Abstract
Keywords
Introduction
When considering effective interventions for individuals with attention disorders such as attention deficit hyperactivity disorder (ADHD), it is crucial to clarify the neural basis of attentional function and its relationship with behavioral performance. ADHD is a developmental disorder characterized by inattention and/or hyperactivity-impulsivity (Virta et al., 2010). It occurs in childhood, but it often continues into adulthood (Leahy, 2018). Although hyperactivity and impulsivity are observed less often in adults than in children, inattention tends to remain in adulthood (Biederman et al., 2000). Therefore, adults with ADHD face difficulties with inattention at work and in family life. In 2020, the worldwide prevalence of persistent adult ADHD was estimated as 2.58%, while symptomatic adult ADHD was estimated as 6.76%, that is, 139.84 and 366.33 million affected adults, respectively (Song et al., 2021).
Currently, there are two types of treatment for ADHD: pharmacological and non-pharmacological. Non-pharmacological therapies include cognitive training, neurofeedback, and psychological interventions such as behavioral interventions (Nazarova et al., 2022). A meta-analysis showed that non-pharmacological therapies for cognitive symptoms in patients with ADHD, such as physical exercise, cognitive behavioral therapy, neurofeedback/biofeedback, and cognitive training, moderately improve their symptoms (Lambez et al., 2020). These interventions are thought to affect higher-order cognitive functions (inhibition and flexibility) rather than attention itself. Therefore, it is necessary to develop new interventions to address the problems with attention in adults with ADHD (Lambez et al., 2020). Problems with attentional function in ADHD are associated with impairments in executive function, which adjusts mental sets in a situation-dependent manner (Luna-Rodriguez et al., 2018). In a task-switching task that assesses executive function, individuals with ADHD have significantly delayed reaction times compared to typical development individuals (Cepeda et al., 2000; Luna-Rodriguez et al., 2018). When required to switch the stimulus set used for the task, subjects with ADHD also have delayed reaction times compared to typical development subjects (Oades & Christiansen, 2008). Even in typical development individuals, reaction times are significantly delayed when switching between two tasks, compared to when the two tasks are executed separately (Bailey & Konstan, 2006; Hung et al., 2016). Furthermore, task switching delays reaction time, even when one task is a rest condition (Chen et al., 2024).
A subject can maintain automatic and sustained focus on a task when it is repeated in a short period of time. In contrast, an unanticipated interruption, even for a brief moment, can distract attention from the task. When this happens, it can be detected as a delay in reaction time, as it takes time to remember what has to be done when the task resumes and to switch attention back to the task (Trafton & Monk, 2007). The cued-switching task (CST) is a cognitive test that deals with the problem of maintaining automatic focus due to repetition of the task and refocusing on the task after an unexpected interruption or very short rest (Sidlauskaite et al., 2014, 2016). In addition to switching between multiple tasks, the CST can evaluate cognitive processes by separating the period of preparation for the execution of a task from the period during which the task is executed, by incorporating a warning for the task. Using this form of the CST to identify differences in cognitive characteristics and associated brain activity patterns in ADHD and typical development subjects may lead to the development of new interventions for inattention in adults with ADHD.
Sidlauskaite et al. (2014) reported that in typical development subjects, reaction time is significantly delayed in the CST when resuming the task after rest compared to performing the task continuously, and that the anterior insular cortex (AIC) is significantly activated during task warning. Furthermore, Sidlauskaite et al. (2016) administered the CST to ADHD and typical development subjects and found that the ADHD group had slower overall reaction times and lower AIC activity during task warning. This suggests that the AIC plays an important role in switching attention from rest to task resumption and in maintaining sustained focus on the task.
Attentional function and executive control are closely related constructs. Executive function has been described as the ability to allocate attention to achieve goals and regulate behavior accordingly, as well as to prioritize tasks and suppress impulses when attention is diverted (Diamond, 2013; Friedman & Miyake, 2017; Molnar-Szakacs & Uddin, 2022; Wager & Barrett, 2017). The AIC plays a critical role as a gatekeeper for the brain regions and neural networks involved in attentional and executive functions (Cai et al., 2016; Molnar-Szakacs & Uddin, 2022; Nomi et al., 2018; Sridharan et al., 2008; Trautwein et al., 2016). Specifically, the AIC exerts a causal influence on the dorsolateral prefrontal cortex (DLPFC), a key region in the central executive network, during tasks that demand attentional control (Sridharan et al., 2008). Furthermore, the strength of this causal influence increases with task demands (Cai et al., 2016). Individuals with ADHD often face daily difficulties related to attentional and executive functions. Therefore, examining AIC activity in situations requiring attention is important for investigating the potential for improving attention in individuals with ADHD.
However, these studies did not investigate brain activity associated with individual differences in behavioral performance, and the relationship between reaction time and AIC activity is not clear. In addition, they investigated brain activity during task warning, but not during task execution. Furthermore, examining which other brain regions are active in coordination with the AIC during task execution in the CST would provide additional insights into the role of the AIC in switching and maintaining attention.
Thus, the present study examined the neural mechanisms underlying attentional switching upon task resumption after rest by investigating brain activity during task preparation and execution using the CST and functional magnetic resonance imaging (fMRI), focusing on the following three points. First, we analyzed the results separately for when the task was resumed after rest (Rest-Task condition) and when the task was performed continuously (Task-Task condition). Second, we examined the relationship between brain activity and reaction time. Finally, we assessed functional connectivity between the AIC and other brain regions during task preparation in both conditions, and we examined its relationship with brain activity during task execution.
Accordingly, we hypothesized the following for these three aspects. First, consistent with the findings of Sidlauskaite et al. (2014), we hypothesized that reaction time would be longer and AIC activity would be greater in the Rest-Task condition than in the Task-Task condition. This is because resuming a task after an interruption requires recalling what needs to be done and redirecting attention to the task. Second, we hypothesized that individuals with higher AIC activity would show better behavioral performance, reflected in a shorter reaction time. This is based on the assumption that the AIC functions as a gatekeeper for the brain regions involved in attentional control. Third, we predicted that functional connectivity between the AIC and other brain regions would be greater in the Rest-Task condition than in the Task-Task condition, given the higher attentional demands associated with resuming a task. In particular, we expected increased connectivity between the AIC and DLPFC, as previous studies have demonstrated a causal influence of the AIC on the DLPFC under high task demands (Cai et al., 2016; Sridharan et al., 2008).
Materials and Methods
Participants
Thirty-two healthy adults participated in this study (24 males and 8 females; age, 23.47 ± 4.24 years [mean ± standard deviation]). Twenty-four participants were right-handed. This study employed the snowball sampling method (Kirchherr & Charles, 2018) to recruit the participants. All subjects gave written informed consent before participation.
This study was performed in accordance with the Declaration of Helsinki, and was approved by the Ethics Committee of Hiroshima University (permission number 172).
Task and Stimuli
We developed a short version of the CST on E-Prime2 (Psychology Software Tools, Inc., Sharpsburg, PA, USA) based on Sidlauskaite et al. (2014) and used it in this study. The participants viewed an LCD monitor (NordicNeuroLab, Bergen, Norway) via a head coil-mounted mirror in the supine position. Whereas the participants judged whether the presented numbers were even or odd in the Task trials, they did nothing until the presentation of the following task warning stimulus in the Rest trials. Before the scanning session, the participants performed a practice session consisting of Rest and Task trials to familiarize themselves with the procedure of the CST.
