Abstract
Bi–Te-based thermoelectric materials with excellent room-temperature properties are difficult to commercialise because of their low dimensionless figure of merit (ZT). In this study, a powder was synthesised by adding an excess amount of Te to improve its thermoelectric performance at room temperature. The synthesised powder was sintered using spark plasma sintering at various temperatures to examine the effect of temperature-dependent behaviour of Te on the electrical resistivity and Seebeck coefficient. The excess Te-containing powder exhibited excellent electrical conductivity and Seebeck coefficient at high temperatures, and the thermal conductivity was significantly reduced. Bi0.4Sb1.6Te3+x (x = 2) showed a maximum room-temperature ZT of 1.57 at a sintering temperature of 450°C.
Keywords
Introduction
The present global energy shortage has led to the burning of fossil fuels in significant amounts, causing serious environmental and energy-related problems. To overcome such challenges, it is necessary to develop an inexpensive, reliable, long-lasting, and eco-friendly energy source that can cope with future energy crises. Thermoelectric materials are semiconductor materials that can reversibly convert thermal and electrical energies according to the Seebeck and Peltier effects.1–3
The dimensionless figure of merit ZT, defined as
Representative thermoelectrics include Bi–Te-based materials in the room-temperature region of up to 127°C, Pb–Te-based materials in the medium-temperature region of 127–327°C, and Si–Ge- and Fe–Si-based materials at high temperatures of 527°C and above. 5 In the case of thermoelectric power generation systems based on a thermoelectric material, power can be generated by applying a temperature difference to the material; therefore, such materials have a simple structure and hold the advantage of utilising waste heat in various temperature ranges.6,7
Among them, Bi–Te-based materials are widely used owing to their excellent characteristics at room temperature, where waste heat is most utilised. 8 Bi–Te-based materials have a trigonal structure with different properties along the a-, b-, and c-axes and constitute a five-layer atomic layer in the order of Te(1)–Bi–Te(2)–Bi along the c-axis. However, breakage easily occurs due to the weak van der Waals bonding between the Te(1)–Te(1) layers; therefore, the durability is low when fabricating a Bi2Te3 single-crystal module.9,10
To overcome such a deterioration in the mechanical properties, a method for synthesising polycrystalline materials using powder metallurgy has been developed. Thermoelectric materials manufactured using the powder method exhibit excellent mechanical properties, and their structures can be easily controlled.11,12 Additionally, in recent times, the spark plasma sintering (SPS) process used for powder synthesis was shown to be more effective than other sintering technologies in terms of generating nanosized particles and effectively scattering phonons because of the high reaction rate and high-density sintering. 13
Despite these methods, because the Seebeck coefficient and electrical conductivity have a trade-off relationship that depends on the charge density, a continuous increase in the ZT is still theoretically impossible.14,15 In addition, Te, which has a higher vapour pressure than Bi or Sb, imparts volatility to the material during sintering, thereby limiting improvement in the ZT value.
16
To address this issue, numerous studies have focused on enhancing the maximum ZT value through the manipulation of lattice thermal conductivity.17–22 Among them, only one study has revealed that by controlling the lattice thermal conductivity of
Therefore, in this study, experiments were conducted to improve the overall physical properties of thermoelectric materials. For this purpose, a powder was prepared by adding a larger amount of Te (11–21 wt%) to the
Experimental procedure
Bi (99.99%, TASCO, UK), Te (99.99%, TASCO, UK), and Sb (99.99%, TASCO, UK) were used as the initial raw materials for alloy manufacturing. The raw materials were weighed according to the composition Bi0.4Sb1.6Te3+x (x = 0, 1, and 2) based on the stoichiometric ratio calculations, placed in a quartz tube, and subsequently sealed in a vacuum (pressure approximately
The Bi0.4Sb1.6Te3+x (x = 0, 1, and 2) alloy samples were powdered by high-energy ball milling. Subsequently, the powder was ground in a planetary mill at a speed of 450 rpm for 4 h, where the ball used was a 10-mm alumina ball, and the ratio of powder to balls was 1:1. At this time, ceramic balls and zar with low reactivity were used to minimise possible contamination during milling. The occurrence of actual contamination can be determined using X-ray fluorescence (XRF) analysis, as shown in Table 1. The results indicate that the use of these Zr jars and alumina balls helps prevent any potential reactions that occur with the thermoelectric material during the milling process. The Bi0.4Sb1.6Te3+x (x = 0, 1, and 2) powders were sintered into cylindrical shapes using the SPS (WT-15.04-202110, WELLTECH) method under the following conditions: a pressure of 50 MPa was applied for 5 min in vacuum in the temperature range of 400–500°C to evaluate the physical properties according to the sintering temperature.
XRF analysis of Bi0.4Sb1.6Te3+x (x = 0, 1, and 2) before and after milling.
After sintering, changes in the phase and orientation factor according to temperature were observed using X-ray diffraction (XRD, D/Max 2500, RIGAKU) analysis, and field-emission scanning electron microscopy (FE-SEM, MIRA3 LM, TESCAN) was used to observe the microstructure of the fracture surface. The calorific value and melting point of the sample were measured through differential scanning calorimetry (DSC, Pyris-1, Perkin Elmer). The Seebeck coefficient and electrical resistivity of the prepared specimens were measured using the series four-terminal sensing equipment (ZEM-3, Ulvac-Riko) after surface polishing. The thermal conductivity of the sample was measured using the laser flash method (LFA-467, Netzsch Co). The Hall effect was evaluated via the Van der Pauw method (HMS 3000, Ecopia), wherein the carrier mobility was measured using a specimen with a diameter of 15 pi and a thickness of 0.5 mm.
