Abstract
Objectives
Although many people who use drugs meet criteria for vaccination against hepatitis A virus (HAV) and hepatitis B virus (HBV), estimates of susceptibility (ie, lack of immunity) are not well established. This study sought to identify the prevalence of and characteristics associated with HAV and HBV susceptibility among people who use drugs attending an urban syringe services program.
Methods
We initiated this seroprevalence study in 2018 among 438 clients of a syringe services program who met study criteria, including provision of a blood specimen and a self-reported history of drug use. We assessed HAV and HBV susceptibility and infection via serological testing. We examined associations between participant characteristics and serology status by using descriptive statistics and multivariable logistic regression models.
Results
Of the initial 438 clients identified, 353 (80.6%) met study criteria. Of 352 participants with conclusive HAV test results, 48.6% (n = 171) were HAV susceptible; of 337 participants with conclusive HBV test results, 32.6% (n = 110) were HBV susceptible, 24.3% (n = 82) showed evidence of past or present HBV infection, and 43.0% (n = 145) had vaccine-derived immunity. Compared with participants born before 1970, participants born during 1980-1989 had 5.90 (95% CI, 2.42-14.40) times the odds of HAV susceptibility and 0.18 (95% CI, 0.06-0.53) times the odds of HBV susceptibility, and participants born during 1990-1999 had 6.31 (95% CI, 2.34-17.00) times the odds of HAV susceptibility. Decreased odds of HAV susceptibility were associated with homelessness (adjusted odds ratio = 0.48; 95% CI, 0.28-0.82).
Conclusion
Despite applicable HAV and HBV vaccination recommendations, substantial gaps exist in HAV and HBV susceptibility among a population of people who use drugs. These findings highlight the need for increased HAV and HBV vaccination efforts among people who use drugs.
New infections of viral hepatitis in the United States are increasing in parallel with the overdose crisis. 1 -4 Upwards of 2.2 million people are estimated to have chronic hepatitis B virus (HBV) infection. 5,6 The lasting effects of past and present HBV infection include cirrhosis and liver cancer, and people who use drugs (PWUD) are disproportionately affected. 7,8 During 2016 through 2018, approximately 15 000 hepatitis A virus (HAV) cases were identified, representing a nearly 300% increase from the preceding 3-year period. 4 These increases were associated with several large HAV outbreaks among PWUD and people experiencing homelessness in the United States. 4,9 In addition, the number of acute HBV infections has increased among PWUD. 10,11 In the absence of effective prevention measures, the transmission and spread of viral hepatitis infections among PWUD and populations experiencing homelessness are likely to continue.
Viral hepatitis exposures among PWUD occur through unsafe sexual practices and the use of contaminated syringes and other equipment to inject, snort, or smoke substances. 1,11 Some PWUD also experience homelessness, which increases the risk of exposure to HAV and other infectious diseases as a result of poor sanitary conditions and inadequate access to health care services. 12,13 HAV and HBV vaccination is essential to prevent transmission among populations at risk of acquiring these infections. 14,15
HBV and HAV vaccines have been routinely recommended for children since 1991 and 2006, respectively. 14,15 HAV and HBV vaccination is recommended for other populations at risk of infection, including people who inject drugs (PWID) and men who have sex with men, and HBV vaccination is recommended for people with hepatitis C virus (HCV) or HIV infections and residents of correctional facilities. 16 People with HCV infection are at an increased risk for becoming infected with other hepatitis viruses and should be vaccinated if they lack immunity for HAV and HBV. 17
Despite the decrease in HAV and HBV transmission attributed to vaccination and infection control practices, gaps exist in vaccine coverage. For example, a study conducted in Kentucky, Tennessee, and West Virginia in 2006-2013 estimated that only one-quarter of adults at risk of infection who were not vaccinated as children adhered to HAV vaccine recommendations, and one-third of adults who initiated the HBV or combination HAV/HBV vaccine series received all doses within the appropriate time frame. 10 Gaps in coverage are especially concerning in light of community-wide HAV outbreaks and the increase in HBV incidence among adult age groups. 4,9 -11 Despite high levels of exposure risk to HAV and HBV among PWUD and people experiencing homelessness, estimates of immunity resulting from vaccination or previous infection are limited.
