Abstract
Objectives
The objective of this study was to characterize the changes in timeliness and completeness of disease case reporting in Texas in response to an increasing number of foodborne illnesses and high-consequence infectious disease investigations and the Texas Department of State Health Services’ new state-funded epidemiologist (SFE) program.
Methods
We extracted electronic disease case reporting data on 42 conditions from 2012 through 2016 in all local health department (LHD) jurisdictions. We analyzed data on median time for processing reports and percentage of complete reports across time and between SFE and non-SFE jurisdictions using Mann–Whitney t tests and z scores.
Results
The median time of processing improved from 13 days to 10 days from 2012 to 2016, and the percentage of disease case reports that were complete improved from 19.6% to 27.7%. Most reports were for foodborne illnesses; both timeliness (11 to 7 days) and completeness (20.9% to 23.5%) improved for these reports.
Conclusions
Disease reporting improvements in timeliness and completeness were associated with the SFE program and its enhancement of epidemiologic capacity. SFEs were shown to improve surveillance metrics in LHDs, even in jurisdictions with a high volume of case reports. Adding epidemiologist positions in LHDs produces a tangible outcome of improved disease surveillance.
Infectious disease reporting and the subsequent investigation of cases is a critical component of the public health system’s ability to track, control, and prevent outbreaks and reduce morbidity and mortality in populations. 1,2 A fundamental principle of disease surveillance is the systematic collection, analysis, and dissemination of information quickly for disease control and prevention. 3 Timely and complete information sharing during routine disease surveillance was crucial to the success of controlling the first new disease epidemic of the 21st century, severe acute respiratory syndrome. 3,4 Gaps in the public health workforce, namely, epidemiologic capacity, are a barrier to the speed at which disease information is processed, whether using traditional forms of disease notification or real-time syndromic surveillance systems. 4 -6
Passive disease reporting (ie, disease reports from reporting entities, typically health care providers or laboratories, to public health departments) is widely used across the United States, including Texas. Passive reporting can slow investigation timelines and increase the response time of public health departments. 7 -9 In nonpandemic times, foodborne illness reporting is a substantial part of the investigative burden of surveillance for local health departments (LHDs). The rates of reported foodborne illnesses increased in Texas from 2008 to 2016, most notably in rates per 100 000 population of campylobacteriosis (9.1-16.5), cryptosporidiosis (1.1-2.6), Escherichia coli infections (1.9-3.6), and shigellosis (7.3-15.5). 10 The workload substantially increased for LHDs in investigating and processing reports for foodborne illnesses from 2012 to 2016. During that time, the expansion of electronic laboratory reporting increased the number of passive surveillance reports to LHDs. Electronic laboratory reporting, commonly the first report of an infectious disease case or incident to the public health system, can improve the timeliness and accuracy of disease reporting to state health departments and LHDs; however, electronic laboratory reporting is often accompanied by a large workload for LHD staff members. 11 -15 As electronic disease surveillance systems become more automated, LHDs may be underresourced and understaffed to meet the need for timely and complete case investigation. 14 -16
Laboratories submit the vast majority of disease reports simultaneously to the relevant LHD and public health region and to the Texas Department of State Health Services (DSHS) through electronic laboratory reporting; these reports are routed from the laboratory to health departments via the National Electronic Disease Surveillance System Base System (NBS; Figure 1). Texas has its own NBS that interacts with and links to the National Electronic Disease Surveillance System. Reporting entities may also report directly to their LHD or public health region by telephone, fax, or an electronic data exchange system. Public health staff members then manually enter these reports into NBS. Based on these initial reports, whether in-person or electronic, public health staff members gather additional data as needed to confirm cases and perform follow-up investigations or interventions. The LHD or public health region enters or updates case information in NBS. DSHS reviews the quality of NBS data before transmission to the Centers for Disease Control and Prevention (CDC). In the examination of the timeliness of such a system, 3 key intervals are (1) the time from a suspected case or diagnosis of a case to the initial case report to the public health system, (2) the time from the initial case report to the case being confirmed and the start of investigation, and (3) the time from investigation completion to being reported to CDC.
