Abstract
Nicotine in electronic nicotine and non-nicotine delivery systems (ENDS/ENNDS) may present a risk of harm to those with cardiovascular disease and the fetuses of pregnant women. We assessed the extent to which adult users of ENDS/ENNDS used these products with nicotine. We obtained data for this study from a national probability survey of 6051 US adults that was conducted in August and September 2015. Of 399 adult ENDS/ENNDS users who were current smokers, 337 (80.7%) used ENDS/ENNDS containing nicotine, whereas only 29 of 71 (36.9%) ENDS/ENNDS users who were never smokers used ENDS/ENNDS containing nicotine. Assessments of the population health impact of ENDS/ENNDS use among never smokers should take into account the extent to which use involves nicotine.
With rapid growth in the use of electronic nicotine and non-nicotine delivery systems (ENDS/ENNDS) and concerns about their potential harm, the US Food and Drug Administration (FDA) extended its regulatory authority over tobacco products to include ENDS/ENNDS in 2016. 1 –4 Among the potential health concerns related to ENDS/ENNDS use, particularly for young people, pregnant women, and never smokers, are risks associated with nicotine exposure and possible nicotine addiction. 4 –7 However, consumers have an option to use ENDS/ENNDS with or without e-liquid containing nicotine. A 2015 study from Monitoring the Future found that only a minority (13%-20%) of adolescents who reported using ENDS/ENNDS reported using ENDS/ENNDS that contained nicotine. 8 The purpose of this study was to estimate the prevalence of ENDS/ENNDS use with nicotine among US adults.
Methods
We obtained unpublished data for this study from the Tobacco Products and Risk Perceptions Survey, which was conducted during August and September 2015 and administered to a cross-sectional, probability sample of 6051 US adults drawn from GfK’s KnowledgePanel, a probability-based web panel designed to be representative of noninstitutionalized US adults. GfK panelists are selected via address-based probability sampling, and only people who are invited can join KnowledgePanel. For this study, we invited 8135 adult panelists to participate in the survey; 6051 (74%) completed the survey. We weighted data using a study-specific poststratification weight to adjust for sources of sampling and nonsampling error; we used demographic and geographic distributions from the March 2015 Current Population Survey as benchmarks for adjustment (https://www.census.gov/programs-surveys/cps/about.html). This study was approved by the Georgia State University Institutional Review Board.
Measures and Statistical Analysis
Participants who reported smoking <100 cigarettes in their life were considered never smokers. Participants who had smoked ≥100 cigarettes in their life were classified as current smokers if they reported that they currently smoked cigarettes “every day” or “some days” and as former smokers if they reported that they did not currently smoke at all. Former smokers indicated the month and year they had quit; we computed duration since last smoking from this date. We defined recent quitters as those who quit smoking within the previous year. Participants who reported that they had not “ever used electronic vapor products” were classified as never users of ENDS/ENNDS. Participants who reported ever using electronic vapor products were classified as current users if they indicated that they now used “every day,” “some days,” or “rarely” and as former users if they indicated that they now used “not at all.” Current ENDS/ENNDS users also reported the age at which they started using ENDS/ENNDS. We assessed nicotine content in ENDS/ENNDS by a yes or no response to the question, “Does the electronic vapor product you usually use contain nicotine?”
We generated weighted prevalence estimates and 95% confidence intervals (CIs) and used weighted logistic regression and Pearson χ2 tests with a Rao–Scott second-order correction to evaluate associations between cigarette smoking and ENDS/ENNDS use and between frequency of ENDS/ENNDS use and cigarette smoking status with use of nicotine in ENDS/ENNDS. We conducted all analyses using the Survey package version 3.30 for the R statistical program version 3.3.1. 9,10
Results
Of the 6051 participants who completed the survey, 6008 participants aged 18-94 had complete data on ENDS/ENNDS use and cigarette smoking status. Half of the participants (n = 3039, 50.2%) were male; 4462 (73.7%) were non-Hispanic white, 519 (8.6%) were non-Hispanic black, 713 (11.8%) were Hispanic, and 357 (5.9%) were other race/ethnicity.
