Abstract
Objectives:
Infants born to mothers who are hepatitis B surface antigen (HBsAg) positive are at risk for perinatal hepatitis B infection. As prevention, these infants receive a series of 3 or 4 doses of hepatitis B vaccine starting at birth and postvaccination serologic testing. Infants with antibody levels <10 mIU/mL are considered vaccine nonresponders and should be revaccinated. The objective of this cost analysis was to assess a single-dose revaccination strategy among infant nonresponders.
Methods:
We used a decision analytic tree to compare the costs of a single-dose revaccination strategy with the costs of a 3-dose revaccination strategy. The analysis consisted of 3 epidemiologic scenarios that varied levels of previous protection among infants indicated for revaccination. We assumed health outcomes in each strategy were the same, and we evaluated costs from the societal perspective using 2016 US dollars. We conducted sensitivity analyses on key variables, including the minimum required efficacy of a single revaccination dose.
Results:
In all analyses, the single-dose revaccination strategy was a lower-cost option than the 3-dose revaccination strategy. Under the assumption that all revaccination visits were previously unscheduled, single-dose revaccination reduced the cost per infant by $119.81 to $155.72 (depending on the scenario). Across all scenarios, the most conservative estimate for the threshold efficacy (the minimum efficacy required to result in a lower-cost option) value of single-dose revaccination was 67%.
Conclusions:
For infants who were born to HBsAg-positive mothers and who were not responding to the initial vaccine series, a single-dose revaccination strategy, compared with a 3-dose revaccination strategy, reduced costs across several scenarios. These results helped inform the Advisory Committee on Immunization Practices’ vote in February 2017 to recommend single-dose revaccination.
Each year in the United States, about 25 000 infants are born to mothers who are hepatitis B surface antigen (HBsAg) positive. 1 Without intervention, 40% to 90% of infants born to HBsAg-positive mothers become infected, 2 primarily through contact with maternal blood or bodily fluids during birth or early-life transmission through contact with infected people. 3,4 Approximately 90% of infected infants develop chronic hepatitis B virus infection; only 5% of children who become infected when they are aged ≥5 years develop chronic hepatitis B virus infection. Infants with chronic hepatitis B virus infection have a 25% risk of premature death from cirrhosis or cancer of the liver. 5
Perinatal hepatitis B virus infection can be prevented through a postexposure prophylaxis regimen that consists of a 3-dose hepatitis B vaccine series and hepatitis B immune globulin starting at birth. Although efficacy varies according to birth weight and age at vaccine administration, hepatitis B vaccination and hepatitis B immune globulin combined can be up to 98% effective in preventing infection in infants. 6 The Advisory Committee on Immunization Practices (ACIP) to the Centers for Disease Control and Prevention (CDC) recommends that all pregnant women are screened for HBsAg to help ensure that infants born to HBsAg-positive mothers receive postexposure prophylaxis within 12 hours of birth, followed by completion of the vaccination series and postvaccination serologic testing (PVST). 5
PVST consists of testing for HBsAg and antibodies to HBsAg (anti-HBs) and should be conducted at age 9 to 12 months, or 1 to 2 months after the final dose if the initial series is delayed. 5 PVST should be performed no earlier than age 9 months to avoid detection of passive anti-HBs from hepatitis B immune globulin administered at birth. Infants with anti-HBs levels ≥10 mIU/mL are considered protected and need no further management. Before February 2017, ACIP recommended that infants who failed to respond to the initial series (HBsAg-negative and anti-HBs <10 mIU/mL) receive a second complete series followed by a second PVST. Beyond 9 to 12 months of age, anti-HBs levels can wane, although protection continues. 7 Therefore, infants who have a delayed initial PVST can have a decrease in anti-HBs to levels <10 mIU/mL and be indicated for revaccination, despite being protected.
Previous data demonstrated protection levels from vaccine series administered to infants. 8,9 Economic analysis, a framework for comparing multiple strategies of allocating resources, 10 can be used to evaluate a single-dose revaccination option. The objective of our study was to analyze the costs of a single-dose revaccination strategy among infants who were born to HBsAg-positive mothers and who were not responding to the initial vaccine series. The ACIP Hepatitis Work Group requested an economic evaluation of single-dose revaccination. We conducted this analysis to inform the ACIP Hepatitis Work Group’s deliberations and presented the results to ACIP in February 2017. Our results helped inform the February 2017 ACIP vote to recommend single-dose revaccination as a revaccination strategy for infants who are born to HBsAg-positive mothers and who are not responding to the initial vaccine series. 11,12
Methods
Study Population and Strategies
From November 2016 to February 2017, we evaluated a decision-analytic model of a hypothetical study population that included all infants who were born to HBsAg-positive mothers and who were not responding to the initial vaccine series. Our study was exempt from institutional review board approval because no human participants were involved.
