Abstract
Metal injection molding (MIM) is a net-forming technology for manufacturing miniature metal parts, which can improve the manufacturing accuracy of complex-shaped parts. In this study, aluminum stearate was used to modify the 17-4PH stainless steel powder, the binder was composed of (PMMA) and (PEG) with the volume ratio of 7:3. The effects of aluminum stearate surfactant amount on the feedstock viscosity, water debinding rate and thermal debinding shape were studied. The results showed that the coating of aluminum stearate breaks the agglomeration between the powders and decreases the viscosity of the feedstock. When the addition of aluminum stearate was 0.6wt% of the powder mass, the melt index, density and flexural modulus of the feedstock were 81.5 g/10 min, 5.44 g/cm3, and 1643Mpa, respectively, and the thermal debinding shape retention was the best of all debinded parts. However, too much aluminum stearate makes the feedstocks thicker and the green part weaker.
Introduction
Metal injection molding (MIM) is a net-forming technology that integrates traditional powder metallurgy and plastic injection molding to manufacture miniature metal parts. It is suitable for producing complex parts with high dimensional precision at a relatively low price.1,2 The process includes mixing feedstock, injection molding, debinding and sintering, and post-treatment. 3 The parts have high precision, excellent mechanical properties, large-scale, fast and efficient production, and it has been applied in many fields.4,5,6 A key problem in MIM is how to improve the shape retention or dimensional stability of the final product. This is the result of comprehensive factors, which are significantly affected not only by critical powder loading, feedstock uniformity, and rheological properties but also by the interface bonding strength between powder and binder and the mechanical strength of green parts.7,8,9
17-4PH is a martensitic precipitation hardening stainless steel with excellent physical and mechanical properties, good toughness, corrosion resistance, easy machining and welding, with excellent combination properties. 10 It is widely used in chemical, petrochemical, and aerospace industries.11,12 Mukund et al. 13 have studied the effect of powder particle size and shape difference in water-atomized 17-4PH powder on MIM parts’ critical powder loading capacity. With the increase of the volume fraction of smaller powder particles and the relative regularization of particle shape, the critical powder loading capacity increases. Ma et al. 14 mixed the feedstock of the 17-4PH powder modified by 3-Aminopropyltriethoxysilane(KH550) and 3-Glycidoxypropyltrimethoxysilane(KH560) and wax-based binder, respectively. The results show that the interaction between powders can improve the green density, and shape retention of the part after thermal debinding. Chen et al. 15 modified the surface of 17-4PH powder by using hydroxy-terminated/carboxy-terminated/epoxy hyperbranched polyester, respectively, and studied the influence of the interaction between the modified powder and the functional binder on the feedstock properties. The results show that the feedstock had a higher critical powder loading, and the green parts had a higher density, mechanical strength, and shape retention, which indicate that the interaction between carboxyl/epoxy hyperbranched polyesters can enhance the properties of MIM feedstocks and the quality of the final products.
In metal injection molding, the powder particles are small and tend to agglomerate. Therefore, to prevent the phase separation between the metal powder and the binder caused by the difference in polarity, the surface of the powder is often modified. 16 Metal powder surface modifiers mainly including coupling agents, such as silane coupling agents, titanate coupling agents, and surfactants, including stearic acid, oleic acid, and so on. Their functions are to keep the polarity of metal powder consistent with the binder system, improve powder agglomeration, enhance the powder-binder compatibility and bonding, and reduce the cracking caused by shrinkage during MIM molding. Ali et al. 17 found that adding 0.2vol% stearic acid (SA) or oleic acid in the wax-based binder system is enough to produce the effect of the surface modifier and significantly improve the fluidity of the feedstock. Momeni et al. 18 found that in the process of 4605 low-alloy steel powder injection molding, with the continuous increase of surfactant SA, the feedstock showed higher sensitivity to the shear rate. When the amount of SA was 9 vol%, the mechanical properties of the feedstock reached the best. Deng et al. 19 studied the injection molding and debinding behaviour of silane coupling agent (A151) modified ZrO2 ceramic powder. The results show that with the addition of A151, the melting index of the ZrO2 feedstock increases, the bending strength of the green part increases and the debinding rate increases.
As an effective surfactant, small organic molecules can change the polarity of the powder and reduce particle agglomeration to a certain extent 20 and enhance the interaction between the powder and the binder. 21 Among a wide variety of surfactants, SA is widely used because of its effectiveness and low cost. Chan et al. 22 studied the effect of SA on alumina injection molding. they found that SA preferentially adsorbed on the surface of alumina powder, which changed the flow behavior of the mixture, increasing the concentration of SA can reduce the uneven distribution of binder in injection molded parts, but it can also increase the possibility of bubble formation. Saberi et al. 23 discussed a SA-assisted mechanical alloying method to prepare Al-SiC composite powders with different morphologies. They found that the addition of SA can reduce particle size and improve the uniformity of the composite powders. However, the effect of ionic surfactants on metal injection molding has been rarely reported. Hwang et al. 24 improved the MgO distributions in ceramic injection-molded (CIM) alumina, among the three additives of Mg-containing precursors, Mg-stearate and Mg-acetate were added during the kneading step, and the Mg-stearate showed the highest sintered density and bending strength, this was attributed to its high molecular weight and it being able to mix with other binder components homogeneously in the liquid state, which leads to a better distribution of the MgO. Dun Wu et al. 25 prepared lead zirconate titanate-based piezoelectric ceramics with bismuth stearate as a surfactant. The results show that the coating of bismuth stearate reduces the agglomeration between ceramic powders and the viscosity of the feedstock. Yang et al. 26 used cetyltrimethylammonium bromide(CTAB) to modify the nano-Sb2O3 surface, CTAB can form a molecular layer on the nano-Sb2O3 surface, making the powder more hydrophobic. The dispersion stability of Sb2O3 powder is improved due to the electrostatic interaction with CTAB.
In this study, aluminum stearate was used as a surfactant for surface coating of stainless steel powder, and PMMA/PEG was selected as a water-soluble binder. The surfactant was added during the powder pretreatment, which could not only improve the dispersion of powder, but also reduce the viscosity of feedstock, increase the bending modulus of the green parts, and improve the shape retention of the debinded parts after thermal debinding. The optimal addition of the surfactant was determined synthetically by the rheological behavior of the feedstock. And this surfactant is suitable for manufacturing metal parts with high loading capacity and good shape retention.
Experimental and method
Materials
The 17-4PH stainless steel powder with a density of 7.89 g/cm3 was purchased from Kotailon Alloy Co., Ltd (Chengdu, China). Table 1 shows the main chemical composition of the powders. The aluminum stearate (AlSA) as surfactant was purchased from Sinopharm Chemical Co., Ltd (Shanghai, China). The polymethyl methacrylate (PMMA. High fluidity injection stage) and the polyethylene glycol (PEG. Molecular weight is 1500) as a multi-component binder system was provided by Aladdin Co., Ltd (Shanghai, China), the antioxidant (AO80) was purchased from Jiedejia New Material Technology Co., Ltd, (Qingdao, China). Aluminum stearate is regarded as the binder part. The melting point of the binder components is shown in Table 2. All the chemical reagents were used as received without any further purification.
17-4PH chemical specification and ingredient analysis.

Melting points of the binder components.
