Abstract
The current interest in Al-Mg-Sc-Zr alloy fabricated by powder bed fusion-laser beam (PBF-LB) is centered around excellent mechanical property without taking into account its corrosion behavior. In this work, gas-atomized Al-Mg-Sc-Zr alloy powder was manufactured by PBF-LB. The influence of heat treatment on microstructure, phase evolution, mechanical properties, and electrochemical corrosion behaviors were investigated and microstructure–property relationship was established to obtain high-performance alloy. The result suggests that the sample heat treated at 350°C for 4 h possesses highest mechanical properties, but is more susceptible to corrosion. When heat treatment temperature reaches 400°C, the loss of coherency for Al3(Sc, Zr) precipitates decreased the strength, but the Al6Mn phase is corroded as cathode instead of the α-Al matrix, thereby inhibiting the occurrence of pitting corrosion. The specimen heat treated at 300°C for 4 h exhibited high strength and high elongation as well as superior corrosion resistance.
Keywords
Introduction
Additive manufacturing (AM) is an advanced digital manufacturing technology involving materials, machinery, and computer science. The process of manufacturing parts is that the computer first slices the three-dimensional structure model into a number of overlapping two-dimensional cross sections and then stacks it into a solid by layering manufacturing. 1 Benefiting from the layer-by-layer building, AM can realize the integrated manufacturing of parts. In addition, it also meets the requirements of near-net-shape forming of complex structures such as lattice, thin wall, and internal hollow. 2 Comparing with the traditional processing, AM eliminates the expensive mold preparation process and produces components on demand, thus shortening the manufacturing cycle and avoiding waste. 3 Powder bed fusion-laser beam (PBF-LB), one of the popular AM processes, can produce fully dense metallic components by laser beam melting. 4
Aluminum (Al) alloys are regarded as good candidates for lightweight structural materials, so the PBF-LB Al alloy components have attracted great attention in the field of aerospace, marine, and automotive. However, the number of Al alloys available for PBF-LB process is rather limited and most of them are near-eutectic Al-Si alloys. With lower solidification shrinkage and narrower solidification temperature range, these alloys show excellent printability. 5 Researches have shown that the PBF-LB processed Al-12Si and AlSi10Mg alloy have superior mechanical properties than traditionally produced counterparts, which is owing to their fine cellular microstructures.6,7 Although PBF-LB Al-Si alloys are beneficial, the coarsening of Si particles during post-process heat treatment results in a decrease in the strength, so these alloys cannot satisfy the requirements of high-strength components. For high-strength 2xxx, 6xxx, and 7xxx series alloys, during the PBF-LB process, high melting point phases precipitate at first, which affects the solid–liquid interface and leads to the growth of dendritic grains. 8 Because of the wide solidification temperature interval, a weak semisolid region accompanied by strain exists between the liquid and those dendritic grains. 9 When the remaining liquid is insufficient to help with grain movement to accommodate stress, volumetric solidification shrinkage, and thermal contraction can produce cracks. 9 In recent years, Sc- and Zr-modified Al-Mg alloys have promoted the development of heat-treatable and PBF-processable Al alloys. 10 Spierings et al. 11 investigated the mechanical properties of commercial Al-Mg-Sc-Zr alloy (Scalmalloy®) after heat treatment at 300∼400°C for 4∼8 h and the results showed that the tensile strength was no less than 450 MPa. In order to improve the comprehensive performance of this novel alloy and extend engineering applications, the corrosion behavior of it has attracted much attention. Gu et al. 12 investigated the microstructures and corrosion behavior on different planes, showing that the XY-plane had a better corrosion resistance than that of XZ-plane in 3.5 wt.% NaCl solution. Gu et al. 13 also compared the corrosion behaviors of PBF-LB fabricated with heat-treated (325°C for 4 h) Al-Mg-Sc-Zr specimens. Nonetheless, there is still a lack of studies focusing the effect of different heat treatments on the corrosion behavior of the alloy, which is essential to comprehensively control its mechanical and corrosion properties, so that the high-strength and corrosion-resistant PBF-LB processed Al alloys can be developed by regulating heat treatment.
In our work, PBF-LB processed Al-Mg-Sc-Zr alloys were heat treated at 300, 350, and 400°C for 4 h. The scanning electron microscope (SEM), transmission electron microscope (TEM), and X-ray diffraction (XRD) were performed to characterize the evolution of the microstructure and phases during post-process heat treatment. The electrical conductivity and microhardness were analyzed to indirectly reflect the precipitation kinetics feature of the samples and the room-temperature tensile properties were performed to study the mechanical properties. Electrochemical corrosion tests including potentiodynamic polarization and electrochemical impedance spectroscopy (EIS) tests were conducted to investigate the corrosion behaviors of samples.