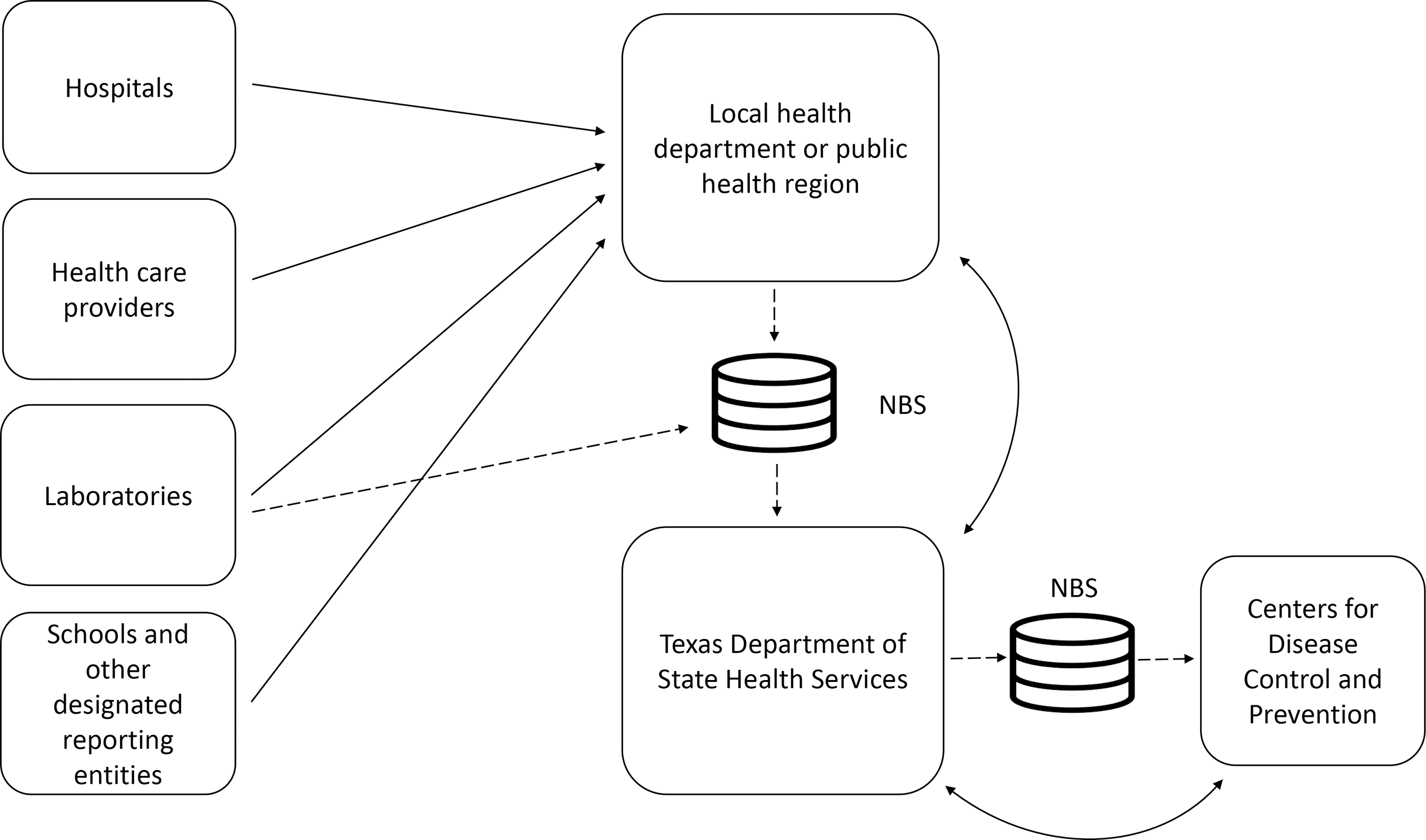
Simplified notifiable disease reporting flow in Texas public health surveillance system, 2017. The solid line indicates reporting via telephone, fax, electronic laboratory report, or electronic medical record. The dashed line indicates electronic reporting. Abbreviation: NBS, National Electronic Disease Surveillance System Base System.
In response to increasing numbers of cases of foodborne illnesses and surge needs created by high-consequence infectious disease investigations in the state, DSHS started the state-funded epidemiologist (SFE) program, described previously. 16 Texas funded 45 epidemiologist positions during 2 years in 31 LHDs in Texas to increase overall epidemiologic capacity and improve the response to infectious disease threats. As in previous studies that compared changes in timeliness and completeness after the introduction of new systems (eg, electronic laboratory reporting), we sought to understand whether the SFE program changed the timeliness and completeness of disease reporting in Texas. 13,14,17
Methods
Data Collection
In collaboration with DSHS, we acquired data on notifiable conditions in the state from the Texas NBS for the years 2012-2016. DSHS staff members queried and extracted de-identified data in a comma-separated value file. The data consisted of infectious diseases typically handled by most SFE positions. 18 The Texas A&M Institutional Review Board reviewed and approved this study and all aspects of data collection, analysis, and reporting.