17-4PH powders coated with aluminum stearate
Firstly, AlSA with 0.4, 0.6, and 0.8wt% of the mass of 17-4PH powders was poured into a beaker containing ethanol (four times the mass of AlSA) and stirred thoroughly at 40°C until the AlSA was completely dissolved. Secondly, the 17-4PH powders were poured into a high-speed mixer (800 rpm), when the temperature of powders reached 140°C, the AlSA solution was poured into the high-speed mixer and stirred vigorously for 1 h. Finally, the powders were placed in a vacuum drying oven at 80°C for 6 h, and 17-4PH powders with AlSA surface treatment were obtained.
Feedstock preparation
The feedstock was prepared using a batch mixer (XSS-300, Shanghai Science and Technology Rubber Machinery Corporation, China, with a mixing chamber volume of 54 cm3). The binder ingredients composed with PMMA and PEG with the volume ratio of 7:3 was evenly mixed with the powders, which was poured into the chamber. The temperature of the mixing chamber and the rotor speed were set at 175°C and 50 rpm, respectively. Additionally, 0.1wt% of PEG mass of AO80 was added to prevent the oxidation of PEG under the high temperature during mixing. The preparation of the feedstock was completed when the torque value stabilized with time and the value was recorded.
Preparation and debinding of green parts
The green part was prepared using an XLB-D400 × 400 × 2 flat curing press (Shanghai Rubber Machinery No.1 Factory, China) and a dumbbell-shaped compression mold at 12 MPa and 180°C, respectively. The density of green part is measured using a direct-reading solid density meter. The density of each sample is measured three times, and the average value is the density of the green part. The green part was soaked in deionized water and kept at a constant temperature of 40°C. Every 4 hours, the green part was taken out to dry and weighed, repeated seven times to obtain the water-debinded part and calculate the degreasing rate. Then the water-debinded part was thermally debinded in an argon atmosphere using an XL-GWL04 vacuum atmosphere furnace (XL-GWL04, Luoyang Luke Kiln Co., Ltd, China), and the schedule of thermal debinding was determined by TG curves of binders components(Figure 1) as follows: (a) the green parts were heated at the uniform heating rate of 1 K min−1 from room temperature to 503 K, (b) the samples’ holding time were 30 min at the temperature of 503 K, (c) the samples were heated at the uniform heating rate of 1 K min−1 from 503 K to 593 K, (d) the samples’ holding time were 1 h at the temperature of 593 K, (e) the samples were heated at the uniform heating rate of 1.5 K min−1 from 593 to 723 K, (f) the samples were held for 2 h at the temperature of 723 K, and (g) the samples were cooled from 723 K to the room temperature naturally.
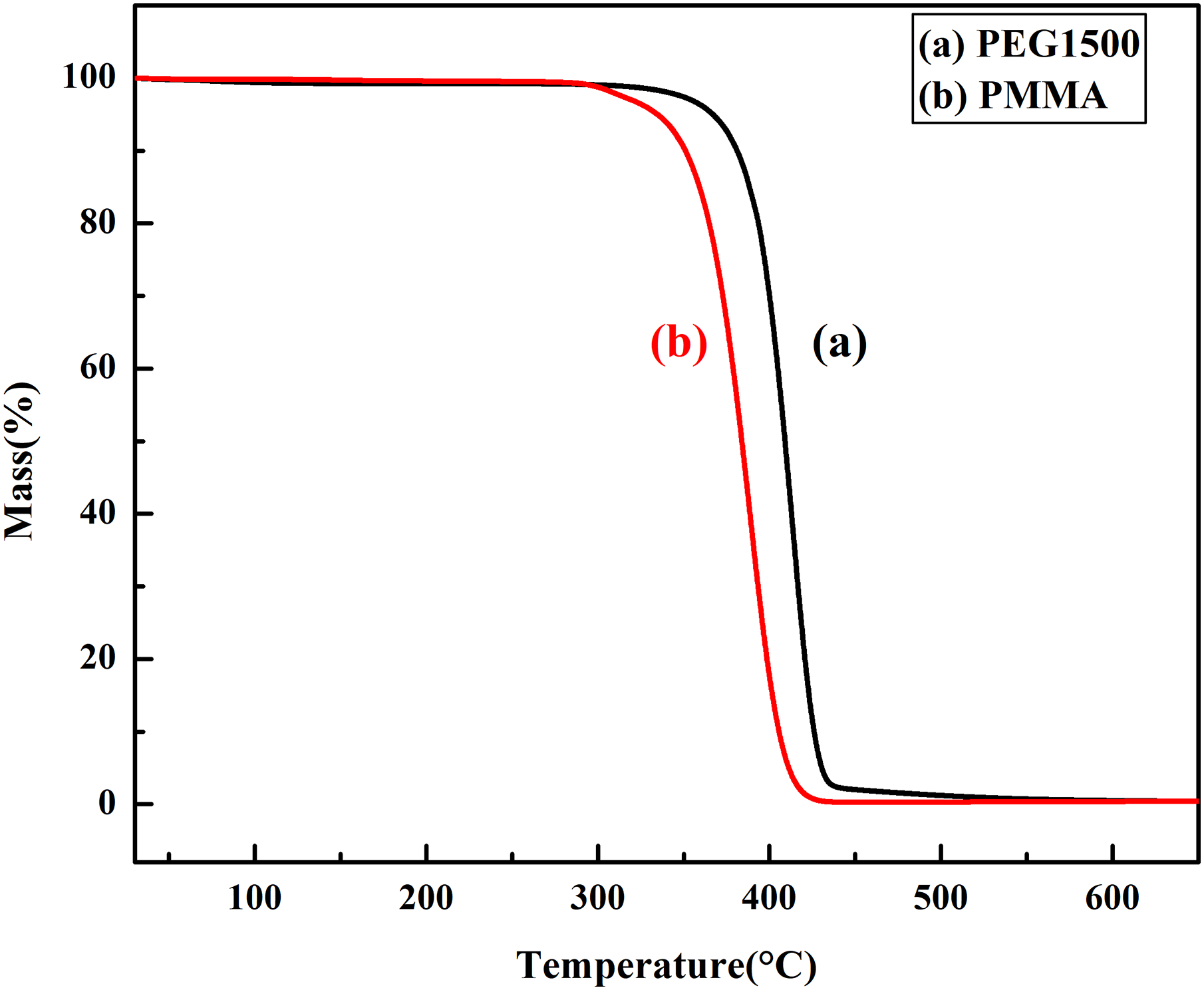
The TG curves of PMMA and PEG.
Characterization
Surface characterization of powders was performed using a Fourier infrared spectrometer (Nicolet iS50, Nicolet, USA). The microscopic morphology of powders, green parts, and debinded parts sections was observed using a thermal field emission scanning electron microscope (Gemini 500, Carl Zeiss AG, Germany) with an accelerating voltage of 2 kV. The powder’s particle size distribution was analyzed by a laser particle size analyzer (MS2000, China). The powder’s thermal weight loss was analyzed using a thermogravimetric analyzer (TGA8000, PE, US) with an argon atmosphere and a heating rate of 10°C/min. The surface potential of the powders in deionized water solvent was determined using a zeta potential meter (NanoZS90, Malvern, UK). A melt index meter (XRL-400, Mts system Co., Ltd, China) was used to test the melt index value of the feedstock at 190°C with a weight load of 5 kg. The automatic density measuring instrument (DH-300, Taizhou Ketuo Instrument Equipment Co., Ltd, China) was used to test the density of the feedstock. The feedstock shear viscosity was tested using a capillary rheometer (XLY-II, Scientific Instrument Factory, Jilin University, China). The green parts were submerged in deionized water at a constant temperature of 40°C, dried and weighed every 4 h. The debinding rate was calculated by dividing the mass loss of the billets before and after debinding by the mass of PEG in the billets. A universal electronic tensile machine (CMT4304, Shenzhen, China) was used to test the bending modulus of the part, following the ISO 178:2001 standard with a span and a test speed of 64 mm and 0.4 mm/min, respectively.