Syringe services programs (SSPs) are community-based programs that provide a range of services to PWUD. Some SSPs offer clinical services, including HAV and HBV vaccination. To date, few viral hepatitis serology studies, particularly studies that focus on HAV and HBV immunity, have been conducted among populations that use SSPs. The ongoing overdose crisis makes it critically important to both understand and improve HAV and HBV immunity among people who use SSPs. 10,13 In this study, we assessed HAV and HBV susceptibility among clients of an urban SSP.
Methods
Study Design and Participants
We performed a seroprevalence study using a convenience sample at an SSP in Philadelphia. The SSP provides comprehensive services for PWUD, people experiencing homelessness, and people who exchange sex for money or drugs. Services include routine screening for HIV and HCV, case management, and linkage-to-care coordination for people who receive positive test results for these infections. In 2018, the SSP served approximately 17 000 people, of whom an estimated 14 000 participated in syringe exchange services.
We enrolled study participants and conducted testing from January through April 2018 among people meeting inclusion criteria. The 2 inclusion criteria were being able to speak English or Spanish and providing written consent to participate. We excluded from analysis participants with unsuccessful blood collection. Our initial enrollment oversampled people aged 18-39 to ensure adequate inclusion of younger people. After we obtained 50% of the enrollment goal (n = 400), we opened participation to anyone aged ≥18. We obtained written consent from all participants before study enrollment. We offered all study participants a small incentive (a $5 gift card). Our analytic sample included only participants with a self-reported history of drug use, including heroin, prescription opioids, crack cocaine, and/or benzodiazepines. We excluded people reporting a history of marijuana use with no other drug use.
Measures
Study staff members orally administered a brief questionnaire to participants in either English or Spanish. Questionnaire items included self-reported demographic characteristics, HAV and HBV vaccination history, and behavioral risk factors.
Study staff members obtained blood specimens from participants and transported the specimens to the Philadelphia Department of Public Health Laboratory. Serological testing included measurement of HAV immunoglobulin G antibody, HAV immunoglobulin M antibody, HBV surface antigen (HBsAg), HBV surface antibody (anti-HBs), total HBV core antibody (anti-HBc), and HCV antibody (anti-HCV). We ordered HCV ribonucleic acid (HCV RNA) for participants with reactive anti-HCV and hepatitis D virus (HDV) total antibody for participants with reactive anti-HBc. We coded data as missing when serological results were inconclusive or when participants refused or were unable to answer survey questions.
HAV serological testing cannot distinguish between vaccine-derived immunity and infection-derived immunity; therefore, we defined people with HAV susceptibility as those having a nonreactive HAV immunoglobulin G test result. 18,19 We defined people with an acute HAV infection as those having a reactive HAV immunoglobulin M with jaundice or gastrointestinal symptoms.
HBV serological testing can distinguish between vaccine-derived immunity and other outcomes. HBV outcomes of interest included susceptibility, vaccine-derived immunity, and past or present infection. We defined HBV-susceptible people as those having nonreactive serological results for the following: anti-HBs, anti-HBc, and HBsAg. We defined people with detectable vaccine-derived immunity as those having reactive serological results for the following: anti-HBs, nonreactive anti-HBc, and nonreactive HBsAg. 20 Anti-HBc results indicate whether a person has a chronic, acute, or cleared HBV infection; therefore, we defined past or present HBV infection by a specimen with a reactive anti-HBc. We excluded participants with inconclusive test results for anti-HBs or anti-HBc from an outcome group.
We categorized participants with reactive anti-HCV as having a past or present HCV infection. 21 Confirmed HCV infections were those with positive HCV RNA tests. We categorized participants with nonreactive anti-HCV tests as having no history of HCV infection. We categorized participants with reactive HDV total antibody, in conjunction with results indicating past or present HBV infection, as having a past or present HDV infection. 22
Result Distribution and Vaccination
We continuously disseminated serological results to study participants onsite whenever possible. In July 2018, study staff members attempted to contact by telephone uninformed participants who were not available onsite. We provided harm reduction education, safe sexual practice education, and linkage to care for participants with infections when we communicated test results. We also offered HAV and HBV vaccination onsite to unvaccinated participants and other SSP clients for 6 days in July 2018. We did not obtain information on vaccinations that participants may have obtained elsewhere.