The Emerging and Acute Infectious Disease Unit of DSHS managed all notifiable conditions in this study as part of various prevention and control teams, including foodborne illness, health care safety, high-consequence infectious disease, invasive and respiratory infectious disease, and vaccine-preventable disease (Figure 2). Mandatory reporting for multidrug-resistant organisms did not begin until 2015; staffing and guidance were in pilot testing during our study period. Therefore, we excluded these case reports from our analysis.
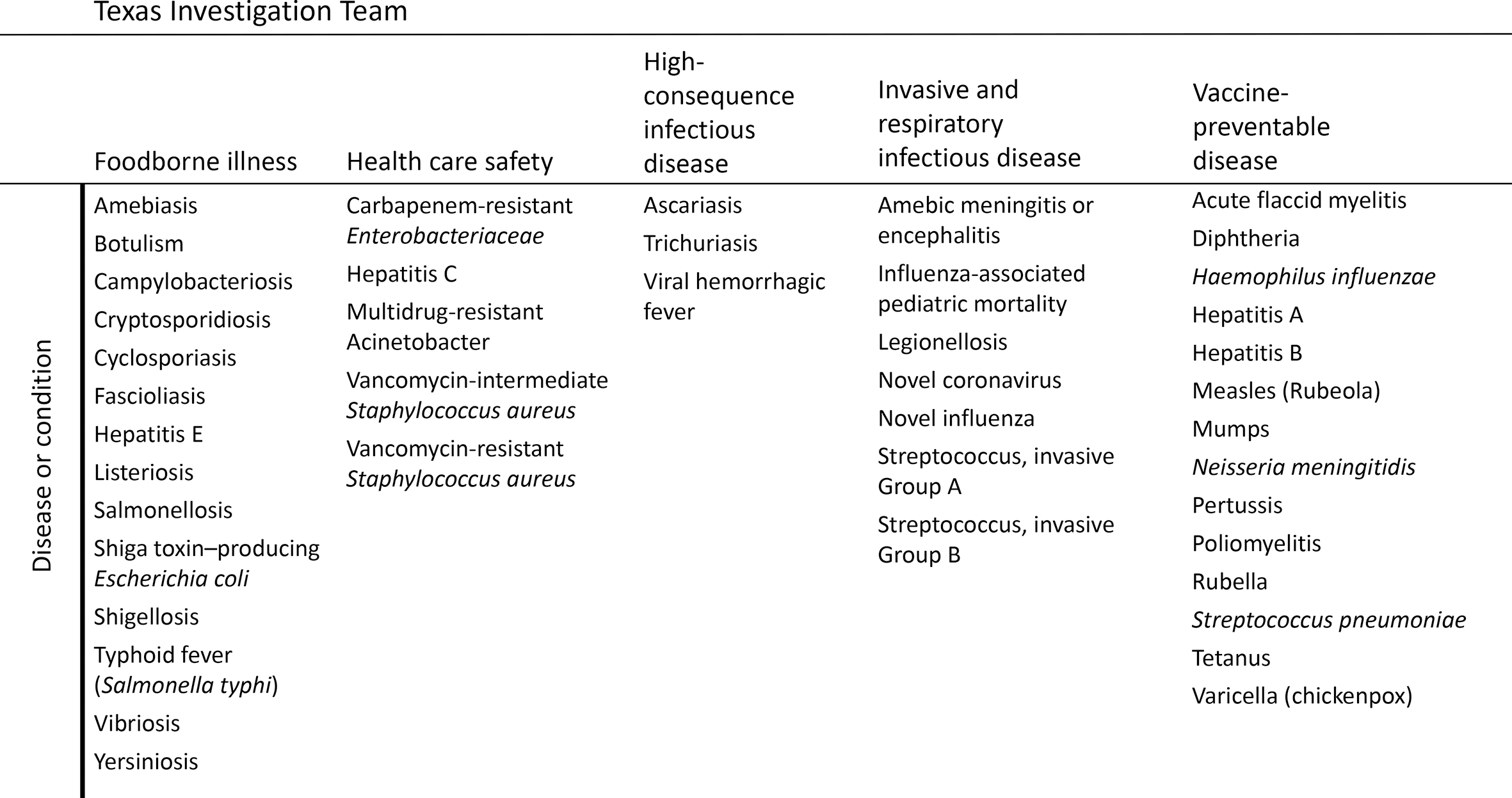
Notifiable conditions investigated by local health departments and the Emerging and Acute Infectious Disease Unit at the Texas Department of State Health Services, 2017. Hepatitis C was investigated by the vaccine-preventable disease team in 2014.