Results and discussion
Characterization of AlSA-coated powders
Figure 2 shows the infrared spectra of AlSA, pristine 17-4PH powder, and 17-4PH powder coated with AlSA. From (Figure 2(c)), AlSA has the carbonyl stretching vibration absorption peak at 1586 cm−1, which corresponds to the carbonyl peak at 1648 cm−1 in Figure 2(b). Compared with uncoated powders (Figure 2(a)), the coated powder (Figure 2(b)) also shows the stretching vibration peak of the hydroxyl group at 3490 cm−1. In addition, the peaks located at 2851 and 2920 cm−1 which corresponded to long alkyl chains (-CH, –CH2, and –CH3), can also be found in the absorption peaks of AlSA. By comparing the characteristics of infrared spectra in these samples, it is shown that AlSA is coated on the surface of powders.
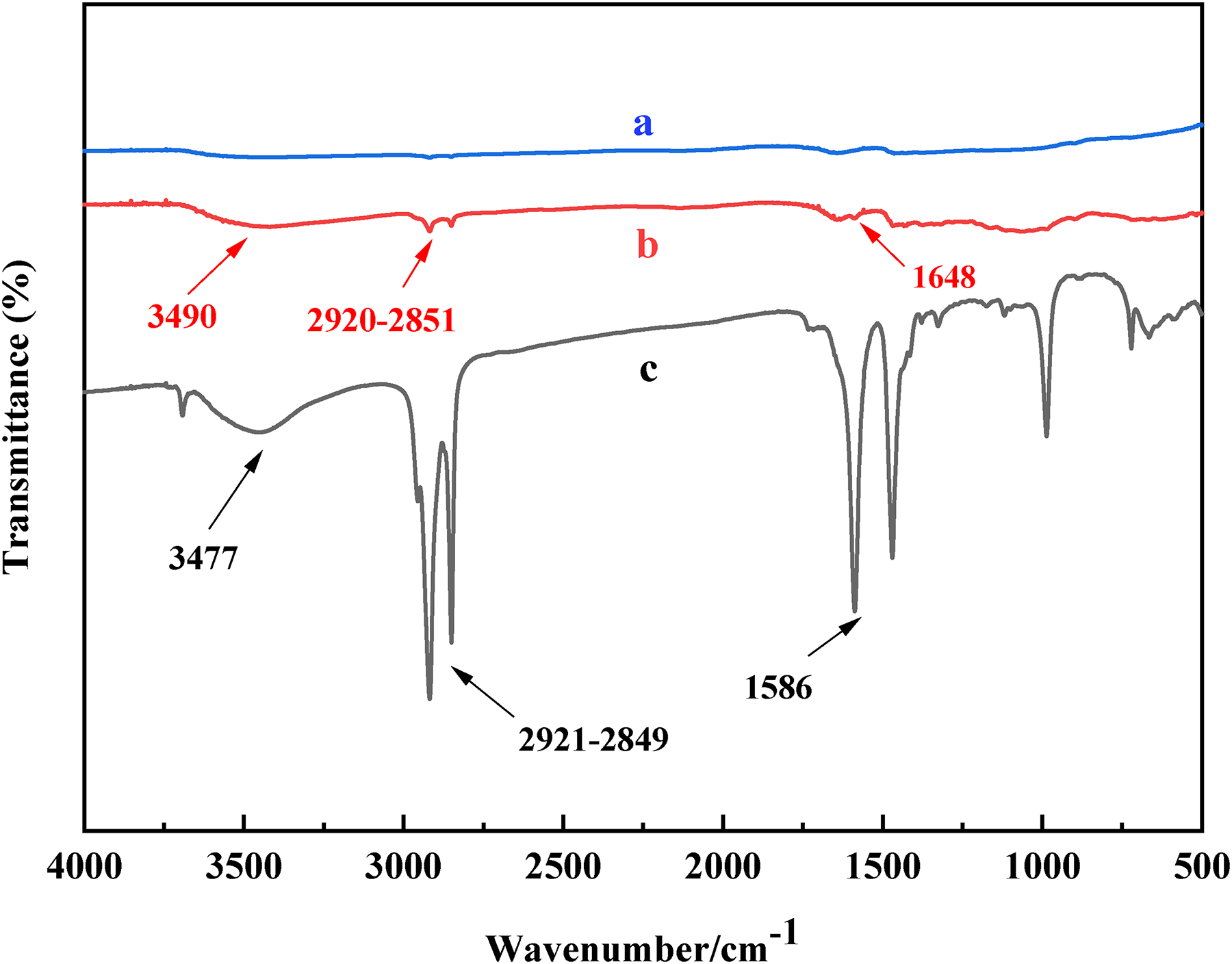
Infrared spectra of the AlSA and 17-4PH powders. (a) uncoated powder, (b) 0.6wt% AlSA coated powder, (c) AlSA.
Figure 3 shows the SEM images of 17-4PH powders before and after surface coated with AlSA. As shown in Figure 3, the surface morphology of the coated powders became smoother than that of the pristine powders. At the same time, the dispersion of the coated powders was improved, the distribution was more uniform, and the agglomeration phenomenon was significantly reduced, which further confirmed the successful coating of AlSA on the powders surface.

SEM images of the pristine powder (a) uncoated powder, (b) 0.6wt% AlSA coated powder.
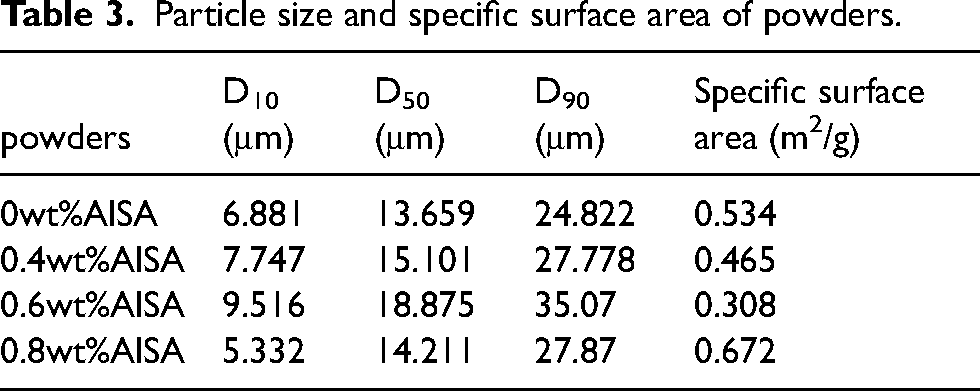
Figure 4 was the particle size distribution curves of 17-4PH powders with different AlSA content. The relevant values are listed in Table 3. It can be seen that compared to the pristine powders, the coated powders have a wider particle size distribution and a larger average particle size. The average particle size of the pristine powders was 13.659 μm, while the average particle size of the powders coated with 0.4, 0.6, and 0.8 wt% AlSA increased to 15.101, 18.875, and 14.211 μm, respectively. The possible reason is that the electrostatic repulsion between the coated powders reduces the powders aggregation and improves the dispersibility. Too little or too much coating cannot achieve a good dispersion effect. Too little coating may not cover the powders surface sufficiently, and too much coating may form multiple layers on the surface of the first layer, which may interfere with the interaction between powder particles and inhibit the increase of particle size. 27 Among the coated powders, the one with 0.6 wt% AlSA has the largest average particle size and the smallest specific surface area.