Prevalence of Smoking and ENDS Use
An estimated 14.1% (95% CI, 13.1%-15.2%) of US adults were current smokers, and 7.4% (95% CI, 6.6%-8.4%) were current ENDS users (Table 1). The association between smoking and ENDS/ENNDS use was significant (F = 166.97, df1 = 3.92, df2 = 23 767.00; P < .001). Although few former smokers were ENDS/ENNDS users (5.3%; 95% CI, 4.0%-7.1%), recent quitters were more likely than those who quit >1 year ago to have used ENDS/ENNDS (df = 1590; P < .001). Among those who quit smoking 1-5 years ago, 16.5% (95% CI, 11.0%-23.9%) were current ENDS/ENNDS users and 32.6% (95% CI, 25.7%-40.5%) were former ENDS/ENNDS users. Among former smokers who quit smoking >5 years ago, only 2.0% (95% CI, 1.1%-3.5%) reported current ENDS/ENNDS use. Among 79 former smokers/current ENDS/ENNDS users, we found that 51 likely used ENDS/ENNDS before quitting and 23 likely started using ENDS/ENNDS after quitting (5 were missing information on dates for quitting smoking or initiating ENDS/ENNDS).
Use of electronic nicotine and non-nicotine delivery systems by smoking status among US adults, 2015a
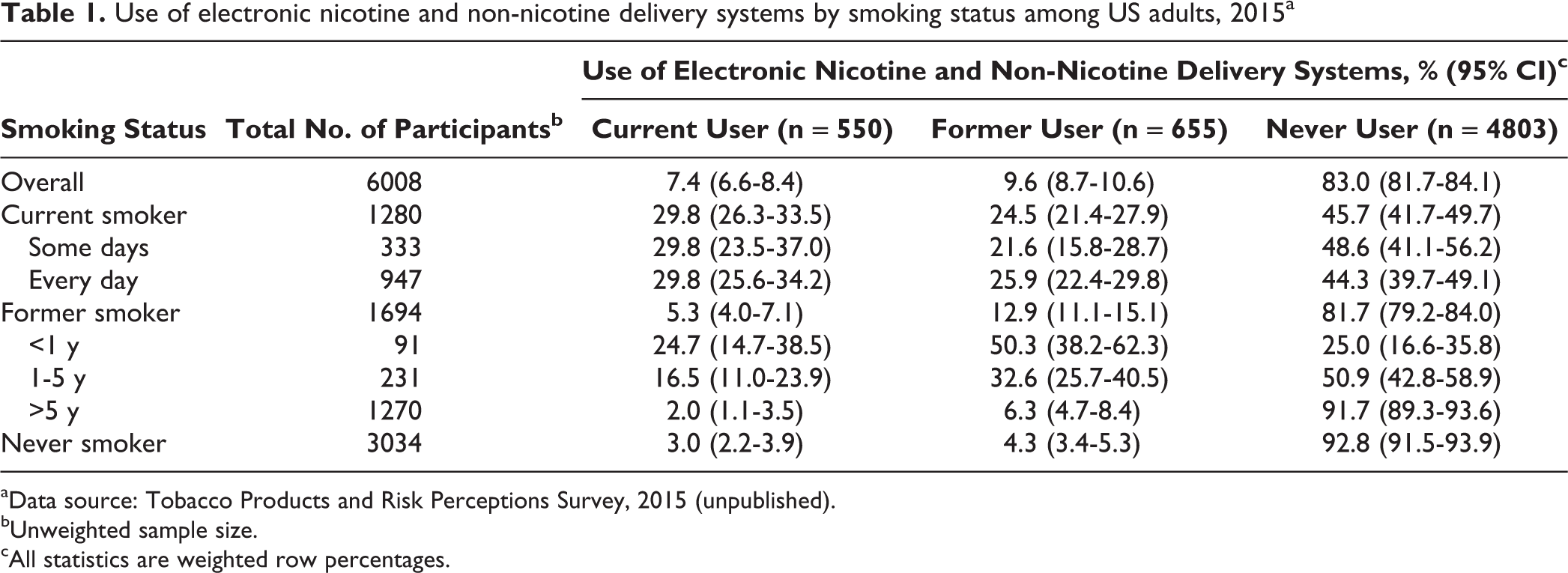
aData source: Tobacco Products and Risk Perceptions Survey, 2015 (unpublished).
bUnweighted sample size.
cAll statistics are weighted row percentages.