Each case report included the following variables: jurisdiction in which the case was investigated, case status (not a case, suspected, probable, confirmed), onset date, diagnosis date, year assigned in Morbidity and Mortality Weekly Report (MMWR; 2012-2016), SFE status (yes/no, where yes included having an SFE in 2013, in 2015, or in both years), the investigation start date, the initial report time to the public health system (either DSHS or LHD, in days), the report time from LHD to DSHS (in days), the report time from DSHS to CDC (in days), and the case report approval dates from DSHS and CDC. Hereinafter, we refer to LHDs with SFEs as SFE–LHDs and LHDs without SFEs as non-SFE–LHDs. Definitions of a case, a suspected case, a probable case, and a confirmed case were based on DSHS notifiable condition guidelines. The SFE program put in place 17 SFEs in 2013 and 28 SFEs in 2015; some LHDs had multiple SFEs, and all LHDs kept their SFE positions after their initial placement.
Data Cleaning
The data set contained 139 035 electronic disease reports on 43 conditions in 71 unique local reporting jurisdictions. Because not all jurisdictions consistently report suspected cases (n = 2367) or noncases (n = 20 235), we included only probable and confirmed cases in our analysis. In addition, we did not include case reports with a time interval of more than 365 days from the first report to the public health system and first notification to DSHS in this study (n = 1497; 1.1% of all case reports). Most (71.2%) case reports that had processing times greater than 365 days (range, 366-7800 days) were cases of foodborne illnesses with missing data. Finally, we restricted data to MMWR years 2012, 2014, and 2016 because of the timing of the SFE program. In 2012, the SFE program was not in place. By 2014, LHDs had fully incorporated the first phase of the SFE program. By 2016, all SFEs were in their respective LHDs. Because the timeline for hiring and training SFEs varied by LHD and the process was only partially completed in 2013 and 2015, we did not use data for those years (resulting in 20 917 exclusions in 2013 and 26 776 exclusions in 2015).
The processing of data on health care–associated diseases is different from the processing of data on diseases in other categories (ie, foodborne, respiratory, vaccine preventable) in this study because it depends more on local health care facilities and requires additional approvals from DSHS. Many health care–associated diseases are reported and investigated by regional and central DSHS staff members in collaboration with hospital personnel; therefore, they do not provide a clear link to LHD disease surveillance activity. A sensitivity analysis that excluded health care–associated diseases found an increased median time of reporting in LHDs by 1 day in 2014 and 2016 and decreased completeness in 2014. We found no changes in timeliness of reporting of health care–associated diseases in non-SFE–LHDs, nor did completeness change in SFE–LHDs in 2016. Therefore, we excluded these case reports (n = 4158) from the study. The final data set included 63 085 confirmed and probable case reports from 2012 (n = 18 308), 2014 (n = 20 126), and 2016 (n = 24 651).
Data Analysis
During 2 periods, we assessed the timeliness and completeness of NBS data and compared SFE–LHDs with non-SFE–LHDs. For this study, we defined timeliness as the interval from the earliest time a case was reported to the public health system from a reporting entity to notification to DSHS. We defined completeness as the percentage of case reports submitted to DSHS that were submitted only once (ie, the minimum requirements for submission were satisfied so that DSHS could submit the case report to CDC). More specifically, we considered a case report complete if the time stamp of the first notification from an LHD to DSHS matched the time stamp of the reported last notification from an LHD to DSHS. We defined public health processing as the time from the public health system first being notified of a potential case to notification of CDC. We also assessed changes in timeliness and completeness before, during, and after implementation of the SFE program. We used Mann–Whitney tests to determine significant differences in median time intervals for reporting and z scores to determine differences in proportions of completeness. We considered a p-value < .05 to be significant and performed all data analyses using Stata version 14 (StataCorp LLC).
Results
The median number of days required for public health processing in SFE–LHDs in 2012 (before implementation of the SFE program) was 1 day more than the median number of days in non-SFE–LHDs (14 days vs 13 days; Figure 3). However, in 2012, these SFE–LHDs handled 75% of all reported cases. In 2012, 2014, and 2016, all time intervals showed significant differences between SFE–LHDs and non-SFE–LHDs, with the exception of the time from the state receiving a report to notifying CDC; this interval was the same from year to year and across SFE status. In 2016, when all SFEs were working in their respective LHDs, the overall median number of days for public health processing was 10 days for SFE–LHDs and non-SFE–LHDs, an overall reduction from 2012. The total number of case reports increased in both SFE–LHDs and non-SFE–LHDs, from 18 308 reports in 2012 to 24 612 reports in 2016. As before the SFE program, the SFE–LHDs processed most (76.1%) reports overall.
