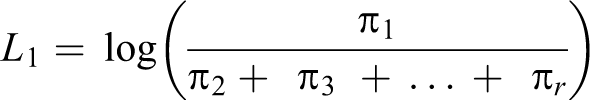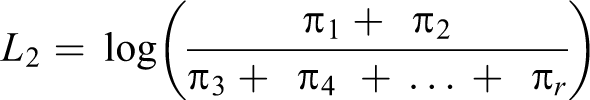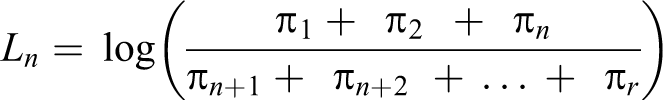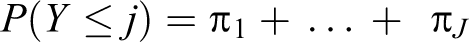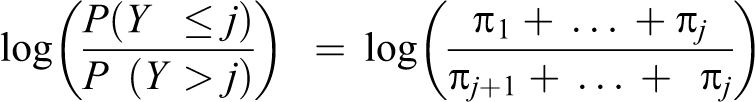Abstract
Objective:
Cardiometabolic disease is the leading cause of mortality in the United States. Cardiometabolic function during adolescence predicts future cardiometabolic disease, yet few studies have examined early determinants of cardiometabolic function. Informed by evidence of sex differences in the prevalence and severity of cardiometabolic disorders and evidence of sexual dimorphism in the stress response, we examined sex differences in the association between living in poverty and cardiometabolic function during adolescence, a precursor of later cardiometabolic disorders.
Methods:
We linked data from 10 415 adolescents aged 12-19 in the National Health and Nutrition Examination Survey (1999-2012) with US Census–tract data on area-level poverty (percentage of the population living in poverty, grouped into quartiles). We parameterized cardiometabolic dysfunction by summing the z scores of 6 cardiometabolic biomarkers, grouped into quintiles. Hierarchical ordinal models estimated associations.
Results:
Compared with residents in low-poverty areas, residents in high-poverty areas had elevated odds of cardiometabolic dysfunction (highest quartile of poverty odds ratio [OR] = 1.27; 95% confidence interval [CI], 1.08-1.50). This association was more pronounced among boys than girls (highest quartile of poverty for boys: OR = 1.36; 95% CI, 1.10-1.70; highest quartile of poverty for girls: OR = 1.17; 95% CI, 0.94-1.47).
Conclusion:
Our study supports the existence of sex-specific associations. These results highlight the potential for community-based programs, such as housing assistance, to improve population health.
Cardiometabolic disease, the leading cause of mortality in the United Sates, 1 has pronounced sex differences. Men have earlier onset of, higher prevalence of, and higher mortality from cardiovascular disease than women. 1 Sex differences in precursors of cardiometabolic disease, however, are underexamined. 2 This scarcity of research led the American Heart Association to call for an investigation of sex differences in precursors of cardiometabolic disease, with a focus on early experiences of adversity. 2
Although the literature on physiologic sequalae of adverse childhood events provides compelling evidence of an association between exposure to adversity and cardiometabolic function, 3 adverse childhood events focus on individual-level experiences (ie, sexual abuse, parental divorce) 4 and not on contextual adversity. Residence in economically deprived areas, a well-recognized contextual source of adversity, 2 may be linked with cardiometabolic function in adolescence. Exposure to economic deprivation during childhood 5 may be associated with the physiologic stress response, which in turn may be associated with increased cardiometabolic risk, 6 but this hypothesis is under investigation.
To our knowledge, only 5 studies have examined the link between residence in economically deprived areas and cardiometabolic function in adolescence. 7 -11 These studies found that residents of economically deprived areas are more likely than residents of economically disadvantaged areas to have adiposity, 7 -9 high allostatic load (a construct related to cardiometabolic function), 10 or cardiometabolic dysfunction. 11 Four of these studies examined differences by sex. Among participants aged 12-19 in the National Longitudinal Study of Adolescent Health (waves 2 and 3 [1996-2002], n = 7788) 7 and among Canadian adolescents aged 11-16 in the Health Behavior in School-Aged Children Survey (2001-2002, n = 6684), the studies found no differences by sex. 8 In contrast, among participants aged 2-18 in the National Longitudinal Survey of Youth (1986-2010, n = 11 504) 9 and participants aged 12-20 in the National Health and Nutrition Examination Survey (NHANES; 1999-2006, n = 11 886), 10 the association between neighborhood-level deprivation and obesity 9 and allostatic load 10 was higher among girls than boys. Both studies with positive findings had large samples and measured their outcome variables according to objective criteria. In contrast, both studies with negative findings 7,8 had self-reported outcomes. Random measurement error likely contributed to these null findings.