Results and discussion
Figure 1(a) shows the XRD pattern of the Bi0.4Sb1.6Te3+x (x = 0, 1, and 2) powder. The analysis results confirmed that all the XRD patterns matched the standard pattern of the Bi0.5Sb1.5Te3 phase regardless of the composition. However, when an excess amount of Te was added, an additional Te phase was observed next to the main peak. This could be attributed to the addition of excess Te, and the intensity of the peak increased with increasing amount of Te.
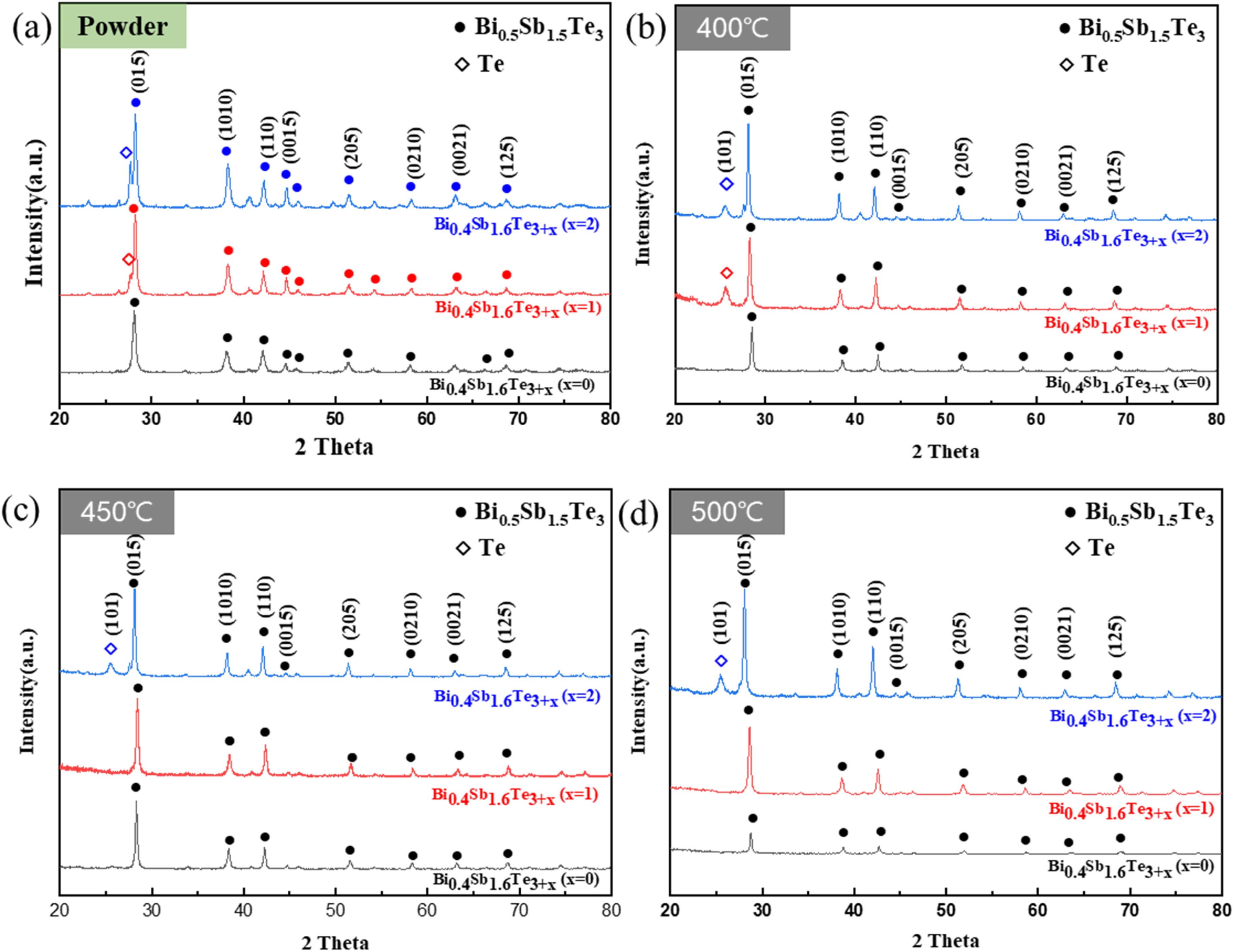
X-ray diffraction patterns of (a) Bi0.4Sb1.6Te3+x (x = 0,1, and 2) powder and Bi0.4Sb1.6Te3+x (x = 0,1, and 2) body sintered at (b) 400, (c) 450, and (d) 500°C.