Experimental materials and methods
Raw material and specimen preparation
Gas-atomized powder raw material with chemical composition of Al-4.66Mg-0.54Mn-0.76Sc-0.37Zr (wt.%) was used for this study. Figure 1(a) shows the secondary electron image of the sieved powder. The illustration presents a cellular dendritic structure. Particle size distribution of the powder was analyzed by Beckman Coulter LS 13 320, with a mean particle diameter d50 = 35.28 um (Figure 1(b)). PBF-LB process was performed on CASIC ASA-260 M machine equipped with a 500 W fiber laser under argon atmosphere, and oxygen content was kept less than 0.5%. The tripe hatch strategy was adopted with 67° laser beam rotation between adjacent layers.
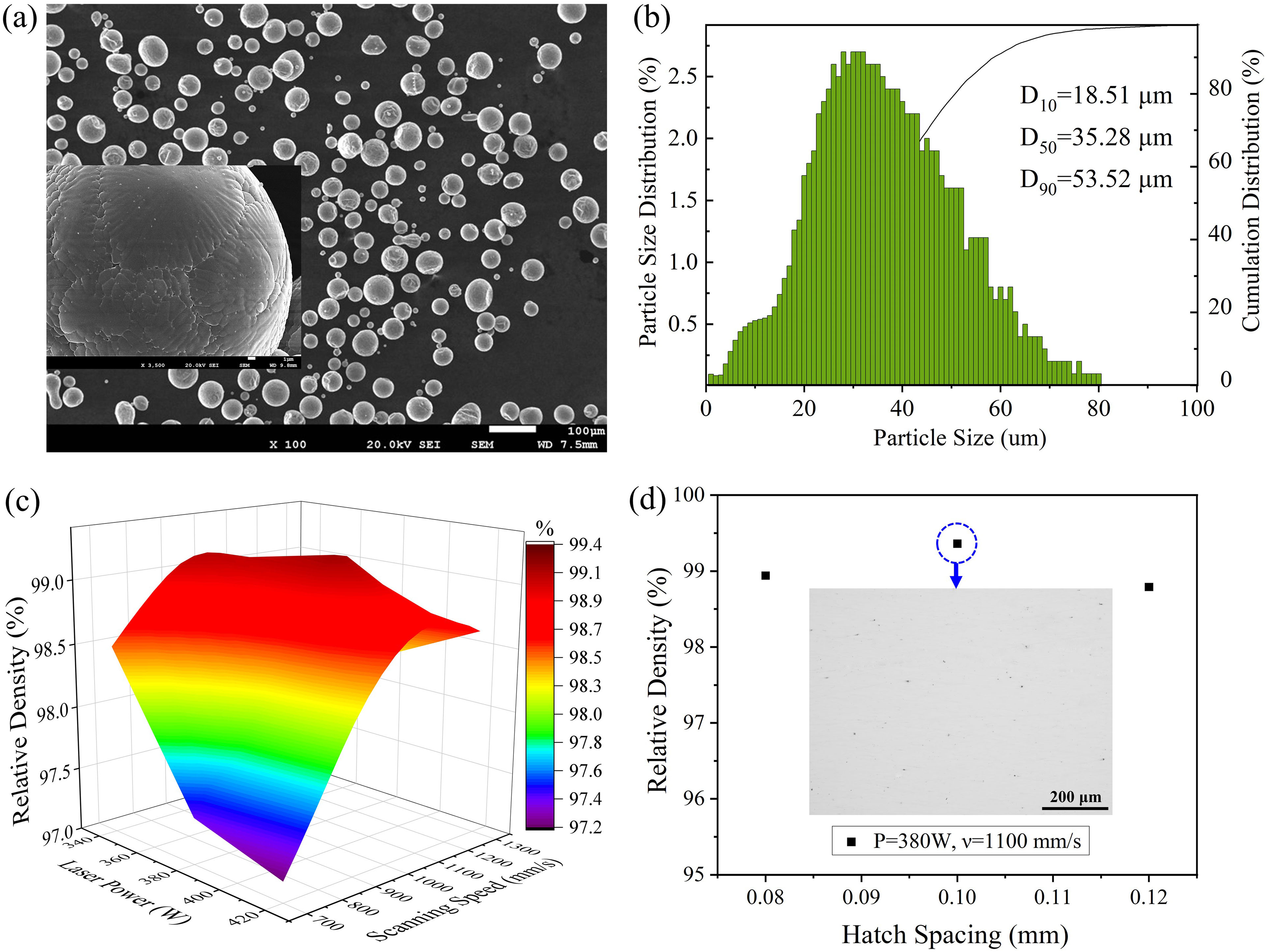
Characterization of the gas-atomized Al-Mg-Sc-Zr alloy powder (a) morphology; (b) particle size distribution; (c) relative density of samples with different laser power and scanning speed; (d) the influence of hatch spacing on relative density of samples.
Cubic samples sized of 10 × 10 × 10 mm3 were produced to optimize process parameters based on the relative density and the density of samples was measured by Archimedes method.