Another reason to suspect that the association between adversity and cardiometabolic function is sex specific is that the human stress response is sexually dimorphic. Important sex differences exist in the functioning of the brain’s stress circuitry (ie, the hypothalamic-pituitary-adrenal [HPA] axis). 12,13 This dimorphic HPA-regulated stress response is primarily mediated by changes in hormones released during critical periods of development. 14 Sex differences in placental and embryonic responses to stressors are evident during gestation and become more pronounced during adolescence. 12 -14 Connectivity and functionality of the HPA axis is modified by a rise in gonadal hormones during adolescence. 14 For example, the growth rate of certain regions of the adrenal cortex diverges among boys and girls: in 1 study, the growth rate significantly decreased among boys, and boys also had a diminished HPA stress response compared with girls. 13 These sex-specific changes are predominantly mediated by increased testosterone production during adolescence. 12 Despite the relevance of sex in the stress response, only 4 studies have investigated sex differences in early precursors of cardiometabolic disease. 7 -10
We examined sex differences in the association between residence in economically deprived areas and cardiometabolic dysfunction among US adolescents to better understand cardiometabolic consequences of sexually dimorphic stress responses in the face of contextual-level adversity. Cardiometabolic dysfunction during adolescence is a marker for onset of cardiometabolic disease during adulthood. 15,16 Although adolescents rarely meet diagnostic criteria for cardiometabolic disease, those with cardiometabolic functioning deviating from population-level norms are at elevated risk of developing cardiometabolic disease during adulthood. 16 -18 We hypothesized that the cardiometabolic consequences of residence in an economically deprived area differ by sex.
Methods
Sample
We examined data from the 1999-2012 NHANES, a cross-sectional survey that provides vital and health statistics from a nationally representative sample of the noninstitutionalized US population conducted continuously in 2-year cycles. 19 We selected the analytic sample from 13 343 adolescents aged 12-19. We excluded respondents who reported current pregnancy (n = 181) or a diagnosis of hypertension or diabetes or using medication for hypertension or diabetes (n = 83). We excluded participants with missing data on any of the cardiometabolic function variables (n = 2664), leaving an analytic sample of 10 415 adolescents aged 12-19.
We drew area-level data from the 2000 US decennial census 20 and 5-year estimates (2005-2009; 2009-2013) from the American Community Survey. 21 We linked individual- and area-level data with contemporary census tract identifiers.
Cardiometabolic Dysfunction Score
As detailed elsewhere, 11 we created an index of cardiometabolic dysfunction because composite measures of cardiometabolic dysfunction during adolescence predict later cardiometabolic health better than any individual biomarker alone. 17 Adolescents with cardiometabolic functioning deviating from population-level norms are at elevated risk of developing cardiometabolic disease during adulthood. 16,17 Our choice of biomarkers was informed by the literature on their predictive utility and the degree of missingness for the biomarkers available in NHANES. 15,22,23 We selected 6 biomarkers, all of which were obtained during the mobile examination center component of NHANES 19 : glycosylated hemoglobin A1c (HbA1c; 3-month average blood glucose level, %), mean systolic and diastolic blood pressure (mm Hg) when ≥2 measurements were available (n = 10 266; 98%), high-density lipoprotein (HDL) cholesterol (mg/dL), total cholesterol (mg/dL), and waist circumference (cm). We chose total cholesterol as a measure of lipid metabolism because it tracks over time and allows for sufficient sample size compared with other measures of lipid metabolism (ie, triglycerides), which were available for only a fasting subsample of NHANES participants. 22 We chose HbA1c as a measure of glucose metabolism because it tracks well over time and is less influenced than fasting glucose by recent events. 23
Sex-specific physiologic differences during adolescence contribute to sex-specific cardiometabolic profiles. For example, compared with boys, girls typically enter adolescence earlier, achieve their adult height at an earlier age, and develop a greater percentage of adipose tissue, 24 which is associated with lower HDL cholesterol levels among boys than among girls. 25 Compared with girls, boys have greater growth velocity and develop more lean muscle, a more dense skeleton, and greater blood mass, which is related to higher blood pressure levels. 24 Thus, we standardized cardiometabolic biomarkers by age and sex, except for blood pressure, which we standardized by age, sex, and height, consistent with guidelines for measuring hypertension in adolescents. 26 We summed z scores for each biomarker to create an overall cardiometabolic dysfunction index. This method of calculating the cardiometabolic dysfunction index can result in negative numbers, because the 2 blood pressure variables are standardized on an external reference category. 26 Higher cardiometabolic dysfunction scores indicate greater dysfunction (HDL was multiplied by –1 for directionality). We parametrized quintiles of cardiometabolic dysfunction, with the lowest quintile as the reference group.
