Abstract
Objectives:
Uptake and completion of the human papillomavirus (HPV) vaccine series among adolescents are suboptimal in the United States. We examined immunization registry data to determine completion of the 3-dose HPV vaccine series among adolescents in Seattle, Washington, born during 1995-2000 who received ≥1 dose of HPV vaccine.
Methods:
Immunization data included the administrating facility, which identified adolescents who used school-based health centers (SBHCs) for any HPV vaccine dose. We calculated completion of the 3-dose series at any time and on time by the 13th birthday. We stratified analyses by sex and assessed differences in on-time and any-time completion between users and nonusers of SBHCs.
Results:
Overall, 67.9% (8612 of 12 676) of females and 41.8% (3560 of 8521) of males with ≥1 dose of HPV vaccine completed the 3-dose series. Compared with female SBHC nonusers, female SBHC users had 37% higher odds of completing the series at any time (adjusted odds ratio [aOR] = 1.37; 95% CI, 1.19-1.58) and 33% higher odds of completing the series on time (aOR = 1.33; 95% CI, 1.08-1.64). Compared with male SBHC nonusers, male SBHC users had 45% higher odds of completing the series at any time (aOR = 1.45; 95% CI, 1.23-1.70) and 79% higher odds of completing the series on time (aOR = 1.79; 95% CI, 1.11-2.89).
Conclusion:
Adolescent SBHC users had higher odds of completing the HPV vaccine series than adolescents who received all doses in traditional health care settings. SBHCs should be leveraged to increase adolescent immunization rates.
Human papillomavirus (HPV) causes multiple types of cancer and is the most common sexually transmitted infection in the United States. 1 The HPV vaccine, initially licensed as a 3-dose and subsequently a 2-dose series in the United States, is safe and effective at protecting against HPV-associated cancers. 1,2 HPV vaccine was first recommended as a routine vaccination for female adolescents aged 11 or 12 by the Advisory Committee on Immunization Practices in 2006 and for male adolescents aged 11 or 12 in 2011. 1 Despite being proven to prevent cancers in both males and females, uptake of HPV vaccine is suboptimal nationwide and is far below the Healthy People 2020 objective of 80% series completion for male and female adolescents by age 13-15. 3 National Immunization Survey–Teen results from 2015 indicated that only 63% of females and 50% of males had received ≥1 dose of the HPV vaccine series. 4,5 The completion rate for the 3-dose series is also low: in 2015, 71% of females and 61% of males who received ≥1 dose of HPV vaccine completed the 3-dose series. 5
Factors associated with failure to initiate the HPV vaccine series tend to be rooted in parental beliefs, lack of knowledge about the vaccine, or the absence of a strong provider recommendation. 5 -12 Substantial progress toward the Healthy People 2020 objective can be made by increasing completion rates among adolescents who start the HPV vaccine series. Many barriers to completion of the HPV vaccine series are structural, including infrequent preventive health care visits during adolescence, missed clinical opportunities, poor communication about the need for multiple HPV vaccine doses, and the lack of reminder/recall systems for follow-up doses. 6 -22 Overcoming barriers to HPV vaccine series completion requires a multipronged approach. Offering vaccines at convenient settings where adolescents are known to access health care services, as in school-based health centers (SBHCs), could play an important role in improving HPV vaccination coverage rates. 8,12,22
SBHCs are a key component of the nation’s health care safety net. 23 SBHCs have been shown to improve health and educational outcomes and are especially useful at reducing health disparities and increasing health equity. 23,24 SBHCs provide convenient and immediate access to the health care system and are intended to supplement, not replace, an adolescent’s primary care provider. 25 A recent study found that providers and parents accepted and supported the provision of HPV vaccine in a school setting. 26
Twenty-six SBHCs are located on elementary school, middle school, and high school campuses in Seattle, Washington; they are sponsored and staffed by 7 local health care organizations. HPV vaccine was available at SBHCs located in 12 high schools and 6 middle schools beginning in 2007. SBHC clinicians provide a range of services to students regardless of their health insurance status, including preventive care, urgent care, chronic disease management, mental and behavioral health counseling, family planning, and drug and alcohol counseling. In Washington State, vaccines are provided at no cost for all children aged <19 through a universal vaccine program in which childhood vaccines are purchased by the state with funds from the national Vaccines for Children (VFC) program and assessments from health plans and other payers. 27,28 The Public Health–Seattle & King County Immunization Program manages the VFC program in King County and Seattle. Health care providers participating in the VFC program are encouraged to enter all immunization records into the Washington State Immunization Information System (WAIIS), which allows for coordination of care across multiple health care systems. Although not all health care providers participate in VFC, nearly all routinely enter data on immunizations into WAIIS.