14
All the process parameters are as follows: laser power (
Characterization of properties
The Vickers microhardness was performed on a Buehler Wilson HV1150 hardness tester with a load of 5 kg for 15 s. Static tensile testing was carried out on a CMT4105 tester at a rate of 1 mm·min−1. The electrical conductivity was measured at room temperature by a Sigmatest 2.069 eddy current instrument. Electrochemical tests containing the EIS and potentiodynamic polarization were conducted on a Princeton PARSTAT 3000A electrochemical workstation. The tested surface of every sample was mechanically ground and polished. Corrosion process was operated in 3.5 wt.% NaCl solution at 25°C using the conventional sealed traditional three-electrode system. A clean specimen surface, a platinum foil, and a saturated calomel electrode (SCE) acted as the working electrode, the counter electrode, and the reference electrode, respectively. EIS tests were carried out under a stable open circuit potential value and the frequency ranged from 100 kHz to 10 mHz with an amplitude of 10 mV. After EIS tests, potentiodynamic polarization curves were scanned from −1.7 to −0.4 V with a scanning rate of 2 mV·s−1. All the electrochemical measurements were repeated three times to ensure data accuracy.
Microstructure observation
Phases were characterized by a Rigaku SmartLab X-ray diffractometer (XRD) with Cu Kα radiation at 40 kV, 200 mA. Surface morphologies of specimens after potentiodynamic polarization tests were observed by a Zeiss Axio Vert. A1 optical microscopy. Backscattered electron (BSE) images, electron backscattered diffraction (EBSD) images, and fracture surfaces of specimens were examined using JEOL JSM-7900F SEM. Precipitates were examined by a Talos F200X emission TEM. The TEM foils were mechanically ground down to ∼50 μm, followed by punching to 3 mm discs and then the twin-jet polishing was conducted in an electrolyte solution (30% nitric acid and 70% methanol) cooled to −30°C at a voltage of ∼20 V.
Results and discussion
Microstructure features
In Figure 2(a)–(d), EBSD inverse pole figure (IPF) maps and the corresponding grain size distribution results of the as-built specimen show the typical known bi-modal grain size distribution. 10 The fine-grained (FG) region is attribute to the heterogeneous nucleation of high melting point phases and the coarse-grain (CG) region consists of epitaxial columnar grain.15,16 Figure 2(e)–(h) shows the BSE images of as-built specimen, heat-treated samples at 300°C (HT300), 350°C (HT350), and 400°C (HT400). It can be observed that as the heat treatment temperature increases from 300 to 400°C, both the size and quantity of precipitates of samples gradually increase, especially for the FG region. In addition, no cracks or large-sized holes are observed in the SEM images, which also proves that the PBF-LB processed Al-Mg-Sc-Zr alloys has a high density.
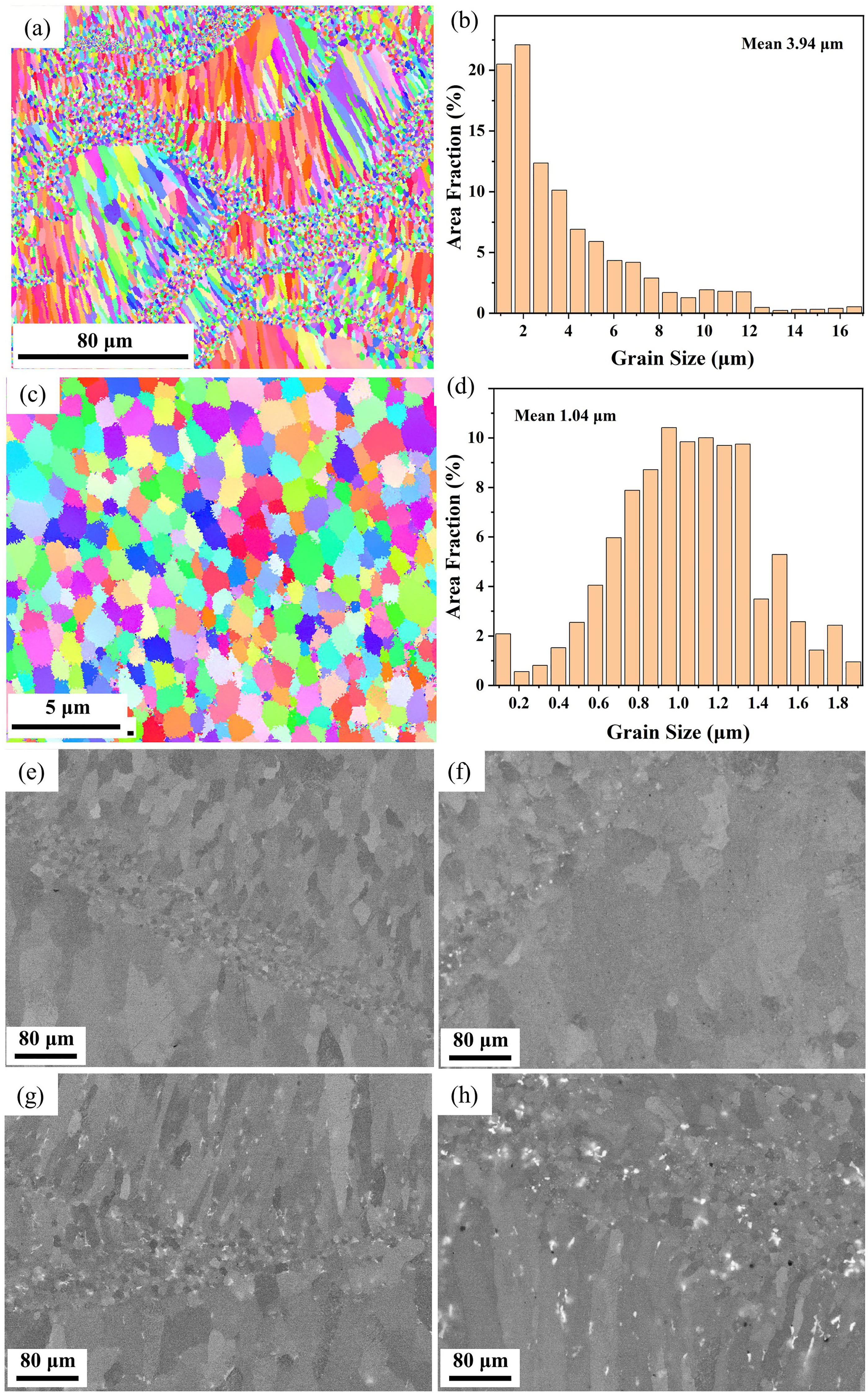
Microstructure of specimens: (a)–(b) IPF maps and the grain size distribution of as-built specimen; (c)–(d) IPF maps and the grain size distribution of FG region for as-built specimen; (e)–(h) BSE images of as-built, HT300, HT350, and HT400 specimen, respectively.