Figure 1(b)–(d) shows the XRD patterns after SPS sintering of each powder at 400, 450, and 500°C. The analysis results revealed the presence of the same Bi0.5Sb1.5Te3 powder phase regardless of the sintering temperature; however, when Te was added in excess, Te existed separately as a single phase, unlike the powder. This single phase exists because the liquid phase formed during SPS is extruded to the outer surface of the graphite die and separated during rapid solidification. In addition, when x = 1, the Te single phase disappeared with increasing sintering temperature, indicating that this phenomenon occurred due to the volatility of the material, apart from the melting of Te at a high sintering temperature. Interestingly, when x = 2, it was observed that the Te single phase did not disappear even when the sintering temperature increased. This appeared to be caused by the relatively excessively added Te compared to x = 1, which can compensate for volatilisation even at high temperatures. Therefore, to confirm the existence of the Te liquid phase and single-phase peaks, Figure 2 shows the XRD results of the die state after sintering and the spilled liquid phase. The analysis revealed that the leaked liquid phase was mostly Te with a small amount of Bi0.5Sb1.5Te3 phase, consistent with the XRD analysis results. In addition, the reason the peak sizes of the same three-component and Te phases are different in the extruded liquid and bulk samples is due to the different relative amounts of the included crystal structures in each specimen.
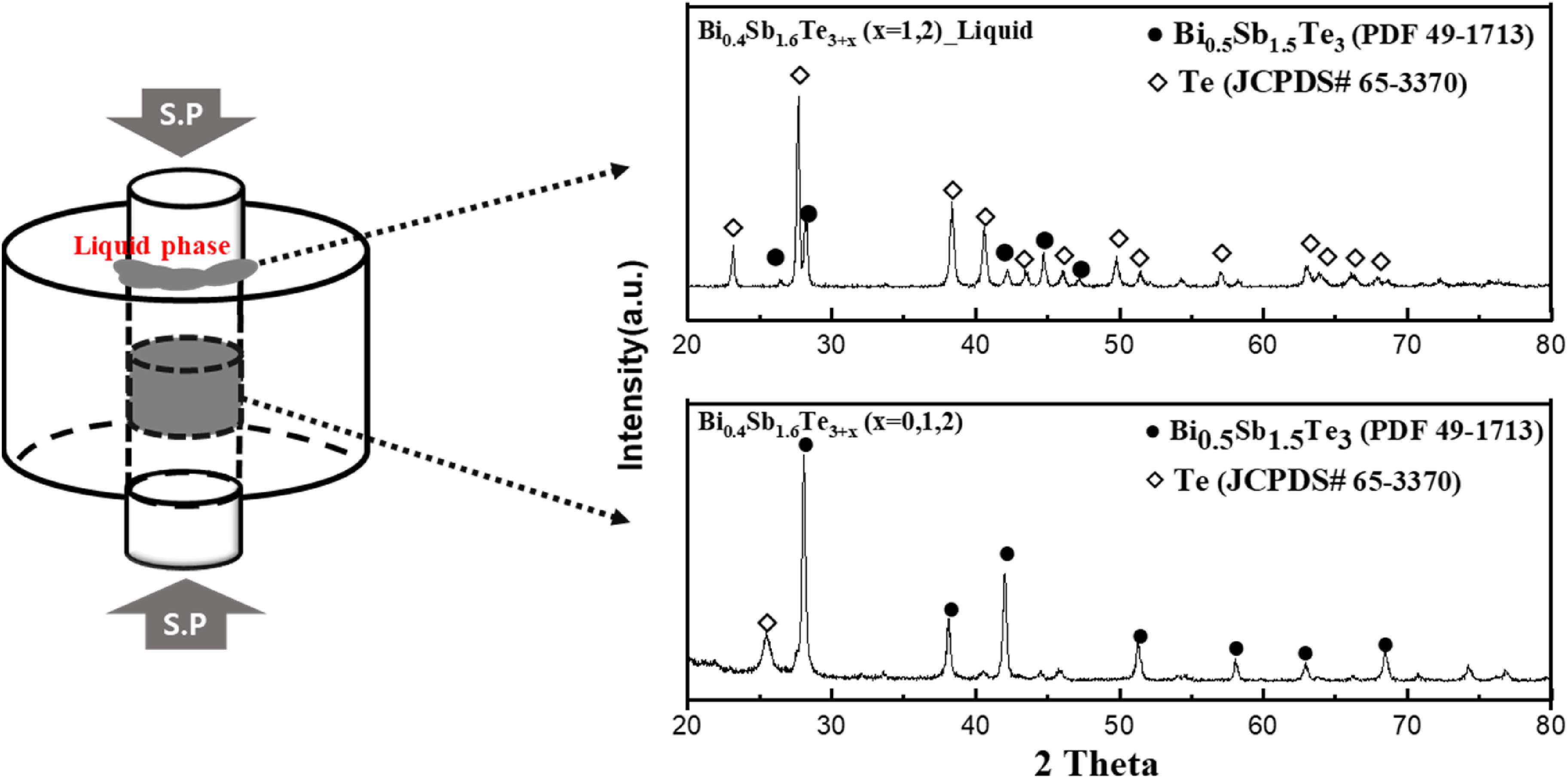
X-ray diffraction pattern of graphite die and Te liquid for Bi0.4Sb1.6Te3+x (x = 1 and 2) after the SPS process.
The density of all the sintered bodies was measured using the Archimedes method after completely drying the specimens, and the results are shown in Figure 3. The measured density was converted to percent density by calculating the ratio of the measured density to the theoretical density. Regardless of the composition, all the sintered bodies exhibited a density of over 98%.