Studies that examined the use of SBHCs to increase HPV vaccine uptake among adolescents found the rates of series completion among SBHC users who initiated the series to be as high as 80%. 29 -31 Although immunization registry data are useful for assessing immunization coverage, immunization information system–based denominators overestimate adolescent populations at the county level because of duplicate and unresolved records and poor tracking of persons who move. 32 To mitigate these denominator problems, we used data from WAIIS on adolescents with ≥1 dose of HPV vaccine to compare completion rates of adolescents who used SBHCs for ≥1 HPV vaccine dose with completion rates of adolescents who received HPV vaccine doses only in traditional health care settings, such as pediatric or family medicine clinics.
Methods
To examine completion of the HPV vaccine series among adolescents, we obtained WAIIS data on HPV vaccine doses administered through December 31, 2013, for Seattle residents born during January 1, 1995, through December 31, 2000 (age cohort, 13-18). WAIIS data included patient information (sex, date of birth, race/ethnicity, health insurance status) and vaccine information (type of vaccine administered, date of vaccination, provider and facility administering the vaccine). Because the Advisory Committee on Immunization Practices recommendation for the HPV vaccine was published in different years for males and females, we stratified all analyses by sex, and we excluded adolescents who did not specify sex in WAIIS.
In 2008, the Public Health–Seattle & King County VFC program created a database to track enrolled clinics’ vaccine orders, monthly vaccine inventory and accountability reports, and annual VFC contracts that contain self-reported information, such as clinic type, hours of operation, address, and number of health care providers in the practice. We used the VFC database to identify SBHCs and other VFC-enrolled clinics in Seattle and match them with their corresponding facility name in WAIIS.
We included adolescents with ≥1 dose of HPV vaccine documented in WAIIS in our analysis, and we analyzed completion of the 3-dose series. We assessed series completion in 2 ways: any-time completion and on-time completion. We defined any-time completion as the percentage of adolescents who received all 3 doses of the series at any time before December 31, 2013, among adolescents who had received ≥1 dose of HPV vaccine. We defined on-time completion as the percentage of adolescents who received all 3 doses of the series at the recommended age of 11 or 12 by assessing completion at the patient’s 13th birthday among adolescents who had started the HPV vaccine series before their 13th birthday. We examined completion of the HPV vaccine series by birth-year cohort to understand how uptake changed since the vaccine’s introduction to the recommended adolescent vaccination schedule.
We defined SBHC users as adolescents who received ≥1 dose of HPV vaccine from an SBHC as documented in WAIIS, and we defined SBHC nonusers as adolescents who received all doses from any other type of clinic as documented in WAIIS. SBHC users included adolescents who received HPV vaccine doses from SBHCs located in middle schools (M-SBHCs) or high schools. Because the recommended age to receive the HPV vaccine series is typically before an adolescent begins high school, we conducted separate analyses of on-time completion rates for users of M-SBHCs to distinguish them from users of SBHCs located in high schools. In the on-time completion analyses, we defined M-SBHC users as adolescents who received ≥1 dose of HPV vaccine from any M-SBHCs; we defined M-SBHC nonusers as adolescents who did not receive any doses of HPV vaccine from an M-SBHC, including adolescents who received HPV vaccine doses from only SBHCs located in high schools and adolescents who received all doses from any other type of health care setting. To identify differences in on-time and any-time completion of the 3-dose series between SBHC users and SBHC nonusers, we stratified all analyses by sex and by birth-year cohort.