Phase analysis
XRD spectra of PBF-LB processed Al-Mg-Sc-Zr alloys with different heat treatment temperatures are depicted in Figure 3. During the high solidification rate (103–105 K·s−1) of PBF-LB process, most of the Mn, Sc, and Zr atoms solute in the Al matrix and those supersaturated solute atom have a spontaneous tendency to precipitate. As the heat treatment temperature rise, the diffusion of atoms is accelerated, thus promoting the precipitation of secondary phases. The Al3(Sc, Zr) phase, with ordered L12 structure, is coherent with the Al matrix, 17 so its diffraction peak is difficult to distinguish from the Al matrix. In the detailed drawing, it can be seen that the diffraction peak of Al6Mn gradually highlights with temperature rising.
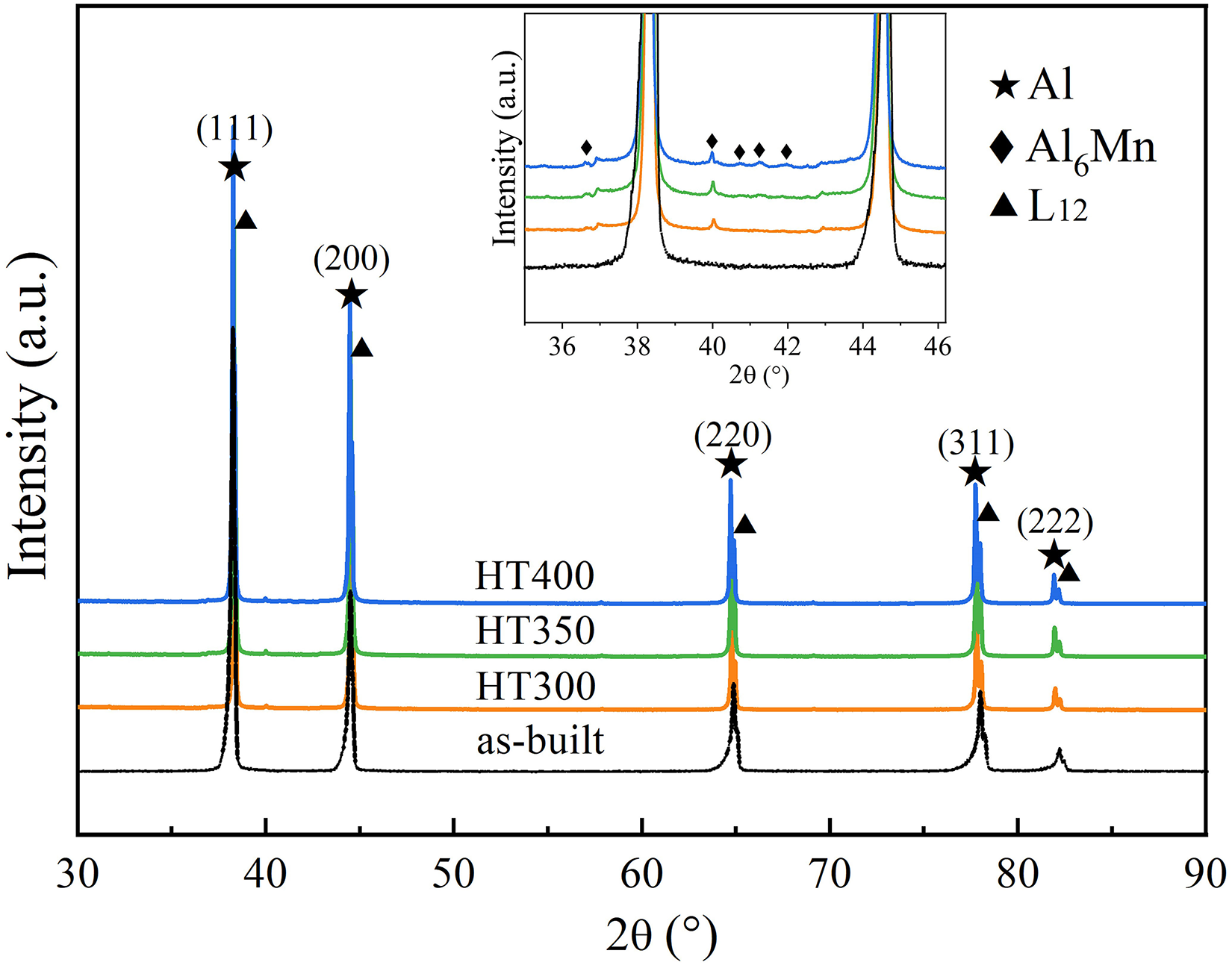
X-ray diffraction patterns of samples.
