Abstract
Objectives:
Public health laboratories (PHLs) provide essential services in the diagnosis and surveillance of diseases of public health concern, such as tuberculosis. Maintaining access to high-quality laboratory testing is critical to continued disease detection and decline of tuberculosis cases in the United States. We investigated the practical experience of sharing tuberculosis testing services between PHLs through the Shared Services Project.
Methods:
The Shared Services Project was a 9-month-long project funded through the Association of Public Health Laboratories and the Centers for Disease Control and Prevention during 2012-2013 as a one-time funding opportunity to consortiums of PHLs that proposed collaborative approaches to sharing tuberculosis laboratory services. Submitting PHLs maintained testing while simultaneously sending specimens to reference laboratories to compare turnaround times.
Results:
During the 9-month project period, 107 Mycobacterium tuberculosis complex submissions for growth-based drug susceptibility testing and molecular detection of drug resistance testing occurred among the 3 consortiums. The median transit time for all submissions was 1.0 day. Overall, median drug susceptibility testing turnaround time (date of receipt in submitting laboratory to result) for parallel testing performed in house by submitting laboratories was 31.0 days; it was 43.0 days for reference laboratories. The median turnaround time for molecular detection of drug resistance results was 1.0 day (mean = 2.8; range, 0-14) from specimen receipt at the reference laboratories.
Conclusions:
The shared services model holds promise for specialized tuberculosis testing. Sharing of services requires a balance among quality, timeliness, efficiency, communication, and fiscal costs.
Public health laboratory (PHL) testing in the United States is evolving because of advances in technology, workforce shortages, varying testing needs and volumes, new public health threats, and the potential impact from health care reform. PHLs currently perform a variety of functions, including diagnostic testing, applied research, and testing conducted for purposes of surveillance and outbreak response. 1 –4 However, the reality of ongoing budgetary constraints, which are projected to continue into the near future, has prompted policy makers to consider eliminating or consolidating select laboratory services in PHLs as a way to maintain high-quality essential laboratory services while reducing costs. Because PHLs are often the only source for these services in a jurisdiction, critical capabilities must be maintained to ensure that PHLs meet their mission of providing accurate and timely laboratory testing to aid in the diagnosis and control of communicable diseases. 2
A competent, technical PHL workforce must be available to use new and emerging technologies, such as molecular detection of pathogens and drug susceptibility testing (DST), and understand the role that whole genome sequencing plays in both. However, as laboratorians with years of technical expertise retire, finding laboratorians with conventional and molecular laboratory proficiencies, as well as knowledge of quality testing systems, is crucial and becoming increasingly difficult for PHLs. 5 In addition, changes in testing volume alter how PHLs might effectively and efficiently serve the nation’s public health needs by affecting workload and turnaround times (TATs). Questions remain about whether clinical laboratories will adjust testing of public health importance and how that may affect volumes in PHLs.
PHLs will continue to play an important role in the diagnosis and surveillance of diseases of public health concern even with the possible flux in laboratory workforce, testing technologies, and volume. PHLs often address, but are not limited to, low-incidence diseases with high rates of morbidity or mortality or diseases with high rates of transmissibility. Tuberculosis (TB) is a prime example because it continues to be a burden for the United States despite continuing declines in TB incidence. 6 Maintaining access to high-quality laboratory testing is critical for disease detection and continued decline of TB cases in the United States. In 2010, the Association of Public Health Laboratories and the Centers for Disease Control and Prevention (CDC) developed the National TB Laboratory Services Survey to characterize the overall ability of US private, clinical, or commercial laboratories and PHLs to provide diagnostic and reference services for TB. A 2012 summary report detailed the responding laboratories’ testing capabilities. 7 Compared with private, clinical, or commercial laboratories in the United States, PHLs represented a higher proportion of laboratories performing specialized testing, such as direct detection of Mycobacterium tuberculosis complex in clinical specimens using nucleic acid amplification testing, as well as first-line and second-line DST. DST is critical for TB control because rapidly detecting drug resistance ensures that clinicians have results for implementing effective treatment regimens as soon as possible. Current recommendations for maintaining DST proficiency include performing ≥50 DSTs annually. 8 According to the 2010 survey, 39 of 91 (43%) responding laboratories and specifically 19 of 58 (33%) responding PHLs that performed first-line DST tested ≤5 isolates per month 7 (ie, ≤60 isolates annually); however, the proportion of laboratories performing 50-60 DSTs annually was not determined by the survey. For PHLs that do not meet the recommended testing volume of ≥50 DSTs annually, the cost and quality benefits of sharing specialized testing might outweigh the possible challenges associated with shipping, service delivery, or loss of expertise. In some instances, PHLs have already effectively implemented shared service approaches for other laboratory testing, including vaccine-preventable diseases and newborn screening, and could expand this practice to other areas of public health testing. 9,10 Currently, the California state PHL performs TB testing for participating county and local PHLs through referral of liquid media inoculated with processed specimens for further testing, such as culture incubation, identification, and DST. 11
These successful examples of sharing specialized testing along with the continued decline in TB incidence prompted the Association of Public Health Laboratories and the CDC Division of Tuberculosis Elimination to create the Shared Services Project. The Association of Public Health Laboratories issued a request for proposals from PHLs interested in exploring novel approaches to sharing TB laboratory testing services. The purpose of the project was to document the challenges and successes associated with sharing laboratory testing services. We describe 3 funded Shared Services Projects focused on consolidation of growth-based DST and molecular detection of drug resistance (MDDR).