A schematic diagram of the CST is shown in Figure 1. Each trial began with the presentation of a fixation cross at the center of the monitor for 500 ms, followed by task warning stimulus presentation for 1,000 ms. Two different warning stimuli were used in this study: one was a circle representing a Rest trial, and the other was a square representing a Task trial. A blank screen was presented in the Rest trials until the fixation cross of the next trial was presented. In this case, the inter-trial interval was changed between 5,000 and 7,000 ms with a 500-ms step. In the Task trials, a blank screen was presented (between 3,500–5,500 ms with a 500-ms step). Then, the imperative stimulus was presented for 500 ms, followed by a blank screen for 1,000 ms. The imperative stimulus was a single-digit number from one to nine, except for five. Schematic diagrams of the rest and task trials. The task warning stimulus was presented at 500 ms after the presentation of a fixation cross in all trials. When a square was presented as a warning stimulus (“task cue” in the Task trial), the participants were asked to judge whether a subsequently presented single digit (imperative stimulus) was even or odd. In contrast, an imperative stimulus was not presented when a circle was shown as a warning stimulus (“rest cue” in the Rest trial). Therefore, the participants did not perform the odd/even judgment in this type of trial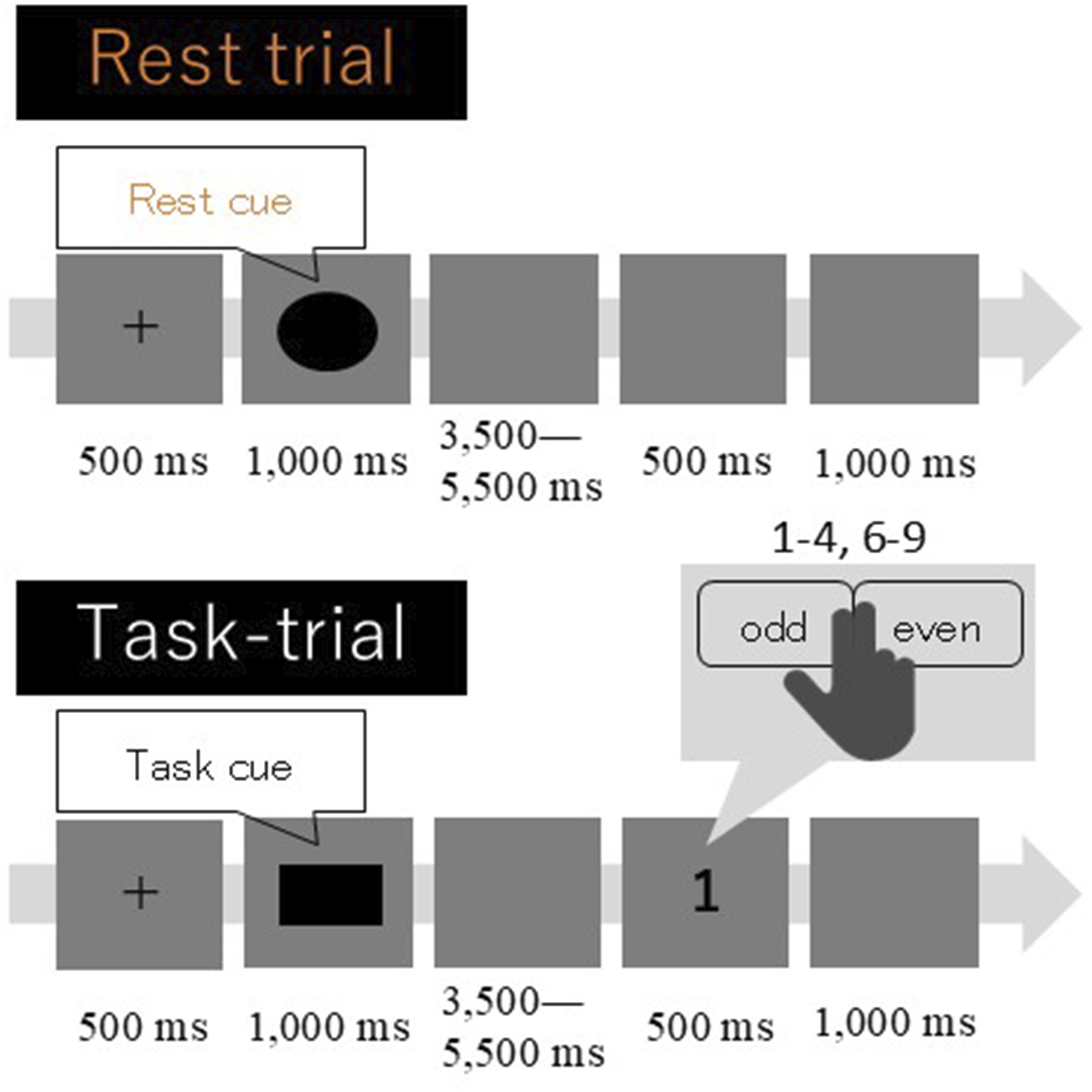
The Task trials were classified into two conditions, Rest-Task and Task-Task, based on the type of the immediately preceding trial. Rest, Rest-Task, and Task-Task conditions accounted for 52% (61 trials), 13% (15 trials), and 34% (40 trials) of all trials (1%, 1 trial was start trial). Each single-digit number, except for five, was presented at an equal frequency throughout the Task trials to each participant. Trial order was initially randomized and then adjusted manually to avoid excessive bias. The task was comprised of a total of 118 trials. The approximate duration of the task was 19 min, which is one-third of the time required to complete the original CST.
The participants were asked to judge whether the presented single-digit number was even or odd and to answer by pressing a button as quickly and accurately as possible (odd/even judgment task). The responses were recorded using an MR-compatible response device (Celeritas Fiber Optic Response Unit–Right hand, 5 buttons; PST-100760; Psychology Software Tools, Inc., Sharpsburg, PA, USA). The participants pressed the button corresponding to the right index finger when an odd number was presented and the button corresponding to the right middle finger when an even number was given. We considered the trials in which participants did not respond within 2,000 ms after imperative stimulus presentation as no-response trials. In the CST, a blank screen was presented for 8 seconds before the onset of the first trial, and a blank screen of at least 4 seconds was presented after task completion.
fMRI Data Acquisition and Analysis
Data Acquisition
All images were acquired using a 3T MRI system (Siemens MAGNETOM Skyra Fit; Siemens Medical Systems, Erlangen, Germany) with a standard 20-channel head coil. Functional images were acquired using a gradient-echo T2*-weighted multiband echo planar imaging sequence. The scan parameters were as follows: single-shot, TR = 1,000 ms, TE = 30 ms, flip angle = 80°, FoV = 212 × 212 mm, matrix size = 64 × 64, slice thickness = 3.2 mm with 0.8-mm gap, and multiband acceleration factor = 3. The number of slices was 42, covering the entire brain, the acquisition order was ascending, and slice orientation was transverse-to-coronal (parallel to the orbitofrontal cortex line). In addition, three-dimensional T1-weighted images were acquired using a magnetization-prepared rapid gradient-echo sequence with the following parameters: TR = 2,300 ms, TE = 2.98 ms, TI = 900 ms, flip angle = 9°, and voxel size 1 × 1 × 1 mm.
Preprocessing
Image preprocessing and analysis were performed using Statistical Parametric Mapping 12 (version 7219; Wellcome Trust Center for Neuroimaging, London, UK) and MATLAB (R2015a; MathWorks, Natick, MA, USA). Image preprocessing steps consisted of realignment, coregistration, normalization, and smoothing. For realignment, all functional volumes in each participant were aligned with the first volume. Next, coregistration was performed to realign the anatomical image with the functional images of each participant. Then, structural images were normalized, and using these deformation parameters, functional images were normalized to the standard Montreal Neurological Institute template and resampled to a voxel size of 2 × 2 × 2 mm. Functional images were then spatially smoothed by convolution using an 8-mm full-width at half maximum Gaussian kernel.
First-Level General Linear Model Analysis (Individual Level)
For each participant, first-level analysis was conducted to detect brain activation corresponding to the preparation and task execution periods using a general linear model. In this analysis, the preparation period was defined as the period between the presentation of the warning and imperative stimuli. The task execution period was defined as the period between imperative stimulus presentation and the participant’s button response. The purpose of the first-level general linear model (GLM) analysis was to model neural responses to the presentation of the warning and imperative stimuli. The task trials were classified as the Task-Task and Rest-Task conditions; therefore, four regressors of interest were set: preparation and task execution periods in the Task-Task and Rest-Task conditions. Moreover, the warning stimulus events in the Rest trials, execution periods of incorrect and no-response trials, and the six realignment parameters were explicitly modeled in the GLM but treated as regressors of no interest, as they were not subjected to hypothesis-driven contrasts. We show the first-level GLM design matrix in the supplemental materials (Figure A1).
The durations of each regressor were defined as follows. For the warning stimulus regressors in both the Rest-Task and Task-Task conditions, the duration corresponded to the jittered blank presentation period (4.5–6 s). For the imperative stimulus regressors in both conditions, the duration was set to the reaction time of each trial. The regressors for the Rest–Rest and Task–Rest conditions were modeled with a duration of 0 s. In trials with incorrect responses, the duration of the warning regressor was set to the blank presentation period of the corresponding trial, whereas the duration of the imperative regressor was set to the reaction time of that trial. In trials with no response, the duration of the warning regressor was set to the blank presentation period, and the duration of the imperative regressor was set to 0 s.
The GLM included six head-motion parameters obtained during the realignment procedure as nuisance regressors. A high-pass filter of 128 s was applied. The canonical hemodynamic response function (HRF) was used as the basis function, and no temporal derivative was included. Model parameters were estimated using Restricted Maximum Likelihood (ReML), and the serial correlation structure was modeled with an AR (1) process.