We defined an infant nonresponder as HBsAg-negative with anti-HBs of <10 mIU/mL at the initial PVST. This cost analysis compared the implementation of 2 strategies: a single-dose revaccination strategy and the previously recommended 3-dose revaccination strategy. In the 3-dose revaccination strategy, infants who were born to HBsAg-positive mothers and who were not responding to the initial vaccine series (anti-HBs <10 mIU/mL) are given 3 more doses (dose 2a, dose 2b, and dose 2c) at 3 separate visits. A final PVST is conducted at the end of the revaccination series. In the single-dose strategy, infant nonresponders are revaccinated with 1 additional vaccine dose (dose 2a) followed by a second PVST. If anti-HBs are ≥10 mIU/mL, infants are considered protected and receive no additional doses. If anti-HBs remain <10 mIU/mL, infants receive 2 more doses (dose 2b and dose 2c) followed by a final PVST. In this strategy, infants who are not protected after a single revaccination dose go on to receive 3 full revaccination doses, mirroring the current 3-dose revaccination strategy. We assumed the cumulative risk of hepatitis B virus infection to be the same in both strategies.
Decision-Analytic Model
We analyzed the costs and outcomes of both strategies from a societal perspective by using a decision-analytic model that consisted of applying a decision tree across various epidemiologic scenarios (Figure 1). The primary output of interest was the cost per infant for each scenario. The time frame and analytic horizon were both 1 year. We performed all analyses in TreeAge Pro 2009. 13
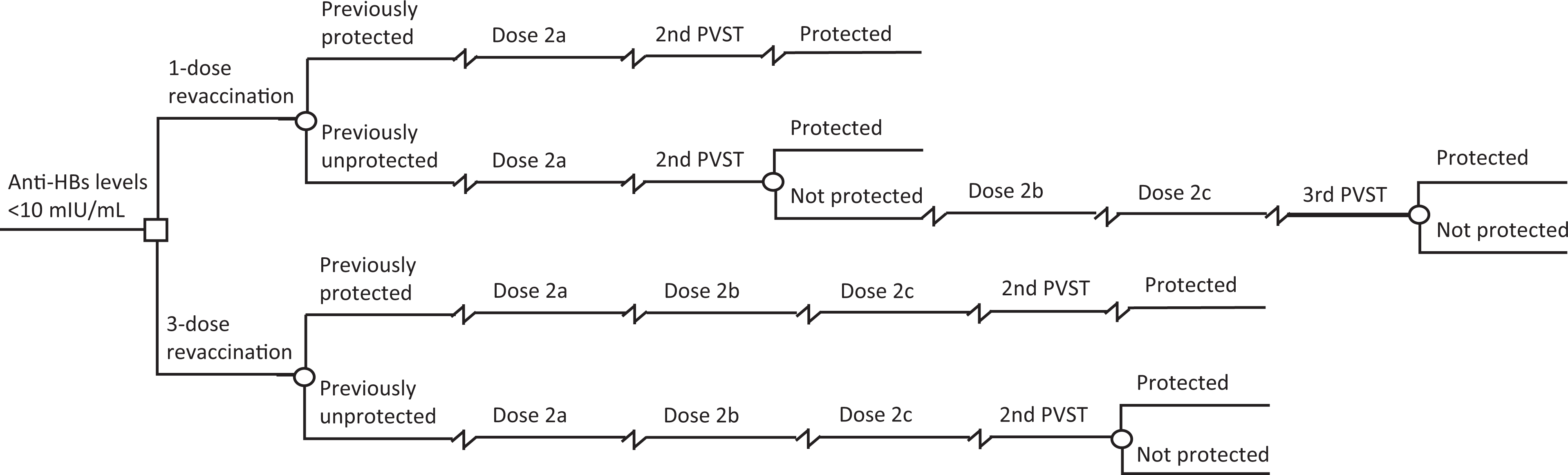
Decision-tree model used in a cost analysis of single-dose hepatitis B revaccination among infants who were born to hepatitis B surface antigen–positive mothers and who were not responding to the initial vaccine series. Terminal nodes represent 2 mutually exclusive outcomes: protected and not protected. Decisions are indicated by squares and chance outcomes by circles. The cumulative risk of hepatitis B infection and health outcomes for each strategy was assumed to be the same. Abbreviations: anti-HBs, hepatitis B antibodies; PVST, postvaccination serologic testing.