Statistical Analysis
The primary outcomes for this study were HAV and HBV susceptibility, as defined previously. We evaluated each outcome separately. We compared HAV-susceptible participants and participants with any HAV immunity. We assessed the frequency of HBV susceptibility, vaccine-derived immunity, and past or present infection. In addition, we compared self-reported HAV and HBV vaccination history with serology-determined status.
We used the Fisher exact test for variables with small cell counts (sex/gender, race/ethnicity, and HIV status) and the Pearson χ2 test for the remaining variables to identify potential associations between susceptibility status and demographic characteristics, drug use history, and risk factors. For each viral infection, we measured differences between susceptible participants and immune participants. To best characterize factors contributing to HBV susceptibility, the comparison group did not include participants with past or present HBV infection.
We assessed all variables with a P value <.20 with univariable logistic regression models to identify participant characteristics and risk factors independently associated with HAV and HBV susceptibility. Next, we used multivariable logistic regression models to assess each outcome. We conducted a complete-case analysis for the regression analyses. We conducted all statistical analyses using SAS version 9.4 (SAS Institute, Inc).
The Philadelphia Department of Public Health Institutional Review Board reviewed and approved this study.
Results
Of the initial 438 clients identified, we obtained blood specimens from 388 (88.6%). Of these 388 participants, 353 (91.0%) reported a history of any drug use and met the inclusion criteria. The median age of the final study sample (n = 353) was 34.7 (interquartile range, 30.1-41.7). Serological test results were inconclusive for 1 HAV test and 16 HBV tests.
Serological Results