To further investigate the precipitates, TEM images of heat-treated samples were displayed in Figure 4. Secondary phases of the FG region are mainly located at grain boundaries and precipitates of CG region are mostly distributed at grain boundaries or intergranular dislocations (Figure 4(a)–(d)). It can be seen that a large number of Al-Mn particles precipitated inside the grain of HT400 specimen are shown in Figure 5(a) and the energy dispersive spectrometry (EDS) result is presented in Figure 5(d)–(i). Figure 5(g) is the enlarged image of a needle-like particle in Figure 5(a) and can be determined as Al6Mn from Figure 5(e)–(l). Figure 4(m)–(o) describes the high-resolution electron microscopy (HRTEM) image of Al3(Sc, Zr) phase, which is validated by the corresponding Fast Fourier Transform (FFT) diffraction patterns (Figure 4(p)). The coherent nano-sized Al3(Sc, Zr) phases are uniformly dispersed in the matrix and their average size increases from 3.72 to 26.09 nm when the heat treatment temperature increases from 300 to 400°C.

TEM images of precipitates: (a)–(c) FG region of HT300, HT350, and HT400 specimen, respectively; (d)–(i) CG-region of HT400 and the EDS mapping for the alloying elements of Mg, Mn, Sc, Zr, and O; (j) HAADF-TEM images of Al6Mn; (k) SAED pattern of Al6Mn; (l) HRTEM image of Al6Mn particle; (m)–(o) HRTEM image of Al3(Sc, Zr) particle of HT300, HT350, and HT400 specimen, respectively; (p) SAED pattern of Al3(Sc, Zr) phase.
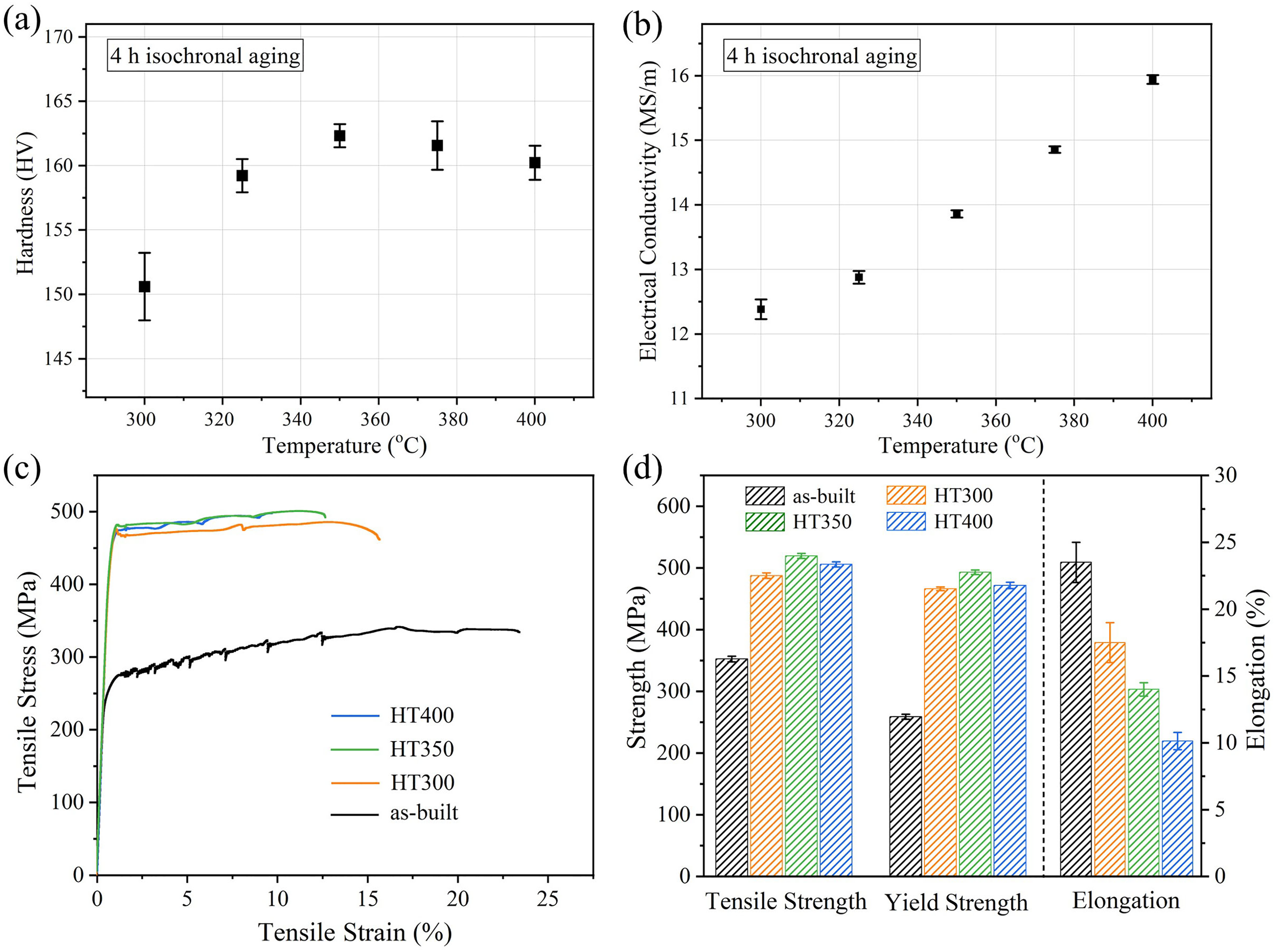