This index is not a validated clinical predictor of disease and was designed to promote a better understanding of preclinical levels of cardiometabolic dysfunction that may increase the risk for future cardiometabolic disease. The biomarkers included in the index are informed by biomarkers used to diagnose metabolic syndrome. 17 Each biomarker included in the index is predictive of future disease, and adolescents with metabolic syndrome are at increased risk for cardiometabolic disease in adulthood. 15,27 -30
Area-Level Poverty
We used the proportion of residents in a census tract living below the federal poverty level as a measure of area-level poverty. Area-level poverty is consistently measured over time, easily interpretable, and relates well to other area-level measures of socioeconomic status (eg, housing quality, crowded living spaces). 31 We measured area-level poverty at the census-tract level, because census tracts are considered more economically homogenous and more socially relevant and produce more consistent observations than other small geographies. 31 We parameterized area-level poverty into quartiles (coded from 1-4, with 1 = low poverty).
Covariates included a 2-year survey cycle, race/ethnicity (non-Hispanic white, non-Hispanic black, Mexican American, and other), family poverty-to-income ratio (ie, ratio of family income to federal poverty level based on family size; continuous), head of household education level (<high school diploma, high school diploma, >high school diploma), serum cotinine levels (continuous), physical activity (with responses to the question, “Do you do any vigorous-intensity sports, fitness, or recreational activities that cause large increases in breathing or heart rate, like running or basketball, for at least 10 minutes continuously?” coded as 1 for “yes” and 0 for “no”), and prevalence of non-Hispanic black population at the census-tract level (continuous). We did not include age and sex in the models because cardiometabolic dysfunction was standardized by age and sex.
Statistical Analysis
We obtained descriptive statistics by using survey commands accounting for complex survey design. We did not transform HDL cholesterol levels for directionality for descriptive statistics, so that higher levels of HDL cholesterol indicate better cardiometabolic function. Second, we estimated the association between area-level poverty and cardiometabolic dysfunction with a series of hierarchical ordinal regression models. We made this choice for ease of interpretation of results. Progression from one unit to another on a continuous scale reflects a small change in the risk of cardiometabolic dysfunction; in contrast, progression from one category to the next category arguably corresponds to a more biologically meaningful change in risk, because each ordinal category includes multiple units in a continuous scale.
Moreover, an ordinal variable may better predict the risk of later development of disease than a dichotomous variable. 17 Each level of cardiometabolic dysfunction serves as a threshold sequentially, yielding a series of binary estimates in which the odds of being in categories at or above that threshold is compared with the odds of being in categories below that threshold (Box, equations 1-3). The cumulative odds ratio (OR) reflects the combined odds of worse cardiometabolic dysfunction across all possible thresholds, interpreted as the odds of being in any category of worse cardiometabolic dysfunction (Box, equation 4). We obtained ORs and 95% confidence intervals (CIs) from hierarchical ordinal regression models fit by using PROC GLIMMIX in SAS version 9.3, 32 using the lowest cardiometabolic dysfunction category as the reference category.
Ordinal logistic regression model specification for the association between area-level poverty and cardiometabolic dysfunction among adolescents aged 12-19, United States, 1999-2012
Equations 1-3
Equation 4
Components π1: Probability of being in category 1 of cardiometabolic dysfunction π2: Probability of being in category 2 of cardiometabolic dysfunction π3: Probability of being in category 3 of cardiometabolic dysfunction π4: Probability of being in category 4 of cardiometabolic dysfunction πr: Probability of being in a higher-ordered category of cardiometabolic dysfunction πj: Threshold of interest (cardiometabolic dysfunction category 1)
We conducted overall and sex-specific analyses given previous evidence suggesting sex-specific physiology during adolescence 12,14 and that the experience of residing in a high-poverty area may differ by sex. First, we fit a model that included area-level poverty and indicator variables for survey cycle. Second, we included race/ethnicity, family poverty-to-income ratio, head of household education level, serum cotinine level, and physical activity. Third, we added the prevalence of the non-Hispanic black population to the model.