Inputs and Parameters
We established the base-case parameters and their corresponding plausible intervals (Table 1). The initial cohort of 24 784 was the estimated number of infants born to HBsAg-positive mothers in 2009 and was used in similar analyses. 1,14 We calculated base-case efficacy of the initial vaccine series by creating a weighted average of vaccine efficacy among infants with a birth weight of <2000 g (efficacy = 0.93) and ≥2000 g (efficacy = 0.98), 6 assuming that 14.3% of infants born to HBsAg-positive mothers weigh <2000 g. 15 We set the upper limit for efficacy of the initial vaccine series at the value for the highest-recorded vaccine efficacy among infants, 6 and we calculated the lower limit from data in an analysis of the national Perinatal Hepatitis B Prevention Program (PHBPP). 14 This weighted average incorporated the reported proportions of infants who initiated and completed the initial vaccine series at various ages (and the associated efficacy) both within and outside coverage of the PHBPP. We calculated the proportion of infants who developed protection after dose 2a by pooling frequency data from 2 studies that recorded anti-HBs at 4 points: the initial dose (at birth), a second dose at 1 month, a third dose at 2 months, and a fourth and final dose at 12 months. 6,8,9 To calculate threshold values in a sensitivity analysis, we used a wide plausible range of 0 to 0.980 for efficacy of dose 2a.
Model parameters used in a cost analysis of single-dose hepatitis B revaccination among infants who were born to hepatitis B surface antigen–positive mothers and who were not responding to the initial vaccine series
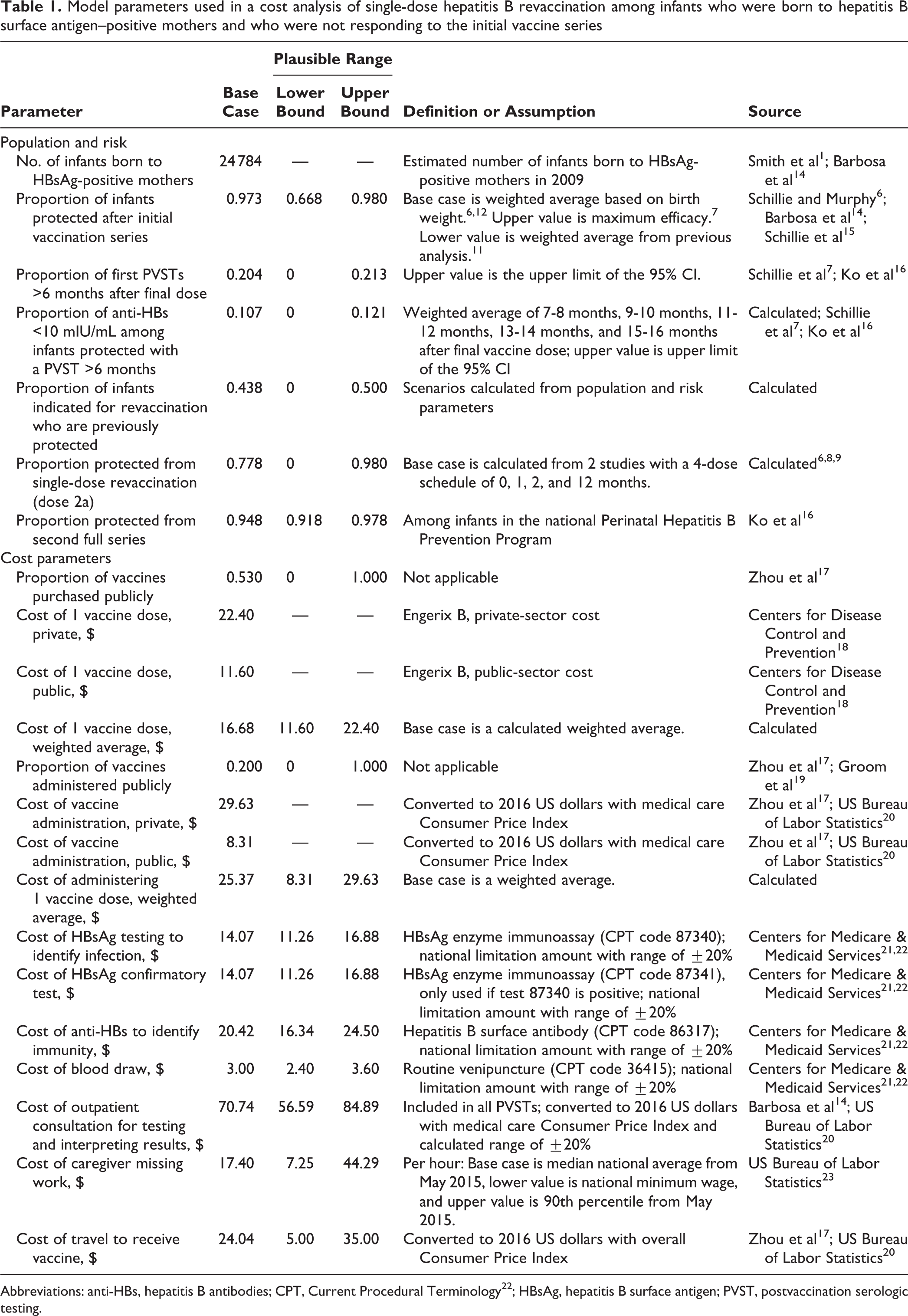
Abbreviations: anti-HBs, hepatitis B antibodies; CPT, Current Procedural Terminology 22 ; HBsAg, hepatitis B surface antigen; PVST, postvaccination serologic testing.