Of 49 290 Seattle adolescents born during 1995-2000 with records in WAIIS, 568 (1.2%) were missing information on sex and were excluded from this study. Among adolescents with sex specified, 24 171 were female and 24 551 were male. Forty-six percent (11 138 of 24 171) of females and 64.3% (15 797 of 24 551) of males had no doses of HPV vaccine and were excluded from this study. Of adolescents with ≥1 dose of HPV vaccine, 357 of 13 033 females (2.7%) and 233 of 8754 males (2.7%) did not have information on the facility that administered ≥1 dose of HPV vaccine and also were excluded. A total of 12 676 females and 8521 males were eligible for inclusion in this study. Data on race, ethnicity, and health insurance status were missing in WAIIS for 67%, 67%, and 42%, respectively, of adolescents, and these covariates were not included in the analyses. Thus, the only covariate tested in logistic regression models was birth cohort.
We determined significance in bivariate analyses by using the Pearson χ2 test or Fisher exact test for small numbers, with P < .05 considered significant. We assessed trends in completion rates by birth cohort by using the Cochran-Armitage test for trend. Because the HPV vaccine was not recommended for males until 2011, sample size was limited and some trend analyses could not be conducted. We used logistic regression models to determine the magnitude of the association between use of SBHCs and completion of the HPV vaccine series, and the models incorporated variables that were found to be significant in bivariate analyses. We conducted data management activities and statistical analyses by using SAS version 9.4. 33 The Washington State Institutional Review Board approved this study.
Results
Of 12 676 females with ≥1 dose of HPV vaccine, 1186 (9.4%) used any SBHC for ≥1 dose of HPV vaccine, and 511 (4.0%) received ≥1 dose of HPV vaccine from any M-SBHC (Table 1). Of 8521 males with ≥1 dose of HPV vaccine, 660 (7.7%) received ≥1 dose of HPV vaccine from any SBHC, and 164 (1.9%) received ≥1 dose of HPV vaccine from any M-SBHC. Overall, 8612 of 12 676 (67.9%) females and 3560 of 8521 (41.8%) males with ≥1 dose of HPV vaccine completed the 3-dose HPV vaccine series at any time. Among adolescents who started the HPV vaccine series on time before their 13th birthday, 3230 of 7773 (41.6%) females and 414 of 1672 (24.8%) males completed the 3-dose series on time. The percentage of adolescents using any SBHC and any M-SBHC varied by birth-year cohort (Table 2).
