reduced the ALT threshold to >200 IU/L and increased the recent seroconversion window to ≤12 months. 8
To maximize case ascertainment based on the expanded 2012 CSTE definition of an acute case of HCV infection and later the 2016 CSTE case definition, the Utah Department of Health (UDOH) used multiple strategies. In 2014, UDOH contacted blood and plasma donation centers to acquire negative testing histories of donors who had a positive test result and to encourage manual reporting of negative HCV test results to the department. For health care and laboratory facilities, UDOH mandated electronic reporting of negative HCV test results beginning in 2015 and LFT results in 2016, when it also added bilirubin measured at >3.0 mg/dL to its own state case definition as a qualifying elevated LFT result. In 2017, UDOH received federal funding 9 to train state and local public health staff to ascertain and investigate new cases, link patients with education and treatment, and detect potential clusters and outbreaks. In 2018, UDOH also engaged Utah’s 3 syringe exchange programs (SEPs) with the highest client volumes (among 9 SEPs in total) to encourage manual reporting of negative HCV test results. This case study reports on the impact of Utah’s investments in enhanced surveillance protocols for acute cases of HCV infection. The objective of this study was to assess the magnitude and composition of acute cases of HCV infection ascertained during 2012-2019 and consider ongoing challenges for surveillance.
Methods
During 2020-2021, we extracted and reviewed acute HCV infection cases reported during January 2012–December 2019 from the Utah National Electronic Diseases Surveillance System. 10 We confirmed cases using the CSTE case definition (2012 or 2016) corresponding to the year in which they were ascertained and subsequently allocated to 1 of 5 ascertainment methods, each comprising a positive HCV test result (antibody test, nucleic acid test, or antigen test) and 1 of the following:
Method 1: clinical evidence (ie, jaundice and ≥1 clinical symptom [fever, malaise, nausea, vomiting, anorexia, or abdominal pain]);
Method 2: an elevated LFT result and ≥1 clinical symptom; or a negative HCV test result during the previous 6 months or 12 months (depending on the corresponding case definition), via 1 of the following methods:
Method 3: reported manually by a blood or plasma donation center,
Method 4: reported manually by an SEP, or
Method 5: reported electronically by a health care or laboratory facility.
We allocated cases to the year in which all requirements for the corresponding case definition were first met. Where multiple ascertainment methods were possible, we allocated ascertainment methods in order of reliability. We prioritized seroconversions, followed by elevated LFT result and ≥1 clinical symptom, and then jaundice and ≥1 clinical symptom, given the need to rule out alternative diagnoses (eg, other hepatitis, alcohol use) and potential variations in interpretation for these latter 2 methods. We also compared the incidence of acute HCV infection in Utah with the incidence in the United States. 11
To identify challenges with case ascertainment and investigation, we conducted 10 interviews with state and local public health department staff involved in HCV surveillance. During the telephone-based or in-person semistructured interviews, each 20-45 minutes in duration, we asked participants for a description of their duties, a general appraisal of Utah’s enhancements to HCV surveillance during 2014-2019, and an evaluation of any changes (eg, to workflow, time required, personnel required) in processes of case ascertainment, case investigation, or linkage to care. Local health department–based participants included 3 epidemiologists and 3 public health nurses engaged in acute HCV case ascertainment and investigation; we interviewed 4 of these participants in pairs. UDOH-based participants included 2 HCV program epidemiologists, a data manager, and an intervention specialist, all interviewed individually. This activity was reviewed by CDC and was conducted consistent with applicable federal law and CDC policy (45 C.F.R. part 46.102(l)(2), 21 C.F.R. part 56; 42 U.S.C. §241(d); 5 U.S.C. §552a; 44 U.S.C. §3501 et seq).