To allow variation in efficacy of the initial vaccine series and potential for delayed PVST, we created 3 epidemiologic scenarios (scenarios A, B, and C). Each scenario had its own unique set of inputs: a plausible value for the efficacy of the initial vaccine series, a proportion of infants with a delayed initial PVST, and a proportion of infants with antibody levels that waned to <10 mIU/mL as a result of a delayed PVST. For each scenario, we used these values to estimate (1) the number of infants indicated for revaccination and (2) the proportion of infants indicated for revaccination who were previously protected. We then used these estimates as inputs in the decision analysis (Figure 2).
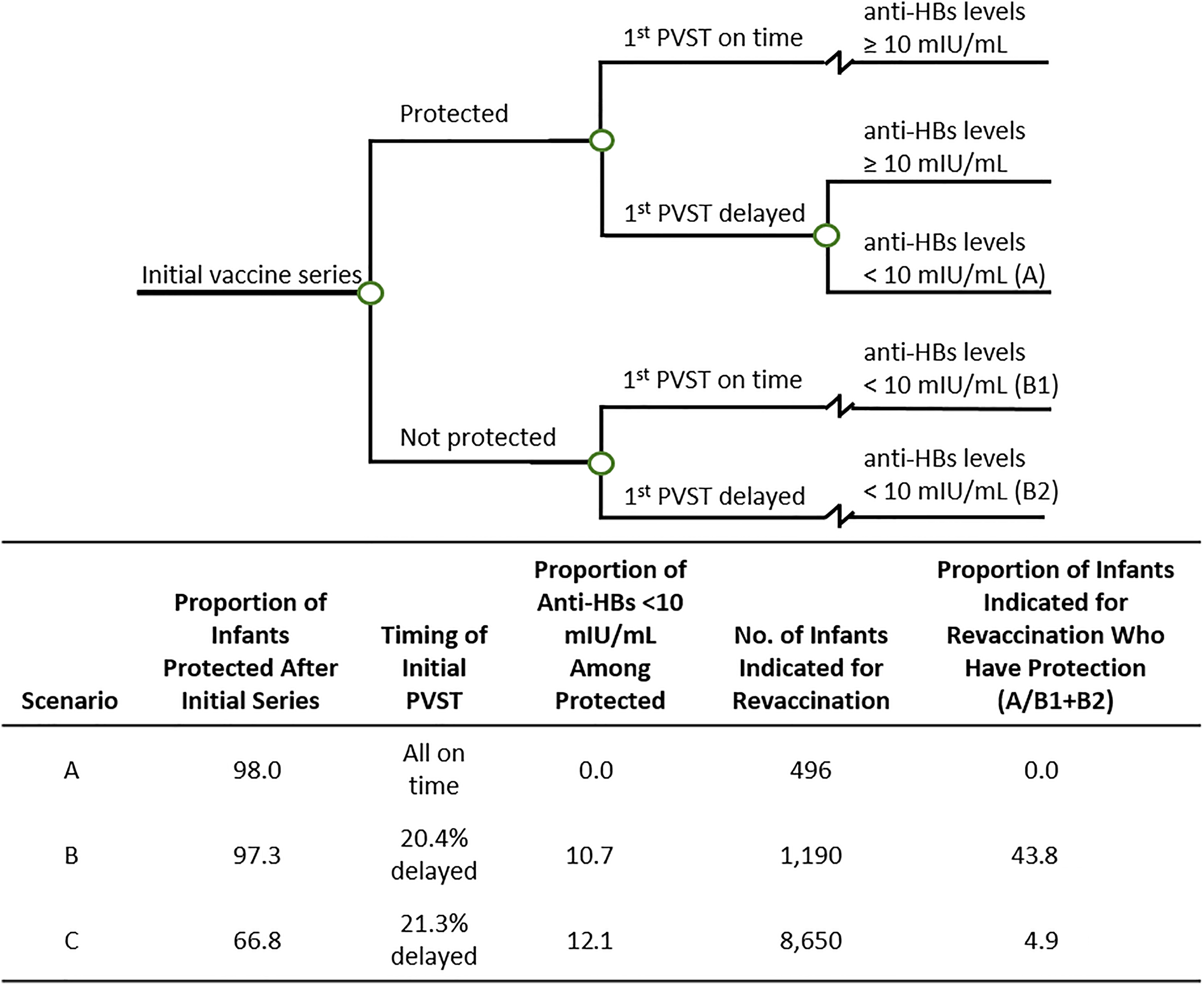
Inputs for creating epidemiologic scenarios A, B, and C for a cost analysis of single-dose hepatitis B revaccination among infants who were born to hepatitis B surface antigen–positive mothers and who were not responding to the initial vaccine series. The tree and set inputs were used to calculate the proportion of infants indicated for revaccination who already have previous protection under each corresponding scenario. Circles in decision tree indicate chance outcomes. Abbreviations: anti-HBs, hepatitis B antibodies; PVST, postvaccination serologic testing.