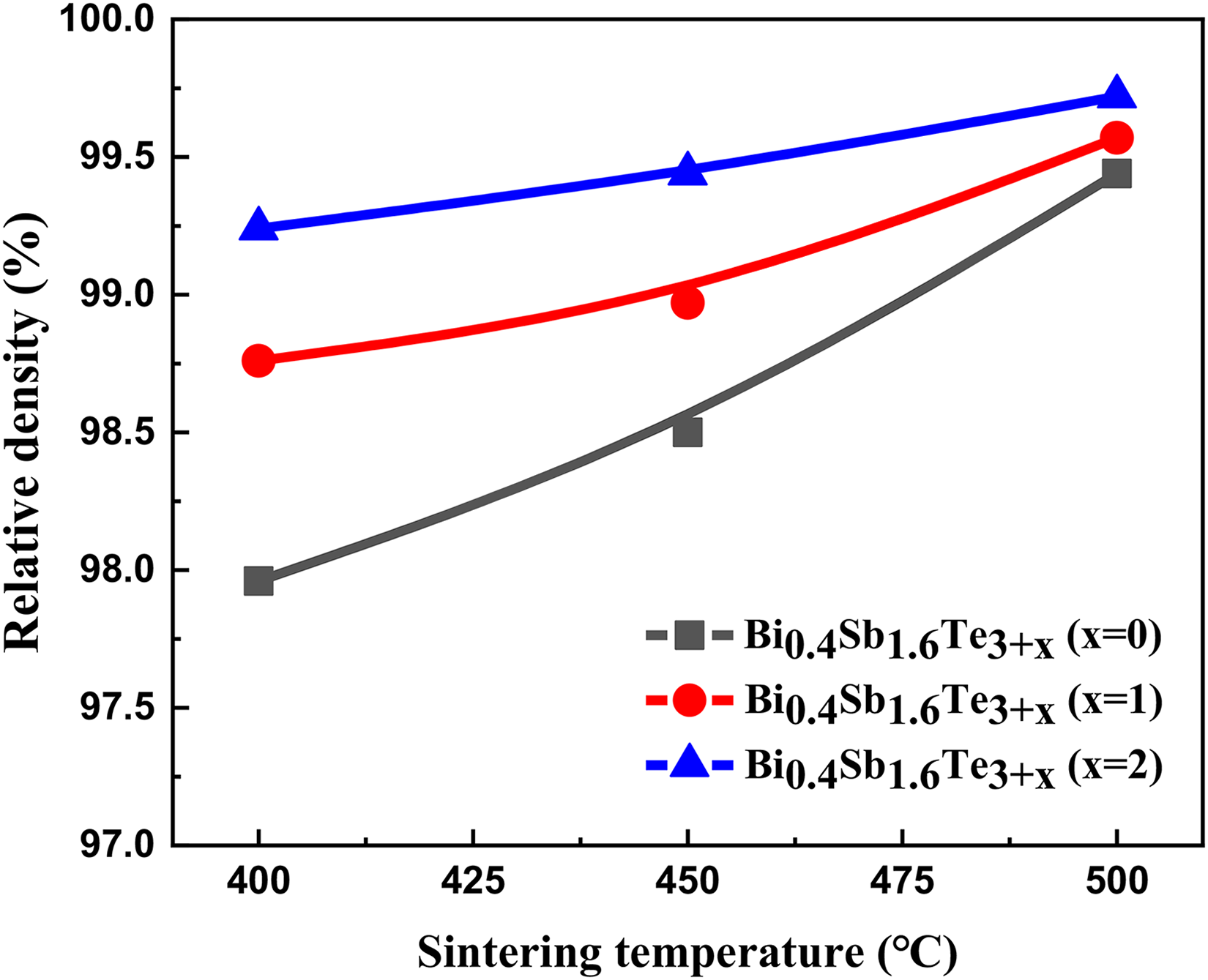
Relative density of Bi0.4Sb1.6Te3+x (x = 0,1, and 2) body sintered at various temperatures.
Figure 4(a) shows the DSC analysis results of the Bi0.4Sb1.6Te3+x (x = 0, 1, 2) sintered bodies; the analysis revealed a large displacement. The displacement occurred at approximately 420°C, and it represents the change in the calories for each sintered body. The absolute magnitude and slope of all displacements increased significantly when an excess amount of Te was added, and it was found that as the amount of Te increased, more liquid phase could be formed at the same temperature. Therefore, to investigate the effect of the increase in the amount of Te liquid phase on the structural changes in the material, we calculated the relative orientation intensity ratio using the formula below:
24

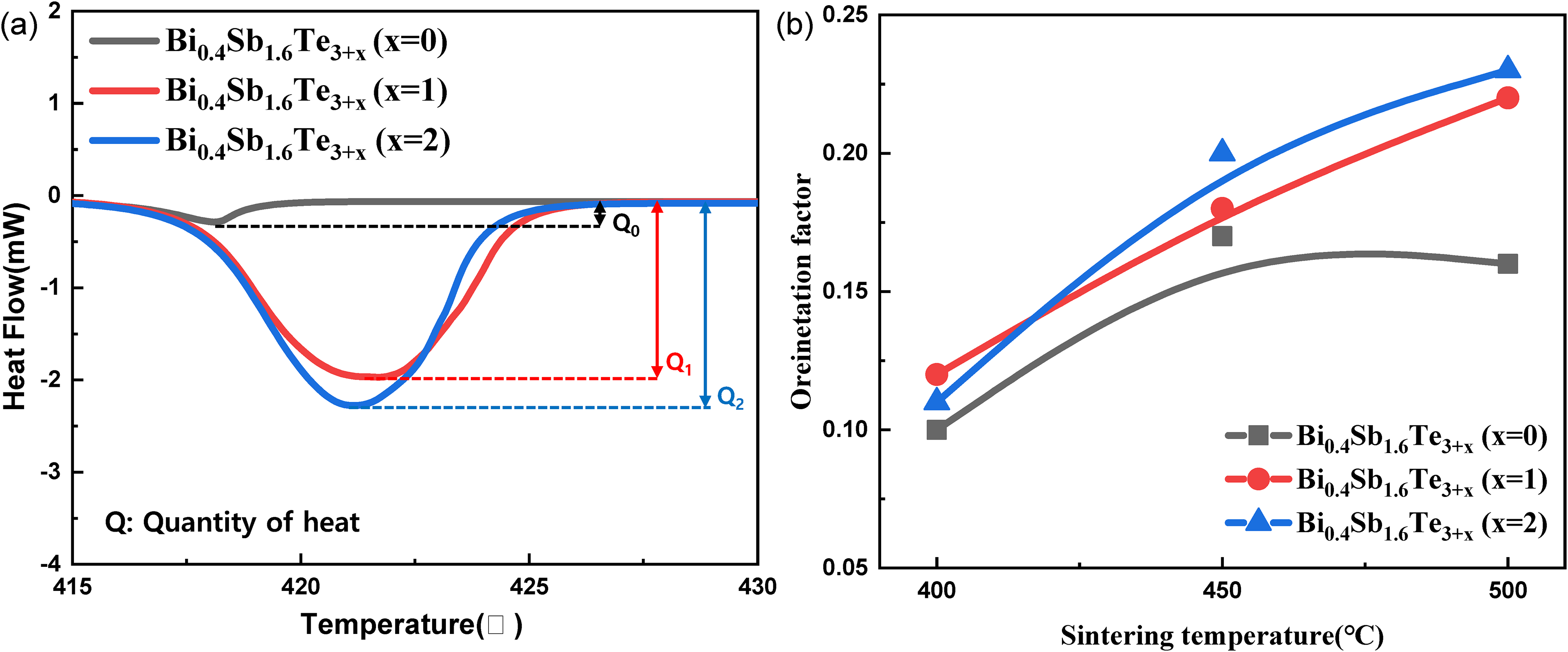
(a) Displacement of calorie as a function of temperature for Bi0.4Sb1.6Te3+x (x = 0,1, and 2), (b) orientation factor of Bi0.4Sb1.6Te3+x (x = 0,1, and 2) sintered body at various sintering temperatures.
As shown in Figure 4(b) and Table 2, the relative orientation intensity ratio gradually increased as the sintering temperature and excess Te increased, indicating that the amount of Te liquid phase has an impact on the structural changes. Specifically, the values increased from F = 0.129 to F = 0.215 with increasing sintering temperature when x = 1 and from F = 0.107 to F = 0.227 when x = 2. However, when x = 0, the value slightly decreased as the sintering temperature increased from 450 to 500 °C, suggesting that the increase in relative orientation strength was affected by the Te liquid phase. Therefore, this orientation advantage can be obtained by adding an excess of Te. In addition, an increase in the orientation intensity means an increase in the orientation of crystal grains, and the orientation of Bi2Te3-based materials is important because it affects the electrical transport properties.
Orientation intensity ratio of Bi0.4Sb1.6Te3+x (x = 0, 1, and 2) according to sintering temperature.