Of 352 participants with conclusive results on HAV serological tests, 48.6% (n = 171) were susceptible to HAV infection (Table 1). We found no acute HAV infections. Of 337 participants with conclusive results on HBV serological tests, 43.0% (n = 145) had evidence of HBV vaccine-derived immunity, 32.6% (n = 110) had evidence of HBV susceptibility, and 24.3% (n = 82) had past or present HBV infection. Among 366 participants with complete HBsAg testing, 1.9% (n = 7) had positive HBsAg, thereby indicating current HBV infection. Overall, 22.1% (78 of 353) of participants were susceptible to both HAV and HBV. Four participants had past or present HDV infection.
Serological results and characteristics of participants in a study on susceptibility to HAV and HBV among clients (n = 353) at a syringe services program in Philadelphia, 2018 a
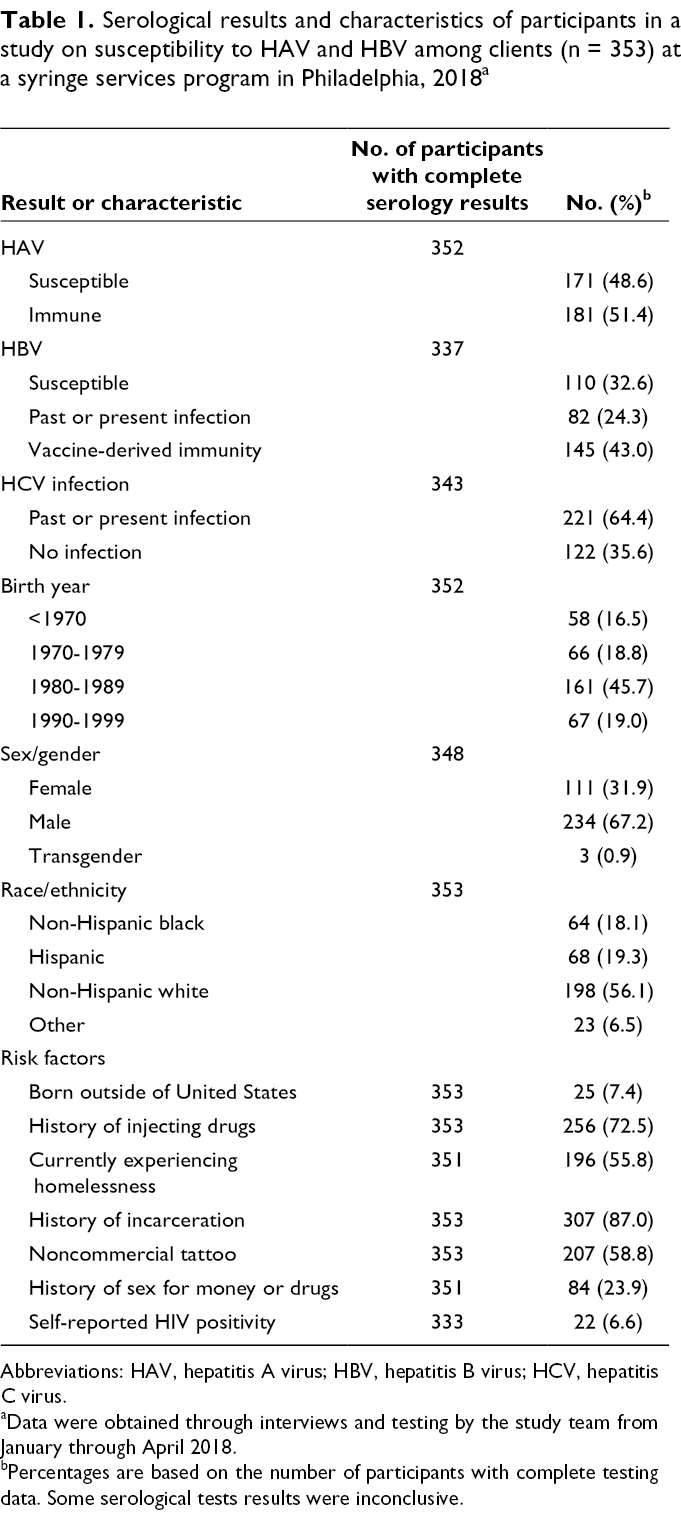
Abbreviations: HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus.
aData were obtained through interviews and testing by the study team from January through April 2018.
bPercentages are based on the number of participants with complete testing data. Some serological tests results were inconclusive.
We communicated serological results to 24.6% (87 of 353) of participants. Of 218 participants with no evidence of HAV or HBV immunity, 10 (4.6%) returned to the SSP for vaccination.
Characteristics of HAV Susceptibility
By age group, proportions of HAV susceptibility were highest among participants born during 1980-1989 (55.9%) and 1990-1999 (62.7%) (Table 2). HAV susceptibility was higher among participants with no history of injecting drugs than among participants with a history of injecting drugs (67.0% vs 41.6%). HAV susceptibility was common among those who were also HBV susceptible (70.9%; 78 of 110). HAV susceptibility was more common among participants with no history of HCV infection (63.9%; 78 of 122) than among participants with past or present HCV infection (40.6%; 89 of 219).