Methods
Proposals
The Shared Services Project was funded by CDC through the Association of Public Health Laboratories as a one-time funding opportunity to PHLs that proposed collaborative approaches to sharing TB laboratory services. Each interested group of laboratories (consortium) submitted a project description. A panel of laboratory subject matter experts objectively reviewed 8 proposals and selected 4 consortium projects. The selected consortiums are hereinafter designated as Consortiums A, B, C, and D. We did not include data from Consortium D in this analysis because the approach (nucleic acid amplification testing only) used by Consortium D was different from the approach (ie, growth-based DST ± MDDR) used by the other 3 consortiums. Consortium A and Consortium C performed both growth-based DST and MDDR; Consortium B performed only growth-based DST (Table 1). MDDR testing was performed only at the reference laboratories; it was not routinely available at the submitting laboratory.
Tuberculosis testing services and methods performed by submitting and reference laboratories participating in the Shared Services Project,a United States, September 2012 through June 2013
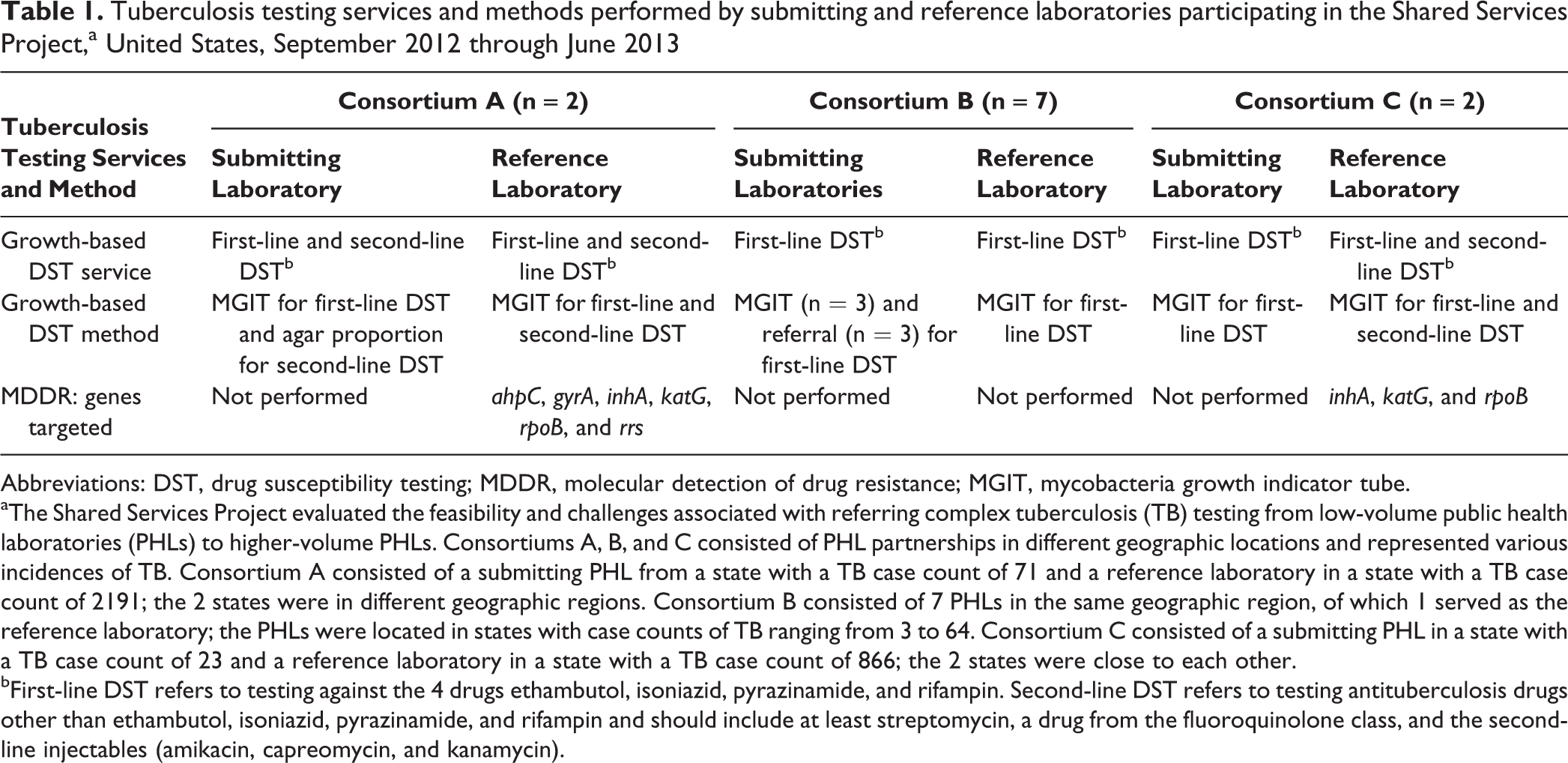
Abbreviations: DST, drug susceptibility testing; MDDR, molecular detection of drug resistance; MGIT, mycobacteria growth indicator tube.
aThe Shared Services Project evaluated the feasibility and challenges associated with referring complex tuberculosis (TB) testing from low-volume public health laboratories (PHLs) to higher-volume PHLs. Consortiums A, B, and C consisted of PHL partnerships in different geographic locations and represented various incidences of TB. Consortium A consisted of a submitting PHL from a state with a TB case count of 71 and a reference laboratory in a state with a TB case count of 2191; the 2 states were in different geographic regions. Consortium B consisted of 7 PHLs in the same geographic region, of which 1 served as the reference laboratory; the PHLs were located in states with case counts of TB ranging from 3 to 64. Consortium C consisted of a submitting PHL in a state with a TB case count of 23 and a reference laboratory in a state with a TB case count of 866; the 2 states were close to each other.