(a) Electrical conductivity; (b) Vickers microhardness; (c) stress–strain curves for samples; (d) tensile properties at room temperature.
Electrical conductivity and mechanical properties
The effect of different heat treatment temperatures on Vickers microhardness and electrical conductivity are depicted in Figure 6(a)–(b). A continuous ascent of electrical conductivity can be seen when heat treatment temperature ranges from 300 to 400°C, which is mainly related to the precipitates. 18 For the temperature–hardness curve, there is a turning point at 350°C. The microhardness increases significantly before 350°C and then decreases. The strengthening mechanism can be classified as shearing mechanism and the looping mechanism depending upon the size of the particles. 19 For the contribution to the strength, Al6Mn phase is calculated by Orowan model and the Al3(Sc, Zr) phase is calculated by the anti-phase boundary (APB) or Orowan model. 20 When the Al3(Sc, Zr) phase is smaller than the critical size, the coherent particle hinders dislocation motion by the formation of an APB and the strengthening effect is stronger, so the loss of coherency for the Al3(Sc, Zr) particle in HT400 specimen leads to the decrease of hardness. 21
(a) Electrical conductivity and Vickers microhardness; (b) tensile properties.
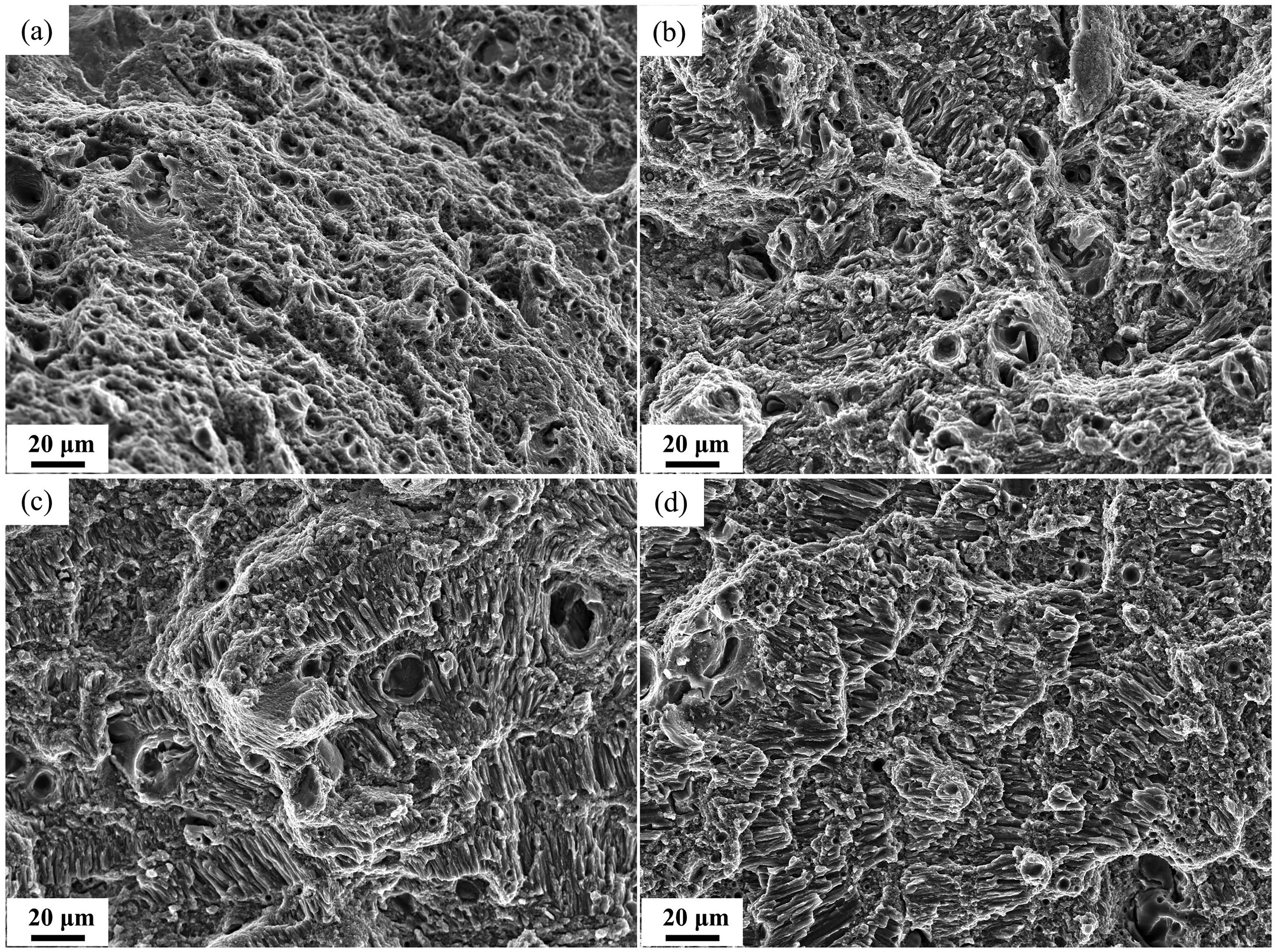