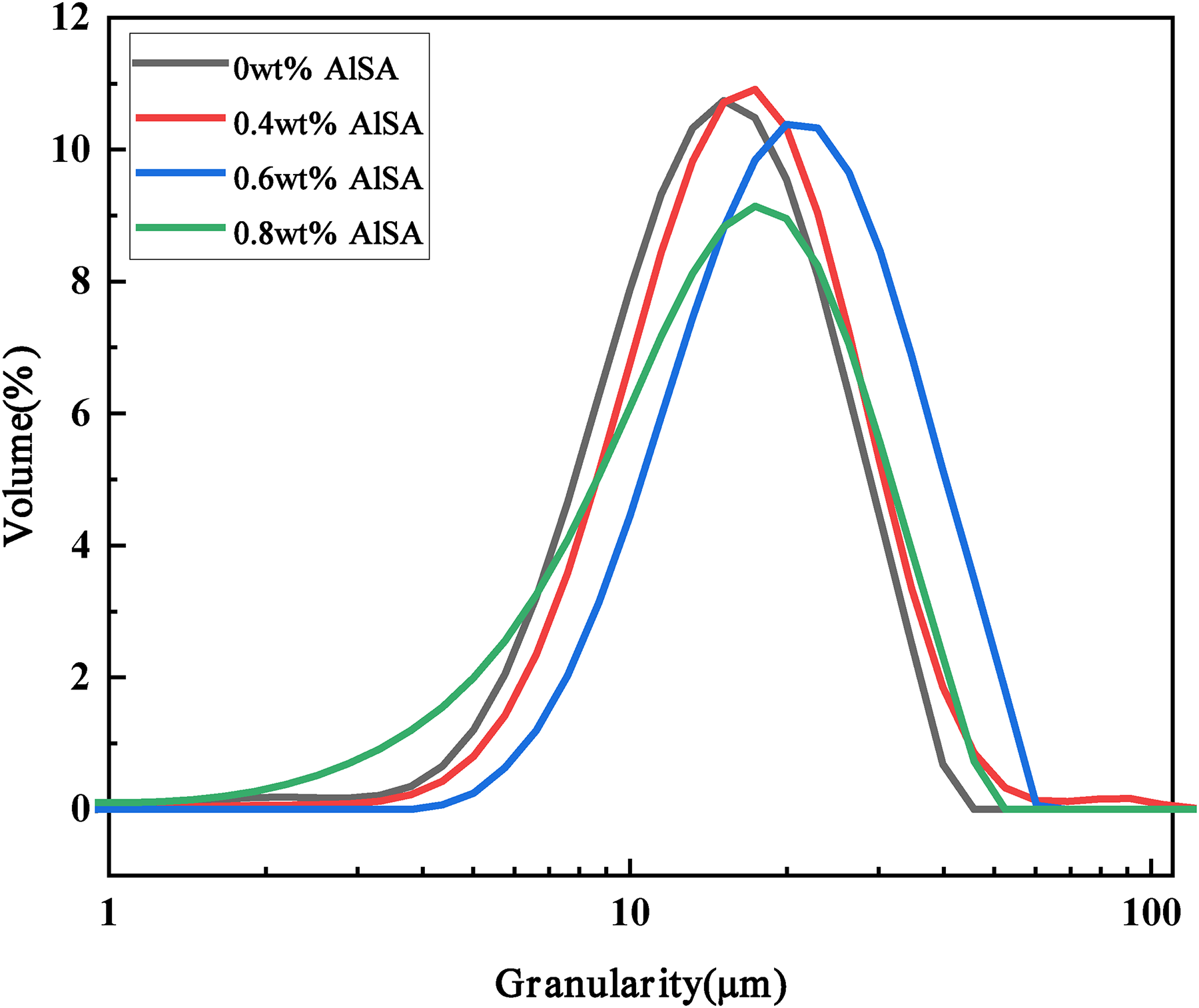
Particle size distribution curve of 17-4PH powder coated with different AlSA content.
Particle size and specific surface area of powders.
Such a situation can also be well represented by zeta potential, which is the potential difference on the particle's surface. The greater the absolute value of zeta potential, the better the stability or dispersion of the particle. 28 Table 4 lists the Zeta potential of powders in a deionized water solvent. Compared with the pristine powders, the powder coated by AlSA has a higher absolute value of zeta potential. Among the coated powders, the one with 0.6 wt% AlSA has the largest absolute zeta value of −11.9 mv, which indicates that the electrostatic interaction between the coated powders makes the powder system stable and the particles more easily dispersed. 29
The zeta potential of sample in deionized water.

Figure 5 shows the TG curve of 17-4PH powders before and after coating by AlSA. The residual mass ratio of pristine powders at 600°C is 99.03%, while the residual mass ratio of the powders coated with 0.4, 0.6, and 0.8wt% are 98.84, 98.69, and 98.31%, respectively. The surface -OH groups and the organic residues of the pristine and coated powders were eliminated at around 600°C. The amount of AlSA coated on the 17-4PH powders could be calculated by comparing the residual mass ratio between the pristine and the coated powders, which are 0.19, 0.34, and 0.72wt% for the powders coated with 0.4, 0.6, and 0.8wt% AlSA, respectively.
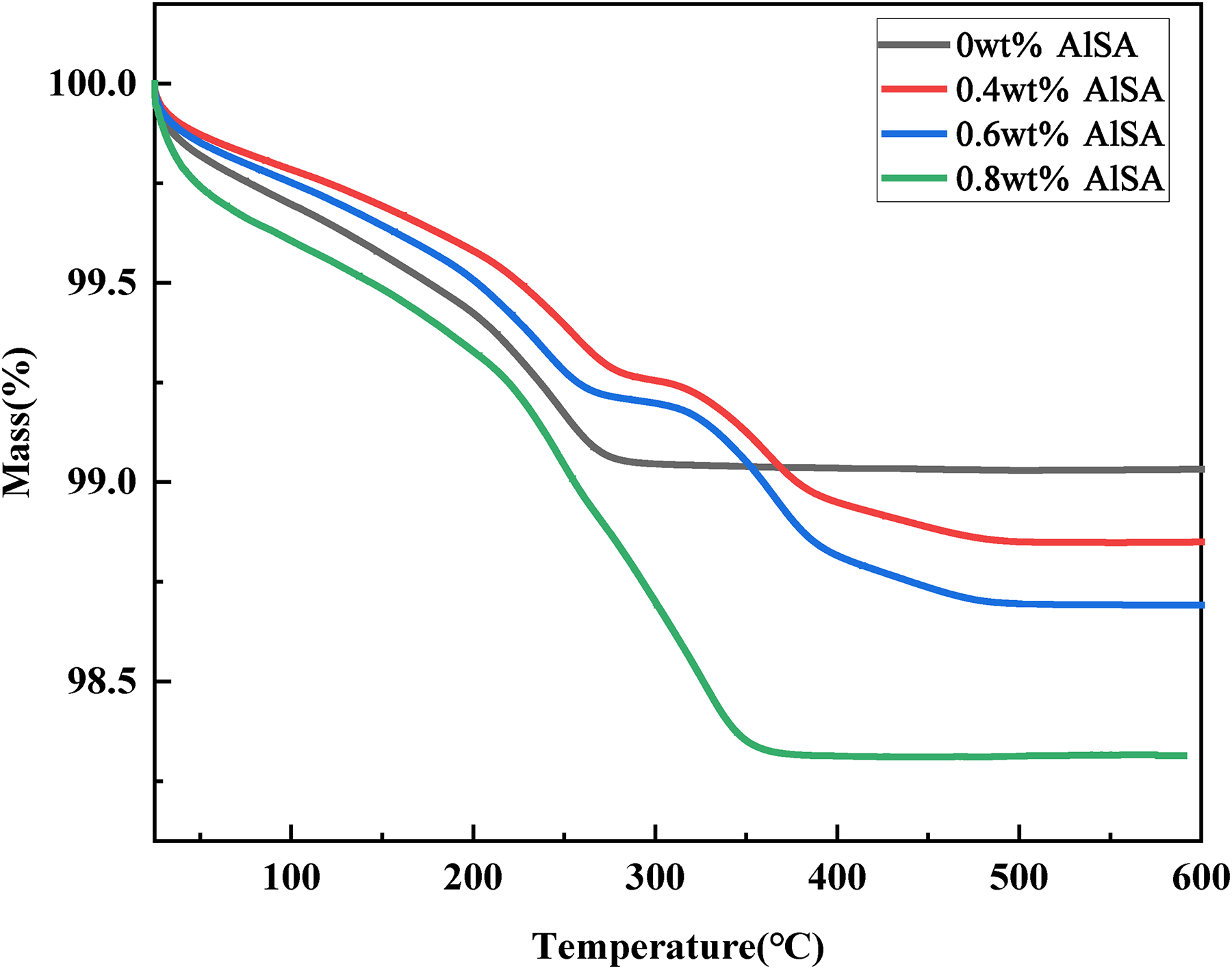
Thermogravimetric analysis of 17-4PH powders coated with different AlSA content.