Approximately half (52.2%; 95% CI, 46.1%-58.1%) of current ENDS/ENNDS users indicated that they used the product rarely, whereas 23.1% (95% CI, 18.1%-28.9%) reported using it daily. More than half (56.9%; 95% CI, 50.6%-62.9%) of current ENDS/ENNDS users were current cigarette smokers (ie, dual users). Among these dual users, 32.1% (95% CI, 25.6%-39.3%) smoked on some days and 67.9% (95% CI, 60.7%-74.4%) smoked daily. Approximately one-fifth (20.3%; 95% CI, 15.6%-26.1%) of current ENDS/ENNDS users were former smokers and 22.8% (95% CI, 17.7%-28.8%) were never smokers. Among current ENDS/ENNDS users, never smokers were more likely than former or current smokers to report using ENDS/ENNDS rarely (75.2% vs 45.4%; odds ratio = 3.64; 95% CI, 1.87-7.10; df = 548; P < .001).
ENDS/ENNDS Use With Nicotine
Of 549 ENDS/ENNDS users, 69.2% (95% CI, 62.9%-74.8%) reported that they usually used ENDS/ENNDS with nicotine (Table 2). Of 106 (23.3%) ENDS/ENNDS users who were daily users, 102 (96.9%; 95% CI, 91.7%-98.9%) used ENDS/ENNDS with nicotine; those who used ENDS/ENNDS only some days (60.2%; 95% CI, 48.9%-70.6%) or rarely (61.0%; 95% CI, 52.0%-69.4%) were less likely than daily users to use ENDS/ENNDS with nicotine (df = 547, P < .001). Although most ENDS/ENNDS users who were current smokers (80.7%; 95% CI, 73.8%-86.1%) or former smokers (73.4%; 95% CI, 56.9%-85.3%) reported usually using ENDS/ENNDS with nicotine, only 36.9% (95% CI, 24.3%-51.7%) of never smokers reported usually using ENDS/ENNDS with nicotine (P < .001). Among former smokers who initiated ENDS/ENNDS before quitting smoking and continued to use them, 76.3% (weighted; 95% CI, 57.7%-88.4%) reported using ENDS/ENNDS with nicotine, whereas of former smokers who initiated ENDS/ENNDS after quitting smoking, 58.8% (weighted; 95% CI, 28.0%-84.0%) reported using ENDS/ENNDS with nicotine (P = .30).
Use of nicotine with electronic nicotine and non-nicotine delivery systems (ENDS/ENNDS), by frequency of ENDS/ENNDS use and smoking status, among US adult current ENDS/ENNDS users, 2015a
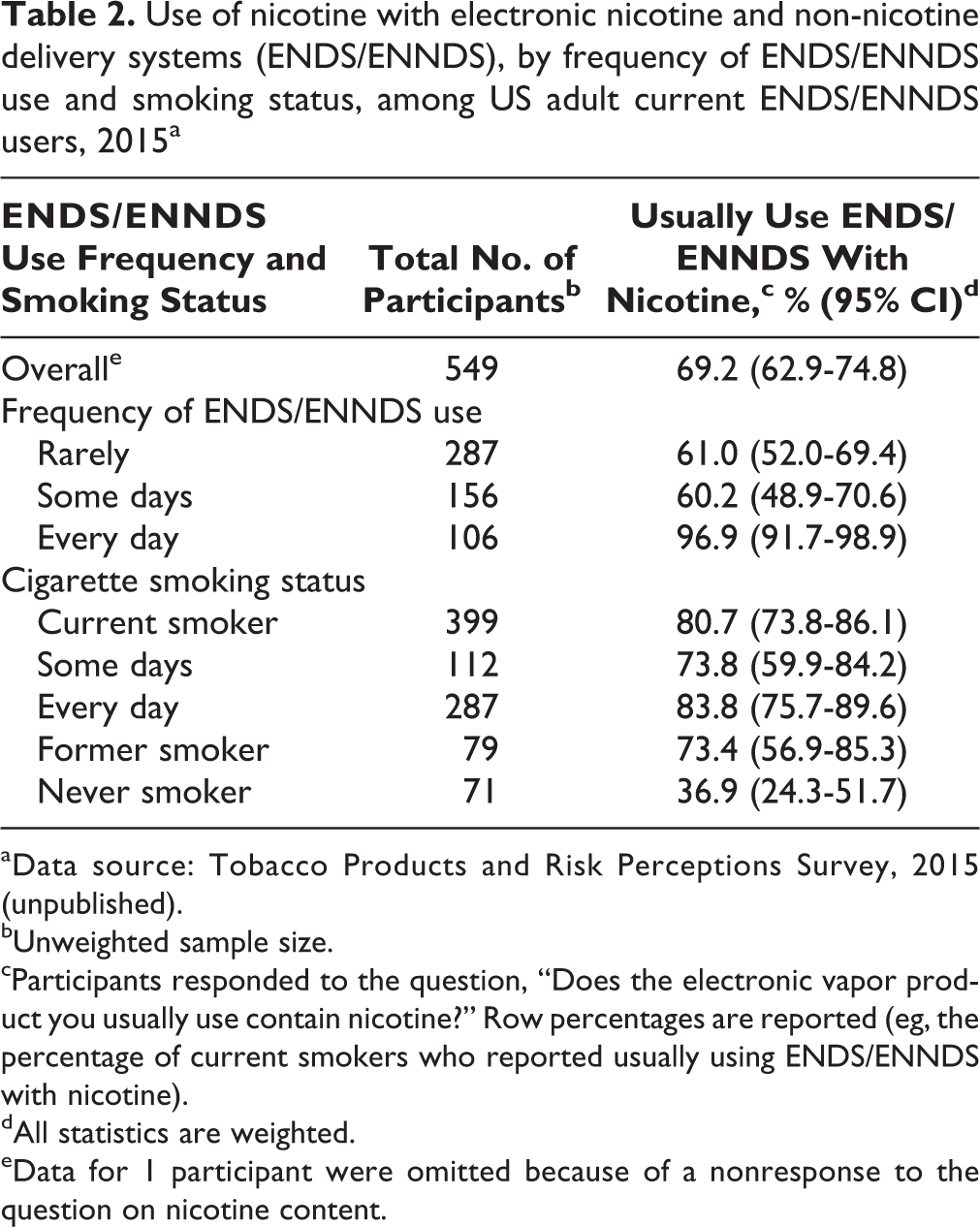
aData source: Tobacco Products and Risk Perceptions Survey, 2015 (unpublished).
bUnweighted sample size.