Outcomes
Among 515 confirmed cases of acute HCV infection extracted from the surveillance system, 503 (98%) met CSTE’s case definition for the year in which they were ascertained (Figure 1). The number of cases generally increased consistently from 2012 (n = 9) to 2019 (n = 127). Compared with the base year of 2012, the number of cases ascertained was 89% (n = 17) higher during 2013, 300% (n = 36) higher during 2014, 278% (n = 34) higher during 2015, 789% (n = 80) higher in both 2016 and 2017, 1233% (n = 120) higher in 2018, and 1311% (n = 127) higher in 2019. During 2012-2019, the incidence of acute HCV infection per 100 000 population increased from 0.6 to 1.3 in the United States and from 0.4 to 4.0 in Utah (Figure 2).
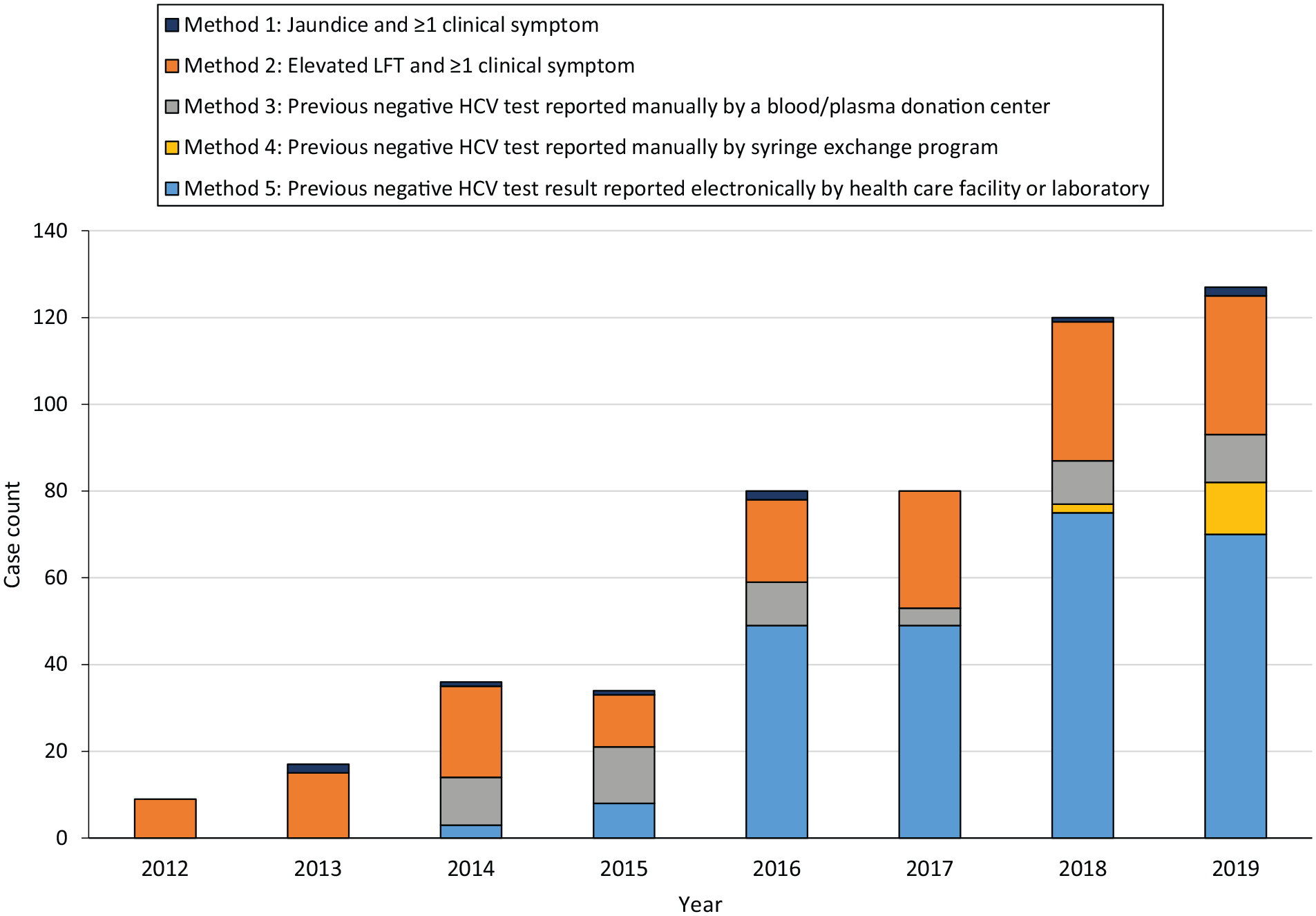
Method of case ascertainment in conjunction with positive HCV test result, acute HCV infections (N = 503), Utah, 2012-2019. Clinical symptoms are fever, malaise, nausea, vomiting, anorexia, or abdominal pain. For LFT, during 2012-2015, indicates ALT >400 IU/L, plus ≥1 symptom reported; during 2016-2019, indicates ALT >200 IU/L or bilirubin >3.0, plus ≥1 symptom reported. For previous negative HCV test result, during 2012-2015, indicates negative HCV test result within previous 6 months; during 2016-2019, indicates negative HCV test result within previous 12 months. Abbreviations: ALT, alanine aminotransferase; HCV, hepatitis C virus; LFT, liver function test; SEP, syringe exchange program. Data source: Utah—National Electronic Disease Surveillance System. 10
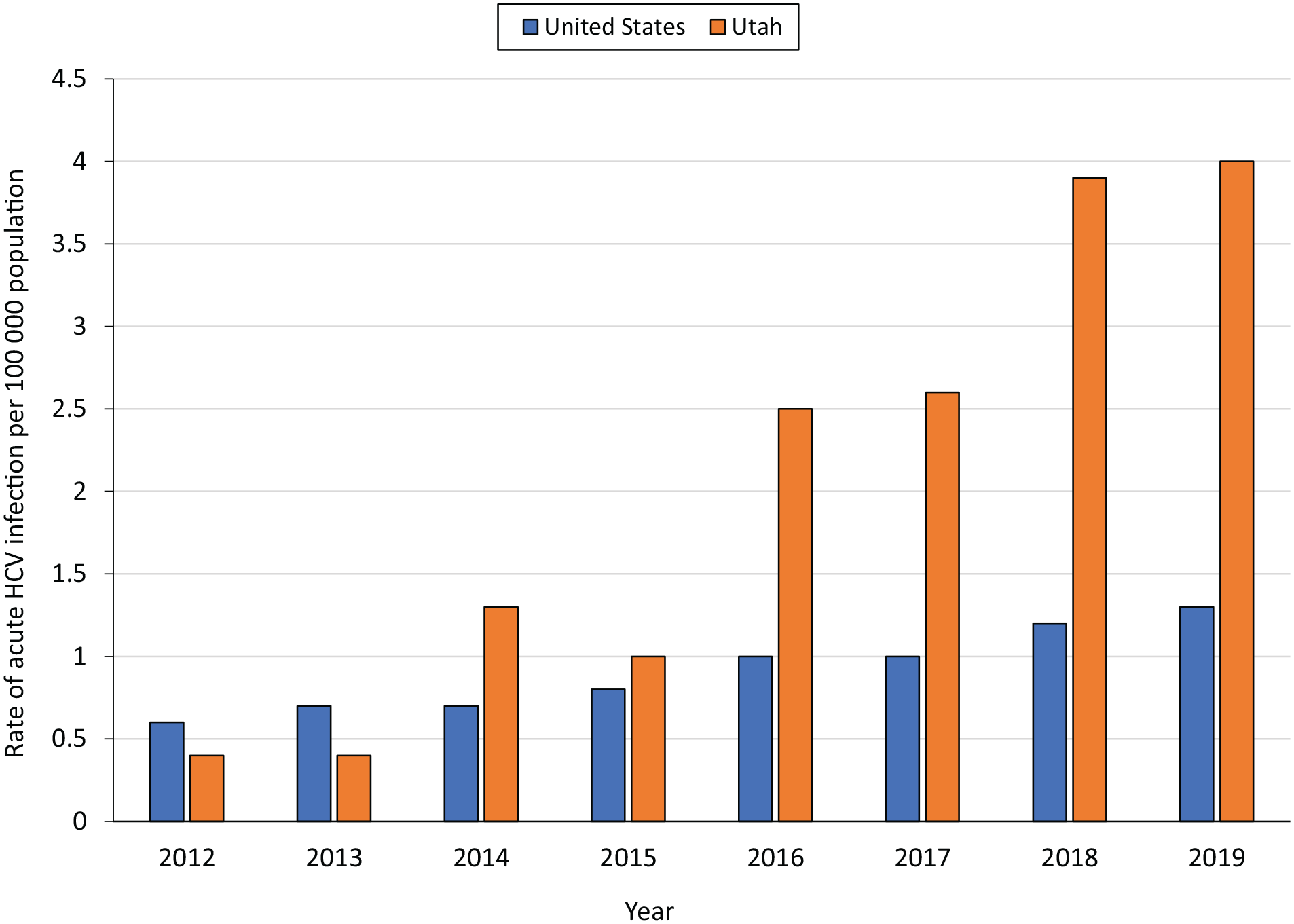
Incidence rate of acute hepatitis C virus (HCV) infection per 100 000 population, United States and Utah, 2012-2019. Data source: Centers for Disease Control and Prevention. 11