Li 30 derived an adsorption model based on a monosized spherical powder, which can estimate the minimum concentration of dispersant to form a dispersant monolayer, thereby reducing viscosity, improving powder dispersibility and increasing the critical filler load. Its calculation formulae are as follows Equation (1) (WS:surfactant content(wt%), Wp: powders content(wt%), M: relative molecular weight of surfactant, NA: Avogadro number(/mol), d1: powders diameter, d2: geometric diameter of hydrophilic groups attached to particles, ρp: powders density). Considering this adsorption model, an optimized AlSA concentration can be estimated, Table 5 lists the AlSA and feedstock parameters necessary for the calculation.
Comparison of theoretical value and experimental value of AlSA concentration.


According to this adsorption model, the theoretical addition amount of AlSA to form a monomolecular layer on the powders surface is about 0.26wt% of the powders mass. The thermal analysis results of the coated powders show that the actual encapsulation rate of AlSA in 0.6wt% AlSA powders is 0.34wt%, which is slightly higher than the theoretical value of 0.26wt%. Although the model agrees well with the numerical results, it should only be applied to estimate the dispersant concentration, because the monolayer adsorption assumption is not realistic, and there may be attraction or repulsion between the dispersant and the binders were neglected. 31 In fact, the powders used in the experiments are not all spherical and their surfaces are not smooth. In addition, the powders are not mono-sized spheres and they have a definite size distribution. Most importantly, the surface of the powder often exhibits multi-layer absorption and then the content of the surfactant adsorbed on the powder surface will increase greatly. 32
Effect of AlSA coating on feedstock performance
It can be seen from Figure 6 that with the increasing temperature, the melt index values of the four types of feedstocks all increase, indicating that the flow properties of the feedstock improve with increasing temperature within a certain range. In particular, when 0.6wt% AlSA was added, the melt index value of the feedstock reached 81.5 g/10 min at 200°C. The reason may be that on the one hand, the AlSA on the surface of the powder's melts, which reduces the agglomeration between powders, so that the powders can be better wetted by the binder. On the other hand, the alkyl chains adsorbed on the powders increase the compatibility of the powders with the binder, so that the collision friction among the original powders particles is transformed into the sliding friction of the powders with the binder, thereby improving the melt flow index of the feedstock, which composed of AlSA coated stainless steel powder and binder.
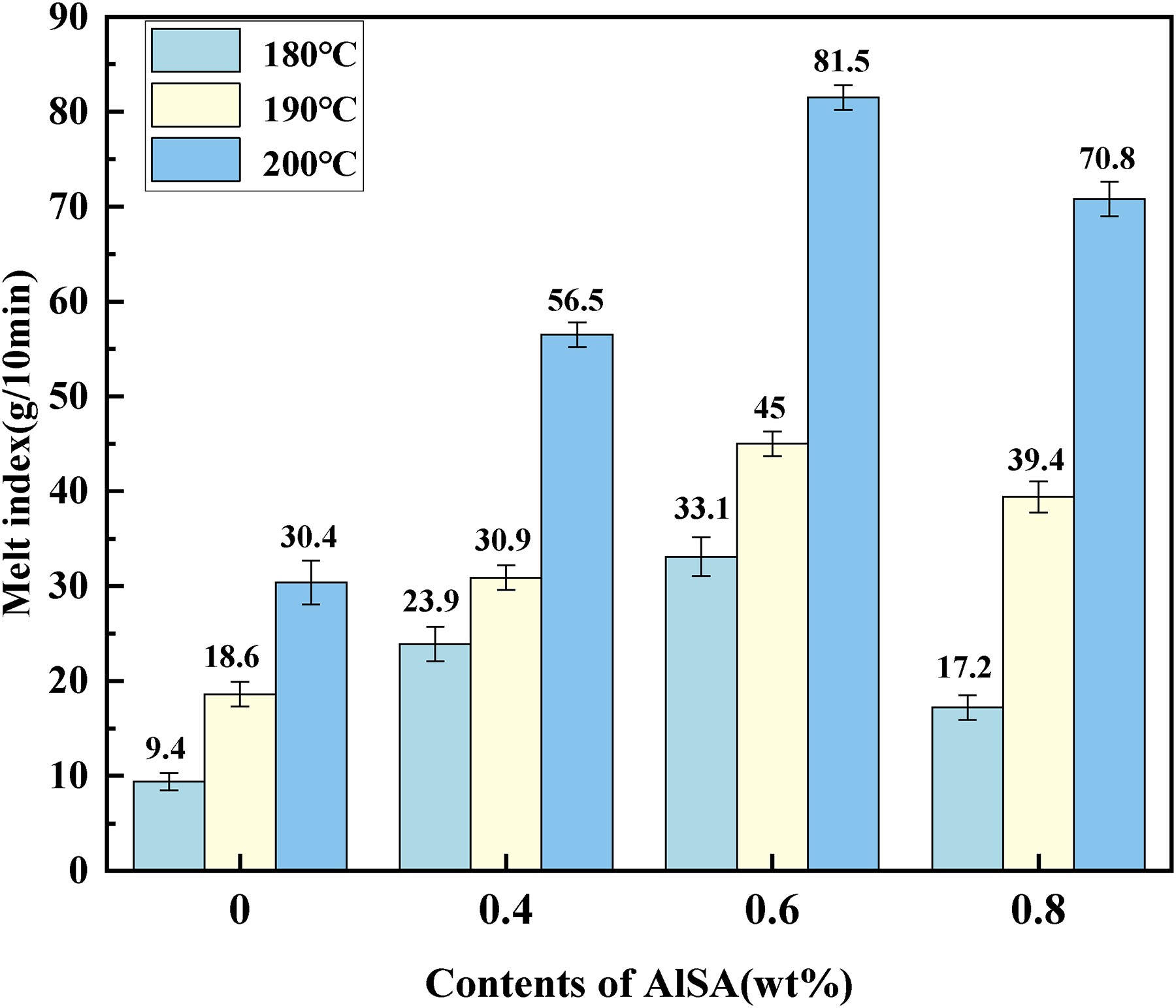
The melt flow index of feedstocks prepared with different AlSA content.