Cost Parameters
We expressed all costs in 2016 US dollars (Table 1). We obtained information on public and private prices for hepatitis B vaccine doses (Engerix B; GlaxoSmithKline Biologicals) from CDC’s 2016 vaccine price list. 18 As in previous analyses, we assumed that 53% of vaccines were purchased publicly, and we created a weighted average for the base-case estimate of vaccine cost. 17 We derived the cost of vaccine administration from previously reported data that we adjusted to 2016 US dollars by using the Consumer Price Index for medical care. 15,20 As in previous studies, we calculated the base-case estimate for the cost of vaccine administration by using a weighted average of the parameters from the private and public cost of vaccine administration and by assuming that 80% of children receive their vaccine from private providers. 17,19 The upper values for vaccine cost and vaccine administration assumed that all vaccines were either bought or administered privately, and the lower values assumed that all vaccines were bought or administered publicly. We did not include costs associated with adverse events of the vaccine because they are extremely rare, 24 and leaving them out conservatively favors the 3-dose revaccination strategy.
The cost of PVST is the sum of the costs for the following items: HBsAg testing to identify infection (Current Procedural Terminology code 87340), anti-HBs testing to identify immunity (Current Procedural Terminology code 86317), a blood draw (Current Procedural Terminology code 36415), 14,22 and an outpatient consultation for interpreting results (from previously reported data, adjusted to 2016 US dollars by using the medical care Consumer Price Index). 14,20 Individual base-case costs are equal to the national limitation amount from the 2016 Clinical Laboratory Fee Schedule. 21 To generate plausible ranges, we adjusted the national limitation amount to plus or minus 20%.
Indirect costs included loss of wages for caregivers taking time off work to take infants to be vaccinated and cost for travel to the clinic. As in previous analyses, we assumed that caregivers missed 2 hours of work, and we used the national median hourly wage of $17.40 per hour. 17,23 For sensitivity analyses, we used the national minimum wage ($7.25 per hour) as the lower bound and the 90th percentile hourly wage ($44.29) as the upper bound of a plausible range. 23 We used the overall Consumer Price Index to adjust previously reported caregiver travel costs to 2016 US dollars. 17,20 We used a plausible range of $5.00 (cost of a round-trip fare on public transportation in Atlanta, Georgia) to $35.00 for caregiver travel costs in sensitivity analyses.
We calculated the base cost of each vaccine visit as the sum of the average cost of a single vaccine dose, the average cost to administer a single dose, the cost of caregiver travel, and the cost of missing 2 hours of work. The base cost of all PVST visits was the sum of the cost of PVST (defined previously), the cost of caregiver travel, and the cost of missing 2 hours of work. We assumed that all PVST visits occurred as separate, previously unscheduled visits (ie, not scheduled well-child visits), and because they were unscheduled, we assumed they incurred indirect costs. In our primary analyses, we assumed that each revaccination visit also occurred as a separate, previously unscheduled visit and did not incur indirect costs. However, in additional analyses, we assumed that 1, 2, or 3 of the revaccination visits took place during a scheduled well-child visit, a visit that would have occurred even if the infant had not needed a hepatitis B revaccination dose. Therefore, we assumed that scheduled visits did not incur indirect costs.
Sensitivity Analyses
We conducted univariate threshold analyses on the proportion of infants protected by a single revaccination dose (dose 2a) in each epidemiologic scenario. Threshold values indicate the minimum efficacy required for the single-dose revaccination strategy to be a lower-cost option. For scenario B, we conducted 1-way sensitivity analyses to analyze the effect of changing each parameter individually. We conducted a 2-way sensitivity analysis to evaluate the results jointly for the efficacy of a single revaccination dose and the proportion of infants indicated for revaccination who were already protected.
Results
A single-dose revaccination strategy had lower costs than the 3-dose revaccination strategy in all scenarios (Table 2). In scenario B, under the assumption that all revaccination visits (visit 2a, visit 2b, and visit 2c) were previously unscheduled, a single-dose revaccination strategy reduced the cost of revaccination per infant by $155.72, which equated to total annual savings of $185 250. A single-dose revaccination strategy reduced the cost of revaccination per infant by $119.81 (total annual savings of $59 388) in scenario A and $123.83 (total annual savings of $1 071 082) in scenario C. Assuming that all vaccine visits were previously unscheduled, the dose 2a efficacy threshold value ranged from 0.03 (scenario B) to 0.45 (scenario A).