Characteristics of study participants by HAV and HBV susceptibility in a study among clients (n = 353) at a syringe services program in Philadelphia, 2018 a
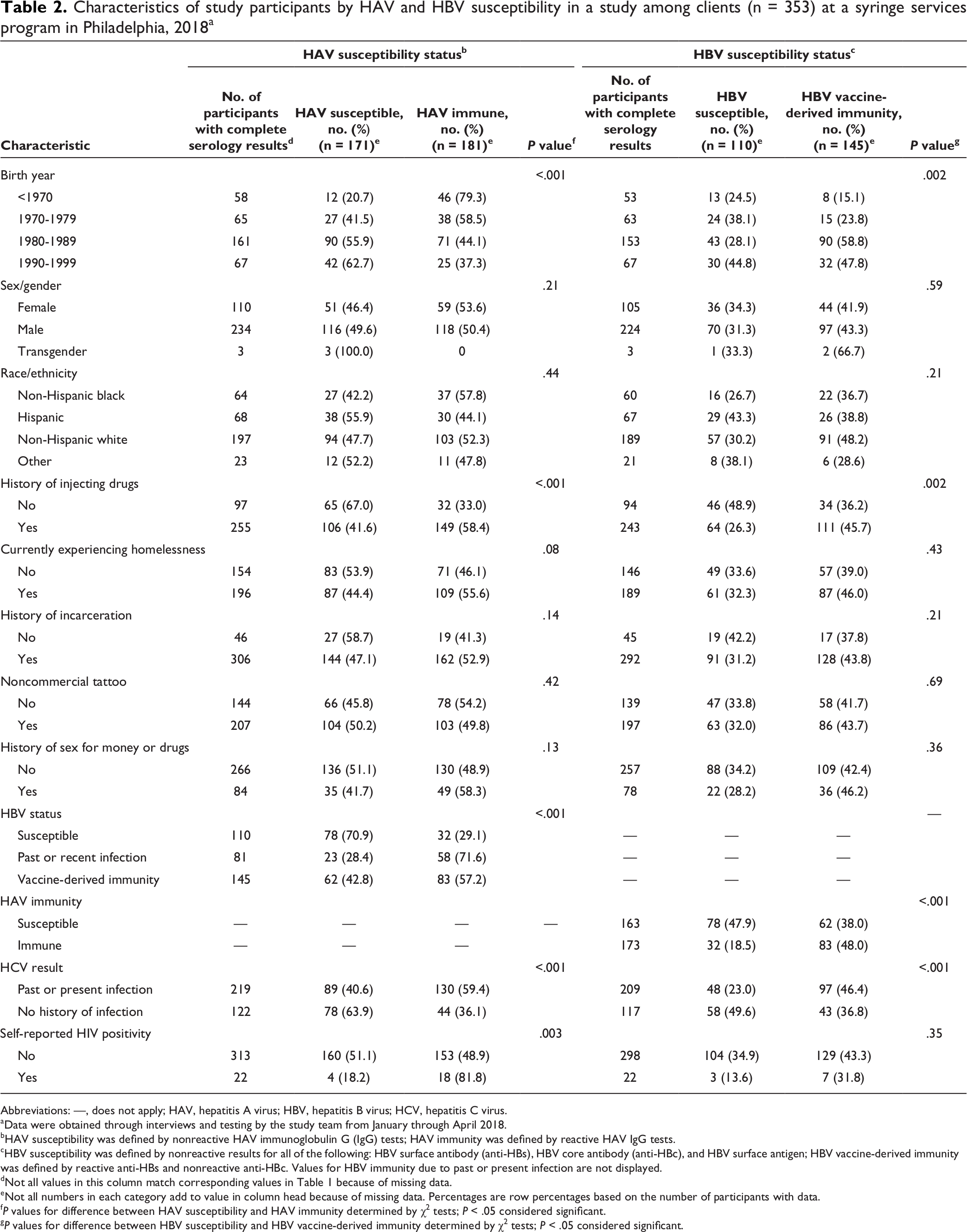
Abbreviations: —, does not apply; HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus.
aData were obtained through interviews and testing by the study team from January through April 2018.
bHAV susceptibility was defined by nonreactive HAV immunoglobulin G (IgG) tests; HAV immunity was defined by reactive HAV IgG tests.
cHBV susceptibility was defined by nonreactive results for all of the following: HBV surface antibody (anti-HBs), HBV core antibody (anti-HBc), and HBV surface antigen; HBV vaccine-derived immunity was defined by reactive anti-HBs and nonreactive anti-HBc. Values for HBV immunity due to past or present infection are not displayed.
dNot all values in this column match corresponding values in Table 1 because of missing data.
eNot all numbers in each category add to value in column head because of missing data. Percentages are row percentages based on the number of participants with data.
f P values for difference between HAV susceptibility and HAV immunity determined by χ2 tests; P < .05 considered significant.
g P values for difference between HBV susceptibility and HBV vaccine-derived immunity determined by χ2 tests; P < .05 considered significant.
Among 171 HAV-susceptible participants, 53.2% (n = 91) reported not previously receiving an HAV vaccination, 30.4% (n = 52) did not know their vaccination status, and 16.4% (n = 28) reported previously receiving an HAV vaccination.