During any year, no more than 2 cases were ascertained based solely on a positive HCV test result and clinical evidence (method 1). The number of cases ascertained from a positive HCV test result and an elevated LFT result (method 2) was higher after electronic reporting of LFT results was mandated, during 2016-2019 (110 of 407), compared with 2012-2015 (57 of 96); however, the proportion of total cases ascertained using this method was lower during 2016-2019 compared with 2012-2015 (27% vs 65%, respectively).
Recent seroconversions determined through manual report of a negative test result (methods 3 and 4) became more common after engagement of blood and plasma donation centers (2014) and SEPs (2018). During 2014-2015, 24 of 70 (34%) cases were ascertained based on manual reports from blood and plasma donation centers, usually obtained during follow-up of a patient’s testing history at the center where their positive test result was reported; this proportion decreased during 2016-2019 (35 of 407; 9%). During 2019, after efforts to engage SEPs in manual reporting were initiated, 12 of 127 (9%) cases were ascertained based on a negative test result reported by an SEP.
Recent seroconversions determined through electronic reports of negative test results from health care and laboratory facilities (method 5), which were made mandatory in 2015, comprised 243 of 407 (60%) cases ascertained during 2016-2019. The time required to initially link testing data between health care and laboratory facilities and state surveillance systems (ie, gathering institutional permissions and joining electronic reporting systems), typically several months, led to a lower proportion of cases (8 of 34; 24%) ascertained this way during 2015, compared with subsequent years.
Interviewees suggested at least 4 ongoing investigation challenges. First, plasma donation centers report many positive HCV test results but are not legally required to report negative test results, thus requiring time-consuming follow-up to determine a patient’s recent testing history at such facilities. In addition, most facilities only have 1 manager authorized to fulfill patient data requests. Second, rurally situated patients at increased risk for HCV infection may have access only to family or general practice clinics and can be hesitant to get tested at such facilities, whereas patients at increased risk who live in urban areas are often tested at mobile clinics that do not always manually report results from the rapid tests typically used there. Third, detailed case reviews are necessary to determine if reports of positive HCV test results signal reinfections of a cleared chronic case, as would be indicated by records of treatment initiation or previous negative test results, and whether elevated LFT results represent a spike in the context of a chronic case, a new acute case, or a reinfection. Such reviews are necessary but can become challenging in the context of understaffing or high staff turnover. Fourth, according to interviewees, the rate of completed investigations with an assessment of risk factors and provision of education to the patient remained low (<50%) because of understaffing, difficulty contacting patients, and potentially less trust among patients in health department staff than in community health care providers such as SEPs.