We treated responses of “don’t know,” “refused,” and ”missing” as missing. We imputed missing values for family poverty-to-income ratio, serum cotinine level, physical activity, and head of household education by using SAS PROC MI in SAS version 9.3. 32 Because NHANES uses a complex, stratified, multistage sample design, all analyses used mobile examination center survey weights. We analyzed restricted-use NHANES data in the Research Data Center at the National Center for Health Statistics. The National Center for Health Statistics reviewed all data, and the University of Maryland Institutional Review Board considered this project exempt from review.
Results
We found a linear association between area-level poverty and cardiometabolic dysfunction (Table 1). The cardiometabolic dysfunction score was on average 20% higher among residents of the highest-poverty areas (ie, quartile 4) than among residents of the lowest-poverty areas (ie, quartile 1; mean scores: –0.69; 95% CI, –0.77 to –0.61 [quartile 4] vs –0.86; 95% CI, –0.94 to –0.79 [quartile 1]).
Weighted mean cardiometabolic function scores and cardiometabolic biomarkers, by demographic characteristics and area-level variables, National Health and Nutrition Examination Survey, 1999-2012a
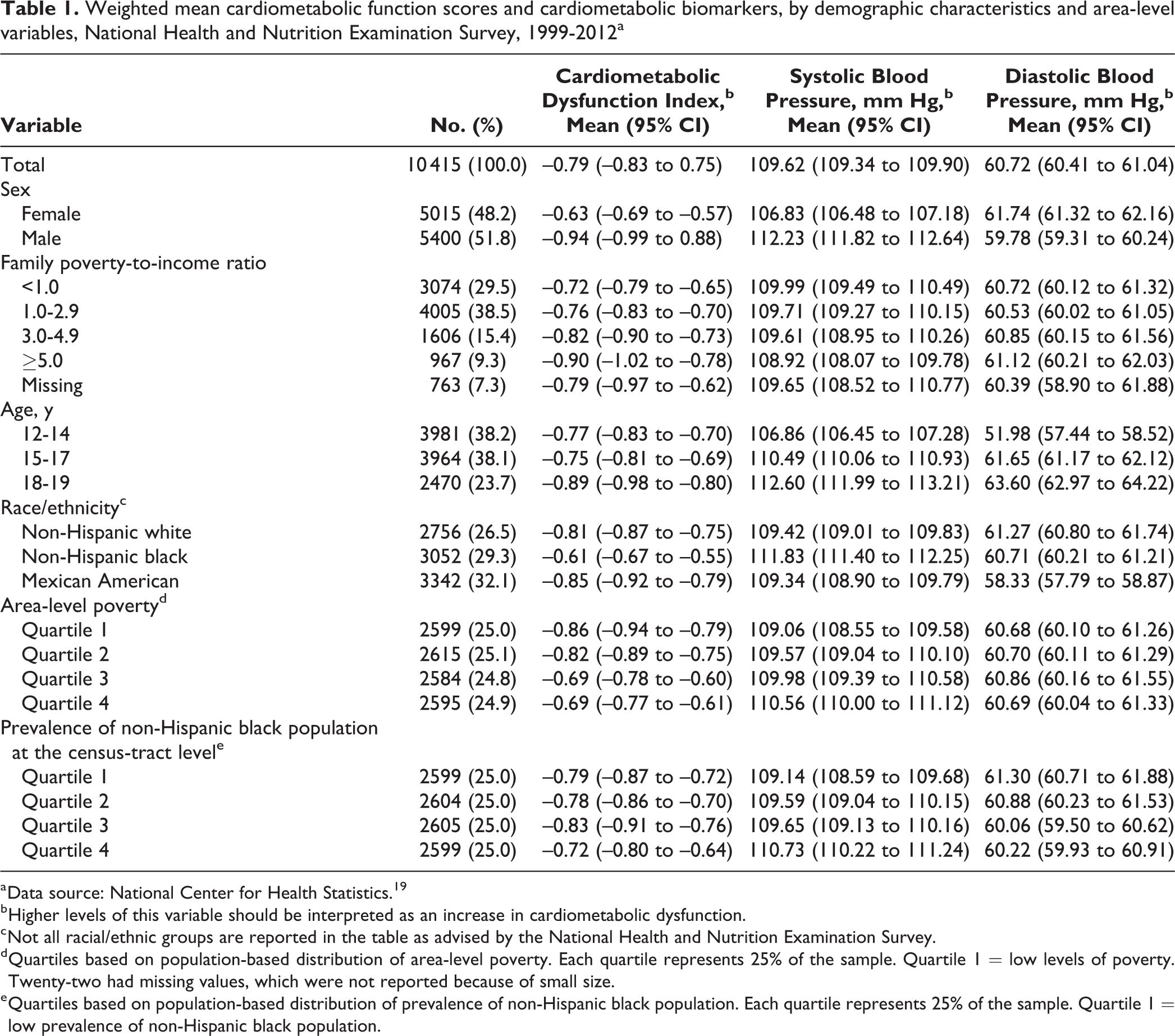
a Data source: National Center for Health Statistics. 19
b Higher levels of this variable should be interpreted as an increase in cardiometabolic dysfunction.