In the first-level GLM analysis, we generated a series of contrasts to characterize neural responses associated with the warning and imperative stimuli across task conditions. To compare task-dependent differences, we created four differential contrasts: (1) Rest-Task condition > Task-Task condition and (2) Task-Task condition > Rest-Task condition for the warning stimulus, and (3) Rest-Task condition > Task-Task condition and (4) Task-Task condition > Rest-Task condition for the imperative stimulus. We also created single-condition contrasts by assigning contrast weights of 1 or −1 to isolate parameter estimates for each event type in each condition; specifically, contrasts (5) and (6) assigned weights of 1 and −1, respectively, to the warning stimulus in the Rest-Task condition, contrasts (7) and (8) assigned weights of 1 and −1 to the warning stimulus in the Task-Task condition, contrasts (9) and (10) assigned weights of 1 and −1 to the imperative stimulus in the Rest-Task condition, and contrasts (11) and (12) assigned weights of 1 and −1 to the imperative stimulus in the Task-Task condition. Furthermore, to examine condition-specific neural responses relative to Rest trials, we generated four additional contrasts for the warning stimulus: (13) Rest-Task condition > Rest-Rest and Task-Rest conditions, (14) Rest-Rest and Task-Rest conditions > Rest-Task condition, (15) Task-Task condition > Rest-Rest and Task-Rest conditions, and (16) Rest-Rest and Task-Rest conditions > Task-Task condition. Together, these contrasts enabled us to dissociate task-dependent differences and to obtain condition-specific parameter estimates for each event type.
Second-Level General Linear Model Analysis (Group Level)
In the second-level GLM analysis, we conducted separate one-sample t-tests for each contrast of interest. For each analysis, the contrast images generated at the first level for all 32 participants were entered into an independent second-level model. Specifically, differential neural responses between conditions were assessed using the first-level contrasts corresponding to (1) Rest-Task condition > Task-Task condition and (2) Task-Task condition > Rest-Task condition for the warning stimulus, as well as (3) Rest-Task condition > Task-Task condition and (4) Task-Task condition > Rest-Task condition for the imperative stimulus. To obtain condition-specific parameter estimates for each event type, we additionally performed separate second-level analyses using the single-condition contrasts assigning a weight of +1 or −1 to each event type (contrasts 5–12).
All second-level models were implemented as one-sample t-tests without covariates. Statistical significance was evaluated using a voxel-wise threshold of p < .001 (uncorrected), combined with cluster-level family-wise error (FWE) correction at p < .05, based on Gaussian random field theory.
First-Level Parametric Modulation Analysis
At the first (individual) level, event-related analyses were conducted using the general linear model. The design matrix included warning and imperative events for the Rest–Task and Task–Task conditions. Event onsets were modeled in seconds and convolved with the canonical hemodynamic response function. The duration of warning events was defined by the jittered blank interval preceding the imperative stimulus, whereas the duration of imperative events corresponded to each participant’s reaction time.
To assess the influence of preceding trial history, parametric modulation was applied to both warning and imperative events. For the Rest–Task condition, the number of consecutively preceding rest trials was entered as a first-order parametric modulator, whereas for the Task–Task condition, the number of consecutively preceding task trials was used. The planned distribution of consecutive trial lengths is summarized in the Supplemental Materials (Table A1), confirming that the parametric modulation was evaluated across a range of sequence lengths rather than being driven by a small number of isolated sequences. Parametric modulators were orthogonalized with respect to their corresponding main regressors.
Incorrect trials and no-response trials were explicitly modeled as separate regressors in the GLM to account for their variance, but were treated as regressors of no interest and not subjected to hypothesis-driven contrasts. These regressors were included only for participants in whom such trials occurred and were omitted otherwise. In addition, six head-motion parameters derived from the realignment procedure were included as nuisance regressors to control for motion-related variance. A high-pass filter with a cutoff period of 128 s was applied, and serial correlations were modeled using an AR (1) process. We show the first-level parametric modulation design matrix in the supplemental materials (Figure A2).
Second-Level Parametric Modulation Analysis
At the group level, second-level analyses were conducted using one-sample t-tests. For the parametric modulation analyses, individual contrast images were generated for each participant to capture the effects of preceding trial history during both warning and imperative events. Specifically, contrasts were created separately for the Rest–Task and Task–Task conditions, and for each condition, parametric modulation effects were modeled at the time of warning stimulus presentation and imperative stimulus presentation.
For each of these four event types, two contrasts were defined to test positive and negative relationships with the parametric modulators, resulting in a total of eight contrasts (i.e., Rest–Task warning, Rest–Task imperative, Task–Task warning, and Task–Task imperative, each with positive (+1) and negative (−1) weights). For each contrast, individual contrast images from all 32 participants were entered into a one-sample t-test to assess whether the parametric modulation effects differed significantly from zero at the group level.
No additional covariates were included in the model. Implicit masking was applied, and global signal calculation and global normalization were not performed.
Second-Level Multiple Regression Analysis
We conducted a multiple regression analysis to examine the relationship between brain activation and behavioral performance. The dependent variable consisted of the first-level contrast images corresponding to imperative stimuli in the Task–Task condition, obtained from all 32 participants. Each contrast image was entered into the model with a weight of plus one. As the explanatory variable, we included the mean reaction time for the Task–Task condition for each participant. This covariate was centered using overall-mean centering, and no interaction terms were specified. An intercept term was incorporated into the regression model. Statistical inference was performed using t-contrasts that tested the effect of the covariate. Specifically, we assessed negative associations between reaction time and brain activation using a contrast that assigned zero weight to the intercept and negative weight to the covariate.
First-Level Psycho-Physiological Interaction Analysis
At the first level, a psycho-physiological interaction (PPI: Friston et al., 1997) model was constructed for each participant. The physiological regressor was obtained by extracting the first eigenvariate from a 5-mm radius spherical ROI located in the AIC. The psychological regressor coded the warning period of the Rest–Task condition as +1 and that of the Task–Task condition as −1. The interaction term between these regressors was entered as the PPI regressor. These three regressors, together with six head-motion parameters and a session constant, were included in the first-level GLM, and contrast images representing the PPI effect were generated for each participant.
Second-Level Psycho-Physiological Interaction Analysis
At the second level, individual PPI contrast images were entered into a random-effects one-sample t-test to identify brain regions showing condition-dependent modulation of connectivity with the AIC. Statistical significance was assessed using a voxel-wise threshold of p < .001 (uncorrected), and clusters were considered significant at p < .05, FWE corrected at the cluster level.
Statistical Analysis
To examine the effect of the immediately preceding Rest trial on task performance, the reaction times between the Rest-Task and Task-Task conditions were compared. In this comparison, a paired t-test was used to analyze the reaction times in the trials in which the participants made a correct response. The significance level was set at p < .05, and the tests were conducted using R version 4.0.5 (R-Tools Technology, Ontario, Canada).
Results
Behavioral Performance
Behavioral Indicators in the Rest-Task Condition and Task-Task Conditions
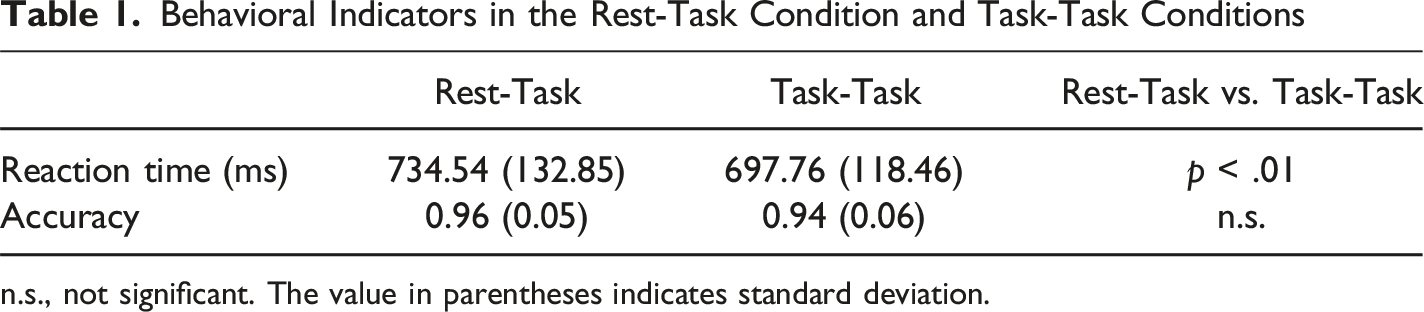
n.s., not significant. The value in parentheses indicates standard deviation.