bFirst-line DST refers to testing against the 4 drugs ethambutol, isoniazid, pyrazinamide, and rifampin. Second-line DST refers to testing antituberculosis drugs other than ethambutol, isoniazid, pyrazinamide, and rifampin and should include at least streptomycin, a drug from the fluoroquinolone class, and the second-line injectables (amikacin, capreomycin, and kanamycin).
We asked the selected consortiums to execute their proposed model for shared services for a period of at least 9 months during September 2012 through June 2013. Each consortium identified a single laboratory that would serve as the reference laboratory to which the other consortium members would refer samples. As a baseline for comparison, submitting laboratories continued to perform in-house testing in real time as submissions were simultaneously sent to the reference laboratory. We instructed submitting laboratories not to send submissions in batches (ie, not to hold multiple submissions to ship together at one time) to the reference laboratory and to treat the referral as real-time diagnostic patient testing. Submitting laboratories identified all samples as Mycobacterium tuberculosis complex before referral. We encouraged laboratories to communicate with their TB control program and establish a communication plan among laboratories.
Participating Laboratories
Consortiums A, B, and C consisted of PHL partnerships in different geographic locations and represented various incidences of TB. As a comparison, the national TB case count during 2012—when the study took place—was 9945. 12 Consortium A consisted of a submitting PHL from a state with a TB case count of 71 and a reference laboratory in a state with a TB case count of 2191; the 2 states were in different geographic regions. Consortium B consisted of 7 PHLs in the same geographic region, of which 1 served as the reference laboratory; the PHLs were located in states with case counts of TB ranging from 3 to 64. As with Consortium A, Consortium C consisted of a submitting PHL in a state with a low TB case count of 23 and a reference laboratory in a state with a higher TB case count of 866; however, the 2 states were close to each other.