cParticipants responded to the question, “Does the electronic vapor product you usually use contain nicotine?” Row percentages are reported (eg, the percentage of current smokers who reported usually using ENDS/ENNDS with nicotine).
dAll statistics are weighted.
eData for 1 participant were omitted because of a nonresponse to the question on nicotine content.
Discussion
To our knowledge, this study is the first national probability study to report the prevalence of ENDS/ENNDS use with nicotine among adult current ENDS/ENNDS users. Particularly noteworthy is the finding that only 36.9% of ENDS/ENNDS users who were never smokers reported using ENDS/ENNDS with nicotine, which mirrors the low prevalence of ENDS/ENNDS use with nicotine among young people found in the Monitoring the Future study. 8 Nevertheless, most ENDS/ENNDS users in this study were current smokers, and most of these adults (80.7%) used ENDS/ENNDS containing nicotine. A recently published study of ENDS/ENNDS sales data concluded that 99% of ENDS/ENNDS products sold in 2015 contained nicotine. 11 However, that study was limited to Nielsen Company sales data collected through tracked channels, which captured an estimated 31% of the ENDS/ENNDS market in 2015. 12 The ENDS/ENNDS products sold by tracked channels are dominated by closed-system products (ie, ENDS/ENNDS where the user purchases cartridges prefilled with e-liquid) produced by the major tobacco companies, which offer limited options for non-nicotine ENDS/ENNDS. 12 Non-nicotine e-liquid is usually purchased for open-system products (ie, the user can refill the ENDS/ENNDS directly with e-liquid) from online stores or vape shops, which are not tracked by Nielsen. Our findings raise implications for current and potential future FDA regulations of ENDS/ENNDS. Users of non-nicotine ENDS/ENNDS may be affected to the extent that FDA regulations adversely affect online stores and vape shops or otherwise limit the availability or affect the price of open-ended systems. As the FDA implements regulations that are likely to affect online stores, vape shops, and the availability or price of open-ended ENDS/ENNDS, how users of non-nicotine ENDS/ENNDS respond, including switching to nicotine-containing ENDS/ENNDS, should be monitored.