Per-infant cost of each revaccination strategy applied, using 3 epidemiologic scenariosa in a cost analysis of single-dose hepatitis B revaccination among infants who were born to hepatitis B surface antigen–positive mothers and who were not responding to the initial vaccine series
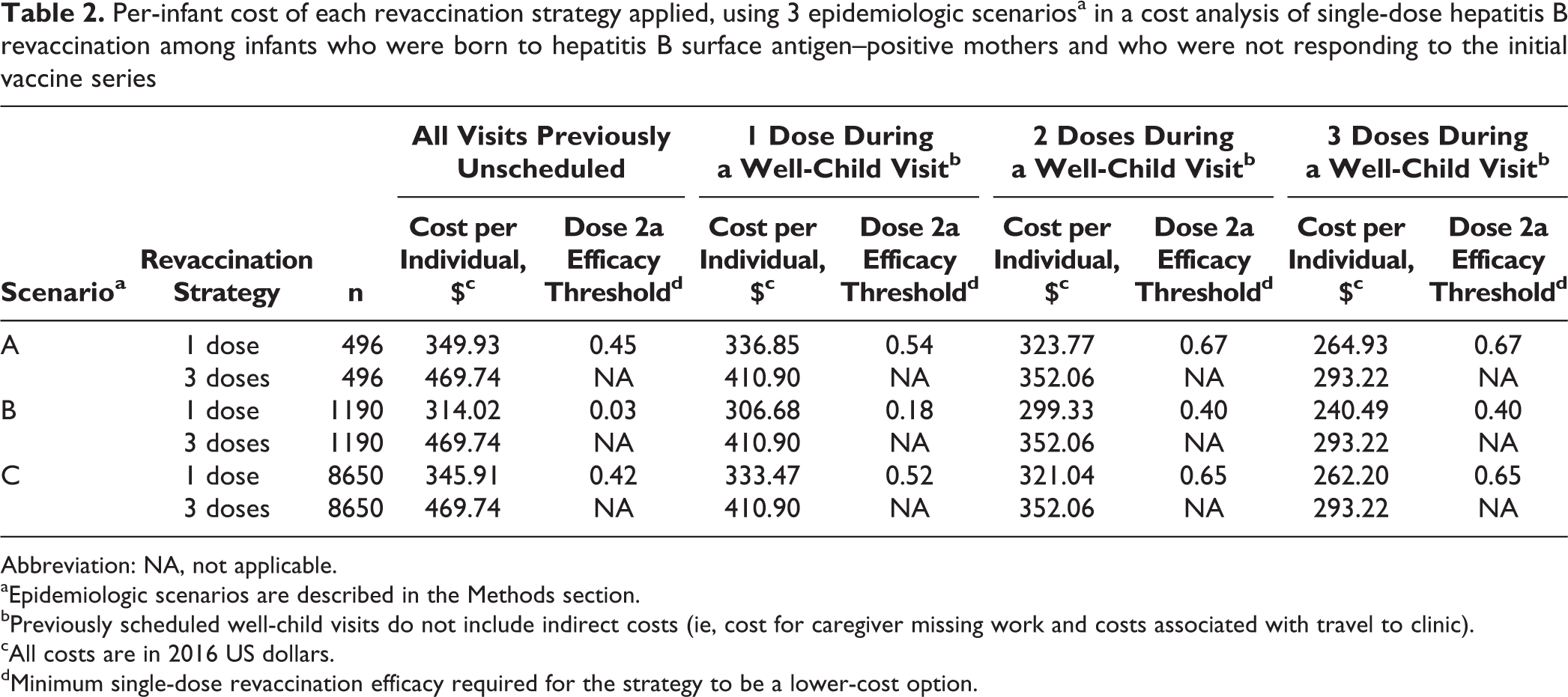
Abbreviation: NA, not applicable.
aEpidemiologic scenarios are described in the Methods section.
bPreviously scheduled well-child visits do not include indirect costs (ie, cost for caregiver missing work and costs associated with travel to clinic).
cAll costs are in 2016 US dollars.
dMinimum single-dose revaccination efficacy required for the strategy to be a lower-cost option.
Assuming that 1, 2, or 3 of the revaccination visits occurred during a scheduled well-child visit, a single-dose revaccination strategy still had lower costs than the 3-dose strategy in all 3 scenarios (Table 2). The incremental cost per infant was smallest when all 3 vaccination visits were assumed to occur during a scheduled well-child visit ($28.29 in scenario A, $52.73 in scenario B, and $31.02 in scenario C), but the single-dose revaccination strategy was still the lower-cost option. When all 3 vaccination visits were assumed to occur during a scheduled well-child visit, the maximum threshold value for efficacy of dose 2a was 0.67 (scenario A). As long as the single revaccination dose had an efficacy of 0.67 or higher, the single-dose revaccination strategy had a lower cost.