HAV susceptibility was more likely among participants born during 1990-1999 (adjusted odds ratio [aOR] = 6.31; 95% CI, 2.34-17.00) or during 1980-1989 (aOR = 5.90; 95% CI, 2.42-14.40) than among participants born before 1970 (Table 3). HAV susceptibility was less likely among participants who were experiencing homelessness (aOR = 0.48; 95% CI, 0.28-0.82) than among participants who were not experiencing homelessness.
Unadjusted and adjusted odds ratios for HAV and HBV susceptibility by participant characteristics in a study among clients (n = 353) at a syringe services program in Philadelphia, 2018 a
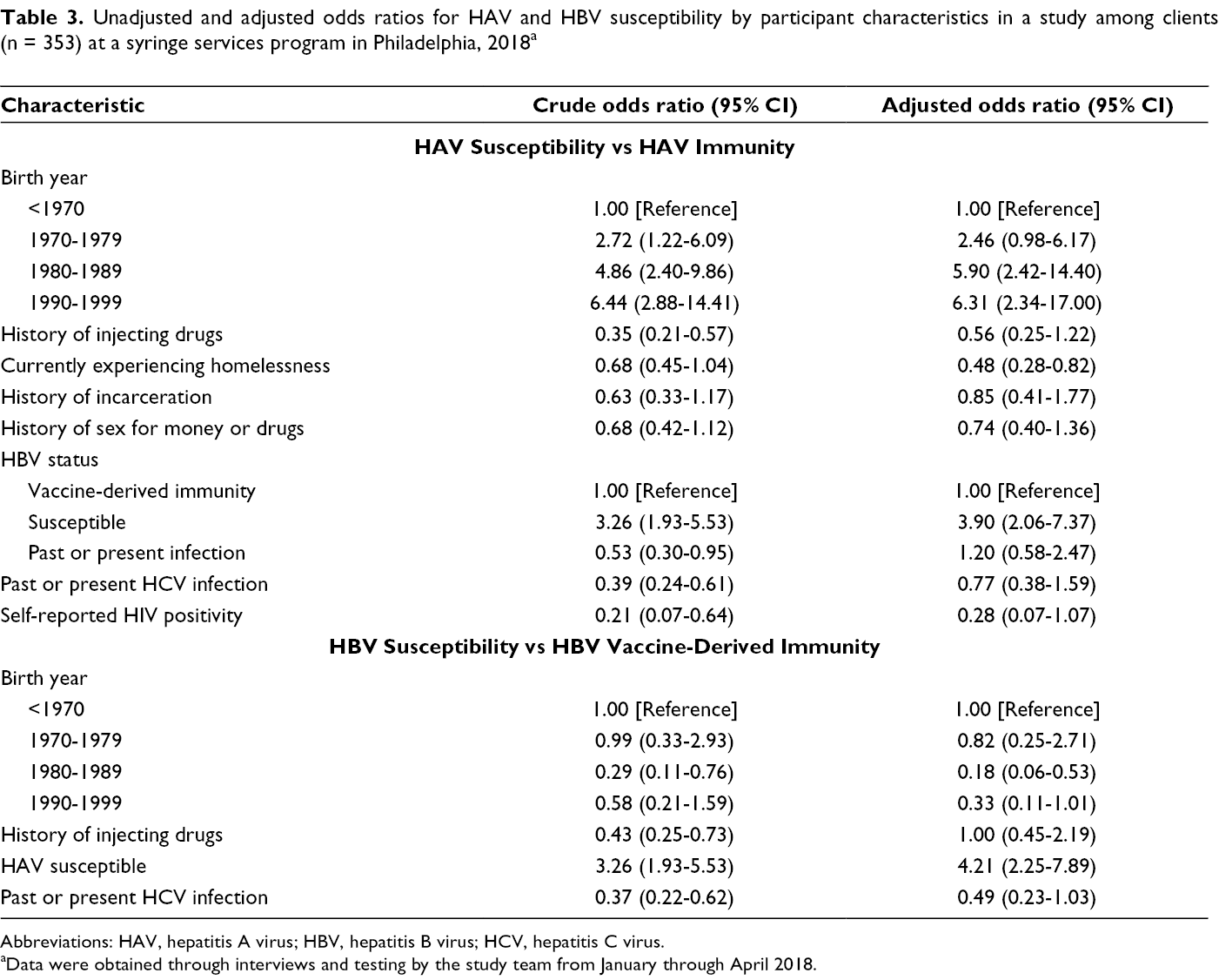
Abbreviations: HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus.
aData were obtained through interviews and testing by the study team from January through April 2018.