Data Collection
Consortiums were required to report data and submit progress reports and final reports to the Association of Public Health Laboratories. We examined the following data for each consortium: TAT, culture contamination rates, correlation of results between submitting laboratories and reference laboratories, challenges and successes, and outcomes. We categorized data on TAT according to several parameters: specimen collection to specimen receipt in the submitting laboratory, specimen receipt in the submitting laboratory to specimen receipt in the reference laboratory, identification of Mycobacterium tuberculosis complex at the submitting laboratory to shipment to the reference laboratory (defined as preshipment TAT), shipment time from submitting laboratory to receipt in the reference laboratory (defined as transit time), and receipt in the reference laboratory to rifampin result report date to the submitting laboratory. We defined DST TAT as the date of specimen receipt in the submitting laboratory to the rifampin result report. We reported TAT in days. Data submitted as part of the Shared Services Project did not include patient identifiers; as such, CDC deemed the project exempt from institutional review board review.
Data Analysis
Two consortiums published consortium-specific analyses and descriptions of experiences. 13,14 Our analysis provides an overview of the Shared Services Project to summarize quantitative data (ie, TAT, contamination rates, and correlation of DST results) and assess common successes, challenges, and lessons learned across the 3 consortiums. We evaluated data using Microsoft Excel and SAS version 9.3. 15
Results
Submissions
During the 9-month study period, 107 sample submissions for growth-based DST and MDDR testing occurred among the 3 consortiums. Referred submissions were classified by submitting laboratories as respiratory (n = 79), nonrespiratory (n = 24), and of unknown origin (n = 4). Submitting laboratories received 38 primary specimens (unprocessed clinical specimens), 33 sediments (processed clinical specimens), and 36 reference isolates (culture growth from sediment) of Mycobacterium tuberculosis complex (Table 2) from health departments, hospitals, and clinical laboratories. After the submitting laboratories tested the samples, the 33 (31%) sediments and 74 (69%) isolates that tested positive for Mycobacterium tuberculosis complex were sent to the reference laboratory, where growth-based DST was performed.
Workload data of Mycobacterium tuberculosis complex submissions at submitting laboratories participating in the Shared Services Project,a United States, September 2012 to June 2013

aThe Shared Services Project evaluated the feasibility and challenges associated with referring complex tuberculosis (TB) testing from low-volume public health laboratories (PHLs) to higher-volume PHLs. Consortiums A, B, and C consisted of PHL partnerships in different geographic locations and represented various incidences of TB. Consortium A consisted of a submitting PHL from a state with a TB case count of 71 and a reference laboratory in a state with a TB case count of 2191; the 2 states were in different geographic regions. Consortium B consisted of 7 PHLs in the same geographic region, of which 1 served as the reference laboratory; the PHLs were located in states with case counts of TB ranging from 3 to 64. Consortium C consisted of a submitting PHL in a state with a TB case count of 23 and a reference laboratory in a state with a TB case count of 866; the 2 states were close to each other.
bPrimary specimens were cultured at submitting laboratories and were sent to reference laboratories as sediments or isolates.
cSediment is defined as a processed specimen and isolate is defined as culture growth from sediment.
Shipment Considerations
Median preshipment TAT was 1.0 (mean = 4.9; range, 0-84) day (Table 3). Median transit time for all 107 submissions was 1.0 (mean = 1.6; range, 0-11) day. Various factors impacted shipping TAT, including weather delays, batched shipments, time or day of shipping from submitting laboratory, time or day of receipt at reference laboratory, and shipment method (eg, express, next day, overnight). Delayed shipments and batching were the most commonly noted shipping-related issues.