In the 1-way sensitivity analysis in scenario B, the factor that had the largest effect on incremental cost differences (per infant) was the efficacy of dose 2a, followed by the hourly wage of caregivers missing work (Figure 3). The only variable that had the potential to make the 3-dose revaccination strategy less expensive than the single-dose strategy was the efficacy of dose 2a (per-infant incremental cost ranged from –$5.50 to $181.10).
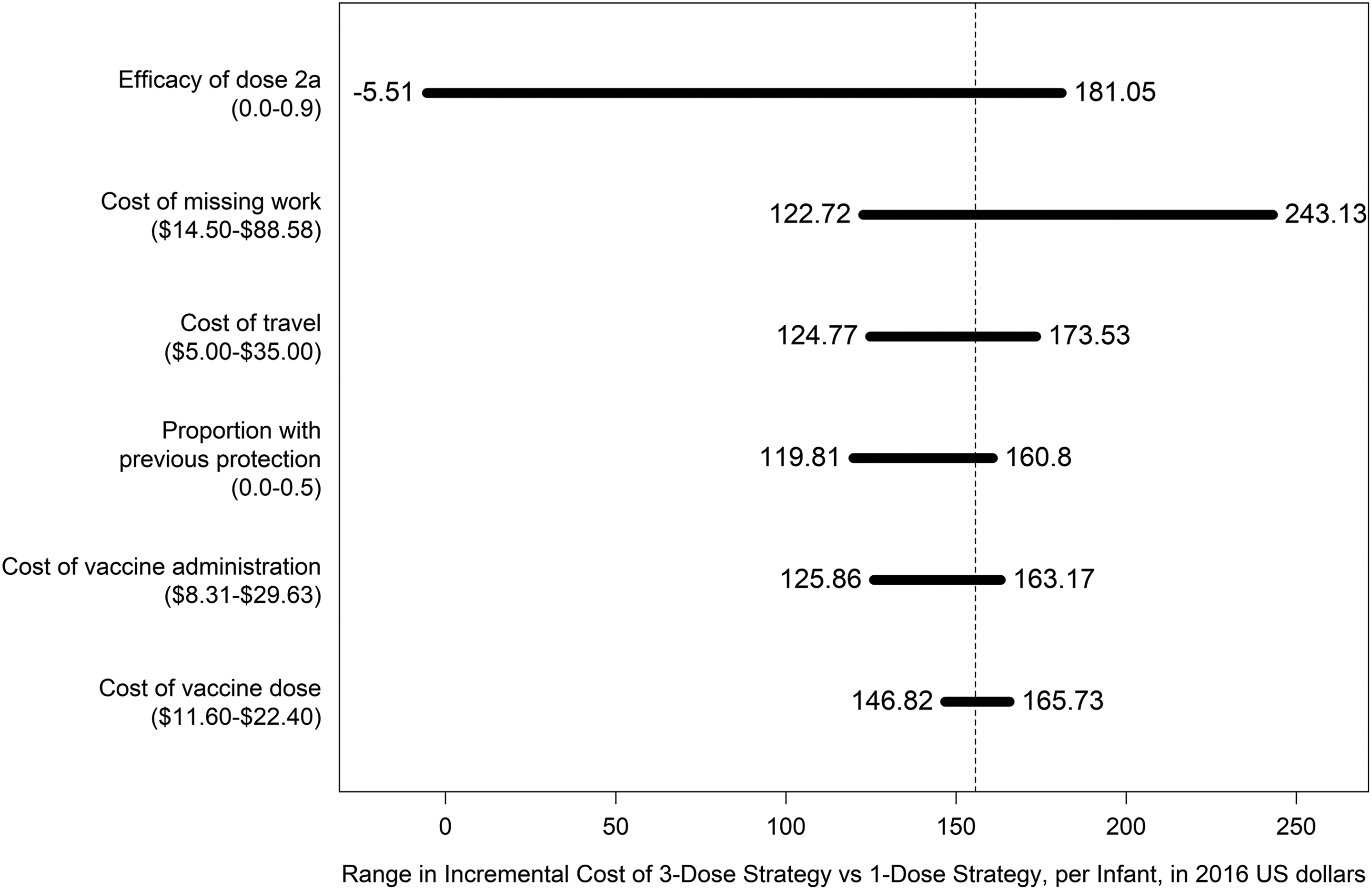
Tornado diagram of variables whose plausible ranges had the largest effect on incremental cost difference (per infant) in a cost analysis of a single-dose hepatitis B revaccination among infants who were born to hepatitis B surface antigen–positive mothers and who were not responding to the initial vaccine series. Analysis was conducted under assumptions described in scenario B, including the assumption that all vaccination visits occur during unscheduled visits. Dashed line indicates the base case at $155.72. Values in parentheses for each cost variable (y-axis) indicate plausible ranges.