Characteristics of adolescents born during 1995-2000 with ≥1 dose of HPV vaccine by December 31, 2013, Seattle, Washington

Abbreviations: HPV, human papillomavirus; SBHC, school-based health center.
a Of adolescents with ≥1 dose of HPV vaccine.
b Of adolescents with ≥1 dose of HPV vaccine, those who received ≥1 dose of HPV vaccine before 13th birthday.
c Of adolescents who started HPV vaccine series on time, those who received 3 doses of HPV vaccine before 13th birthday.
Adolescents born during 1995-2000 with ≥1 dose of HPV vaccine who received any HPV vaccine doses at an SBHC or M-SBHC through December 31, 2013, by year of birth, Seattle, Washington
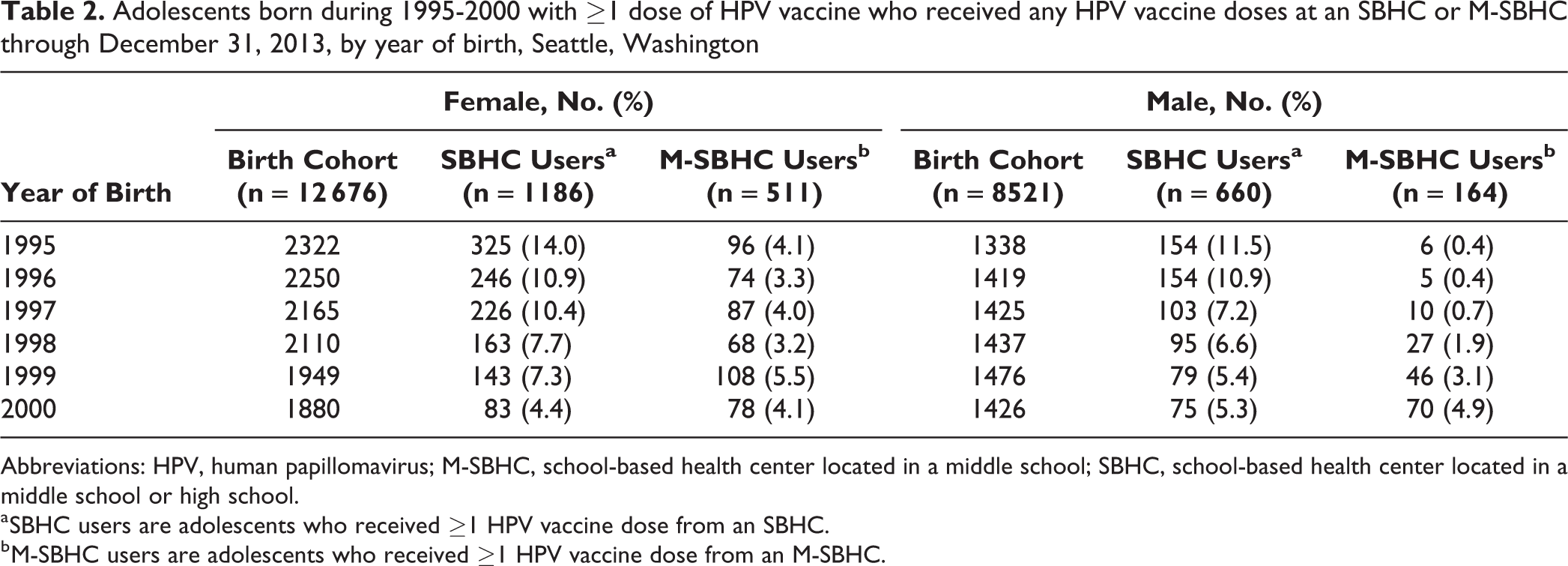
Abbreviations: HPV, human papillomavirus; M-SBHC, school-based health center located in a middle school; SBHC, school-based health center located in a middle school or high school.
a SBHC users are adolescents who received ≥1 HPV vaccine dose from an SBHC.
b M-SBHC users are adolescents who received ≥1 HPV vaccine dose from an M-SBHC.
Any-Time Completion of HPV Vaccine Series Among Females
Nine hundred of 1186 (75.9%) female SBHC users with ≥1 dose of HPV vaccine completed the 3-dose series at any time, which was significantly higher than the 7712 of 11 490 (67.1%) female SBHC nonusers who completed the series at any time (P < .001). Of 900 female SBHC users who completed the 3-dose series, 300 (33.3%) received all doses at SBHCs, and 600 (66.7%) received doses at a mix of SBHCs and other settings. We also found differences in rates of any-time completion among females by birth year (Table 3). We found a significant trend in rates of any-time completion by birth year among female adolescents who did not use SBHCs (P < .001); among SBHC nonusers, older female adolescents had higher any-time completion rates than younger female adolescents. However, among female SBHC users, we found no significant trend in any-time completion rates by birth year (P = .50). Compared with female SBHC nonusers born in the same years, female SBHC users born in 1998, 1999, and 2000 had significantly higher rates of any-time completion (Table 3).