Shipping turnaround times for submitting laboratories participating in the Shared Services Project,a United States, September 2012 to June 2013
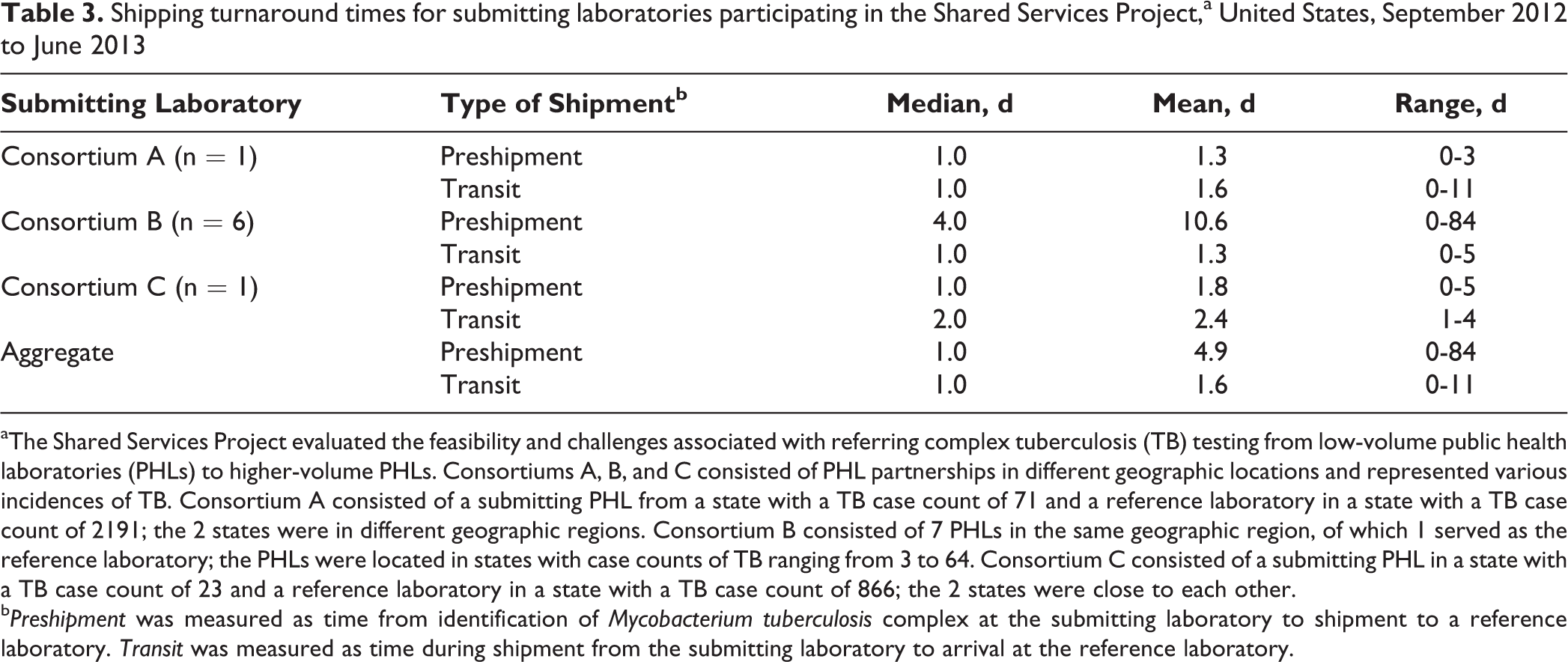
aThe Shared Services Project evaluated the feasibility and challenges associated with referring complex tuberculosis (TB) testing from low-volume public health laboratories (PHLs) to higher-volume PHLs. Consortiums A, B, and C consisted of PHL partnerships in different geographic locations and represented various incidences of TB. Consortium A consisted of a submitting PHL from a state with a TB case count of 71 and a reference laboratory in a state with a TB case count of 2191; the 2 states were in different geographic regions. Consortium B consisted of 7 PHLs in the same geographic region, of which 1 served as the reference laboratory; the PHLs were located in states with case counts of TB ranging from 3 to 64. Consortium C consisted of a submitting PHL in a state with a TB case count of 23 and a reference laboratory in a state with a TB case count of 866; the 2 states were close to each other.
b Preshipment was measured as time from identification of Mycobacterium tuberculosis complex at the submitting laboratory to shipment to a reference laboratory. Transit was measured as time during shipment from the submitting laboratory to arrival at the reference laboratory.
Growth-Based DST
Turnaround times for growth-based first-line DST varied across consortiums primarily because of different testing practices. Overall, median DST TAT for parallel testing performed in house by submitting laboratories was 31.0 days compared with 43.0 days for reference laboratories (Table 4). Median growth-based DST TATs at reference laboratories were longer for sediments (48.0 days) than for referred isolates (42.0 days) for all 3 consortiums.