Characteristics of HBV Susceptibility
Participants with no history of HCV infection had a higher proportion of HBV susceptibility than participants with past or present HCV infection (49.6% vs 23.0%) (Table 2). Of the 109 HBV-susceptible participants, 55.0% (n = 60) reported not previously receiving an HBV vaccination, 29.4% (n = 32) did not know their vaccination status, and 15.6% (n = 17) reported previously receiving an HBV vaccination.
In the adjusted model, participants born during 1980-1989 were less likely than participants born before 1970 to be HBV susceptible (aOR = 0.18; 95% CI, 0.06-0.53) (Table 3). We found no significant differences in HBV susceptibility among participants born before 1970, during 1970-1979, or during 1990-1999. Participants who were HAV susceptible were more likely to be HBV susceptible than participants who were HAV immune (aOR = 4.21; 95% CI, 2.25-7.89).
Discussion
Our study found substantial gaps in HAV and HBV susceptibility among PWUD clients of an SSP in Philadelphia, a population at particular risk for both infections. This study is one of the few that assessed serological markers for HAV and HBV susceptibility among this group. 23 A notable proportion of participants showed evidence of previous HBV and HCV infection, consistent with known associations among PWUD. 10,11 Many participants were also susceptible to HAV. In light of current outbreaks among PWUD and people experiencing homelessness, 2,4 -11,13 our study results are concerning and demonstrate a critical need for improved vaccination efforts in this population. As the overdose crisis continues, the increasing incidence and prevalence of viral hepatitis infection is likely to continue unless improved harm reduction approaches, including vaccination, are widely implemented. 1,24
Although studies on the seroprevalence of HAV and HBV susceptibility among SSP participants are limited, some studies were conducted among similar populations. 23,25,26 One study conducted among PWID found higher proportions of HAV and HBV susceptibility than our study did. 23 A study using a nationally representative sample also found higher proportions of HAV and HBV susceptibility than our study did. 25 Another study among PWID with evidence of HCV infection found higher proportions of HAV and HBV susceptibility; however, these findings were based on records of vaccination history, not serological measures. 26
Although we found that age was inversely related to HBV susceptibility, significant proportions of all age groups in our study did not have HBV immunity. The proportion of HBV susceptibility was higher among participants born during 1990-1999 than among participants born before 1990, suggesting that HBV vaccination may not be resulting in comprehensive coverage. Interestingly, we found no notable differences in demographic characteristics or risk factors (eg, country of birth, race/ethnicity) among participants who were HBV susceptible, between participants born during 1990-1999 and participants born before 1990. However, of participants born during 1990-1999, 20% reported previous HBV vaccination. We found that HAV susceptibility was negatively correlated with age. However, this correlation could be due, in part, to older adults having a longer window of exposure to infection than younger adults. We could not differentiate between immunity from natural infection and immunity from vaccination; however, previous studies showed that naturally derived immunity likely contributed to immunity with increased age. 27
Because national guidelines recommend HAV vaccination for PWUD and HBV vaccination for PWID or any adult wanting vaccination, all participants were eligible to receive both vaccines. 14,15 However, a significant proportion of participants in our study showed no evidence of immunity to either HAV or HBV. Many possible explanations exist for the lack of immunity. First, the stigma associated with drug use may keep people from reporting risk factors to their clinicians. Second, some health care providers may rely on self-reported vaccine history to determine vaccination needs. Several studies, including ours, demonstrated that self-reported vaccination history insufficiently predicts immunity. 23,28 Third, despite well-established risks among people experiencing homelessness, HAV vaccination recommendations did not target this population until 2018. 29 A large proportion of our study population was experiencing homelessness, of whom nearly half were HAV susceptible.