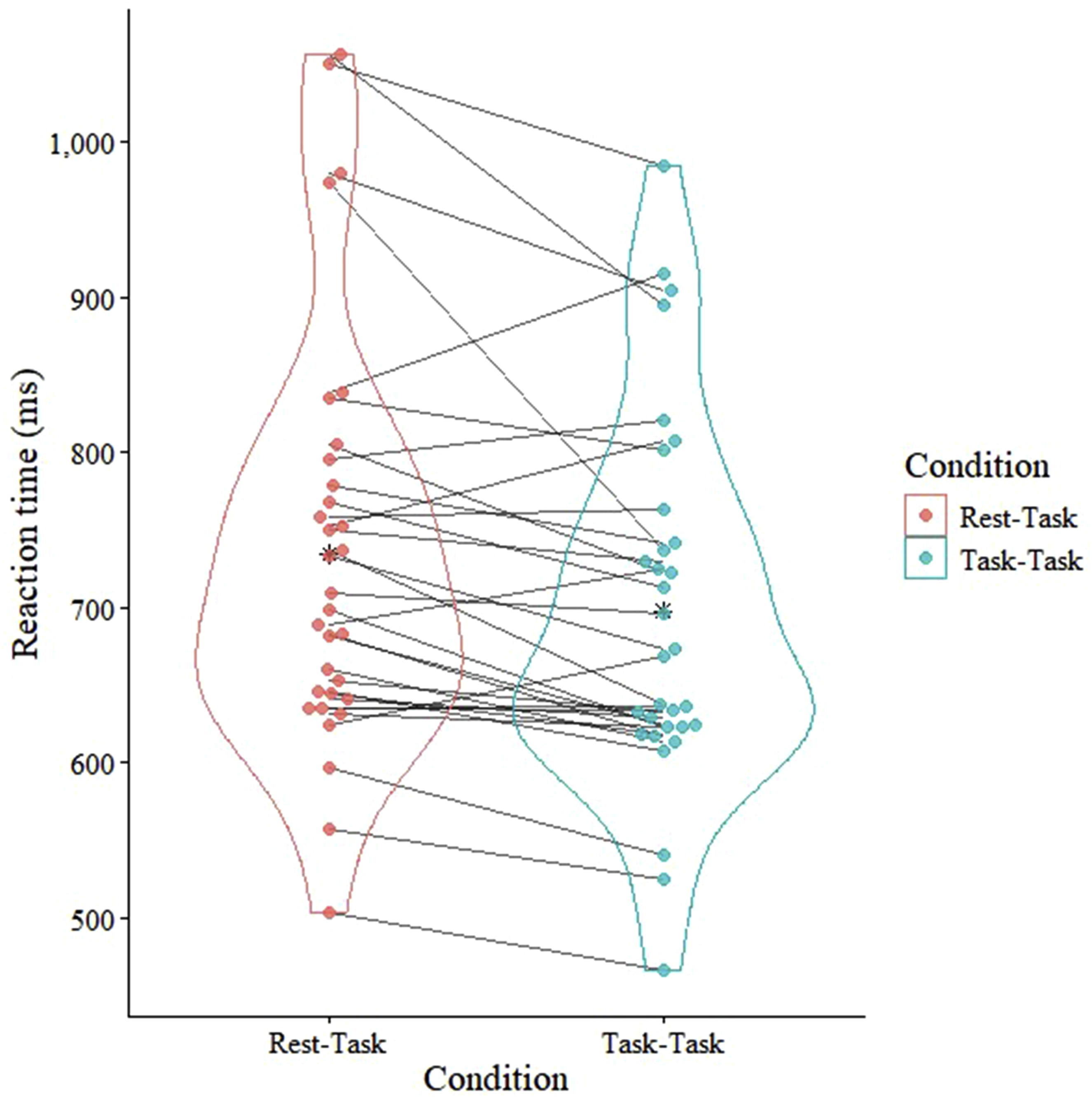
Comparison of reaction time between the Rest–Task and Task–Task conditions. A violin plot shows the distribution of reaction times in both conditions. Data for each participant in both conditions appear, and paired observations are connected by lines to indicate within-participant differences between the two conditions
fMRI Result From General Linear Model Analysis
Brain Activity in Response to the Warning Stimulus
Brain Regions that Responded Significantly to the Task Warning or Imperative Stimulus
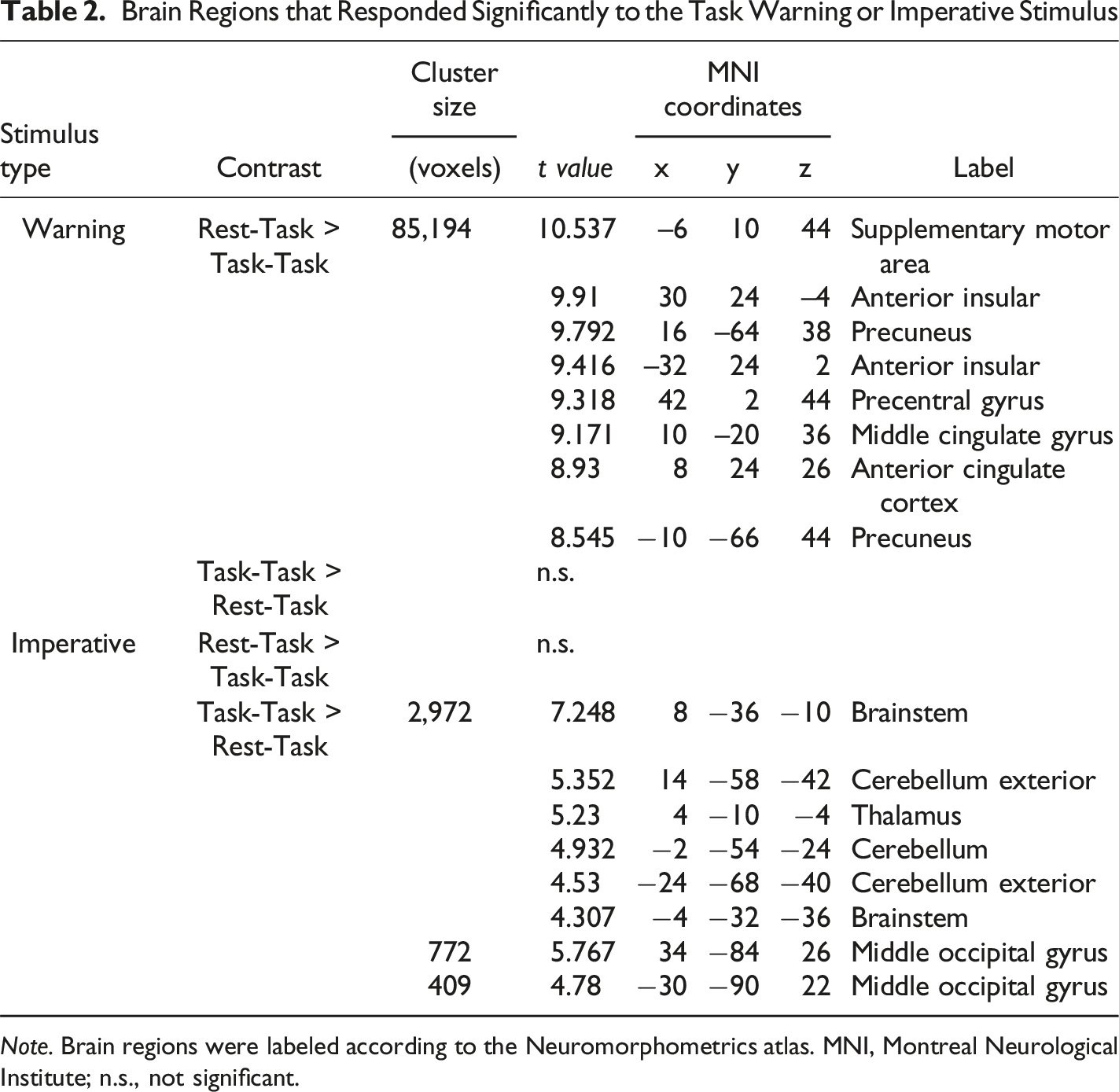
Note. Brain regions were labeled according to the Neuromorphometrics atlas. MNI, Montreal Neurological Institute; n.s., not significant.