MIM feedstock usually exhibits pseudoplastic behaviour within a certain range of shear rates, and shear thinning is necessary to reduce the phase separation of the powders and the binder in the feedstock to be injected. The following Equation (2) describes the general relationship between shear viscosity and shear rate for pseudoplastic fluids at a certain temperature.

where η is the feedstock viscosity, K is the constant related to shear viscosity, γ is the shear rate, and n is the flow behaviour index. The value of n represents the sensitivity to the shear rate, and the lower the value is, the more sensitive the viscosity is to the change of shear rate. The feedstock viscosity decreases rapidly with increasing shear rate in the forming process, and higher shear sensitivity is particularly important for MIM forming.
Figure 7 shows the relationship between the shear viscosity and the shear rate of the feedstock at different temperatures from 170 to 200°C. With the increasing shear rate, the viscosity of all feedstocks decreases rapidly at each temperature, showing pseudoplastic behavior, and the viscosity of each feedstock decreases with increasing temperature from 170 to 200°C, indicating that higher temperature within a suitable range is more favorable for MIM forming.
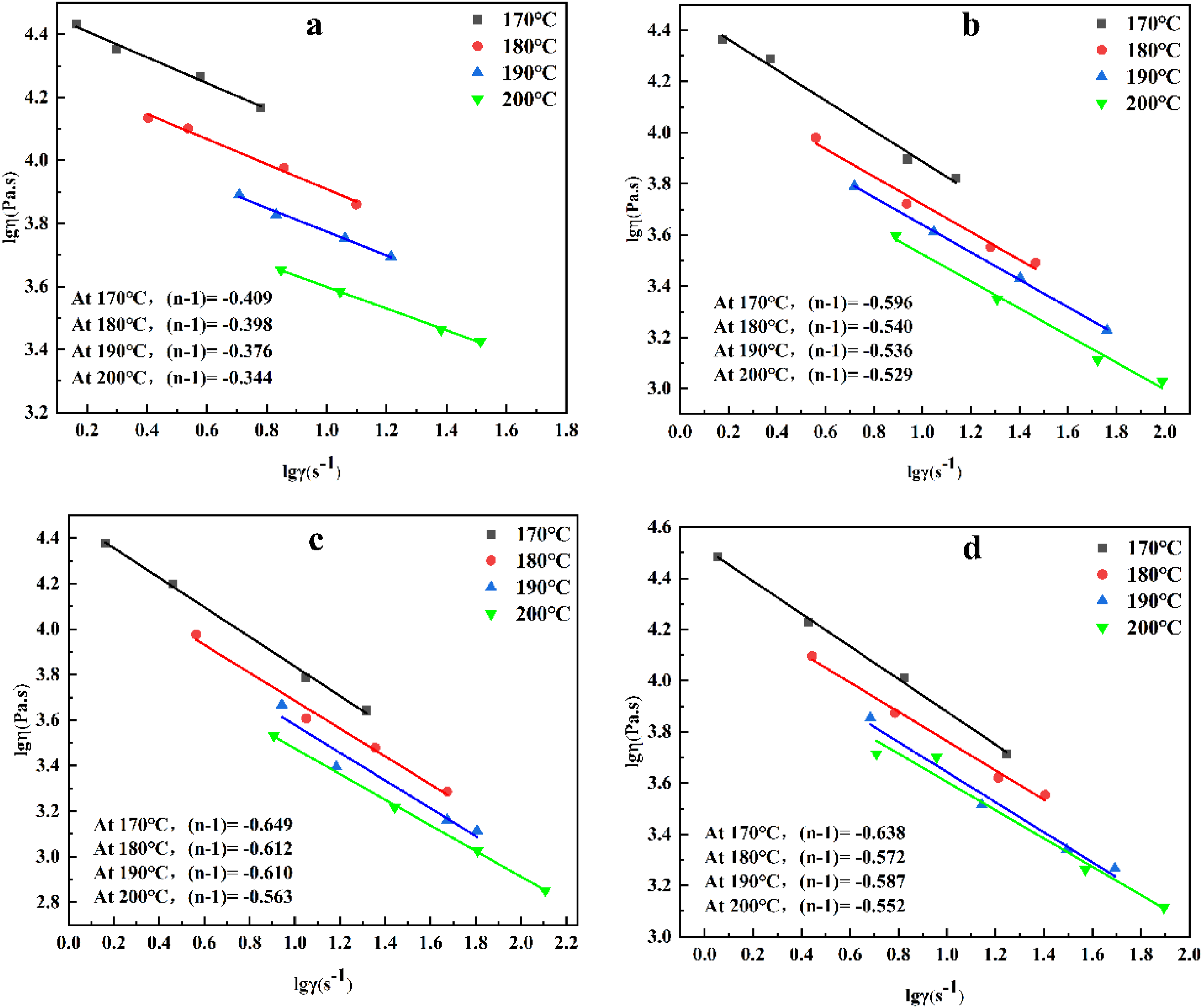
Logarithmic fit plot of feedstock shear viscosity versus shear rate for four different temperatures: (a) 0wt%AlSA, (b) 0.4wt%AlSA, (c) 0.6wt%AlSA, (d) 0.8wt%AlSA.
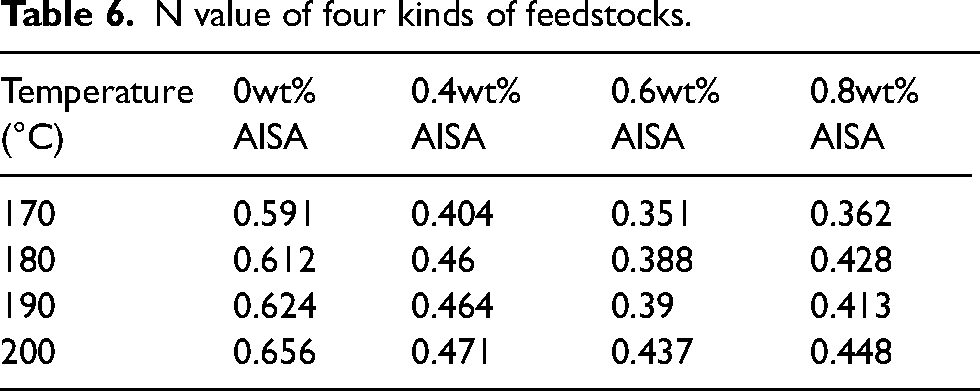
The flow behavior index of feedstock at each temperature can be derived from the slope of lgη – lgγ curve by linear fitting. From Table 6, the flow behavior indices at 170–200°C are all less than 1, indicating that the four types of feedstocks exhibit strong pseudoplastic behavior. The value of n for the feedstock prepared with pristine powders and coated powders is similar, but the viscosity of the feedstock does not decrease monotonically with increasing amount of AlSA, and the viscosity reaches a minimum when 0.6wt% AlSA is coated. This is because the excess AlSA is reabsorbed on the surface of the first layer, which causes the powders particles to attract each other and reduces their moving space, which in turn increases the viscosity. 33
N value of four kinds of feedstocks.