Figure 5 shows FE-SEM images of the fracture surface of the structure with respect to the sintering temperature and the excess amount of Te. The analysis results show that with the increase in the sintering temperature and the excess amount of Te (increase in the amount of Te added), slight grain growth and slip occurred along the cleavage surface in the structure. Consequently, the randomly arranged crystal grains were oriented in one direction. Therefore, because the pressure acts parallel to the fracture surface, the direction of the structure is clearly elongated along the a-b axis, 25 and it has a plate-like structure owing to the splitting characteristics between the Te(1)–Te(1) layers. However, when x = 0, the benefit of Te liquid phase formation is not realised at all sintering temperatures. Except for the slight grain growth with respect to the sintering temperature, the orientation is relatively random despite the layered structure of Bi2Te3.

FE-SEM images of the fractured surfaces of Bi0.4Sb1.6Te3+x (x = 0,1, and 2): surface of a sample with Te excess and no Te excess observed parallel to the pressing direction.
As shown above, an excess amount of Te melted and formed a liquid phase during sintering, which caused the crystal grains to orient along the a-b axis. Therefore, the dependence of the electrical and thermal transport properties on the temperature can be investigated in detail. In Figure 6(a)–(c), the electrical resistivity and Seebeck coefficient of each sample are plotted as a function of the temperature, where the sample without excess Te (x = 0) is used as a reference. First, the electrical resistivities of the samples with x = 1 and 2 tended to decrease with increasing sintering temperature. This is because, as seen from Figure 4, with increasing temperature, the crystal grains clearly reorient in the a-b plane, and the carrier mobility of the oriented a-b plane becomes much higher than that along the c-axis. 26 Interestingly, however, when the sintering temperature was 450°C, the electrical resistivity value at x = 1 was found to increase significantly. This is because, unlike x = 2, excess Te that did not melt acted as a lattice defect and hindered the carrier mobility (Figure 6(d)). This, in turn, is because excess Te occupies the lattice sites of Bi and Sb owing to the antisite defect effect. 27 As a result, at 450°C, the resistivity values for x = 0, 1, and 2 were 0.782, 1.04, and 0.831, respectively.