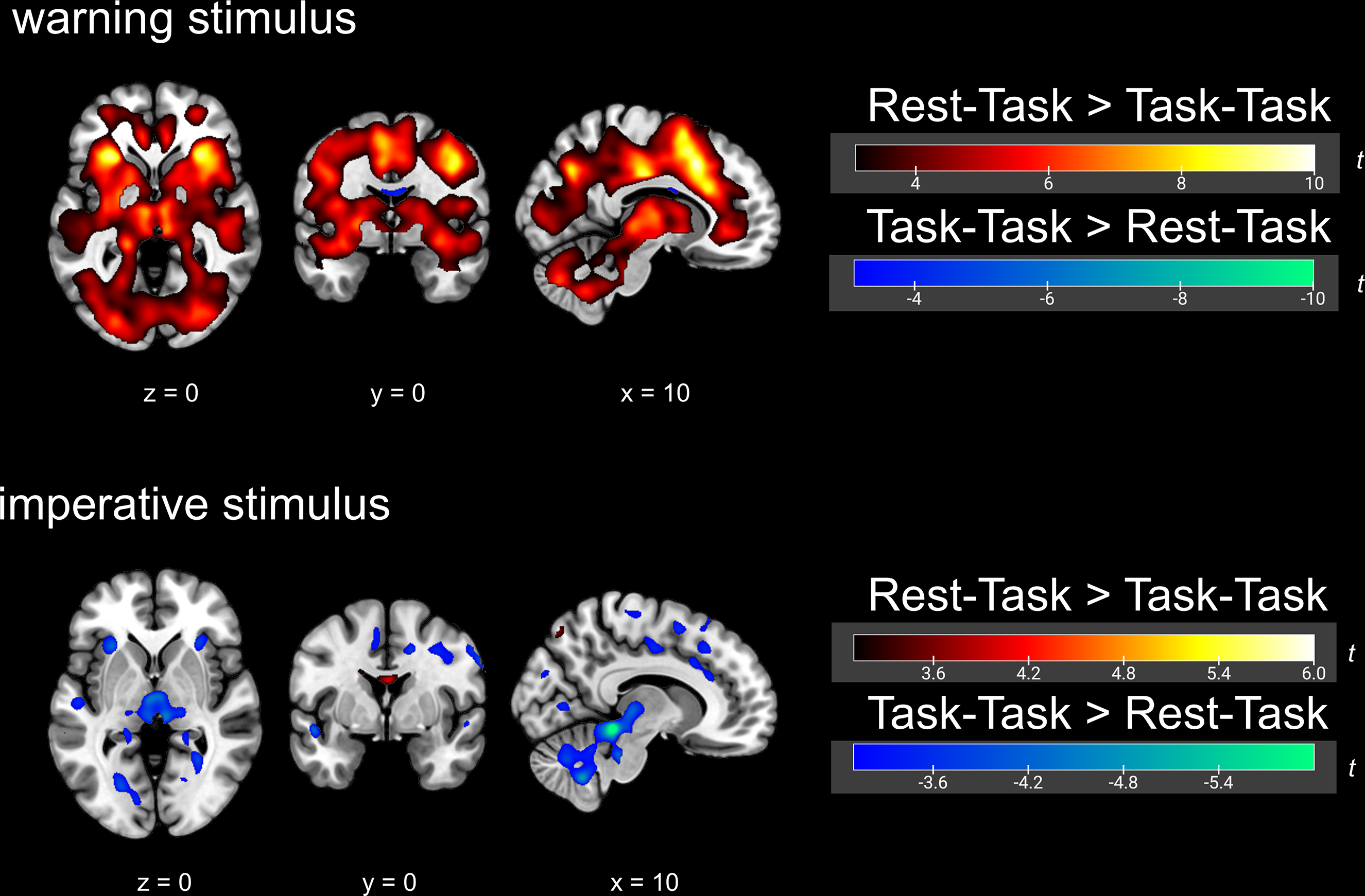
Brain activity in response to the warning and imperative stimuli, showing a comparison between the Rest–Task and Task–Task conditions
In the present study, contrasts of the Rest–Task condition relative to the implicit baseline and of the Task–Task condition relative to the implicit baseline were also examined to confirm the robustness of the analyses. However, to maintain a clear focus on the primary condition comparison (Rest–Task vs. Task–Task) in the main text, these results are presented in the Supplemental Materials (Figure A3).
Brain Activity in Response to the Imperative Stimulus
Brain activity in response to the imperative stimulus is shown in Table 2 and Figure 3. Group-level comparisons between the Rest-Task and Task-Task conditions revealed significantly greater activation in the thalamus and hypothalamus in the Task-Task condition compared with the Rest-Task condition. In contrast, no brain region showed significantly greater activation in the Rest-Task condition compared with the Task-Task condition. With the exception of these regions, the two conditions showed largely overlapping patterns of activation in response to the imperative stimulus. To extract brain regions activated in both conditions during task execution, we additionally performed a second-level analysis of brain responses to the imperative stimulus in the Task–Task condition using an explicit mask derived from the second-level brain responses to the imperative stimulus in the Rest–Task condition (Figure A4).
Similarly, contrasts of the Rest–Task and Task–Task conditions relative to the implicit baseline were also examined for the imperative stimulus, and these results are provided in the Supplemental Materials (Figure A3).
Parametric Modulation Analysis of Task-Evoked Brain Activity by Preceding Trial
Given the pronounced condition difference observed in warning-related activity and the relatively modest condition difference observed in imperative-related activity, parametric modulation analyses were conducted to examine whether these effects could be attributed to differences in preceding trial history rather than to condition differences per se (see Table A1 for the distribution of consecutive trial lengths). Parametric modulation analysis showed within-participant correlations between the magnitude of brain activity and the number of preceding consecutive Rest or Task trials (Figure A5). Positive within-participant correlations between the magnitude of brain activity in response to the imperative stimulus in the Task-Task condition and the number of preceding consecutive Task trials were observed in the premotor area and prefrontal cortex. However, no other within-participant correlations between brain activity and the number of preceding Rest or Task trials were observed in the Task-Task and Rest-Task conditions.
Multiple Regression Analysis of Brain Activity and Reaction Time
To examine whether brain responses elicited by the warning and imperative stimuli in each condition were associated with behavioral performance, we conducted multiple regression analyses. In these analyses, participant-wise mean reaction times calculated separately for each condition and stimulus type were entered as regressors to assess between-participant relationships with the corresponding first-level contrast images. Reaction time regressors were mean-centered across participants prior to inclusion in the model, in accordance with the default settings of SPM.
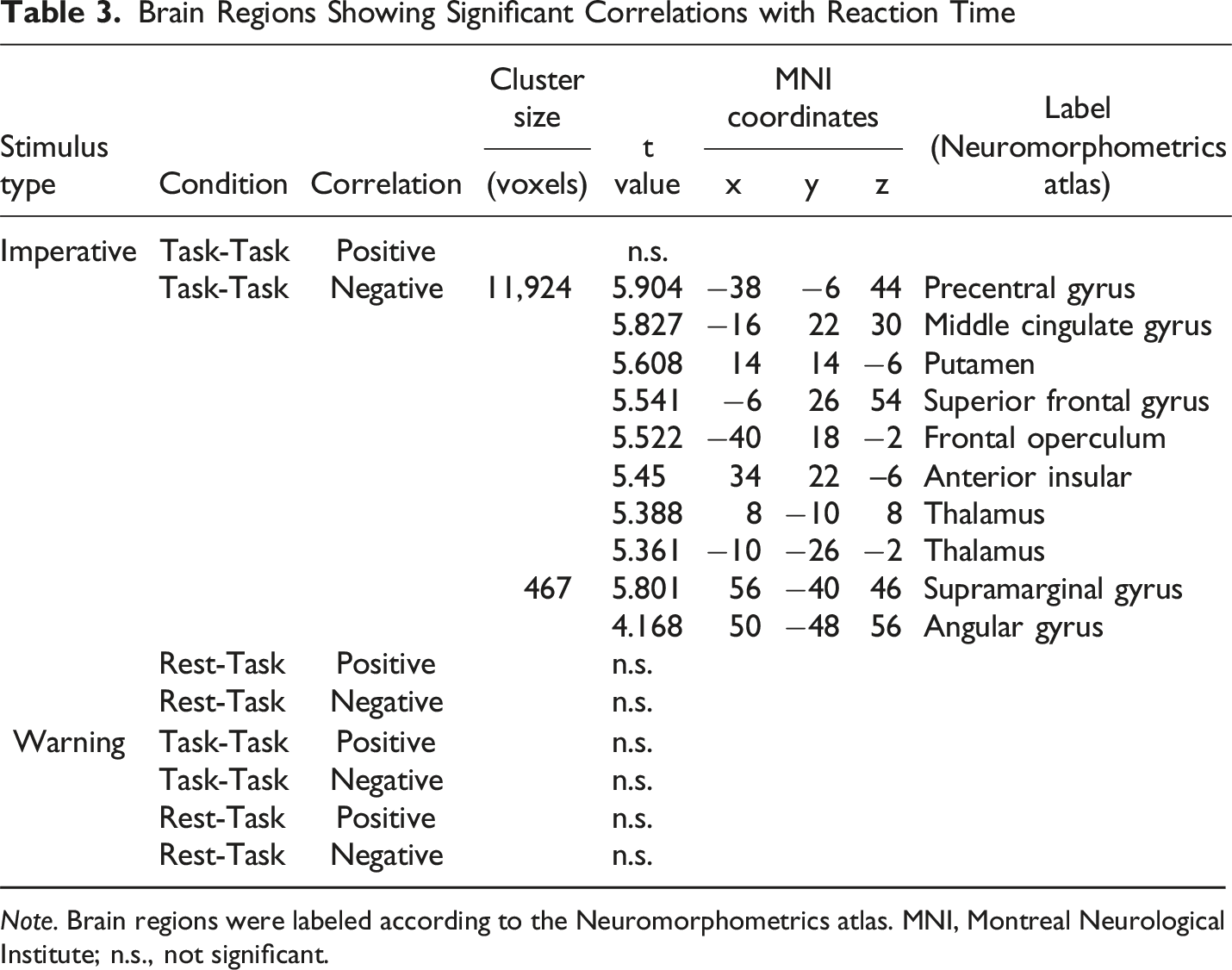
Brain Regions Showing Significant Correlations with Reaction Time
Note. Brain regions were labeled according to the Neuromorphometrics atlas. MNI, Montreal Neurological Institute; n.s., not significant.