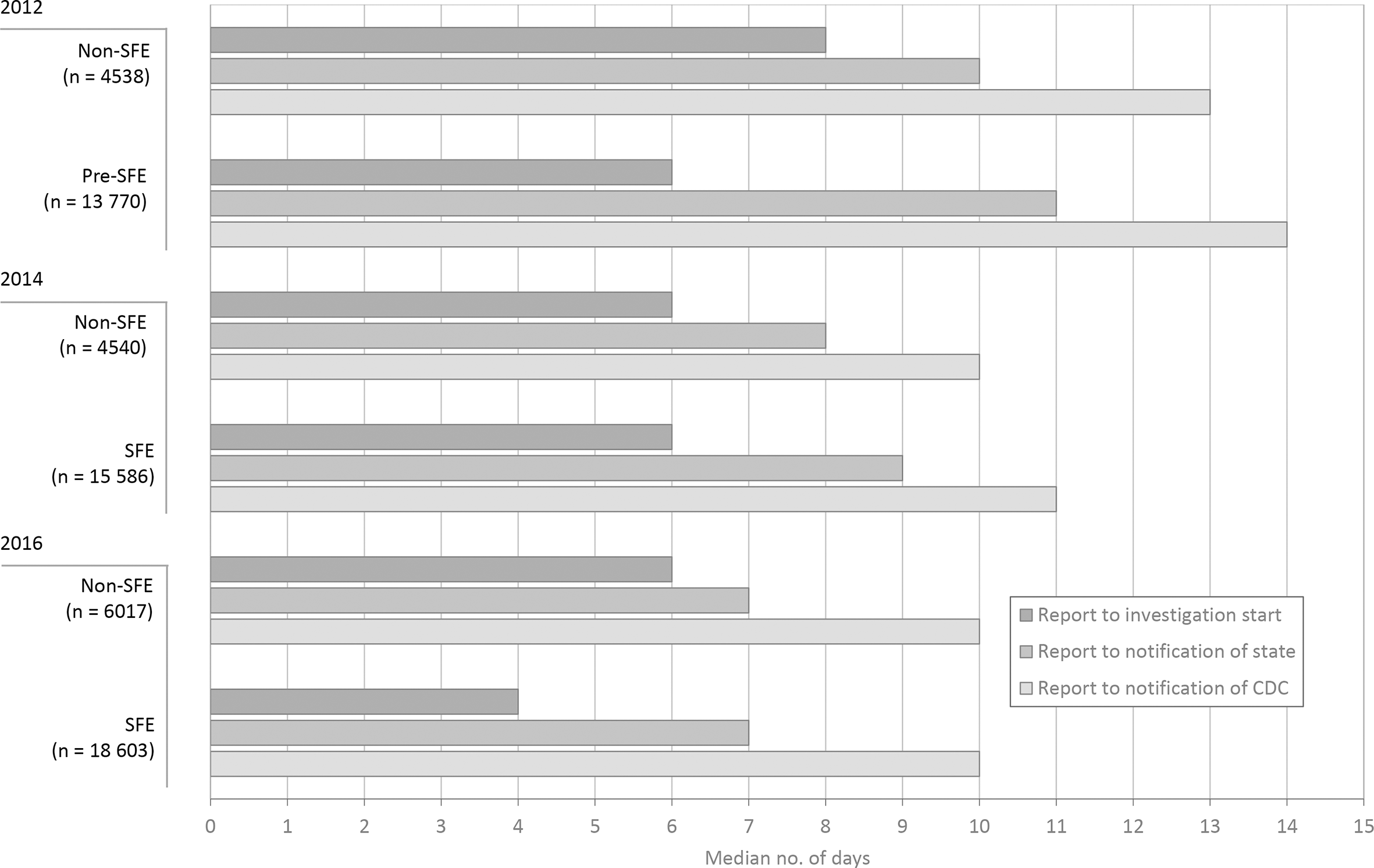
Timeline (median number of days) of public health processing of notifiable conditions in local health departments, by year and whether the department had a state-funded epidemiologist (SFE). 18 Public health processing was defined as the time from notifying the public health system of a potential case to notifying the Centers for Disease Control and Prevention (CDC).
In an examination of SFE–LHDs only, the median number of days from the initial report to notification of DSHS decreased from 11 days in 2012 to 9 days in 2014 and 7 days in 2016 (Table 1). The percentage of completeness also increased from 19.6% in 2012 to 27.7% in 2016.