To further illustrate the effect of different AlSA additives on feedstock viscosity, Figure 8 shows schematic diagrams of powder/water-soluble binder with different amounts of AlSA as a surfactant. When the addition of AlSA is 0.4wt%, the surface of stainless steel particles is not entirely coated with AlSA, which does not reduce the agglomeration of the powder, as shown in Figure 8(b1) and (c1). At this time, the distance between the particles may be shortened in the mixing process, resulting in the decrease of their migration space and the increase of the feedstock's viscosity. As shown in Figure 8(b2) and (c2), when 0.6wt% AlSA is added, the surface of powder particles is covered by a monolayer of surfactant, promoting a uniform arrangement of particles, 34 and the feedstock exhibits the lowest shear viscosity.
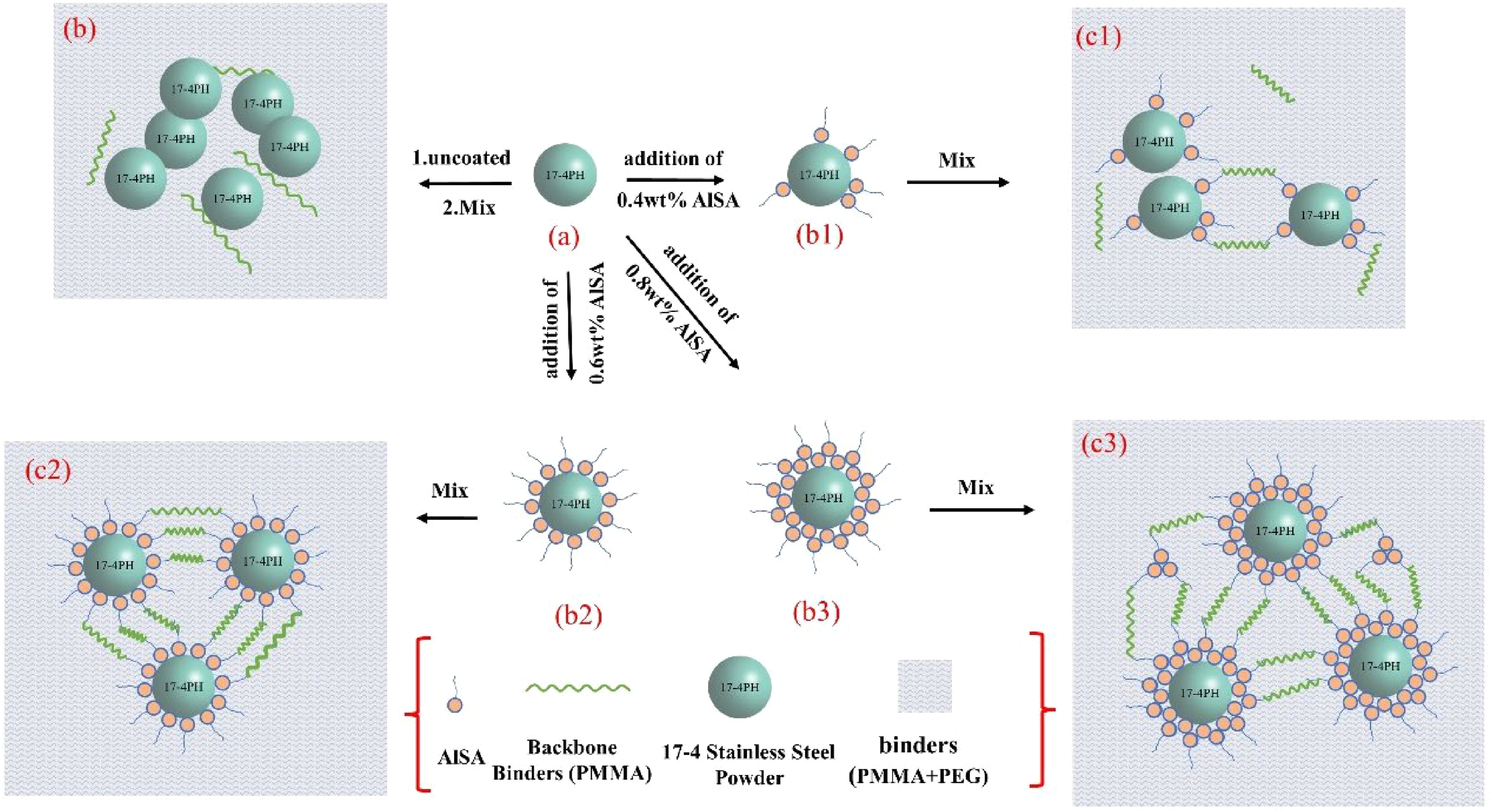
Schematic diagram of powders with different AlSA contents in the binder system for MIM. (a) 0wt% AlSA, (b1) 0.4wt% AlSA, (b2) 0.6wt% AlSA, (b3) 0.8wt% AlSA.
Debinding and microscopic morphology observation of green part
Table 7 shows the bending modulus and density of the four types of green parts. Compared with the pristine parts, the density and bending modulus of the coated parts are higher, especially when 0.6wt% of AlSA is coated. The green density and bending modulus reach 5.44 g/cm3 and 1643 Mpa, respectively, indicating that the AlSA coating on the powders helps to improve the compatibility of the powders with the binder mainly composed of PMMA. However, an overly thick coating may peel off from the surface of the powders under stress, resulting in a decrease in the strength of the green part.
The flexural modulus and density of four green parts.

Figure 9 shows the cross-sectional micro-morphology of the green parts prepared with different AlSA content. The SEM images reveal that the pristine powders have poor compatibility with the binder, the powders particles are separated from the binder and are discrete. The binder cannot fully wrap the powders, and there are some obvious holes, and the powders are relatively rough. Compared with the pristine, the green part powders of (b) and (c) coated with AlSA are more closely connected with the binder; the binder can wrap the wetted powders particles more effectively, increasing the compactness and smoothness of the surface. It is also found that the green part powders particles coated by 0.6wt% AlSA have a closer connection with each other and with the binder, and the powders particles can be well wrapped and filled by the binder. The powders coated with AlSA show no obvious agglomeration, and is closely wrapped by the binder. This demonstrates that AlSA coating enhances the interfacial compatibility between the powders and the binder.

SEM micrographs of green parts. (a) 0wt% AlSA, (b) 0.4wt% AlSA, (c) 0.6wt% AlSA, (d) 0.8wt% AlSA.
The debinding of green parts is the most critical step in the MIM process. Choosing a suitable method can ensure a high debinding rate and dimensional stability of the green parts. 35 Since the binder used in the feedstock is PMMA/PEG, in which PEG is a water-soluble binder that can be removed with deionized water. Using deionized water is more environmentally friendly than other organic solvents. 36 The PEG component in the binder can be discharged out of the green part through dissolution and diffusion under the internal and external concentration differences. At the same time, the remaining resin PMMA helps to maintain the shape of the green part. The effect of different debinding time on the solvent debinding rate of green parts was studied, and the results are shown in Figure 10. After 28 h of deionized water debinding, the PEG removal rate of the green parts reached more than 85%, and the water debinding rate of the green parts prepared by pristine powders was the slowest. This is mainly because the powders coated with AlSA are more uniformly dispersed in the binder, which facilitates the formation of debinding voids in the process of PEG removal, so that PEG will flow out more rapidly. 37 However, the debinding rate decreases when the amount of AlSA is 0.8wt%. This may be because too much hydrophobic coating on the surface of the powders prevents the water molecules from entering the green part, which makes it hard to remove PEG from the green part.
