Abstract
Recent research has suggested that top-down executive function associated with the prefrontal cortex is key to the decision-making processes and pacing of endurance performance. A small but growing body of literature has investigated the neurological underpinnings of these processes by subjecting the prefrontal cortex to functional near-infrared spectroscopy (fNIRS) measurement during self-paced endurance task performance. Given that fNIRS measurement for these purposes is a relatively recent development, the principal aim of this review was to assess the methodological rigor and findings of this body of research. We performed a systematic literature search to collate research assessing prefrontal cortex oxygenation via fNIRS during self-paced endurance performance. A total of 17 studies met the criteria for inclusion. We then extracted information concerning the methodology and findings from the studies reviewed. Promisingly, most of the reviewed studies reported having adopted commonplace and feasible best practice guidelines. However, a lack of adherence to these guidelines was evident in some areas. For instance, there was little evidence of measures to tackle and remove artifacts from data. Lastly, the reviewed studies provide insight into the significance of cerebral oxygenation to endurance performance and the role of the prefrontal cortex in pacing behavior. Therefore, future research that better follows the guidelines presented will help advance our understanding of the role of the brain in endurance performance and aid in the development of techniques to improve or maintain prefrontal cortex (PFC) oxygenation to help bolster endurance performance.
Introduction
Recent research has outlined how self-regulation is crucial to the decision-making processes and pacing of endurance exercise performance (Hyland-Monks et al., 2018). The review conducted by Hyland-Monks and colleagues drew upon various research domains to argue that executive function (EF), predominantly governed by the prefrontal cortex (PFC), underlies the top-down self-regulation of endurance performance. Although EF per se is not explicitly referenced, contemporary thinking concerning the psychology of endurance performance places self-regulatory processes at center stage. For instance, Brick et al. (2014) argue that metacognitive skills (i.e., planning and monitoring) and experiences (i.e., feeling and judgements) are key to the regulation of endurance performance. Providing a foundation for this work, Roebers (2017) presented a unified account of self-regulation by synthesizing the scientific understanding of EF and metacognition. Furthermore, Marcora’s (2010) psychobiological model proposed that the conscious regulation of pace is governed by the integration of potential motivation and effort perception. Perceived exertion, motivational drive and potential modifiers of these variables (e.g., self-talk; Barwood et al., 2015) may be information that is, in part, integrated by the PFC to inform decision-making regarding pace (Robertson & Marino, 2016).
Following on, researchers have theorized that an evolutionary-driven trade-off in efficiency impacts brain activity during exercise. The reticular activating hypofrontality (RAH) model (Dietrich & Audiffren, 2011) proposes that exercise induces a hypofrontality effect. As whole-body dynamic exercise is metabolically costly, the limited resources of the brain are preserved for areas implicated in the control of movement (i.e., motor and sensory areas; Christensen et al., 2000), resulting in a downregulation in the PFC with associated costs to EF. This hypofrontality effect is thought to increase in line with exercise duration and intensity. Therefore, based on the RAH model propositions, the downregulation of the ‘hardware’ to self-regulate during exercise presents a challenge to endurance performers concerning the top-down control of pacing. This has possible implications for the ability to integrate performance feedback by the PFC. Experimental (Wingfield et al., 2018) and field-based (Dukalski et al., 2020) research has demonstrated that the removal of live performance feedback hinders cycling time-trial (TT) performance and pacing. Interestingly, when feedback was provided, power output was strongly associated with PFC activity (Wingfield et al., 2018). This ability to integrate performance feedback may also explain differences in performance. Faster finishers in an ultramarathon displayed superior EF capacities (Cona et al., 2015). Likewise, more experienced cyclists were less susceptible to mental fatigue (induced through EF testing) in subsequent TT performance (Martin et al., 2016).
Given the above, researchers have sought to investigate the PFC and its potential role in endurance performance using functional near-infrared spectroscopy (fNIRS). fNIRS is a non-invasive method reliant on near-infrared light (between 650–900 nm) to measure cerebral hemodyamics by assessing the absorption spectra of oxyhemoglobin (O2Hb) and deoxyhemoglobin (HHb) reflections from cortical tissue (Villringer et al., 1993). fNIRS relies on neurovascular coupling (Filosa & Blanco, 2007), whereby an increase in energy consumption from an active brain region increases cerebral blood flow to the area to supply oxygen and nutrients. As oxygenation outstrips demand, blood oxygenation levels rise in the brain area in question. This interplay between O2Hb and HHb forms the basis by which fNIRS methodology allows inference regarding cortical activity from neurovascular redistribution. fNIRS is used within exercise research because the technology is relatively robust to movement artifacts and offers acceptable spatial resolution in comparison to other available neuroimaging methods (Perrey, 2008).