Timeliness (median number of days) and completeness of infectious disease reporting in the 31 local health departments with a state-funded epidemiologist (SFE) before the SFE program, after phase 1 of the SFE program, and after phase 2 of the SFE program, Texas, 2012-2016 a

aData source: the National Electronic Disease Surveillance System Base System. The Texas Department of State Health Services funded 45 epidemiologist positions in 31 local health departments to increase overall epidemiologic capacity and improve response to infectious disease threats. All changes in days and completeness by year were significant at P < .001. Mann–Whitney tests were used to determine significant differences in median time intervals for reporting, and z scores were used to determine differences in proportions of completeness.
bFrom first case report to public health system to first submission to state health department.
cDefined as the percentage of case reports submitted to the state from the local health department only once (ie, the minimum requirements for submission were satisfied so that the state could submit the case report to the Centers for Disease Control and Prevention).
In 2014 and 2016, three-quarters of case reports were processed by SFE–LHDs across disease categories (Table 2), and SFE–LHDs were responsible for most complete first reports in each disease category in 2014 and 2016. From 2014 to 2016, despite more case reports, the timeliness of case reports for invasive and respiratory infectious disease improved in SFE–LHDs; the timeliness of case reports for foodborne illness and vaccine-preventable disease improved in both SFE–LHDs and non-SFE–LHDs. We observed the greatest improvements in timeliness for foodborne illness at SFE–LHDs (from 11 to 7 days). The timeliness of case reports for high-consequence infectious disease declined.
Timeliness (median number of days) and completeness of reporting of infectious diseases in local health departments with and without state-funded epidemiologists (SFEs), by disease category, Texas, 2014 and 2016 a
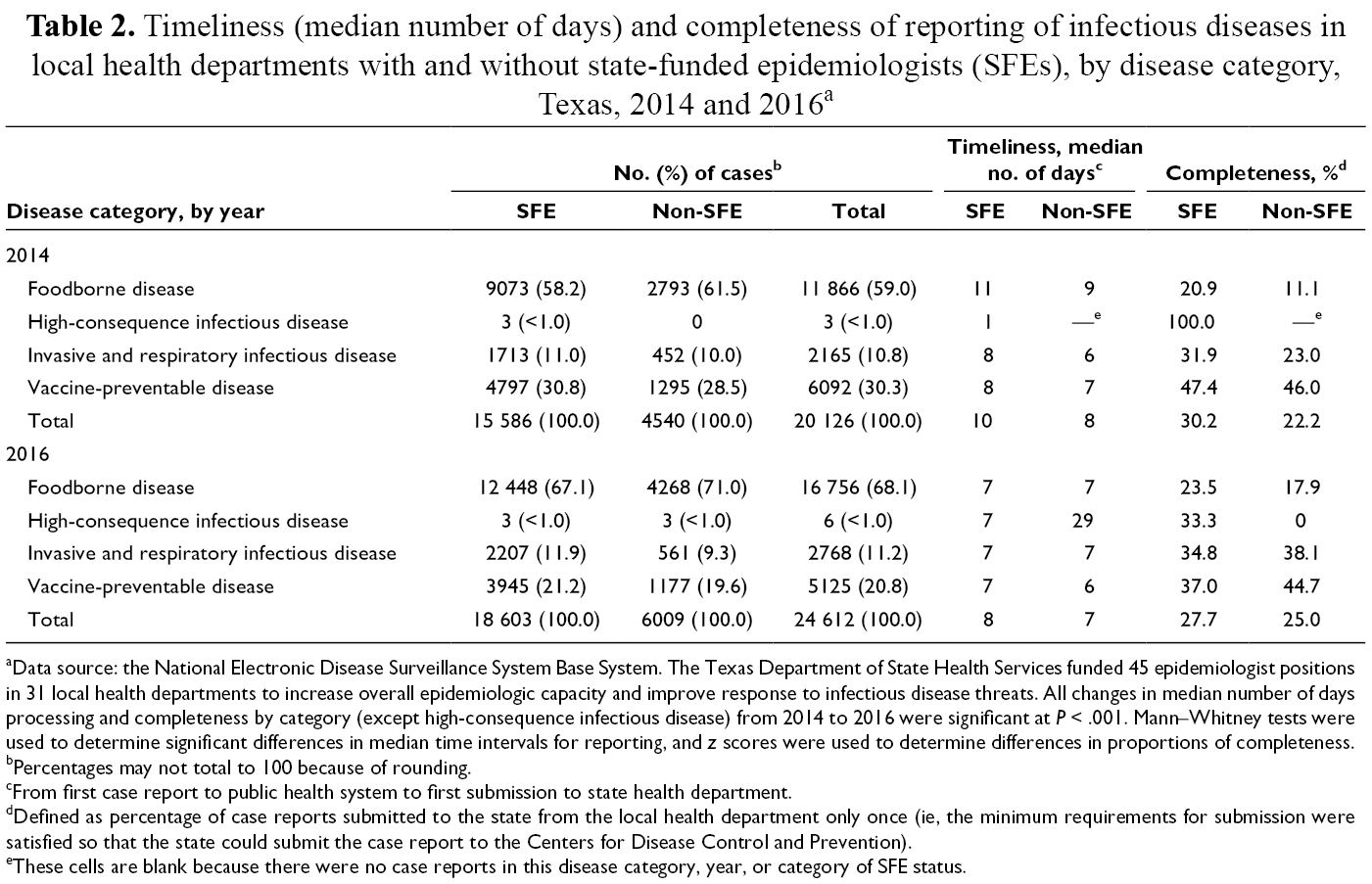
aData source: the National Electronic Disease Surveillance System Base System. The Texas Department of State Health Services funded 45 epidemiologist positions in 31 local health departments to increase overall epidemiologic capacity and improve response to infectious disease threats. All changes in median number of days processing and completeness by category (except high-consequence infectious disease) from 2014 to 2016 were significant at P < .001. Mann–Whitney tests were used to determine significant differences in median time intervals for reporting, and z scores were used to determine differences in proportions of completeness.
bPercentages may not total to 100 because of rounding.
cFrom first case report to public health system to first submission to state health department.
dDefined as percentage of case reports submitted to the state from the local health department only once (ie, the minimum requirements for submission were satisfied so that the state could submit the case report to the Centers for Disease Control and Prevention).
eThese cells are blank because there were no case reports in this disease category, year, or category of SFE status.