Completion of the 3-dose HPV vaccine series at any time before December 31, 2013, among adolescents born during 1995-2000 with ≥1 dose of HPV vaccine, by year of birth and use of an SBHC, Seattle, Washingtona
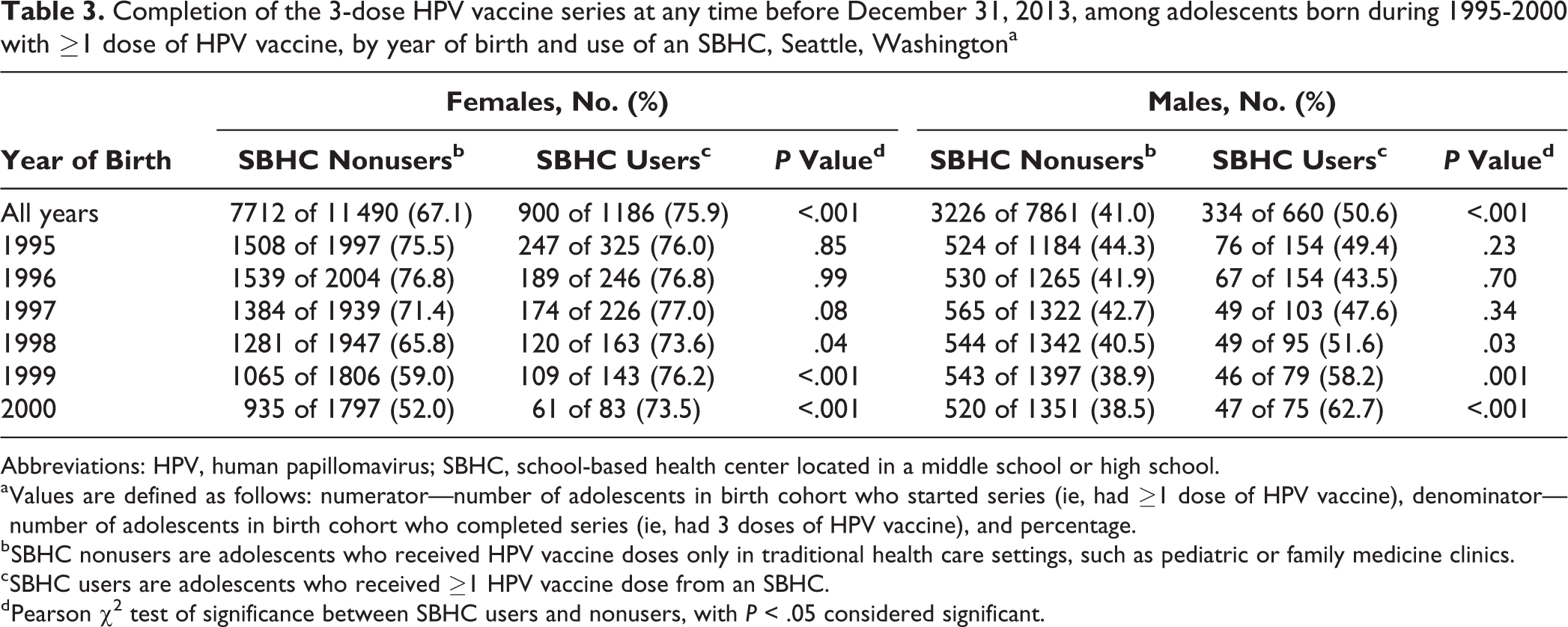
Abbreviations: HPV, human papillomavirus; SBHC, school-based health center located in a middle school or high school.
a Values are defined as follows: numerator—number of adolescents in birth cohort who started series (ie, had ≥1 dose of HPV vaccine), denominator—number of adolescents in birth cohort who completed series (ie, had 3 doses of HPV vaccine), and percentage.
b SBHC nonusers are adolescents who received HPV vaccine doses only in traditional health care settings, such as pediatric or family medicine clinics.
c SBHC users are adolescents who received ≥1 HPV vaccine dose from an SBHC.
d Pearson χ2 test of significance between SBHC users and nonusers, with P < .05 considered significant.
In logistic regression analysis, female SBHC users had 37% higher odds of completing the HPV vaccine series at any time than female SBHC nonusers, after adjusting for birth cohort (adjusted odds ratio [aOR] = 1.37; 95% CI, 1.19-1.58; P < .001; Table 4).
Logistic regression model estimates examining factors associated with completion of the 3-dose HPV vaccine series among adolescents born during 1995-2000 with ≥1 dose of HPV vaccine, Seattle, Washington

Abbreviations: aOR, adjusted odds ratio; HPV, human papillomavirus; M-SBHC, school-based health center located in a middle school; OR, odds ratio; SBHC, school-based health center located in a middle school or high school.
a Wald χ2 test of significance for individual regression coefficients, with P < .05 considered significant.
b Models compare any-time completion of 3-dose HPV vaccine series of SBHC users with SBHC nonusers (reference group) among adolescents with ≥1 dose of HPV vaccine.
c Models compare on-time completion of 3-dose HPV vaccine series of M-SBHC users with M-SBHC nonusers (reference group) among adolescents with ≥1 dose of HPV vaccine before 13th birthday.