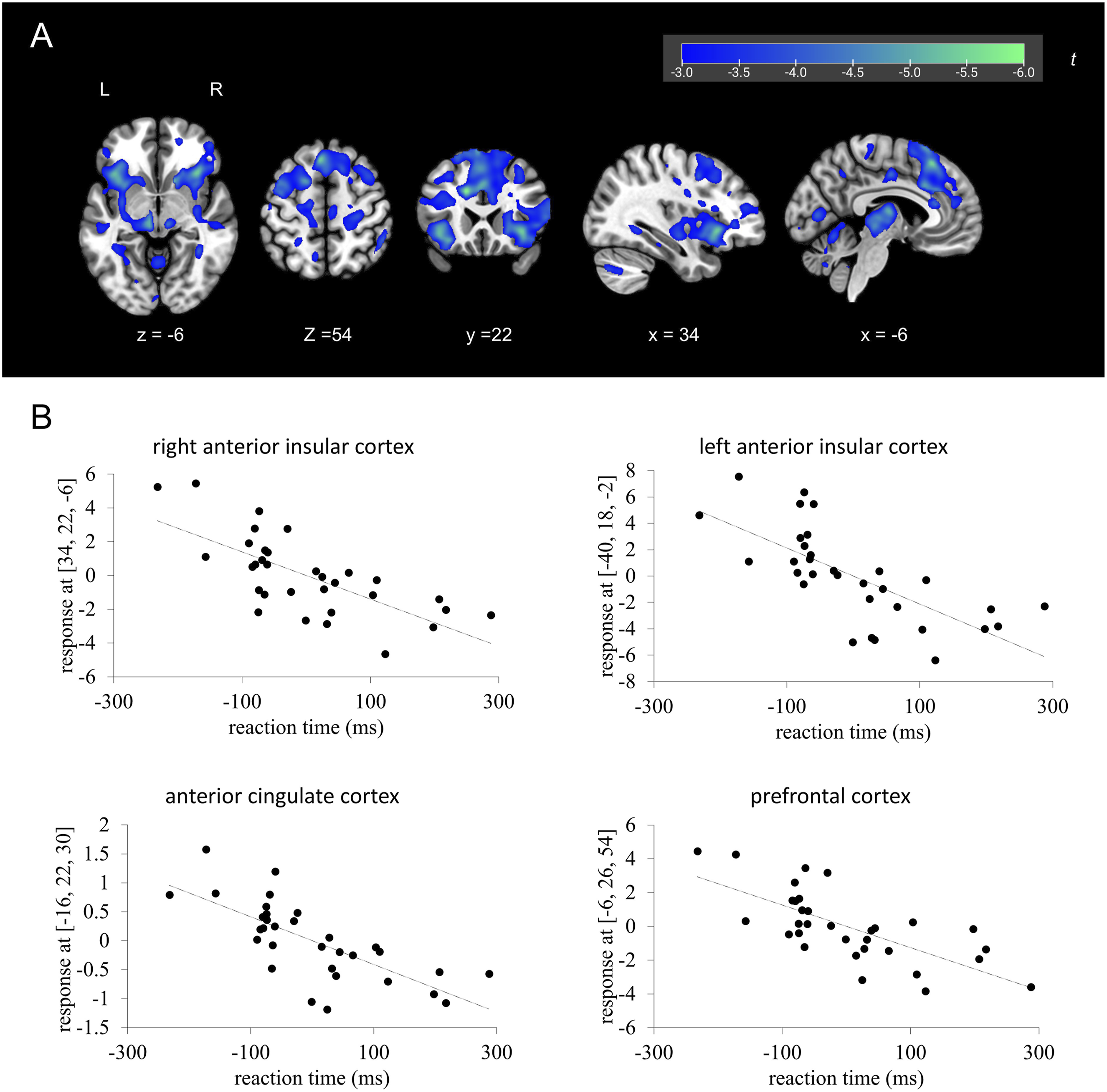
Multiple regression analysis of reaction time in the Task-Task condition versus brain activity in response to the imperative stimulus in the Task-Task condition. (a): Brain regions showing a significant negative correlation between brain activity and reaction time. (b): Scatter plots showing the correlation between brain activity and reaction time. The vertical axis is the beta value at the peak coordinate. The dotted line is a regression line. Reaction time values were mean-centered in accordance with the default settings of SPM; therefore, negative values reflect deviations from the mean reaction time rather than absolute reaction times
Psycho-Physiological Interaction Analysis
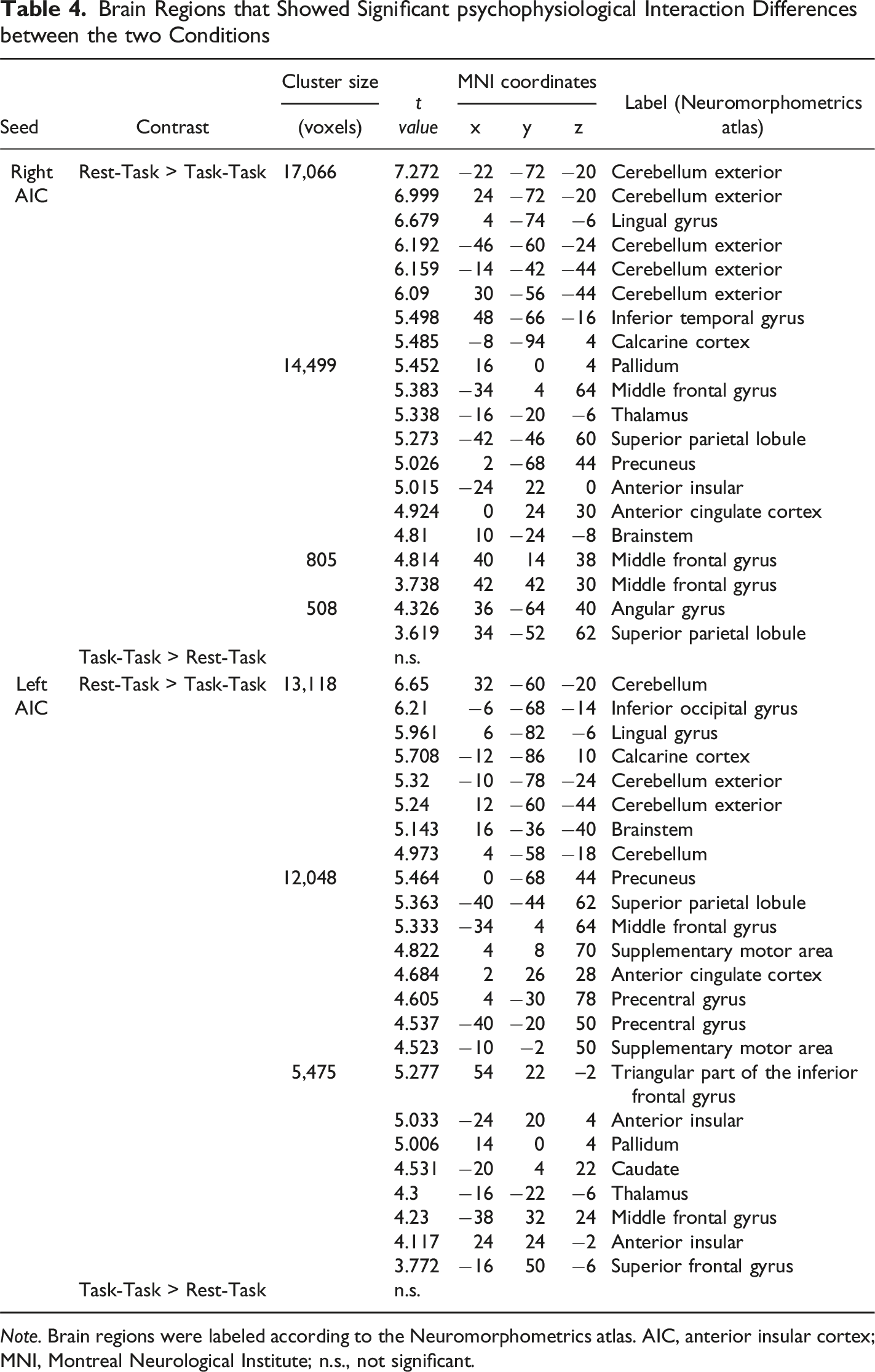
Brain Regions that Showed Significant psychophysiological Interaction Differences between the two Conditions
Note. Brain regions were labeled according to the Neuromorphometrics atlas. AIC, anterior insular cortex; MNI, Montreal Neurological Institute; n.s., not significant.