From 2014 to 2016, completeness improved in SFE–LHDs and non-SFE–LHDs for foodborne illness (58.2% to 67.1% in SFE–LHDs; 61.5% to 71.0% in non-SFE–LHDs) and invasive and respiratory infectious disease (31.9% to 34.8% in SFE–LHDs; 23.0% to 38.2% in non-SFE–LHDs); completeness increased more in non-SFE–LHDs than in SFE–LHDs. Completeness of reporting did not improve in the other 2 disease categories: high-consequence infectious disease and vaccine-preventable disease.
Discussion
Overall infectious disease reporting timeliness and completeness improved in both SFE–LHDs and non-SFE–LHDs from 2012, before the SFE program was implemented, to 2016, when it was fully functional. Both the time interval between receiving a report and starting an investigation at the LHD and the time interval between starting the investigation and reporting to the state decreased from 2012 to 2016 in both SFE–LHDs and non-SFE–LHDs. Reducing the time to beginning disease investigations is of critical importance in identifying and confirming potential outbreaks, especially high-consequence infectious diseases. 19 In 1988, the Institute of Medicine recommended that all public health agencies systematically collect, analyze, and perform epidemiologic investigations on public health problems. 20 LHDs in Texas perform most disease surveillance investigations in the state; therefore, increasing capacity and timeliness at this level is important. Our study showed an association between the SFE program and improvements in timeliness on all time intervals evaluated. Previous research on the timeliness and completeness of disease surveillance systems in relation to outbreak detection and control agreed that improvements in timeliness of at least 1 day reduce the spread of contagion in the community or hospital settings. 13,21 -25 For many notifiable conditions, the incubation periods and infectious time frames are measured in days, not months. Depending on the condition, an improvement of 3 or 4 days can make a vital difference in detecting and starting control procedures before a second or third incubation period or infectious time frame starts, limiting the spread and resources needed to control the outbreak.
The first phase of the SFE program was designed to improve foodborne illness surveillance and response in Texas in response to increases in foodborne illnesses, outbreak investigations, 10 and multistate Salmonella cluster investigations that resulted from improved molecular identification. The epidemiologists deployed in the first phase of the program were assigned to foodborne illnesses. LHDs with the greatest need for improvements in foodborne disease investigation timeliness and completeness and who had the greatest number of disease reports were issued contracts for an SFE. The LHDs that received an SFE in the first phase improved in their timeliness and completeness of foodborne diseases and processed most of the foodborne illness reports in the state, meeting the goals of the program. SFE–LHDs investigate most local-level notifiable diseases in Texas. In addition to the improvements in median days of report processing and completeness between 2012 and 2016 shown in this study, improvements in the proportion and number of cases identified in these jurisdictions are noteworthy. With the aid of the SFE program, local jurisdictions that had the greatest number of disease reports improved timeliness even as disease report counts increased.