Lessons Learned
Utah’s investments in enhanced acute HCV surveillance generated substantial increases in case ascertainment during 2014-2019. It is possible that some of the increase was attributable to an actual increase in acute HCV cases; however, the 10-fold increase in the rate in Utah exceeded the 2-fold increase in rate nationally, which could itself be attributable to surveillance enhancements undertaken in Utah and several other states. 9 The introduction of mandatory electronic reporting likely generated the largest numbers of ascertainments, primarily by negative HCV laboratory test results to determine recent seroconversions and secondarily by elevated ALT or bilirubin (ie, LFTs). In 2019, the CSTE acute case definition was revised 12 to require that cases ascertained through these methods could be confirmed only in the absence of alternative diagnoses, reflecting the rule-out requirement that Utah had adopted during its implementation of enhanced surveillance.
Improved ascertainment has, in the short term, allowed for more accurate estimates of acute HCV infection and potential levels of person-to-person transmission at the state level. Utah’s experience thus has the potential to inform other states and focus federal attention and resources on acute HCV infection and associated public health issues such as the current opioid crisis. Fourteen states implemented enhanced surveillance programs for hepatitis B virus and HCV during the same period as Utah. 9 Although, to our knowledge, no evaluations of these programs have been published, previous evaluations of enhanced HCV surveillance in other states reiterate the substantial benefit of improved informatics such as electronic laboratory reporting (Massachusetts) 13 and the considerable resources required for case investigations in populations that are medically underserved (New York). 14 Other health departments can also improve detection of acute cases of HCV infection through mandatory reporting of negative HCV laboratory test results to identify recent seroconversions, particularly given CDC’s expansion of funding for enhanced HCV surveillance and support to all 50 states in 2021,15,16 but additional changes to staffing and infrastructure at the state and local levels may be necessary to complete investigations and maximize the benefits of case ascertainment. These types of enhancements may increase chances of detecting sentinel cases in outbreak or cluster events for HCV infection or diseases with similar risk factors, such as hepatitis B or HIV infection.
Extensive follow-up was often required to access negative testing histories of blood and plasma donors who had recently received a positive test result for HCV. The follow-up process, however, yielded a substantial number of cases, especially during 2014-2015, when electronic reporting of negative HCV test results was not yet available. Manual reports of negative test results from SEPs generated more than a dozen case ascertainments in the first 2 years of engagement with UDOH, requiring comparatively limited follow-up. Encouraging manual reporting of negative HCV test results from both SEPs and blood and plasma donation facilities, which often work with populations at increased risk of HCV infection, may further improve the ascertainment of acute HCV cases. Jurisdictions without SEPs might consider engaging shelters or community support agencies as partners and physical sites in the provision of HCV testing or piloting point-of-care testing with peer outreach workers. 17
Some interviewees were unsure about whether improved ascertainment translated consistently into education and linkage to treatment given the challenges of case investigations. Although interviewees described difficulties with some processes, such as obtaining comprehensive case information and contacting patients, most of the challenges described were more broadly connected to high staff turnover and underfunding at health departments and other institutions that participate in the workflow of case investigation and patient care for people with acute HCV infection. Additional resources may therefore be necessary to increase HCV staffing resources, train staff on case investigation protocols, and maintain staff continuity in the collection of data on patient contact and investigation completion.
Conclusions
Investments in case ascertainment are necessary to align with broadened definitions intended to identify acute cases of HCV infection that are commonly mildly symptomatic or asymptomatic. This Utah case study demonstrated that implementing mandatory reporting rules for negative HIV test results and LFT results can help identify additional acute HCV cases. However, practical considerations, such as hepatitis staffing at local health departments, should also be considered. Additional resources are necessary to increase HCV staffing and to train staff on case investigation protocols and the rationale for investigating acute HCV cases. To sustain and further improve ascertainment, additional resources may be necessary to promote continued engagement of plasma donation centers and SEPs and the expansion of HCV testing options into rural areas.
Footnotes
Acknowledgements
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