Figure 5(c)–(d) shows the stress–strain curves and results of the tensile test. After heat treatment, the tensile strength of samples increases significantly. It can be seen that the HT350 specimen also has the maximum tensile strength. The reason for strength reduction of HT400 is the loss of coherency for Al3(Sc, Zr) precipitates.20,21 The higher elongation of HT300 specimen is mainly due to the small number of small-sized precipitates, so it is easy for grain boundaries and dislocations to slip. Fracture morphology of samples is shown in Figure 6. There are many dimples of different sizes in Figure 6(a), revealing a ductile fracture mode of as-built specimen. In Figure 6(b)–(c), fracture surfaces of specimens are both consist of dimples and recognizable columnar/equiaxed grains, so the fracture mode of HT300 and HT350 specimens is a mixture of ductileness and brittleness. Almost no dimples can be observed on the fracture surface of HT400 specimen and the poor ductility is related to the intergranular fracture as shown in Figure 6(d). This is because large-sized aggregated precipitates cause stress concentration, which serves as the source of cracks and leads to intergranular fracture.
Corrosion properties
Potentiodynamic polarization and EIS measurements were performed to research the corrosion resistance of as-built and heat-treated alloys. The polarization curves in Figure 7(a) exhibits apparent passivated behavior. Pitting potential (
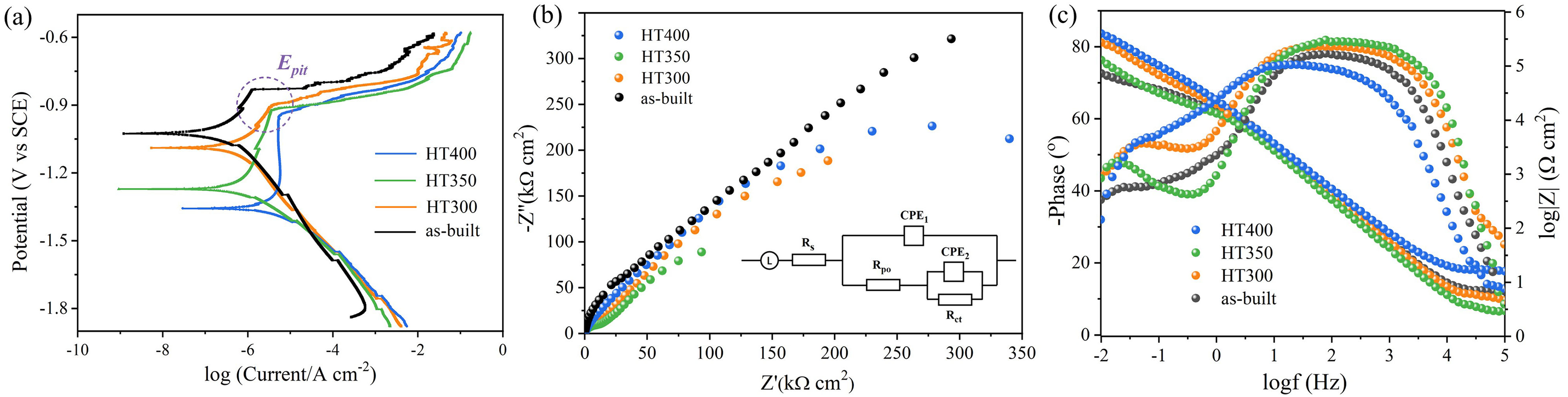
Electrochemical corrosion results: (a) potentiodynamic polarization; (b) Nyquist plots and equivalent circuit model; (c) bode plots.