Any-Time Completion of HPV Vaccine Series Among Males
Of 660 male SBHC users with ≥1 dose of HPV vaccine, 334 (50.6%) completed the series at any time, which was significantly higher than 3226 of 7861 (41.0%) male SBHC nonusers who completed the series at any time (P < .001). Of 334 male SBHC users who completed the 3-dose series, 135 (40.4%) received all doses at SBHCs, and 199 (59.6%) received doses at a mix of SBHCs and other settings. We found differences in rates of any-time completion among males by birth year (Table 3). We also found significant trends in rates of any-time completion for males by birth-year cohort; similar to female SBHC nonusers, older male SBHC nonusers had significantly higher rates of series completion than younger male SBHC nonusers (P = .001). However, among male SBHC users, rates of any-time completion were higher among younger males than among older males (P = .01). Compared with male SBHC nonusers born in the same years, male SBHC users born in 1998, 1999, and 2000 had significantly higher any-time completion rates.
In logistic regression analysis, male SBHC users had 45% higher odds of completing the HPV vaccine series at any time than male SBHC nonusers after adjusting for birth year (aOR = 1.45; 95% CI, 1.23-1.70; P < .001; Table 4).
On-Time Completion of HPV Vaccine Series Among Females
Of 380 female M-SBHC users who started the series on time, 182 (47.9%) completed the series on time, which was significantly higher than 3048 of 7393 (41.2%) female M-SBHC nonusers who completed the series on time (P = .01). Of 182 female M-SBHC users who completed the series on time, 68 (37.4%) received all doses at M-SBHCs, and 114 (62.6%) received doses at a mix of M-SBHCs and other settings. We found differences in rates of on-time completion among females by birth year (Table 5). Younger females had higher rates of on-time completion than older females for both M-SBHC users (P = .002) and M-SBHC nonusers (P = .002). Female M-SBHC users born in 1998 had significantly higher rates of on-time completion than female M-SBHC nonusers born in the same year (P = .04).
Completion of the 3-dose HPV vaccine series on time by the 13th birthday among adolescents born during 1995-2000 with ≥1 dose of HPV vaccine before the 13th birthday, by year of birth and use of an M-SBHC, Seattle, Washingtona

Abbreviations: HPV, human papillomavirus; M-SBHC, school-based health center located in a middle school; SBHC, school-based health center located in a middle school or high school.
a Values are defined as follows: numerator—number of adolescents in birth cohort who started series on time (ie, had ≥1 dose of HPV vaccine before their 13th birthday), denominator—number of adolescents in birth cohort who completed series on time (ie, had 3 doses of HPV vaccine before their 13th birthday), and percentage. Dashes (—) indicate that males born before 1998 were not recommended to receive the HPV vaccine series until 2011.
b M-SBHC nonusers are adolescents who received the HPV vaccine doses only in traditional health care settings, such as pediatric or family medicine clinics, and adolescents who received HPV vaccine doses from an SBHC located in a high school.
c M-SBHC users are adolescents who received ≥1 HPV vaccine dose from an SBHC located in a middle school.
d Pearson χ2 test of significance between M-SBHC users and nonusers, with P < .05 considered significant.
e Fisher exact test was used to calculate P values for small numbers, with P < 0.5 considered significant.
In logistic regression analysis, among females who began the HPV vaccine series on time, female M-SBHC users had 33% higher odds of completing the series on time than female M-SBHC nonusers, after adjusting for birth cohort (aOR = 1.33; 95% CI, 1.08-1.64; P = .01; Table 4).
On-Time Completion of HPV Vaccine Series Among Males
Of 79 male M-SBHC users who started the series on time, 29 (36.7%) completed the series on time, which was significantly higher than 385 of 1593 (24.1%) male M-SBHC nonusers who started the series on time and completed the series on time (P = .01). Of 29 male M-SBHC users who completed the series on time, 13 (44.8%) received all doses at M-SBHCs, and 16 (55.2%) received doses at a mix of M-SBHCs and other settings. We found differences in rates of on-time completion among males by birth year (Table 5). When compared with M-SBHC nonusers born in the same year, male M-SBHC users born in 2000 had significantly higher rates of on-time completion (P = .004).
In logistic regression analysis, among males who began the series on time, male M-SBHC users had 79% higher odds of completing the 3-dose series on time than male M-SBHC nonusers, after adjusting for birth-year cohort (aOR = 1.79; 95% CI, 1.11-2.89; P = .02; Table 4).