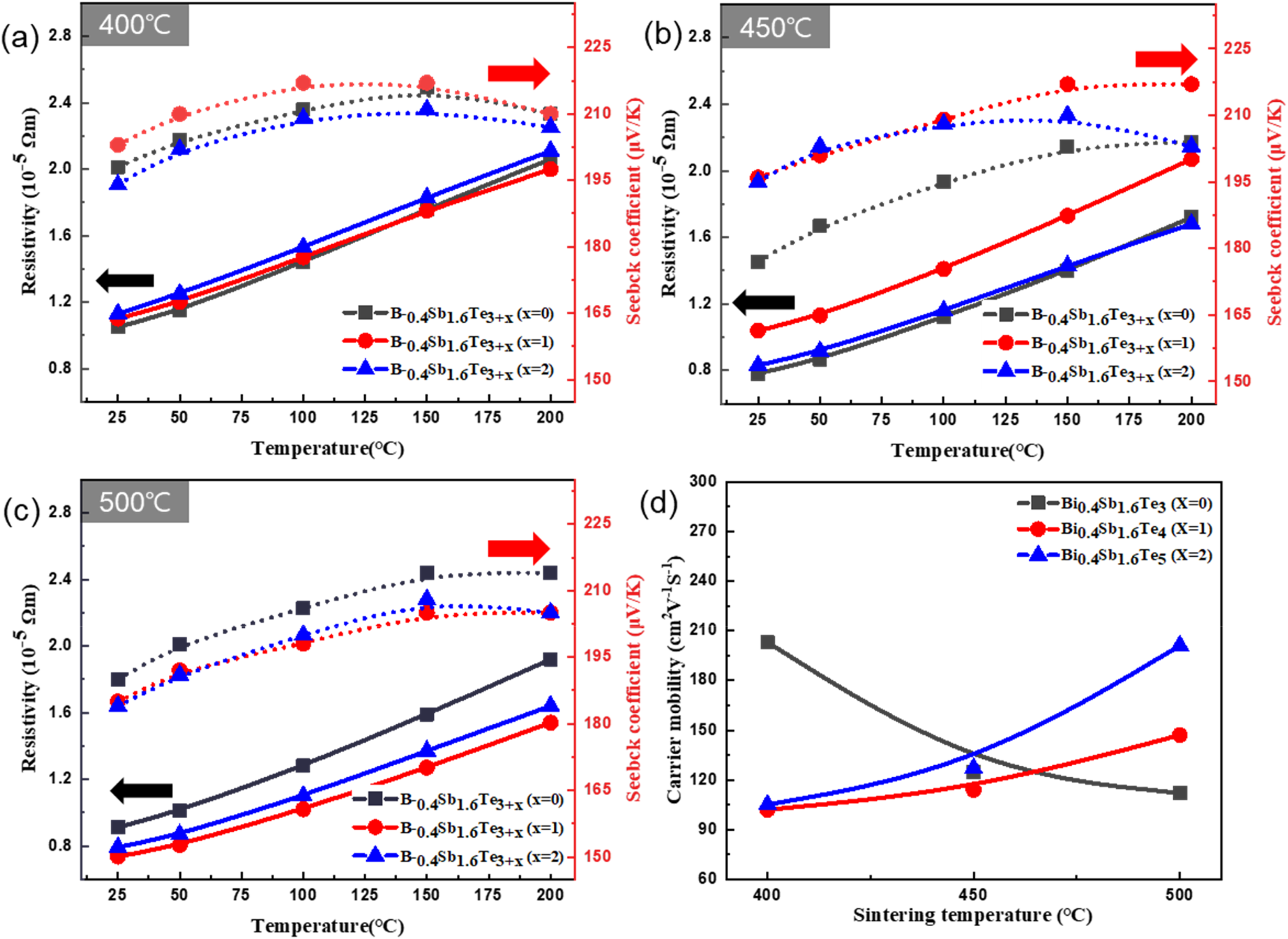
(a)–(c) Temperature dependence of electrical resistivity and Seebeck coefficient for Bi0.4Sb1.6Te3+x (x = 0,1, and 2) at various sintering temperatures (d) carrier mobility of Bi0.4Sb1.6Te3+x (x = 0,1, and 2) at various sintering temperatures.
The analysis of the Seebeck coefficient revealed that all the sintering temperatures exhibited a trade-off relationship, except for the sample with x = 2. Therefore, it is necessary to investigate why the opposite phenomenon occurs at x = 2. Generally, the Seebeck coefficient is expressed as follows:
28

Figure 7 shows the thermal conductivity k of the sample with respect to the excess amount of Te as a function of the temperature. The measured thermal conductivity decreased with increasing amount of added Te. Generally, an excess amount of Te can independently decrease the lattice thermal conductivity by forming dislocations and nanosized defects in the tissue.