c Not all racial/ethnic groups are reported in the table as advised by the National Health and Nutrition Examination Survey.
d Quartiles based on population-based distribution of area-level poverty. Each quartile represents 25% of the sample. Quartile 1 = low levels of poverty. Twenty-two had missing values, which were not reported because of small size.
e Quartiles based on population-based distribution of prevalence of non-Hispanic black population. Each quartile represents 25% of the sample. Quartile 1 = low prevalence of non-Hispanic black population.
We observed differences in mean cardiometabolic dysfunction by sex. Boys had lower mean cardiometabolic dysfunction scores (mean: –0.942; 95% CI, 0.998 to –0.887) than girls (mean: –0.634; 95% CI, –0.691 to –0.577). Boys also had higher mean systolic blood pressure, waist circumference, and HbA1c than girls, whereas girls had higher mean diastolic blood pressure, total cholesterol, and HDL cholesterol than boys (Tables 1 and 2).
Weighted mean cardiometabolic function scores and cardiometabolic biomarkers, by demographic characteristics and area-level variables, National Health and Nutrition Examination Survey, 1999-2012a
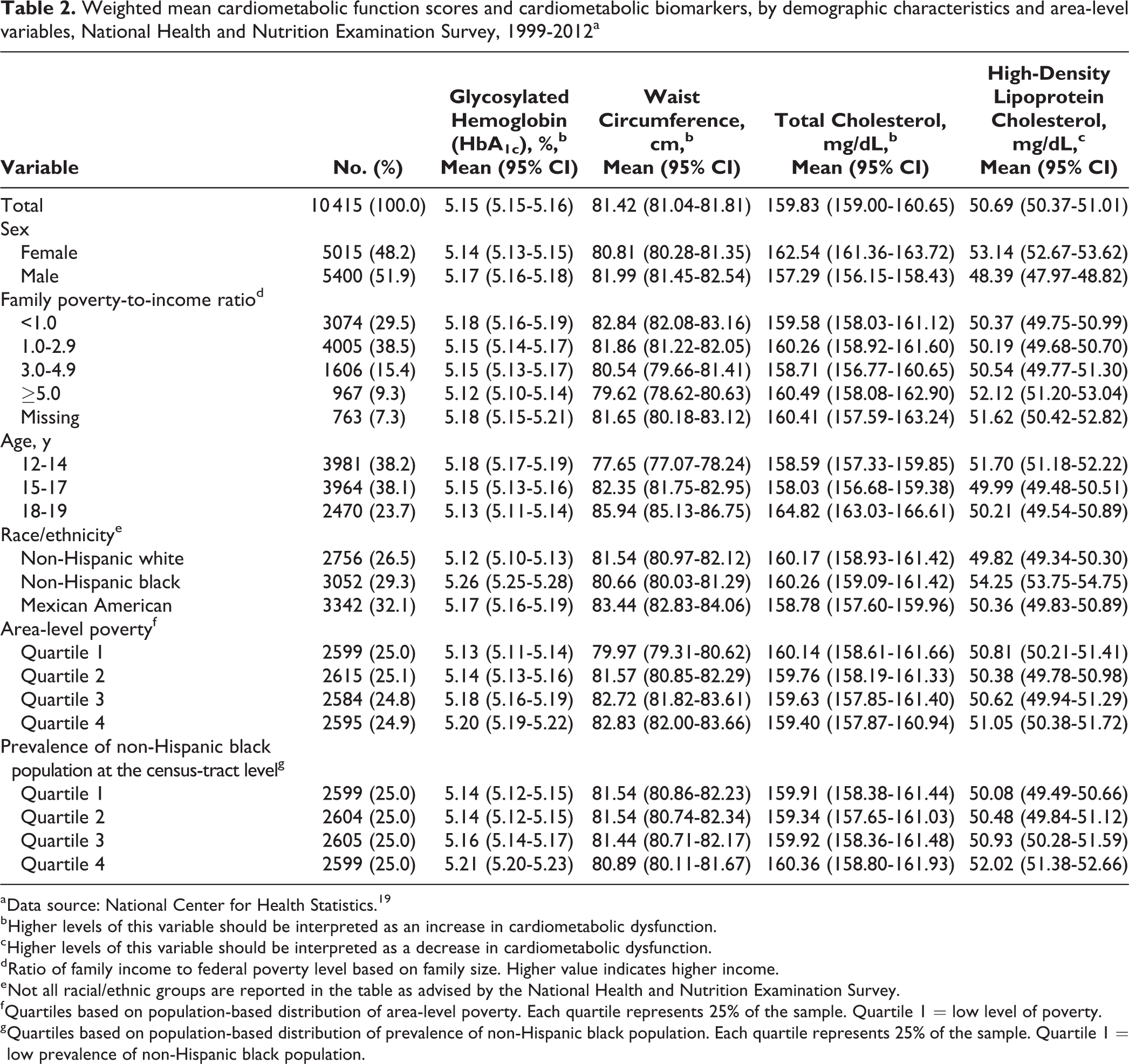
a Data source: National Center for Health Statistics. 19
b Higher levels of this variable should be interpreted as an increase in cardiometabolic dysfunction.