Discussion
We found that adolescents who used SBHCs for HPV vaccine doses had significantly higher series completion rates than their counterparts who received all their doses in traditional health care settings. After controlling for differences in birth year, SBHC users had higher odds of completing the HPV vaccine series at any time than SBHC nonusers. The odds of completing the series on time were 33% higher among female M-SBHC users than among female M-SBHC nonusers. Male M-SBHC users had 79% higher odds of completing the series on time than male M-SBHC nonusers, which is especially important when considering the lagging uptake of HPV vaccine among males. These findings demonstrate that by encouraging series completion through the use of alternative health care settings such as SBHCs, measurable progress can be made toward achieving the Healthy People 2020 objective of 80% of males and females aged 13-15 with 3 doses of HPV vaccine. 3
The potential for SBHCs to improve adolescent health by increasing rates of HPV vaccine series completion is supported by other studies. 29 -31 Emphasizing HPV vaccine series completion is a realistic and tangible goal that can be accomplished by reducing structural barriers. This study suggests that the need for return visits to a traditional health care setting for follow-up doses might be a barrier to HPV vaccine series completion. SBHCs provide vaccinations in a setting that is convenient for parents and adolescents. 12,22,26 Our study found that most SBHC users also received doses from other health care settings, providing evidence that SBHCs supplement the adolescent’s medical home. Among SBHC users who completed the series, 33% of females and 40% of males received all 3 HPV vaccine doses from SBHCs; it is unknown if these adolescents were connected to care in a medical home where they might also have received HPV vaccine.
Recently, the Advisory Committee on Immunization Practices updated the recommendations for HPV vaccine among adolescents: those who begin the series between ages 9 and 14 are required to receive only 2 doses of HPV vaccine; 3 doses are still required for those initiating the series at or after age 15 and for immunocompromised persons. 2 The updated recommendation should reduce the burden of follow-up doses and should result in more adolescents completing the series on time. Regardless of the number of doses required, SBHCs are an important resource to assist with improving HPV vaccine series completion.
Limitations
This study had several limitations. First, because a small percentage of adolescents received doses of HPV vaccine from SBHCs, some analyses were limited by a small sample size, especially for male adolescents. Despite the small number of SBHC users, our analysis found a significant improvement in rates of HPV vaccine series completion for both males and females. Second, the percentage of adolescents born in 2000 who used an SBHC for any doses of HPV vaccine was lower than the percentage of adolescents born in other birth-year cohorts, because nearly all of these adolescents had not yet started high school, which is where most adolescents access SBHCs. Third, data on race, ethnicity, and health insurance status in WAIIS were missing for most adolescents. Differences in HPV vaccine coverage may exist among various demographic groups; had we had information on race/ethnicity and insurance status, our results may have been different. We also excluded adolescents whose sex was not specified in WAIIS and adolescents who had documented doses of vaccine but were missing information on the facility that administered the doses. It is possible that those with missing sex or facility information had differences that could not be accounted for in our analysis. Fourth, the total number of adolescents with records in WAIIS exceeded population estimates. Some of these adolescents may have moved from the study area after the creation of their WAIIS record or were mistakenly entered as duplicates. Our analysis was limited to adolescents with a documented dose of HPV vaccine to account for these population issues and to better understand low rates of series completion. However, it is possible that some adolescents included in the analysis who did not appear to have received 3 doses of HPV vaccine had moved elsewhere and completed the series there.
Conclusion
We found that male and female adolescents who used SBHCs for HPV vaccine doses had significantly higher completion rates than adolescents who did not use SBHCs. This research adds HPV vaccine series completion to the many other benefits that SBHCs have been shown to provide to students, families, and communities. 23 Using SBHCs to provide immunization services is an important strategy to protect adolescents against serious vaccine-preventable diseases, including HPV-related cancers. SBHCs should be expanded and promoted nationwide to provide more adolescents with access to these services.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by an appointment to the Applied Epidemiology Fellowship Program administered by the Council of State and Territorial Epidemiologists and funded by the Centers for Disease Control and Prevention (cooperative agreement 5U38HM000414-5).