Growth-based drug susceptibility testing turnaround times for referred sediments and isolates of Mycobacterium tuberculosis complex performed in parallel by submitting and reference laboratories participating in the Shared Services Project,a United States, September 2012 to June 2013
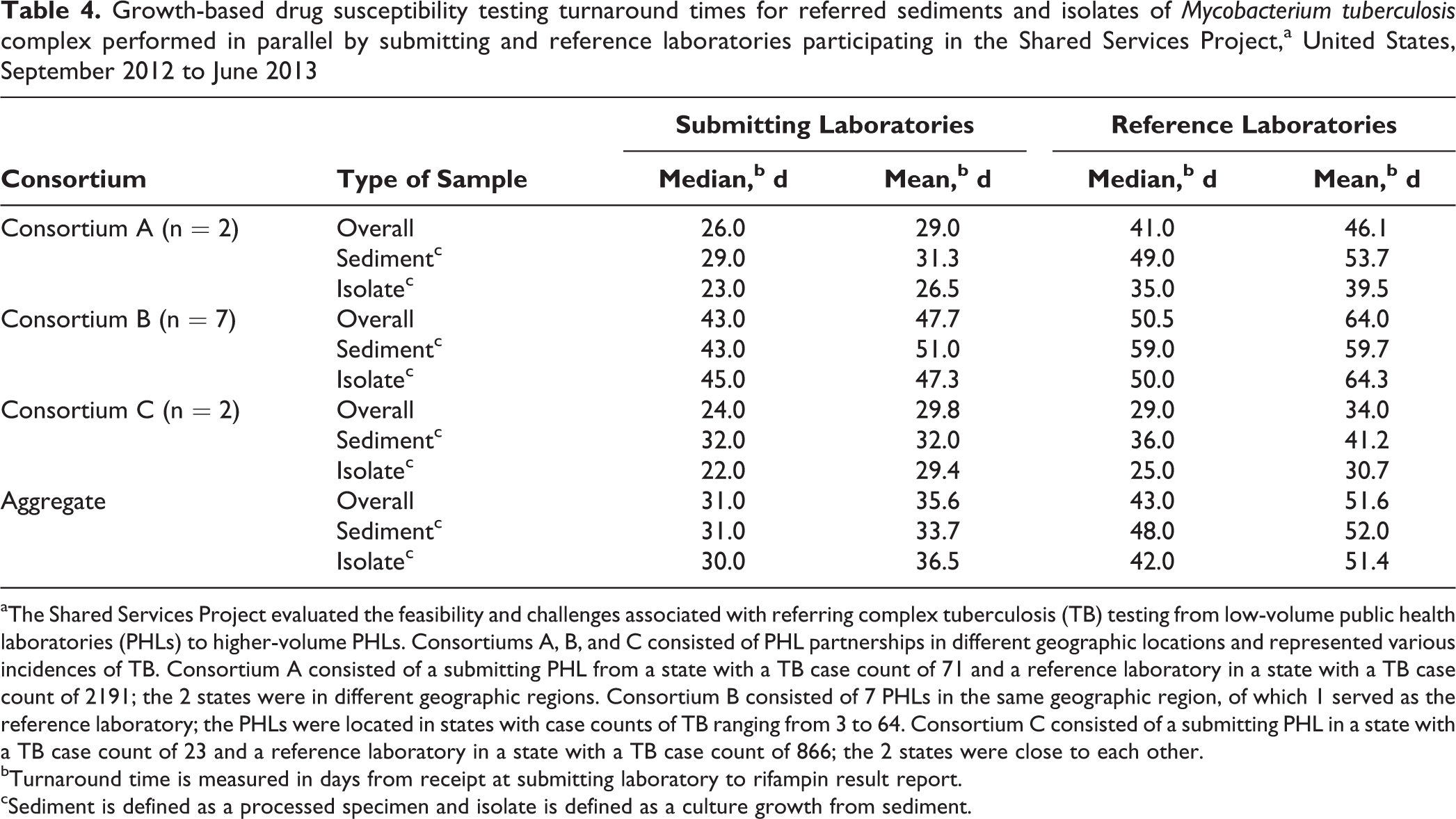
aThe Shared Services Project evaluated the feasibility and challenges associated with referring complex tuberculosis (TB) testing from low-volume public health laboratories (PHLs) to higher-volume PHLs. Consortiums A, B, and C consisted of PHL partnerships in different geographic locations and represented various incidences of TB. Consortium A consisted of a submitting PHL from a state with a TB case count of 71 and a reference laboratory in a state with a TB case count of 2191; the 2 states were in different geographic regions. Consortium B consisted of 7 PHLs in the same geographic region, of which 1 served as the reference laboratory; the PHLs were located in states with case counts of TB ranging from 3 to 64. Consortium C consisted of a submitting PHL in a state with a TB case count of 23 and a reference laboratory in a state with a TB case count of 866; the 2 states were close to each other.
bTurnaround time is measured in days from receipt at submitting laboratory to rifampin result report.
cSediment is defined as a processed specimen and isolate is defined as a culture growth from sediment.