c Higher levels of this variable should be interpreted as a decrease in cardiometabolic dysfunction.
d Ratio of family income to federal poverty level based on family size. Higher value indicates higher income.
e Not all racial/ethnic groups are reported in the table as advised by the National Health and Nutrition Examination Survey.
f Quartiles based on population-based distribution of area-level poverty. Each quartile represents 25% of the sample. Quartile 1 = low level of poverty.
g Quartiles based on population-based distribution of prevalence of non-Hispanic black population. Each quartile represents 25% of the sample. Quartile 1 = low prevalence of non-Hispanic black population.
Overall, residents of highest-poverty areas had 27% higher odds of cardiometabolic dysfunction (quartile 4: OR = 1.27; 95% CI, 1.08-1.50) than residents of lowest-poverty areas, independent of individual- and area-level covariates (Table 3). In the adjusted sex-specific model (Table 3, model 3), boys residing in the highest-poverty areas had 36% higher odds of cardiometabolic dysfunction (quartile 4: OR = 1.36; 95% CI, 1.10-1.70) than boys residing in the lowest-poverty areas, and girls residing in the highest-poverty areas had 17% higher odds of cardiometabolic dysfunction than girls residing in the lowest-poverty areas (quartile 4: OR = 1.17; 95% CI, 0.94-1.47), but the difference was not significant (Table 3).
Hierarchical models estimating the association between area-level socioeconomic status and cardiometabolic function, overall and by sex, National Health and Nutrition Examination Survey (NHANES), 1999-2012a
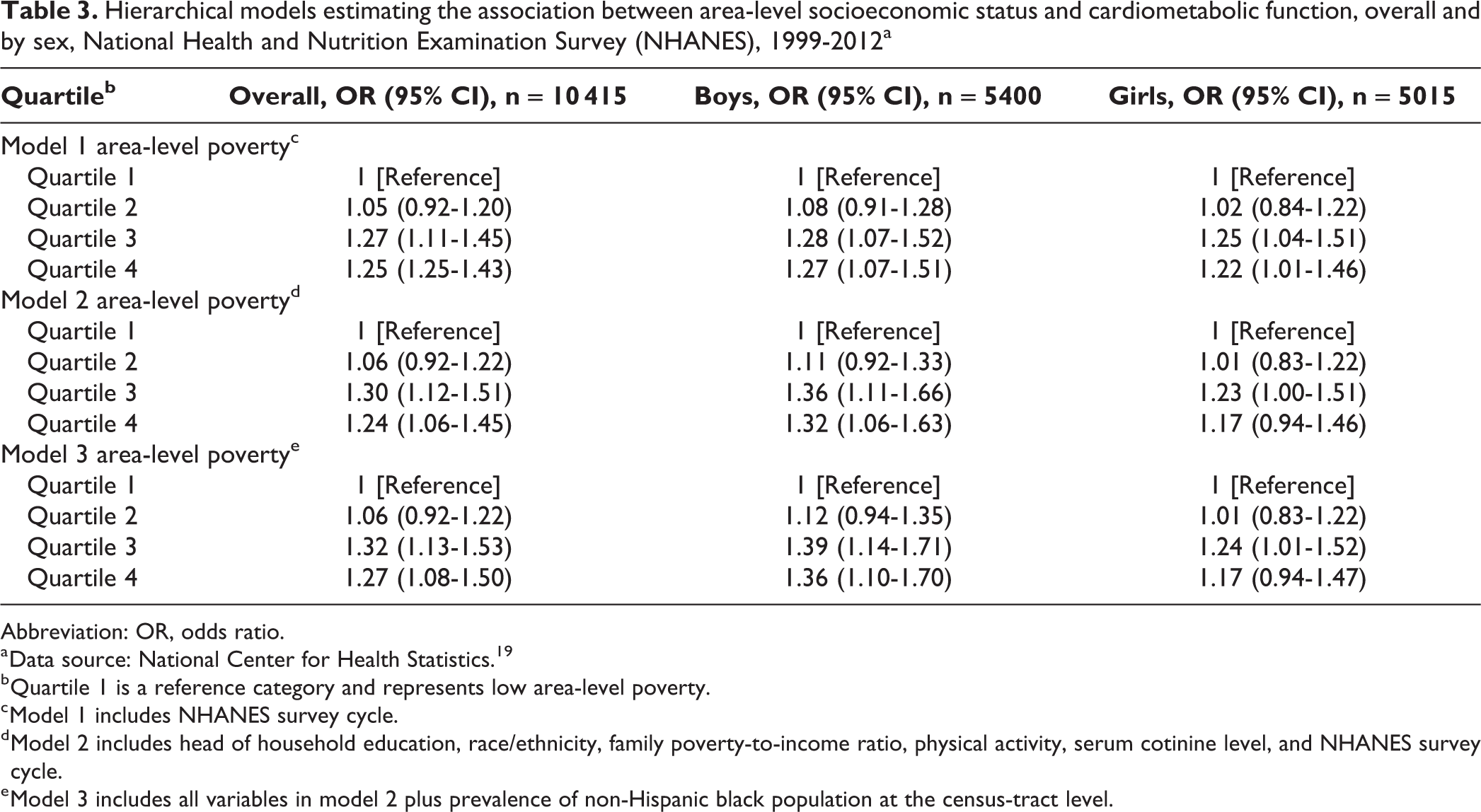
Abbreviation: OR, odds ratio.