These findings also have implications for estimating the population health impact of ENDS/ENNDS use. 13,14 Assessments of this impact among never smokers should account for the extent to which use involves nicotine, which this study finds to be a minority of never-smoking ENDS/ENNDS users. Accordingly, the concern that never smokers using ENDS/ENNDS will lead to eventual cigarette smoking may be diminished, although additional research is needed to confirm this finding. It is important that although current ENDS/ENNDS use among never smokers is low and a minority of those ENDS/ENNDS users reported using ENDS/ENNDS with nicotine, this study estimates that a nontrivial 1.5 million (95% CI, 786 245-2 159 846) never smokers are using ENDS/ENNDS with nicotine. Although most tobacco-related mortality and morbidity are not directly attributable to nicotine, evidence suggests that nicotine is addictive and is harmful to both the fetuses of pregnant women and adults with cardiovascular disease. 4 –7,15 –17 A recent report on ENDS/ENNDS by the US surgeon general cautions pregnant women and those women who intend to become pregnant “against using e-cigarettes to avoid unnecessary nicotine exposure to their baby.” 12 Future research should assess the extent to which pregnant women and women of childbearing age intending to become pregnant are using ENDS/ENNDS with nicotine.
This study had several limitations. The use of an internet panel may raise concerns about sample representativeness. Although sampled to be nationally representative, participants may have been conditioned by previous participation in tobacco research as a member of the GfK KnowledgePanel. However, internal research by GfK suggests minimal panel conditioning from participation in previous tobacco research and that our prevalence estimates of ENDS/ENNDS use and smoking among ENDS/ENNDS users track closely with other national estimates after adjusting for measurement differences. 18,19 An additional limitation was that the data were based on self-report, and biochemical verification of cigarette smoking and ENDS/ENNDS use was not conducted for this survey. The question used to assess current ENDS/ENNDS use included a “rarely” response option. Caution is warranted when comparing the prevalence of ENDS/ENNDS use found in this study with the prevalences found in other surveys that omitted this response option. The accuracy of self-report of ENDS/ENNDS use and nicotine content has not been verified. Previous research found that some e-liquids were inaccurately labeled as nicotine free and that many young adults, who are often experimental users, were unaware of whether or not the ENDS/ENNDS they used contained nicotine. 20,21 Related, this survey did not provide participants with a “don’t know” option for ENDS/ENNDS use with nicotine. It is possible that given their greater experience with nicotine, current and former smokers can more accurately discern whether their ENDS/ENNDS contain nicotine than can never smokers. Our results do not support a conclusion that use of nicotine vs. non-nicotine containing ENDS depends on whether former smokers first used ENDS before or after quitting smoking. However, caution is needed because the limited statistical power and the imprecision in which we were able to determine the date at which participants started using ENDS/ENNDS.
Further validation research on self-report nicotine measures for ENDS/ENNDS is warranted. Nonetheless, the findings emphasize the importance of assessing whether use of ENDS/ENNDS involves nicotine or non-nicotine e-liquid.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by grant no. P50DA036128 from the National Institutes of Health, National Institute of Drug Abuse, and the US Food and Drug Administration, Center for Tobacco Products. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the US Food and Drug Administration.