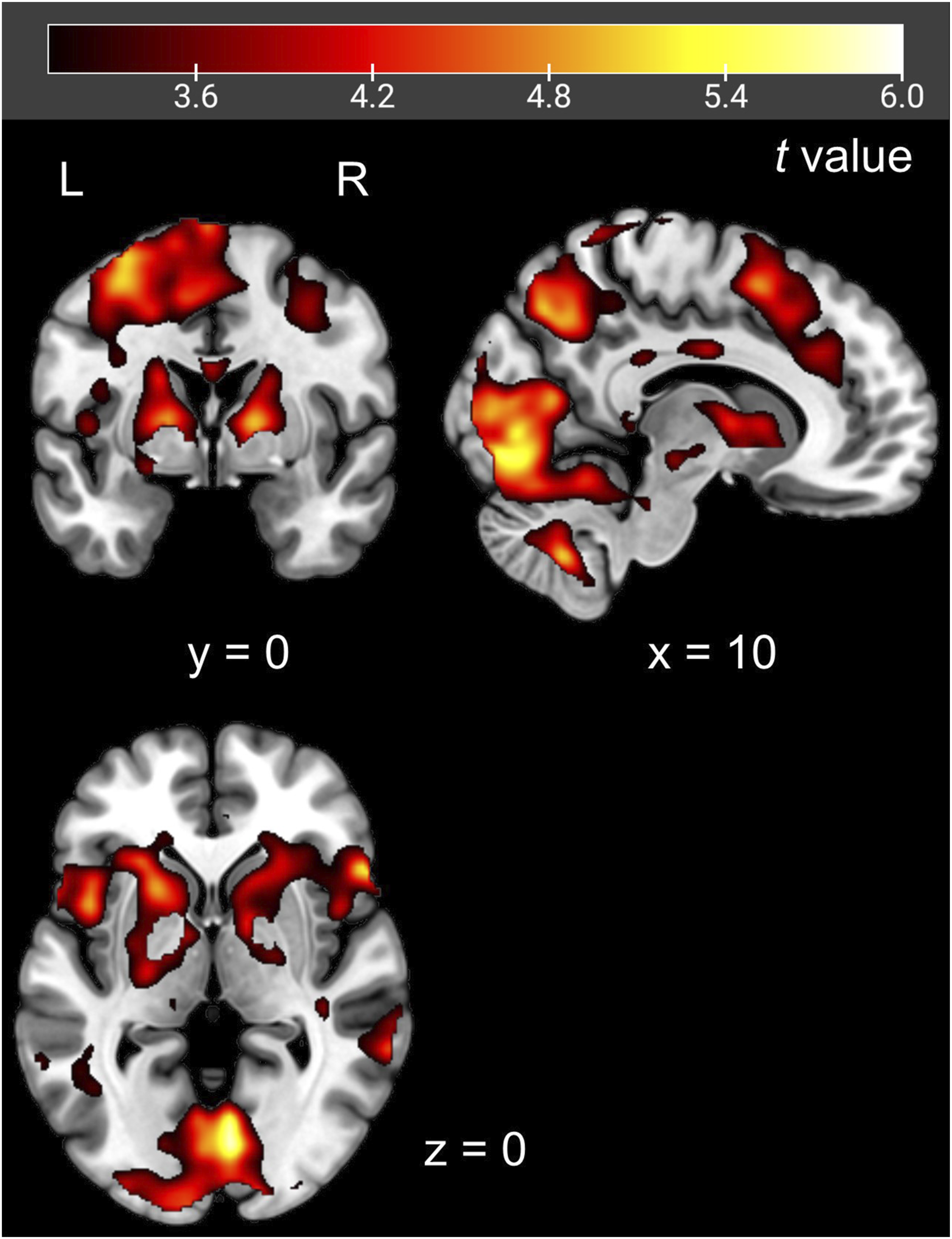
Psychophysiological interaction analysis using the left anterior insular cortex as a seed. Red-colored areas showed significantly stronger functional connectivity with the left anterior insular cortex in the Rest-Task condition than in the Task-Task condition. The seed was defined based on the regions that showed significantly higher activation for the task warning stimulus in the Rest-Task condition than in the Task-Task condition
Discussion
This study not only replicated the results of previous studies showing that the AIC is important when resuming a task after rest but also revealed that AIC activation promotes activity in the salience network, prefrontal cortex, and motor areas. We also revealed that AIC activity is negatively correlated with reaction time when there is no confounding due to attentional switching.
Specifically, we examined the differences in the neural processes involved in task preparation and execution between the Rest-Task and Task-Task conditions. In the Rest-Task condition, the AIC, ACC, and prefrontal cortex were activated during task preparation and execution. Conversely, in the Task-Task condition, a wide range of brain regions including the DMN showed a signal decrease during task preparation, whereas similar brain activity in response to the imperative stimulus was observed as in the Rest-Task condition. Brain activity in response to the imperative stimulus in the Task-Task condition was negatively correlated with reaction time. Moreover, functional connectivity from the AIC to the contralateral AIC, ACC, prefrontal cortex, and motor areas during task preparation was stronger in the Rest-Task condition than in the Task-Task condition. These results demonstrate that brain activity differs during task preparation with and without attentional switching, and that the AIC has a critical role in producing behavioral responses in the Task-Task condition as well as in attentional switching in the Rest-Task condition.
Brain activity to the task warning stimulus in the Rest-Task condition differed from that in the Task-Task condition, indicating that the Rest-Task condition, in which the participants resumed the task after a rest, required a shift in attention. In the Rest-Task condition, the participants were presented with the task warning stimulus when they were not focused on the task. Thus, the task warning stimulus strongly urged them to shift their attention to the task in this condition. Taken together, the present findings regarding warning-related brain activity indicate that the Rest-Task condition–Task-Task condition contrast during the warning period does not reflect differences in the strength of a common warning-related network, but rather reflects differences between two distinct brain states: a state requiring attentional reorientation in the Rest–Task condition and a state characterized by sustained task engagement in the Task–Task condition. The AIC switches brain states from rest to task (Sridharan, 2008), and also facilitates attentional information processing (Menon & Uddin, 2010). Therefore, the response of the AIC to the task warning stimulus in the Rest-Task condition reflects a shift in attention. In addition, the salience network, including the AIC and ACC, detects contextually salient stimuli, not just perceptual pop-ups (Cai et al., 2017; Knudsen, 2007; Sterzer & Kleinschmidt, 2010). Therefore, the response of the salience network to the task warning stimulus indicates that the task warning stimulus in the Rest-Task condition was sufficiently salient to induce a response from the salience network. In contrast, we did not observe a response of the salience network to the task warning stimulus in the Task-Task condition, suggesting that the participants were fully focused on the task in this condition, and the task warning stimulus was not sufficiently salient to induce the response of the salience network.
The salience network and DLPFC responded to the task warning stimulus in the Rest-Task condition. Moreover, PPI analysis revealed stronger functional connectivity between them in the Rest-Task condition than in the Task-Task condition. These regions overlapped with those regions in which a correlation between brain activity and reaction time was observed in the Task-Task condition. These findings suggest a role for the AIC in task preparation with a shift in attention, i.e., detection of the imperative stimulus, switching attention to the cognitive task, and increasing DLPFC activity, which is necessary for cognitive processing during imperative stimulus presentation. That is, it requires more effort to resume cognitive processing during task preparation after taking a rest than in the continuous task situation. Therefore, in the Rest-Task condition, the DLPFC responded not only to the imperative stimulus but also to the task warning stimulus, and functional connectivity between the AIC and DLPFC was strengthened during the presentation of the task warning stimulus.
Our findings demonstrate that the salience network is involved not only in a shift in attention during the presentation of the task warning stimulus after rest but also in a rapid behavioral response to the imperative stimulus. The AIC and ACC showed a significant response to the imperative stimulus in the Rest-Task and Task-Task conditions, and their activity was negatively correlated with reaction time in the Task-Task condition. The ACC is involved in motor behavior, and this area has direct projections to the premotor and primary motor cortices that facilitate action selection and motor responses (Rudebeck et al., 2008). Furthermore, it has been suggested that damage to the ACC leads to delayed reaction times and increased reaction time variability when subjects are required to press a button in response to a stimulus (Picton et al., 2007). The ACC also has direct projections to the spinal cord and brainstem, which are important for motor function (Vogt, 2009). In addition, the cingulate cortex contains a motor-related area, the so-called “cingulate motor area,” which is associated with the planning, preparation, and execution of motor behavior (Dum & Strick, 1993). In fact, ACC activity has been shown to be negatively correlated with reaction time (Naito et al., 2000).
In contrast to the ACC, there is little evidence about the relationship between the AIC and motion (Uddin, 2015). However, Mashour et al. (2020) argued that the AIC plays a gating role to make workspace information available from local to global processing when performing higher-order cognitive processing compared to during perception, including motion and attention. Furthermore, several models have been proposed in which the AIC performs mediated processing leading to movement (Huang et al., 2021). There is a possible need for the AIC to be activated as a gate for processing, including motion. Regarding the role of the AIC and ACC in adaptive behavioral change, Sterzer and Kleinschmidt (2010) demonstrated that the AIC increases sensory arousal and sensitivity to sensory information, while the ACC promotes autonomic activity and motor responses to initiate and control behavioral change. In other words, the AIC is involved in attention and awareness mediated by the expression of bodily responses, while the ACC has a role in intention and action, which are interrelated and complex activities. Considering these findings and theoretical models, the AIC and ACC may be involved in the detection of salient stimuli and the regulation of behavioral responses to imperative stimuli.