a Data source: National Center for Health Statistics. 19
b Quartile 1 is a reference category and represents low area-level poverty.
c Model 1 includes NHANES survey cycle.
d Model 2 includes head of household education, race/ethnicity, family poverty-to-income ratio, physical activity, serum cotinine level, and NHANES survey cycle.
e Model 3 includes all variables in model 2 plus prevalence of non-Hispanic black population at the census-tract level.
The inclusion of individual- and area-level covariates differentially influenced the association between area-level poverty and cardiometabolic dysfunction by sex (Table 3). In the unadjusted sex-specific analysis (model 1), the association between residence in high-poverty areas (quartiles 3 and 4) and cardiometabolic dysfunction was similar among boys and girls. Among girls, the inclusion of individual-level covariates reduced the ORs for all quartiles of area-level poverty by up to 5% (ie, model 1 quartile 4, OR = 1.22 [95% CI, 1.01 -1.46] vs model 2 quartile 4, OR = 1.17 [95% CI, 0.94-1.46]), and the OR for quartile 4 became nonsignificant. In contrast, among boys, with the inclusion of individual-level covariates, the ORs for all quartiles of area-level poverty were slightly higher (ie, model 1 quartile 4, OR = 1.27 [95% CI, 1.07 -1.51] vs model 2 quartile 4, OR = 1.32 [95% CI, 1.06-1.63]), and the ORs for quartiles 3 and 4 were significant. The inclusion of percentage of non-Hispanic black residents (model 3) resulted in no change among girls (model 3 quartile 4, OR = 1.17 [95% CI, 0.94-1.47]), yet the ORs for quartiles 3 and 4 were higher by up to 3% among boys (model 3 quartile 4, OR = 1.36 [95% CI, 1.10-1.70]).