A systematic review of cerebral oxygenation (Cox) during incremental exercise found that oxygenation in the PFC typically increases between low and moderate exercise intensities, remains stable from moderate-to-hard intensities, and decreases when nearing exhaustion (Rooks et al., 2010). At very hard intensities, compared to their less trained counterparts, aerobically trained participants obtained higher absolute values of O2Hb, HHb and cerebral blood volume, as approximated by total hemodynamics (tHb). Further research has also focused on cerebral hemodyamics to shed light on the psychology of endurance performance. Investigators have observed changes in PFC activity as a function of exercise intensity and cognitive load (Tempest et al., 2017), deception regarding exercise duration (Radel et al., 2017) and using imagery to aid performance (Tempest & Parfitt, 2013). However, these investigators adopted methodologies that featured prescribed exercise intensities. Therefore, self-paced protocols are also needed to gain insight into the self-regulation of endurance performance via brain-based executive processes.
A small and growing cohort of researchers are interested in self-paced endurance performance and the cerebral hemodyamics implicated in EF. This work has included exploratory investigations of PFC oxygenation during full-effort treadmill TT running in trained (Billaut et al., 2010) and elite (Santos-Concejero et al., 2015) athletes. Several studies have investigated simulated altitude effects (i.e., hypoxia) on PFC hemodyamics during self-paced endurance performance (Bourdillon et al., 2014; Fan et al., 2013; Ferguson et al., 2018; Hamlin et al., 2010; Nielsen et al., 1999), while others have investigated the potential for ergogenic aids to ameliorate hypoxic effects or increase cerebral perfusion (Decroix et al., 2016, 2018; Fan et al., 2018; Liao et al., 2019; Shannon et al., 2017; Shaw et al., 2020). In addition, new research has provided insight into the effect of feedback blinding (Wingfield et al., 2018) and distance deception (Wingfield et al., 2019) on PFC oxygenation during cycling TT performance.
The methodology for self-paced protocols used in the studies mentioned above presents several challenges for fNIRS measurement of the brain. Most pertinent is the high-intensity effort performed around and above the ventilatory threshold (Goldberg et al., 1988). Although most athletes distribute their resources to avoid complete catastrophic failure and collapse (St Clair Gibson et al., 2013), the strain of high-intensity exercise results in biomechanical changes in motor performance, for instance, at the hip and knee joint during cycling (Bini et al., 2010). This has likely implications for whole-body movement, as evident in the ‘swaying’ of the upper body observed in cyclists when fatigued. This creates the potential for movement artifacts in addition to those presented by lower intensity exercise. Although analytic techniques may mitigate the effect of artifacts, they still present a possible confounding effect by reducing the signal-to-noise ratio (Pinti et al., 2019). A second challenge relates to experimental control. Many protocols using fNIRS for exercise-based research have adopted block or event-related designs (Herold et al., 2018), allowing researchers to relate cortical hemodyamics to discrete events (e.g., assessment at various prescribed exercise intensities). Although experimental manipulations can be used in self-paced protocols, participants are typically free to pace activities as they wish, giving added importance to how researchers analyze fNIRS data to compare experimental conditions (e.g., recording windows). To date, however, there have been no extensive efforts to specifically review this body of research and the unique challenges it presents.
Several recent efforts have been made to review both the methodology and findings of exercise-based research using fNIRS to assess cortical hemodyamics. Perrey and Besson (2018) compared this methodology to other neuroimaging methods and highlighted attempts at using fNIRS to answer longstanding unknowns regarding exercise physiology and neurological phenomena (e.g, acute and long-standing effects of exercise on the brain). Herold et al. (2018) systematically reviewed the methodological rigor of studies using fNIRS to investigate cerebral hemodyamics, cognition and exercise. Most recently, Wachter et al. (2021) reviewed the literature on endurance performance and PFC oxygenation, organizing their findings by the nature of the exercise completed (i.e., intensity). However, this review only featured three of the studies included in the present review. Since no review has primarily focused on studies using fNIRS during self-paced endurance performance, we aimed in this review to assess both the methodology and findings of investigations using fNIRS to study the relationship between cortical hemodyamics of the PFC and self-paced endurance performance. More specifically, we sought to review best practice evidence for fNIRS use and the adherence of reviewed studies to these guidelines, and to provide a theoretical and practical commentary on study findings to date.
Method
To ensure robustness and adherence to discipline standards, where appropriate, in this scoping review, we followed the guidelines provided by the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) pertaining to scoping reviews (Tricco et al., 2018). To ensure transparency, the review was preregistered on the Open Science Framework website (DOI: 10.17605/OSF.IO/7XDBJ).