Inconsistent access to health care services is a critical barrier to HAV and HBV vaccination among many populations at risk of infection. In addition to being available in primary care, HAV and HBV vaccines should be provided to PWUD in a wider range of medical settings, including emergency departments, social service organizations, and correctional facilities. Although a large majority of our study participants had a history of incarceration, many participants were HAV and HBV susceptible. Vaccination in jails and prisons could increase population-level immunity among people at high risk of infection. The new 2-dose HBV vaccination can be implemented within 1 month, whereas the previous 3-dose series occurred during a 6-month period. SSPs have been established as a successful venue to promote and provide vaccination, and federal resources exist to support vaccination access at SSPs with clinical services. 30 -32 Resource constraints, however, may still prevent some SSPs and other organizations from initiating vaccination services; local and state governments should work with these providers to facilitate access to vaccination among populations at high risk of infection. To streamline efforts across services, all providers should use their state or county immunization information system, the population-based immunization database, to record and reference a patient’s vaccination history. 33 Other barriers to vaccination may include provider- and system-level issues in vaccine reimbursement, administration, and documentation.
Because of the difficulty in engaging PWUD in health care, providing HAV and HBV booster vaccinations, without reliance on serological test results, should be evaluated and considered. Because serology-based immunity testing does not provide point-of-care results, vaccination follow-up requires an additional visit, which can introduce an additional barrier to care. In our study, follow-up among participants for test results and subsequent vaccination was poor, such that the opportunity was lost to vaccinate many people at risk of HAV and HBV infection. When people are at high risk of infection and are hard to reach (ie, loss to follow-up is likely), clinicians should provide vaccination before receiving serological test results, if delayed results of serological testing may interfere with subsequent vaccine initiation. 15,17 The cost/benefit of potentially revaccinating people at high risk for HAV and HBV infection, in the absence of serological testing, should be considered. One dose of HAV vaccination without serological testing is already recommended and is frequently administered in outbreak environments. 14
Limitations
Our study had several limitations. First, the study indicated seroprevalences at a single point in time and did not account for all aspects of temporality in virus exposures in relation to individual risk factors. Although our study results should not be interpreted etiologically, they can be used to inform public health interventions. Second, given that the prevalence of viral hepatitis susceptibility and disease may vary geographically, our results may not be generalizable to all PWUD populations. In addition, the study population included only PWUD who were engaged with the SSP and, thus, may not be representative of other PWUD populations. However, national increases in HAV and HBV infection among PWUD suggest similarly high proportions of susceptibility. 2,4,10,11,13 Third, we were unable to obtain blood samples from 11% of prospective participants because of damaged veins or dehydration, and the risks of viral hepatitis infection among these participants may differ from the risks among our study participants. Fourth, we used nonreactive anti-HBs to define whether a participant had vaccination-based immunity for HBV. This criterion may have underestimated the true prevalence of vaccination-based immunity; anti-HBs is not detectable in some vaccinated people even though they have viable immune memory to protect them from HBV infection. 34
Conclusions
Our study suggests the need for increased HAV and HBV vaccination services among PWUD, particularly people who are experiencing homelessness. Despite national risk-based vaccination recommendations, many PWUD do not have evidence of immunity to both HAV and HBV infection. Providing vaccination services in a comprehensive range of settings may be an important strategy to build protective levels of immunity in drug-using and homeless communities and to prevent future HAV and HBV outbreaks from occurring.
Footnotes
Acknowledgments
We thank Kerry Bucks, MS, Philadelphia Public Health Laboratory; Chari Cohen, Hepatitis B Foundation; and Kristen Feemster, MD, MPH, Philadelphia Department of Public Health, for their important feedback on our study design and interpretation of results.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Centers for Disease Control and Prevention Cooperative Agreement for Improving Hepatitis B and C Care Cascades (NU51PS005109) and a grant from the Hepatitis B Foundation. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Centers for Disease Control and Prevention.