Of the 107 samples tested, 78 were analyzed for correlation of DST results between the submitting laboratory and the reference laboratory. DST was not performed on the remaining 29 specimen submissions at both the submitting and reference laboratories because of culture contamination, growth issues, or unspecified reasons at one of the laboratories; these samples were distributed proportionately among consortiums. The correlation of DST results between the submitting laboratory and reference laboratory for first-line drugs showed that 69 of 78 (88%) isolates had the same results. The 9 discordant results occurred for the drugs pyrazinamide (3/78; 4%) and isoniazid (6/78; 8%); isolates with discordant results for isoniazid received molecular testing to clarify results. Across the consortiums, the reported contamination rate was 7% (7/107); Consortium C did not note any contamination issues. Consortiums A and B observed growth issues for isolates in delayed shipments and sediments. Referred sediments and delayed isolates were sometimes contaminated; because of this contamination, subcultures were needed for pure growth, thereby contributing to extended growth-based DST TATs.
Molecular Detection of Drug Resistance
In addition to shared service testing of growth-based DST, 2 of the reference laboratories also provided MDDR testing to submitting laboratories where this testing was not routinely available in house (Table 1). The reference laboratory for Consortium A analyzed ahpC, gyrA, inhA, katG, rpoB, and rrs genes to assess resistance to the following drugs: fluoroquinolones, isoniazid, rifampin, and second-line injectables. The reference laboratory for Consortium C analyzed inhA, katG, and rpoB genes to assess resistance to isoniazid and rifampin. The MDDR median TAT for Consortium A was 1.0 (mean = 1.4; range, 0-7) day and for Consortium C was 4.0 (mean = 6.8; range, 1-14) days from specimen receipt at the reference laboratories.
Communication
Each consortium used different communication strategies for the project. Consortium A held routine conference calls to discuss challenges and to assess any need for corrective action. Consortium B coordinated routine conference calls with all partners but found it challenging to have representatives from all submitting laboratories on the calls. Consortium C participated in frequent informal communication through telephone calls as needed. Two consortiums used conference calls during the project period to adjust workflow caused by delays and challenges. Consortiums A and C performed mid-project evaluations of practices and workflow. Several practices were adjusted to minimize TAT and maximize efficiency, such as whether a sediment or isolate was referred, submission of an isolate as follow-up to sediments shipped, timing and type of shipment from submitting laboratory, and timing of testing at reference laboratory.
Results Reporting
Three submitting laboratories planned to receive results from their reference laboratory via electronic reporting. However, only 2 laboratories successfully implemented electronic reporting because of delays in information technology changes. One submitting laboratory in Consortium B achieved capacity to electronically receive Health Level 7 messaging from the reference laboratory, and Consortium C’s submitting laboratory participated in a secure internet-based portal to retrieve electronic reports. Consortiums B and C noted that electronic reporting was a valuable tool for timely and efficient reporting from reference laboratories to submitting laboratories. Submitting laboratories not using electronic reporting received results via secure facsimile.
Discussion
As laboratory testing continues to change, PHLs will need to consider modifications to testing practices, including the possibility of sharing services. In this study, the 3 participating consortiums provided evidence that, even during a relatively short period of time, a shared services model was a viable alternative to continued complex TB testing in house in low-volume laboratories. As a result of the study, the consortiums recognized that with adjustments to workflow practices to minimize shipping time and maximize efficiency, with ongoing communication, and with development of efficient mechanisms for reporting results, low-volume laboratories may benefit from a shared services model for complex TB testing, while maintaining in-house smear, culture, and identification. Consistent communication and collaboration between partners was central to the success of the Shared Services Project, because these laboratories had not previously established relationships for shared services. Flexibility, by both the submitting and reference laboratories, contributed to productive partnerships in which challenges were addressed and TAT was reduced. Shared services also allowed low-incidence submitting laboratories access to MDDR testing, thereby providing them with results not routinely available to them. Consequently, partner TB programs received molecular results weeks before growth-based DST results would have been available, thereby potentially allowing patients to start appropriate therapy earlier. 14
Shared services models will inevitably have challenges. 10,13,14 Once partnerships had been developed, timely shipment of specimens, as acknowledged by all consortiums, was a critical component. Consortiums indeed encountered numerous shipping-related issues, including delays because of severe weather, time and day of shipping from the submitting laboratory and of receipt at the reference laboratory, shipment method (eg, express, next day, overnight), and batched shipments (despite instruction not to batch). Specifically, the chosen shipment method affected the timing of delivery at the reference laboratory and batched shipments of multiple isolates at once resulted in a wide range (0-84 days) in preshipment TAT, ultimately delaying testing and results at the reference laboratory. Efficient reporting of results is important because delayed reporting negates timely testing performed at reference laboratories. As such, the use of electronic reporting was noted as an advantageous tool for timely reporting. Each challenge was met with collaboration and protocol adjustments to reduce shipping-related TAT, thus improving overall TAT and reporting that ultimately impacted patient management outcomes.