In contrast to the salience network, the DMN showed a significant signal decrease in response to the task warning stimulus in the Task-Task condition, indicating that the participants were already focused on the task when the task warning stimulus was presented in this condition. Previous research has shown that DMN activity decreases during the execution of tasks that require cognitive activity, such as attentional tasks (Lin et al., 2011). Menon and Uddin (2010) reported that the salience network plays a role in switching the activated network between the central executive network and DMN in a situation-dependent manner. In other words, the salience network suppresses the activity of the DMN during cognitive activity. During task execution, the AIC, ACC, and DLPFC were activated in both conditions, indicating that these regions are directly related to the performance of motor responses and the cognitive task. This suggests that the DMN, which was activated during rest, was suppressed during the presentation of the task warning stimulus in the Task-Task condition. Considering that the Task-Task condition does not require a shift in attention to the task warning stimulus, the deactivation of the DMN to the task warning stimulus in the Task-Task condition suggests that the participants maintained their alertness at a high level so that they could identify the imperative stimulus, make the motor response, and perform the cognitive task promptly.
In this study, we observed a correlation between brain activity and reaction time for the imperative stimulus only in the Task-Task condition. In the Task-Task condition, brain activity in response to the imperative stimulus was negatively correlated with reaction time in the AIC, ACC, and prefrontal cortex, whereas these correlations were not observed in the Rest-Task condition. These regions were significantly activated by the imperative stimulus in both conditions. However, comparing these two conditions, despite the similar activity pattern, the activated areas and the intensity of their activation were smaller in the Rest-Task condition than in the Task-Task condition, especially in the AIC and a part of the thalamus. In the Rest-Task condition, the cognitive process of switching attention from rest to the task may have made the relationship between brain activity and motor response more complex, and it is possible that the intensity of AIC activity was not sufficient compared to the Task-Task condition, or that it was not directly reflected by reaction time. In addition, no correlation was observed between brain activity in the task warning stimulus phase of the Task-Task condition and reaction time. This may be attributed to the lack of activation in the salience network, DLPFC, and motor regions in response to the task warning stimulus, unlike the imperative stimulus in the same condition. It is possible that, even if only the DMN shows a signal reduction during the presentation of the task warning stimulus, this may not be sufficient to produce a correlation with reaction time without concurrent activation in the salience network and DLPFC.
Similar to Sidlauskaite et al. (2014), the present study showed that the AIC and ACC were activated during the presentation of the task warning stimulus in the Rest-Task condition, and DMN activity decreased during the presentation of the task warning stimulus in the Task-Task condition. However, we did not observe activation of the precuneus during task warning stimulus presentation in the Rest-Task condition, as reported by Sidlauskaite et al. (2014). This discrepancy may be due to the differences in the CST procedure between the two studies. We employed a shortened version of the CST. The original CST has two task conditions and requires participants to switch from one task to the other, whereas the shortened version has only one task condition and does not require task switching. In addition, in the present study, the period of the blank screen after the presentation of the imperative stimulus was shortened, the number of trials was reduced, and two breaks were inserted during performance of the CST. These changes possibly prevented the participants from becoming fatigued and less alert, which, in turn, prevented them from engaging in mind-wandering during the CST. Considering the fact that the precuneus is a part of the DMN, we think this explanation is plausible.
This study has several limitations that should be addressed. It has been pointed out in studies dealing with the resting state that there are individual differences in what state participants are in during the rest period and that it is uncontrollable (Doucet et al., 2012). In this study, we were also concerned that the participants may not have been resting during the Rest trials. To address this concern, we asked the participants to evaluate how well they maintained a calm state until the subsequent trial during the Rest trials using a 101-point scale (0 = not at all, 100 = fully successful). The average score was 83.3 points (standard deviation = 20.0), indicating that the participants considered they were in a resting state during the Rest trials. In addition, we did not observe a difference in accuracy between the Rest-Task and Task-Task conditions. Considering that we found a difference in reaction time between these two conditions, as reported previously (Sidlauskaite et al., 2014), the cognitive load of the CST after the Rest trials was sufficiently high to delay reaction time, but insufficient to affect accuracy. In a future study, a cognitive task with higher difficulty should be employed to examine the relationship between accuracy and brain activity.
The shortened version of the CST used in the present study included approximately one-third the number of trials compared with that used in Sidlauskaite et al. (2014), which may have reduced the statistical power to detect significant differences between conditions. The number of trials was reduced because the original CST required 1 hour to complete, and during pilot testing, most participants reported pronounced drowsiness and difficulty maintaining alertness. In the pilot study, we sought to determine the number of trials that would preserve the ratio of Rest–Task to Task–Task conditions from the original CST while still producing a significant difference in reaction time between conditions, with a longer response time in the Rest–Task condition. On the basis of these considerations, the shortened CST used in the present study was developed.
In addition to the reduction in trial number, the present study also differed from Sidlauskaite et al. (2014) in the duration of each trial. Whereas the previous study employed relatively long trials (6,000–19,200 ms), the present study used shorter trials (6,500–7,500 ms). Because the primary focus of the present study was neural activity during the warning and imperative stimulus periods of Task trials, the blank interval following task execution was treated as a period of no interest and was therefore fixed across trials. As a consequence of this shortened trial structure, the present task emphasized situations in which participants had more recently and continuously engaged in task performance, particularly in the Task–Task condition. Accordingly, the results of the present study should not be directly compared with those of Sidlauskaite et al. (2014), and these methodological differences should be taken into account when interpreting the findings.
Finally, the participants in this study were not assumed to have ADHD, nor were they screened for ADHD characteristics. In this regard, we cannot conclude that the results can be immediately applicable to the treatment of ADHD. However, our findings indicate that the salience network plays a crucial role in situations requiring attentional switching and those demanding the maintenance of attention, offering potentially meaningful clinical implications. Specifically, for individuals with attentional deficits, such as those with ADHD, these findings could support therapeutic interventions, such as neurofeedback training, aimed at increasing salience network activity specifically in response to target stimuli.
In summary, by using a shortened version of the CST, we found that the AIC, ACC, and prefrontal cortex were activated during task execution in the Rest-Task and Task-Task conditions. AIC, ACC, and prefrontal cortex activity during task execution in the Task-Task condition was negatively correlated with reaction time. During task preparation in the Rest-Task condition, the contralateral AIC, ACC, prefrontal cortex, and motor areas were more synchronously activated with the AIC. Our findings suggest that the AIC plays a crucial role in maintaining continuous task-related concentration and in task resumption after rest. Although further research is needed, this study shows that AIC activity may be an effective indicator of training in attentional interventions such as biofeedback in the future.
Supplemental Material
Supplemental Material - Activation of the Anterior Insular and Prefrontal Cortices During Attention Switching and Task Execution: A Study Using a Cued-Switching Task and Functional Magnetic Resonance Imaging
Supplemental Material for Activation of the Anterior Insular and Prefrontal Cortices During Attention Switching and Task Execution: A Study Using a Cued-Switching Task and Functional Magnetic Resonance Imaging by Miyuki Kato, Shigeyuki Kan, Naho Ichikawa, Masahiro Takamura, Go Okada in Perceptual and Motor Skills
Footnotes
Acknowledgements
We would like to thank to Dr. Yasumasa Okamoto, Dr. Alan Fermin, Dr. Satoshi Yokoyama, and Mr. Yuki Mitsuyama for their insightful comments, and Dr. Masahito Sakakibara for valuable advice and encouragement from the beginning. Additionally, we appreciate the participants, and the proofreaders from NAI, Inc. (Yokohama, Japan).
Ethical Considerations
This study was performed in accordance with the Declaration of Helsinki, and was approved by the Ethics Committee of Hiroshima University (permission number 172).
Consent to Participate
All subjects gave written informed consent before participation, in written form.
Authorship Contribution
Miyuki Kato: Writing – original draft, Conceptualization, Validation, Investigation, Formal analysis, Data curation. Shigeyuki Kan: Writing – review & editing, Supervision, Formal analysis, Methodology. Naho Ichikawa: Supervision, Investigation. Masahiro Takamura: Supervision. Go Okada: Writing – review & editing, Supervision, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by AMED under grant number JP24wm0625204. Miyuki Kato would like to thank JSPS KAKENHI grant number JP22H04926, Grant-in-Aid for Transformative Research Areas ― Platforms for Advanced Technologies and Research Resources “Advanced Bioimaging Support” for the opportunity to learn from the tutorial on fMRI analysis methods.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that supports the findings of this study are not publicly available at this time due to the absence of a data-sharing platform currently in place. However, the data can be provided by the corresponding author upon request. The data presented this study are available from the author on request.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