Fitting results of polarization curves for as-built, HT300, HT350, and HT400 samples in 3.5 wt% NaCl solution (average value for three parallel specimens).

The corrosion current is directly proportional to the electrochemical corrosion rate, which can directly determine the level of corrosion resistance.
23
The value of
EIS analysis can evaluate the condition of electrode surface. Nyquist plots in Figure 7(b) consist of two capacitive loops deviating from semicircle in first quadrant and the corresponding phase angle and impedance bode plots are presented in Figure 7(c). There are also two peaks in the phase angle bode plot. One peak at ∼80° is originated from the formation of oxide film on the surface. The other one is at ∼50°, relating to the blocking effect of Al(OH)3 on Cl− migration.
24
The impedance moduli with a wide linear part at middle frequency reveals that the property of oxide film dominants the impedance behavior. Based on the EIS characteristic, an equivalent circuit model of Figure 7(b) was established.
Fitting results of EIS measurements for as-built, HT300, HT350, and HT400 samples in 3.5 wt% NaCl solution (average value for three parallel specimens).
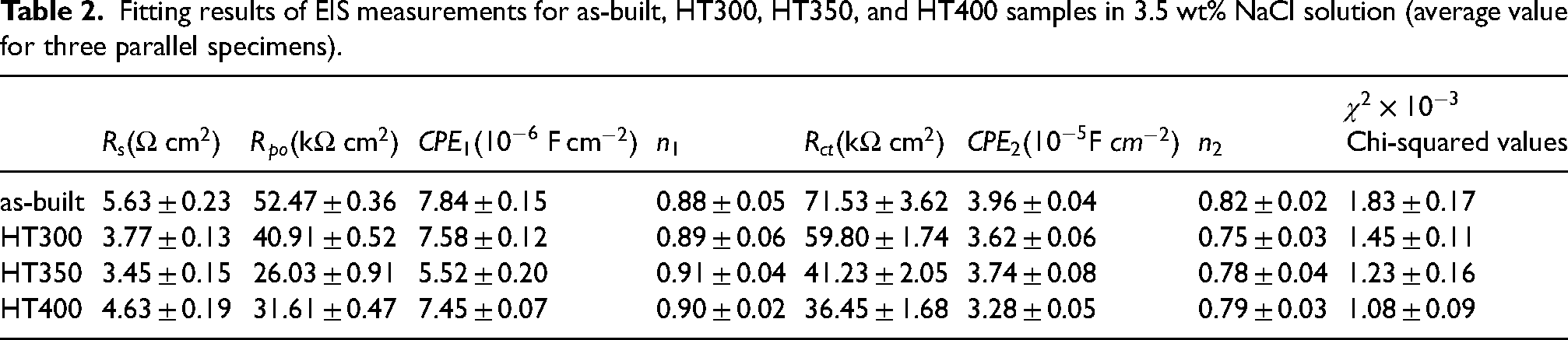
Surface morphologies of the as-built and heat-treated samples after polarization tests are presented in Figure 8. Some large holes originating from the PBF-LB process can be observed and they tend to be large corrosion pits by enrichment of Cl−. For the heat-treated samples, molten pool boundaries seem to be corroded preferably. This is because precipitates mostly distribute in the FG regions along the molten pool boundary and they have been confirmed to act as the cathodic to accelerate the corrosion of the surrounding α-Al matrix. 25 It is worth noting that the surface of HT400 specimen is slightly corroded compared with HT350 specimen, because the Al6Mn phase possesses higher potential than α-Al matrix 26 and can moderate the corrosion reaction between Al3(Sc, Zr) phase and α-Al matrix.

Optical micrographs of samples after polarization tests in 3.5 wt.% NaCl solution (a) as-built; (b) HT300; (c) HT350; (d) HT400.
Conclusion
In this work, the effect of different heat treatment temperatures on microstructure, mechanical properties, and corrosion behavior of PBF-LB processed Al-Mg-Sc-Zr alloy was investigated. The following conclusions can be drawn:
The mechanical and corrosion properties of samples are closely related to the evolution of Al3(Sc, Zr) and Al6Mn particles, which can be tuned through heat treatment.
As the heat treatment temperature increases from 300 to 400°C, both the size and quantity of those precipitates gradually increase. HT350 specimen possesses the highest tensile strength owing to more coherent Al3(Sc, Zr) particles (compared with HT300 specimen) and less small-sized Al6Mn particles (compared with HT350 specimen).
With higher potential, Al6Mn particles preferentially forms a corrosive galvanic cell with Al3(Sc, Zr) particles rather than the surrounding α-Al matrix, so the HT400 specimen shows superior corrosion resistance compared with the HT350 specimen.
The HT300 specimen shows excellent mechanical properties and corrosion resistance mainly due to the strengthening effect provided by the coherent Al3(Sc, Zr) particles and the small total number of precipitates.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Key R & D Program of China [2021YFB3701201].