Different testing practices across the 3 consortiums resulted in variable TATs for growth-based DST. Although the TAT for growth-based DST at reference laboratories was longer than at submitting laboratories, Consortium C showed promise that this lag could be overcome in a shared services model even when factoring in shipping TAT. A potential consideration for a TB shared services model is the referral of sediments versus isolates. Turnaround times for growth-based DST at reference laboratories were expected to be longer for sediments than for isolates because of growth effects due to shipment at a time of low bacterial burden and the need for reference laboratories to first culture growth from sediments before performing DST. However, we found variability in the TATs between sediments and isolates at the consortium level. Two consortiums improved TAT for growth-based DST when testing was performed from referred isolates rather than from sediments. Isolates may be more amenable for referral of growth-based DST; however, there are times that referral of sediments for MDDR followed by a shipment of culture growth for testing could be more successful than shipping only culture growth, as demonstrated by Consortium A during the project.
Conclusions
In a shared services model, most challenges can be overcome with defined roles, responsibilities, and expectations as well as clearly detailed agreements and protocols. Defined protocols related to workflow, delivery of specimens, and specimen type should be developed and agreed upon before implementation. Expectations of testing practices, performance measures, and timely results reporting should be clarified and accepted by all. Submitting laboratories should be vocal on behalf of their patients and ensure quality on both ends of the partnership. Partners must be accessible, willing to collaborate, and willing to engage in discussions to adjust practices as needed. Finally, trust must be established between reference and submitting laboratories through transparent and frequent communication to ensure that quality testing is performed with timely and reliable results.
Partially based on lessons learned through the Shared Services Project, the CDC Division of Tuberculosis Elimination funded the Association of Public Health Laboratories to establish a TB DST Reference Center in 2015 to provide an alternate resource for high-quality DST for PHLs that perform <50 TB DSTs annually. Shared services could also be used on a smaller scale for defined periods. To help mitigate the effects of staff turnover and loss of expertise, shared services may allow continuity of operations while laboratories train new personnel. Laboratories could also implement shared services agreements during temporary laboratory closures, such as a move to a new laboratory building, replacement/repair of air-handling systems, or equipment replacement.
Sharing of services requires a balance among quality, timeliness, efficiency, and fiscal impacts. Referral of services may require an increased financial investment, 13,14 but assured quality of testing and reliability of results should be of highest concern for laboratories supporting the diagnosis and care of TB patients. Outcomes from this study showed that the shared services model is feasible and practical for laboratories performing low volumes of growth-based DST and MDDR testing for TB, when common challenges are addressed to ensure a successful model.
Footnotes
Acknowledgments
The authors thank the tuberculosis laboratorians from the California Department of Public Health Microbial Diseases Laboratory, Colorado Laboratory Services Division, Denver Health Medical Center, Montana Public Health Laboratory, New York Department of Health, Wadsworth Center, North Dakota Department of Health, Rhode Island State Health Laboratory, South Dakota Public Health Laboratory, Utah Public Health Laboratory, Wisconsin State Laboratory of Hygiene, and Wyoming Public Health Laboratory; Anne Gaynor and Paul Zell from the Association of Public Health Laboratories; and Frances Tyrrell from the Centers for Disease Control and Prevention. The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention, the Association of Public Health Laboratories, or the US Department of Health and Human Services.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was 100% funded with federal funds from a federal program of $1 227 171. This publication was supported by cooperative agreement no. U60HM000803 funded by the Centers for Disease Control and Prevention.