19
Therefore, to quantitatively find the actual lattice thermal conductivity kL, we used the Wiedemann–Franz law (κe = σLT), where ke is the thermal conductivity of electrons, σ is the electrical conductivity, L is the Lorentz number, and T is the absolute temperature. The Lorentz number varies within the range from
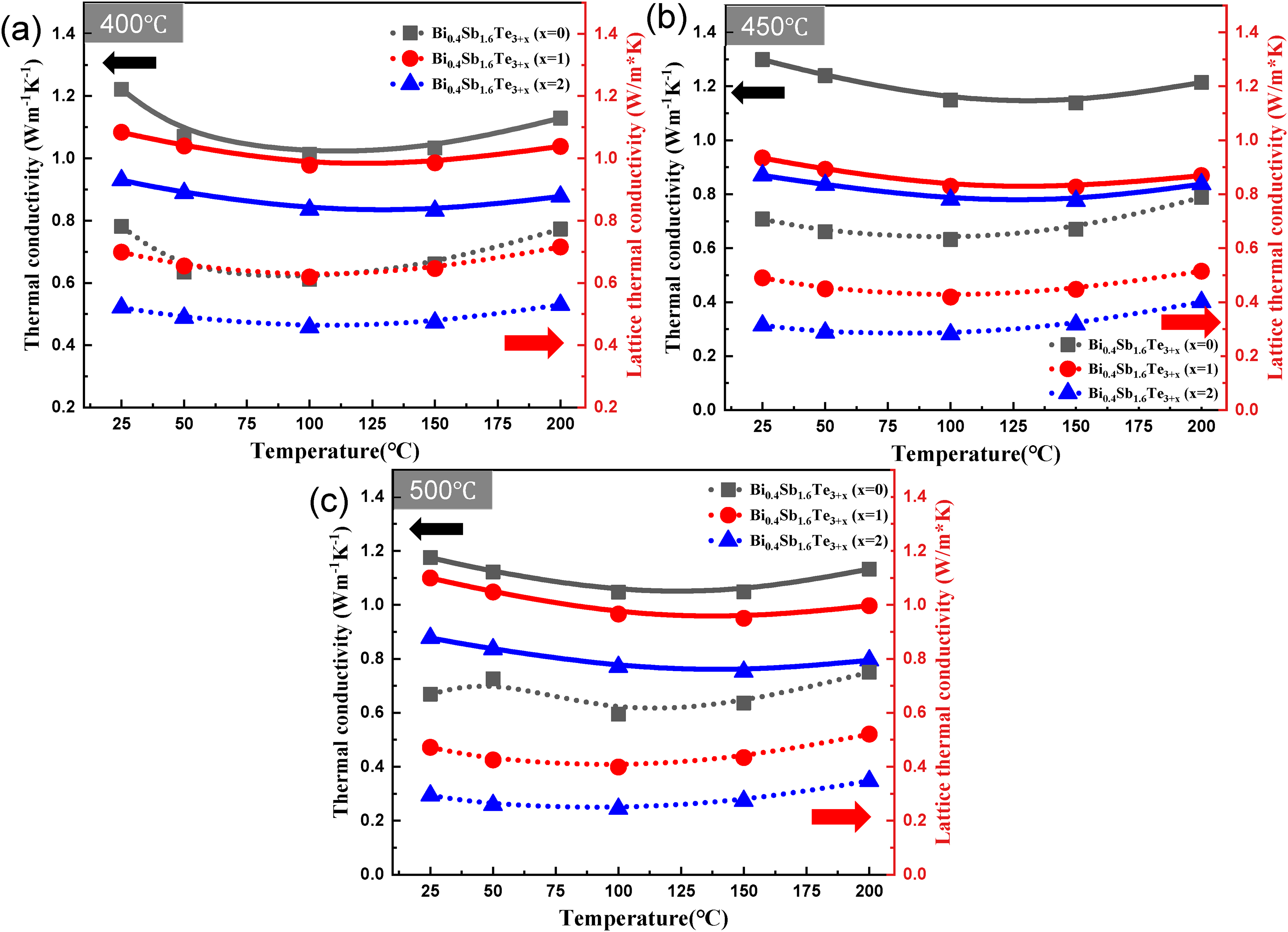
Temperature dependence of thermal conductivity and lattice thermal conductivity of Bi0.4Sb1.6Te3+x (x = 0,1, and 2) at various sintering temperatures: (a) 400, (b) 450, and (c) 500°C.
The calculated lattice thermal conductivity values at each sintering temperature were as follows: at 400°C, the values for x = 0, 1, and 2 were 0.78, 0.70, and 0.52, respectively; at 450°C, the values were 0.71, 0.49, and 0.31; and at 500°C, they were 0.69, 0.47, and 0.29. In other words, the lattice thermal conductivity appears to have lower values with increasing amounts of Te and higher sintering temperatures. It appears that more Te liquid phase is formed with increasing sintering temperature, leading to the creation of more defects and potential sites within the material. 29 Consequently, increased phonon scattering reduces the lattice thermal conductivity, resulting in reduced overall thermal conductivity. This reduced thermal conductivity impacts the improvement of the ZT value.
Figure 8 shows the dimensionless figure of merit ZT, calculated using the Seebeck coefficient, electrical resistivity, and thermal conductivity as a function of the temperature. The calculation results confirmed that the higher the amount of Te added, the better the ZT value at all sintering temperatures, which can be attributed to the significantly increased Seebeck coefficient and low thermal conductivity. In addition, because of this, it was possible to secure an excellent room-temperature ZT value. Moreover, when the sintering temperature was 450°C, the highest room-temperature ZT value of 1.57 was obtained in the specimen with x = 2, which is a significant improvement compared to the ZT value at room temperature for Bi–Te thermoelectric materials.