High-consequence infectious diseases were a notable exception to the overall and categorical improvement in timeliness and completeness. This exception can be explained by the changing content of this disease category. In 2014, all cases of high-consequence infectious diseases were cases of Ebola, which was investigated immediately. In 2015, several neglected tropical diseases were added to the list of notifiable conditions. Despite these diseases being categorized as high-consequence diseases, health care providers and epidemiologists do not generally consider gastrointestinal parasites an immediate or high-consequence concern. The reason for the decline in percentage completeness for vaccine-preventable diseases is less easily explained. Vaccine-preventable disease investigation is supported by federal and state contracts, but changes in federal requirements were beyond the scope of our study. Similarly, the marked improvement in completeness of invasive and respiratory infectious disease reporting among non-SFE–LHDS would require additional inquiry.
Another important element of the SFE program was the effect of the SFEs on the shared disease investigation duties at the local and regional health department levels. In Texas, approximately 180 of the 254 counties do not have LHDs, and not all LHDs have epidemiologists or disease investigation staff members for notifiable diseases. DSHS public health regions provide epidemiology and investigation support to all LHDs in their region, including LHDs that have limited or no epidemiologic capacity. The SFE program provided epidemiologists to some jurisdictions that previously had no epidemiologist or disease investigation staff members, and these jurisdictions had relied on the public health region for support. The increased capacity at the local level from the SFE program enabled public health regions to help other jurisdictions without SFEs. This increased capacity could explain why non-SFE–LHDs also saw increases in timeliness and completeness during the study period.
Limitations
Our study had several limitations. First, to mask the identity of reporting jurisdictions, a limited number of variables and characteristics were made available by the Texas NBS (Box). One important omission was SFE status by intervention phase matched to jurisdiction to estimate the effects of adding an SFE over time. We simply characterized SFE status as having received an SFE in phase 1, phase 2, or both phases. The second limitation was the way we assessed completeness. The data set did not contain individual fields in case reports to precisely measure the level of completeness in each report. Instead, the percentage of case reports that were submitted only once to the state without further submissions was deemed complete. Although this measure was likely a good proxy for completeness, we do not know with certainty that the report was complete; we know only that the minimum fields in the submission were met to be processed from DSHS to CDC. The need for additional submissions is typically due to having to provide information that was missing on the first submission or correcting information. Although other reasons for needing additional submissions may exist, we believe the best indicator for a complete report is the need for only 1 submission. In addition, different conditions have different mandatory fields for submission. Two jurisdictions in Texas do not directly use NBS. These jurisdictions use their own surveillance systems and submit their reports to DSHS, which then inputs case reports into NBS. Because of this process, the time intervals in this study are potentially longer than they are in reality. We did not have information on other LHD variables, such as total caseload, number of disease investigators, or number of epidemiologists at each LHD. The number of probable and confirmed cases does not reflect the entire caseload managed by an LHD; many noncase reports are not entered into NBS. For this reason, we did not include noncases in our analysis. However, reports of probable and confirmed case-reporting timeliness and completeness are ultimately of interest.
Despite these limitations, our data suggest that disease-reporting timeliness and completeness improved in LHDs in Texas after implementation of an SFE program, even with larger caseloads in those jurisdictions. These data do not provide the information needed to understand the underlying mechanisms for improvements to timeliness and completeness, but they demonstrate improvements at the system level. Alerting public health officials to cases and outbreaks allows the public health system to respond to disease threats promptly.
Conclusion
Epidemiologic capacity is linked to quality infectious disease surveillance and is crucial to preventing and controlling outbreaks in their early stages. In this study, changes in the timeliness and completeness of disease case reports followed improvements in epidemiologic capacity in the Texas public health system. These improvements in timeliness and completeness promote rapid response to outbreaks, quicker implementation of disease control measures, and enhanced ability to monitor emerging and re-emerging high-consequence diseases. The Texas SFE program demonstrates how state-level appropriations for LHD epidemiologic capacity have local, state, and national implications to disease control and prevention by improving overall disease response capacity at all levels.
Infectious disease variables extracted from the Texas National Electronic Disease Surveillance System Base System, 2012-2016
Abbreviation: LHD, local health department.
aAll cases are assigned a year in the Centers for Disease Control and Prevention’s Morbidity and Mortality Weekly Report.
bDate reviewed and determined to have sufficient data for full report to Centers for Disease Control and Prevention.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