In the 2-way sensitivity analysis of dose 2a efficacy and the proportion of infants who were previously protected but indicated for revaccination, the maximum threshold value for dose 2a efficacy was 0.45, which occurred when the proportion of infants with previous protection was 0. Under the assumption that all infants who were revaccinated were not previously protected, the 1-dose revaccination strategy was the least costly option as long as the efficacy of 1-dose revaccination was not less than 0.45.
Discussion
Our results indicate that the single-dose revaccination strategy is a less costly option than the 3-dose revaccination strategy under various epidemiologic scenarios and assumptions. Our results largely depended on the efficacy of dose 2a, a single revaccination dose. We calculated the highest, and most conservative, threshold value for dose 2a efficacy to be 0.67, which occurred under the extreme assumption that all infants indicated for revaccination received their initial PVST on time and were correctly indicated for revaccination (scenario A) and that 2 or 3 revaccination visits occurred during previously scheduled well-child visits. The threshold value for dose 2a was inversely related to the number of infants with a delayed initial PVST, because infants who were incorrectly indicated for revaccination (ie, previously protected, but anti-HB levels had waned to <10 mIU/mL) were correctly identified as previously protected in the single-dose revaccination strategy. In an analysis of more than 8000 infants enrolled in the PHBPP, nearly one-fourth of infants had PVST more than 6 months after the final vaccine dose. 24
No studies have specifically measured the efficacy of a single revaccination dose among infants not responding to the initial vaccine series, but related data indicate that efficacy is likely to be higher than this threshold. Pooling data from the only 2 studies that recorded anti-HBs in this population at 4 vaccine intervals 8,9 allowed us to calculate an efficacy of 0.78 (95% confidence interval, 0.40-0.97). Data from auxiliary sites in the PHBPP indicated an efficacy of 0.93 for a single revaccination dose (S.S., unpublished data, February 2017). The only other data on the efficacy of a single hepatitis B revaccination dose were from studies of teenagers and adults (median age, 46 years; range, 17-66 years). 25 –27 In these studies, 46.6% to 92.3% of participants responded to a single additional hepatitis B vaccine dose. However, the difference in age between infants and these older study participants limited the comparability of these data.
Limitations
A few assumptions and limitations should be considered when interpreting our results. First, the lack of data on the additional protection provided by a single revaccination dose led to large variability in the calculated efficacy of dose 2a. However, with the consideration of available data sources and the expectation of some infants incorrectly being indicated for revaccination as a result of a delayed PVST, it is likely that the actual efficacy of dose 2a is higher than the minimum threshold value needed for a single-dose strategy to be a lower-cost option. Second, we assumed that the risk of infection was the same in both the single-dose revaccination strategy and the 3-dose revaccination strategy. However, we based this assumption on the design of the single-dose revaccination strategy in which infants who remained unprotected after dose 2a would receive doses 2b and 2c. Third, we assumed that the dropout rate would be the same in both the single-dose revaccination strategy and the 3-dose revaccination strategy. We had no reason to believe that the single-dose strategy would increase the likelihood of dropout. Finally, our modeling assumed that all infants received 3 hepatitis B vaccine doses in their initial series; we did not model data on infants who received 4 doses in their initial series (eg, the recommended series for infants weighing <2000 g).
Conclusions
Single-dose hepatitis B revaccination is an appropriate strategy for infants who are born to HBsAg-positive mothers and who are not responding to the initial hepatitis B vaccine series. This strategy can conserve public health resources, shorten the duration of follow-up, and reduce the overall number of vaccine doses needed for infants born to HBsAg-positive mothers.
Our results can help guide revaccination strategies for infants across the United States. CDC funds the PHBPP in 64 US public health jurisdictions (50 states, 6 cities, and 8 territories). PHBPPs provide case-management services to HBsAg-positive women and their infants up to age 24 months; case management includes following the infant through completion of PVST and ensuring the infant’s test results are correctly interpreted. PHBPPs also search for infants who are lost to follow-up and refer infants to resources for complete follow-up when families move to a different public health jurisdiction or country. 14 Single-dose revaccination can conserve public health resources by shortening the duration of PHBPP follow-up for infants not responding to the initial vaccine series.
In February 2017, partially informed by these results, ACIP voted to approve single-dose revaccination for infants who were born to HBsAg-positive mothers and who were not responding to the initial vaccine series. 11,12 Three-dose revaccination will be maintained as an ACIP-recommended alternate strategy, depending on clinical circumstances or family preference (eg, some families may prefer possible additional vaccine doses instead of an additional blood draw).
Footnotes
Acknowledgments
The authors thank the Applied Public Health Advisory Group, a collaborative set of state and local public health professionals convened for this project, for its guidance and expertise. We especially thank Dr Greg Felzien for reviewing our article and providing feedback.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by funding from the CDC National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention Epidemic and Economic Modeling Agreement (NEEMA) (U38 PS004646). The findings and conclusions in this article are those of the authors and do not necessarily represent the views of CDC.