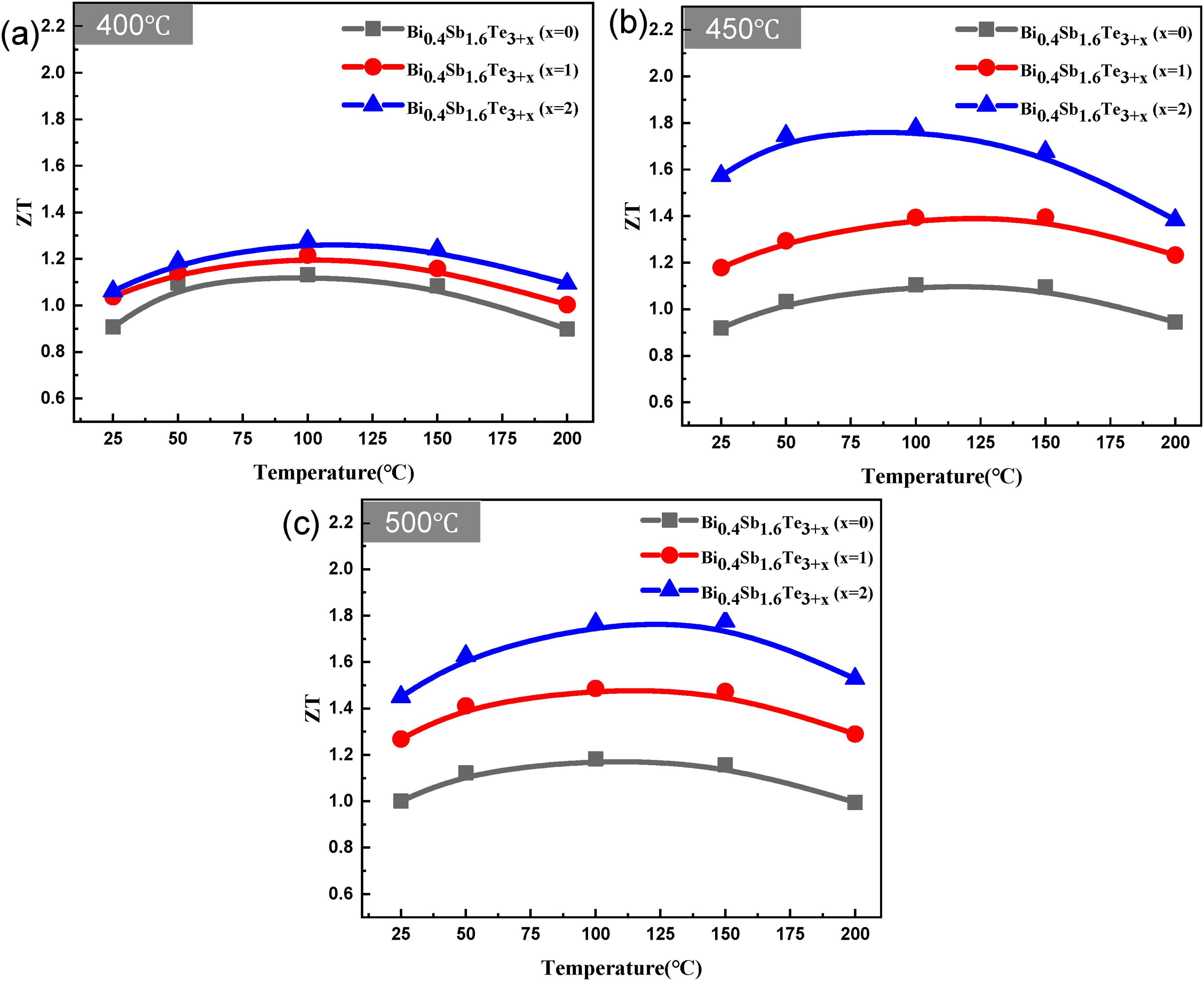
Temperature dependence of ZT values of Bi0.4Sb1.6Te3+x (x = 0,1, and 2) at various sintering temperatures: (a) 400, (b) 450, and (c) 500°C.
Conclusions
Adding an excess amount of Te to Bi0.4Sb1.6Te3 affected the improvement in the figure of merit ZT after sintering. First, as the sintering temperature increased, the orientation factor of the structure increased, and the electrical conductivity and Seebeck coefficient of the sample with excess Te improved. Moreover, the lattice thermal conductivity decreased significantly with increasing amount of Te. Thus, excess addition of Te is limited in terms of improving the thermoelectric performance at low temperatures, although it compensates for the volatilisation of Te with respect to the increase in sintering temperature and an increase in the orientation factor of the structure at a relatively high temperature. Therefore, this study showed that it could be possible to obtain excellent thermoelectric properties at room temperature using a material with a relatively high Seebeck coefficient and electrical conductivity.
The best room-temperature ZT value observed was 1.57, which was obtained for the sample with x = 2 when the sintering temperature was 450°C. The possibility of manufacturing a commercially available thermoelectric material by adding an excess amount of Te to a thermoelectric powder was confirmed.
Footnotes
Acknowledgments
This work was supported by the Fundamental Research Program of the Korea Institute of Material Science (PNK9010) and the Defense Acquisition Program Administration (DAPA) under a Korea Research Institute of Defense Technology (KRIT) planning and advancement grant (KRIT-CT-21-024).
Authorship
Hyo Jeong Kim is an MS student at Pusan National University. Min Soo Park is a senior researcher at Korea Institute of Materials Science. Gook Hyun Ha is a principal researcher at Korea Institute of Materials Science. NamHyun Kang is a professor at Pusan National University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 52079108).