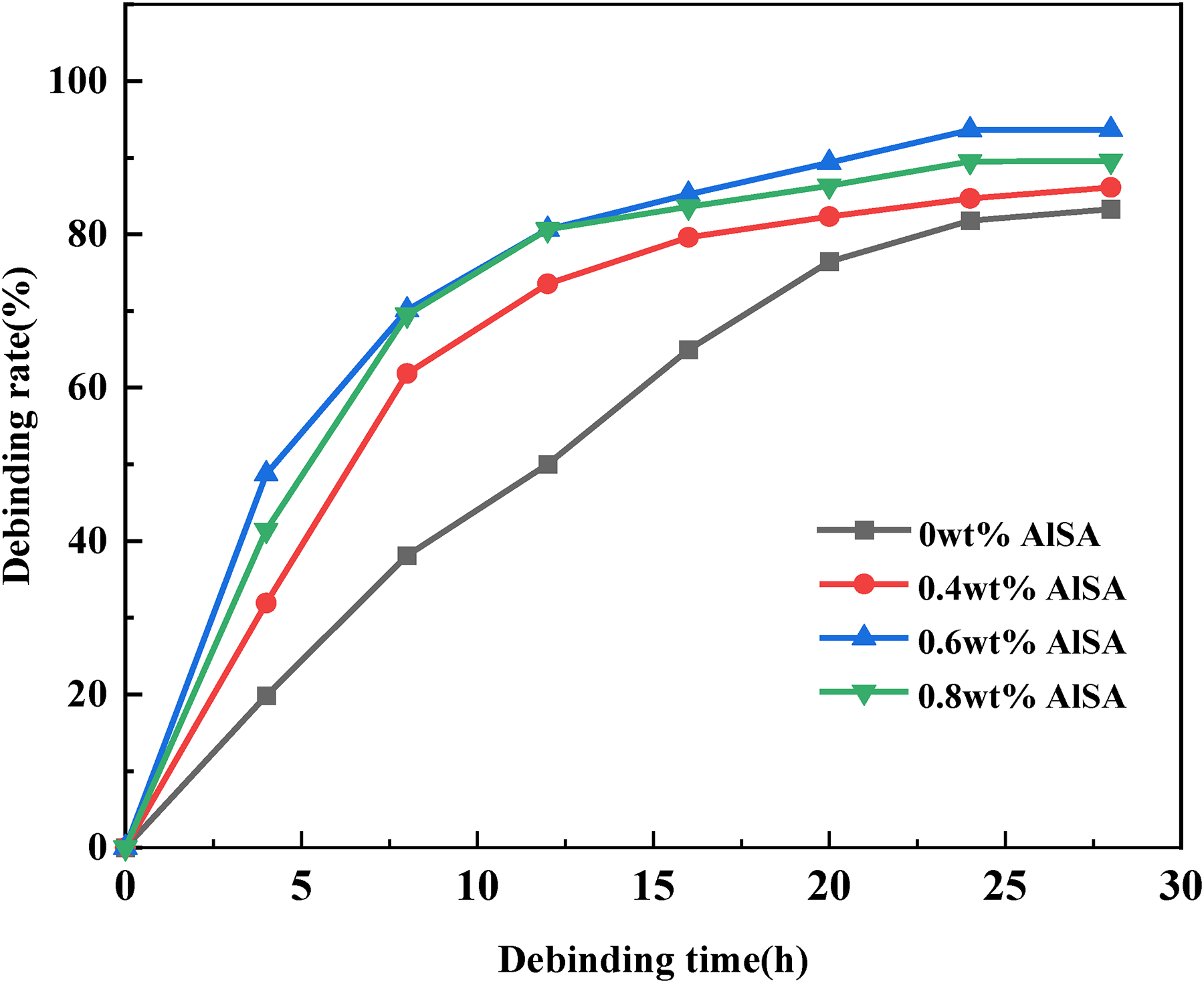
Variation curve of debinding rate with debinding time.
Figure 11 shows the cross-sectional SEM images of the debinding parts. There is almost no difference in the morphology of the powders after thermal debinding, indicating that the powders coated with AlSA do not affect the state of the powders after thermal debinding. As shown in Figure 11(a), there are large pores on the surface of the pristine blank, because the compatibility between the pristine powders and the binder is poor, the powder dispersion is uneven, and the capillary pore channel cannot be formed in time, Therefore, the gas generated by the high-temperature decomposition of the binder in argon atmosphere cannot be discharged from the green part along with the argon flow in time. This accumulation pressure causes holes, cracks, and other defects in the green part, which are very detrimental to debinding and shape retention. Compared with the pristine parts, the coated have better compatibility with the binder, so the morphology shows that the defects of the coated green parts are reduced after thermal debinding, although local smaller pores can also be seen. Among the components coated with AlSA, the powders with 0.6wt% addition has the most complete morphology, Figure 11(c) after thermal debinding. The connection between the powders particles is more compact and orderly, and the pores and defects are further reduced, which is beneficial to the improvement of debinding and shape retention. It is worth noting that when the content of AlSA is 0.8wt%, the residue on the surface of the powders after thermal debinding can be clearly observed. This may be because when the amount of AlSA is too high, alumina may be formed at high temperature and adsorbed on the surface of powders particles. These results are consistent with the research findings of Liu's, 38 who found that adding aluminum stearate can improve the density of carbonyl iron compacts. Some aluminum from the stearate dissolves in the iron matrix, while some other aluminum forms alumina during debinding and sintering.

SEM micrographs of debinded parts. (a) 0wt% AlSA, (b) 0.4wt% AlSA, (c) 0.6wt% AlSA, (d) 0.8wt% AlSA.
The vertical and side views of the debinded parts is shown in Figure 12, and the amount of expansion of the debinded parts is shown in Table 8. After removing the binder, the strength of the green part depends only on the meshing action between the powders. As shown in Figure 12(a), the part after debinding has a certain expansion, and the expansion degree of the part coated with AlSA is lower than that of the pristine part. As the AlSA content increases, the expansion degree of the debinded parts decreases, and the amount of expansion of the part coated with 0.6wt% AlSA is the lowest, only 0.82%. As shown in Figure 12(b), obvious bubbles and cracks appear after the debinding of the pristine part. At the same time, the AlSA-coated parts have fewer defects and are less deformed and distorted. The reason may be that AlSA forms alumina at high temperatures and fills in the voids left by the removed PMMA, resulting in the lowest rate of length change and a further improvement in integrity. On the other hand, too much alumina formation may cause the green part to shrink after debinding. For 0.6wt% AlSA coating, the surface integrity of the debinded part is the best, the expansion rate is the smallest, and the shape retention is the best, the shape retention during thermal debinding can be further improved by coating 17-4PH powders with an Appropriate amount of AlSA.

The photograph of the debinded parts after thermal debinding: (a) vertical view, (b) side view.
Length change rate of green parts after thermal debinding.

Conclusion
In this study, the effect of AlSA coated on 17-4PH powders was systematically analyzed from powders surface coating to shape retention after thermal debinding. The AlSA coating reduces the agglomeration of 17-4PH powders. When the AlSA coating amount is 0.6wt%, the feedstock has the lowest shear viscosity, while excessive coating increases the viscosity and slows down the water debinding. The theoretical calculation shows that 0.26wt% AlSA can form a monolayer on the powder surface. The SEM images reveal that the green part coated with 0.6wt% AlSA has the highest density, and the powders is more uniformly coated by the binder. It is worth emphasizing that the dimensional expansion rate in the length direction of debinded parts prepared by 0.6wt% ALSA decreases from 1.71% to 0.82% after thermal debinding. This study contributes to improving the performance of MIM feedstock and provides a new method for improving the accuracy and quality of the final product.
Footnotes
Acknowledgment
The authors are grateful for the support and funding from Anhui Provincial Department of Science and Technology, China (17030901076).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