Discussion
The greater cardiometabolic dysfunction among girls than among boys in our study aligns with evidence of a higher prevalence of metabolic syndrome among girls than among boys in the literature. 33 Also in line with other studies, 7 -11 we observed a positive association between residence in economically deprived areas and cardiometabolic dysfunction. Although girls had greater cardiometabolic dysfunction than boys, the association between residence in high-poverty areas and cardiometabolic dysfunction was more pronounced among boys. Our findings also align with clinical evidence that men are more prone to develop stress-induced metabolic syndrome than women. 34 Although sexual dimorphism in the stress response generally presents as heightened vulnerability to stress-induced mood and autoimmune disorders among females, 35 males exposed to stress, particularly chronic stress, are more susceptible to cardiometabolic effects of stress, including visceral adiposity, hyperglycemia, and hyperinsulinemia, than females. 36
These findings are noteworthy for at least 2 reasons. First, our findings advance our understanding of the link between exposure to contextual adversity and cardiometabolic function during adolescence by examining sex differences in this association for the first time. Second, our findings contribute to the sparse literature of population-based studies on precursors of cardiometabolic disease. Of the 5 extant population-based studies in this area, 4 examined sex differences, and 2 reported sex differences in the association between stress and cardiometabolic function. 9,10 Aligned with the 2 positive studies, 9,10 our study benefited from a large sample of adolescents with objectively measured cardiometabolic variables rather than self-reported outcomes. Our findings support the notion that low statistical power and random measurement error likely contributed to the null findings 7,8 and tip the balance toward the existence of sex-specific associations; however, this finding is not yet conclusive, and more research is warranted.
Boys and girls residing in economically deprived areas are subjected to distinct social experiences and influences. 37 -39 For example, boys generally have more unsupervised time than girls 37 -39 and, thus, may be more exposed to stressors in these areas. Indeed, the association between parental supervision and unhealthy behaviors, including risky sexual behaviors, smoking, and violence, is stronger among boys than among girls in such areas. 38,39 In addition, preliminary evidence suggests that gender role socialization (whereby boys and girls are socialized to be seen as or act more masculine or feminine based on their biological sex) may influence sex differences in the stress response by influencing the perception of a situation as threatening or by influencing the ameliorating effect of social support. 40
Limitations
This study had several limitations. First, the associations observed in this study are likely to be explained, in part, by societal processes, such as disinvestments in areas of concentrated disadvantage, 41 and related sex-specific health behaviors, such as physical activity and diet. 42 We lacked data on the full complement of health behaviors relevant to cardiometabolic dysfunction, and the available variables (eg, physical activity) were subject to errors inherent in large epidemiologic surveys. However, these errors likely occurred at random; thus, our estimates were likely to be conservative. Second, inclusion in the model of area-level percentage of non-Hispanic black residents, a widely used proxy for societal disinvestments (eg, access to health care, education), 43 strengthened the effect among boys and did not influence the null effect among girls. The apparently sex-specific effects of societal disinvestment were unexpected and were not anticipated by theory. Further investigation of this interaction is warranted. Third, examinations of area-level determinants of health can produce various results depending on how areas of residence are defined. Thus, our findings may diverge from analyses using different geographic units of analysis. However, census tracts produce more consistent observations than other small geographies. 31 Fourth, persons residing in high-poverty areas may be at risk for greater exposure to adverse childhood experiences than those residing in low-poverty areas, 4 and we did not account for adverse childhood experiences in our analysis.
Fifth, our cardiometabolic dysfunction score is not a validated predictor of future disease, and we used empirical quartile cut points that do not represent a clinically meaningful increase in risk. Our cardiometabolic dysfunction score reflects levels of 6 biomarkers of metabolic syndrome, 17 and these biomarkers are predictive of future disease. Some persons with hypercholesterolemia may have been included in our analyses, because a clinical cut point for hypercholesterolemia among adolescents does not exist. Finally, inclusion of obese persons likely increased the prevalence of cardiometabolic dysfunction. Obese persons were not excluded because doing so would have reduced our sample by approximately 20%, thereby lowering statistical power.
Conclusions
The accumulating evidence linking area deprivation with cardiometabolic dysfunction 7 -11 emphasizes the potential of community-based health promotion programs targeting adolescents 44 as a way to improve health. Poverty is linked with housing instability. 45 -47 Of relevance are interventions that help to mitigate housing instability by linking at-risk families with social workers who facilitate eligible families’ access to public housing 48 and programs that subsidize heating fuel, control rent to improve accessibility, and provide tax incentives to promote home maintenance. These social benefits could have long-term health benefits through their influence on early precursors of cardiometabolic health, in addition to their well-documented more immediate health benefits. 49 More structural interventions include national policies, such as progressive tax codes, that are intended to mute the effects of relative poverty linked to income inequality. 50,51 Finally, broader neighborhood investments in safety, 52 parks and other recreational facilities, walkability, and ready access to public transportation can improve public health regardless of individual income levels. 47
Through a rigorous epidemiologic investigation, we found more pronounced associations between residence in high-poverty areas and cardiometabolic dysfunction among adolescent boys than among adolescent girls in the United States. Funding of evidence-based community health programs is warranted to improve cardiometabolic health among adolescents, which may have lifelong benefits.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.