Search Process and Criteria
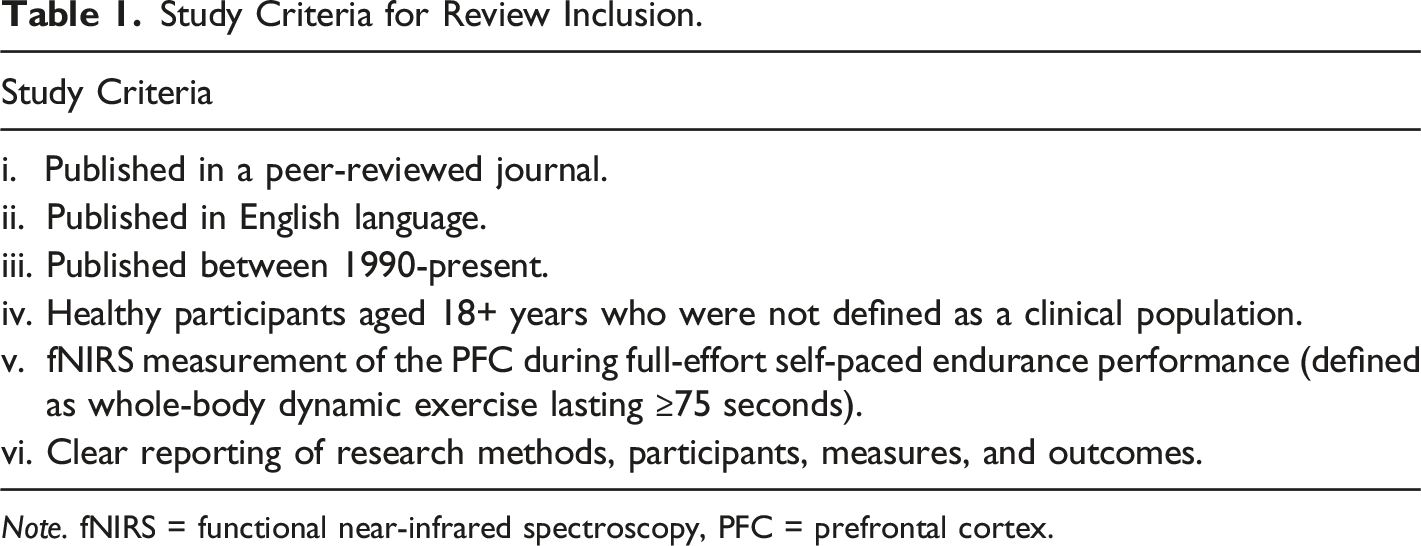
In November 2021, we performed a systematic literature search across two electronic databases (PsychINFO and SPORTDiscus) to identify relevant studies using the following criteria: (i) Inclusion of at least one self-paced, full-effort performance-focused endurance exercise task, defined as whole-body, dynamic exercise that involved continuous effort and lasted for 75 seconds or longer (McCormick et al., 2018), and (ii) Use of fNIRS to assess cortical hemodyamics over any area of the PFC during exercise. Searches were restricted to article titles of studies published in English language only. As fNIRS use in human research began in the early 1990s (Ferrari & Quaresima, 2012), we also restricted articles to those published between 1990-present. For SPORTDiscus, the search was further restricted to journal articles with abstracts available. For PsychINFO, the search was restricted to articles using human participants aged 18+ years. The following search strings were used in both databases: aerobic OR cycli* OR endurance OR exercis* OR pace OR pacing OR physical activity OR run* OR time trial* OR time-trial* OR training AND cerebral OR cortical OR fNIRS OR functional near-infrared spectroscopy OR near-infrared spectroscopy OR neural OR NIRS OR oxygenation OR PFC OR prefrontal cortex NOT cerebral palsy
1
All articles derived from the search strategy described above were databased in Mendeley Desktop (v1.19.8). To remove any duplicates, we performed a ‘check for duplicates’ within the software. We used the study criteria described above to conduct a careful review of the articles’ titles, abstracts, and keywords. We excluded any articles that fell unambiguously outside these criteria at this early stage. The remaining articles were then subject to full-text review against the study criteria. We provided a reason for any articles excluded from review at this stage. We then screened the references and citations of articles that reached the full-text review stage. Any of these articles that met the study criteria were then also subject to full-text review. Figure 1 outlines this search process and Table 1 outlines the inclusion criteria. Flowchart Detailing the Selection Process. Study Criteria for Review Inclusion. Note. fNIRS = functional near-infrared spectroscopy, PFC = prefrontal cortex. 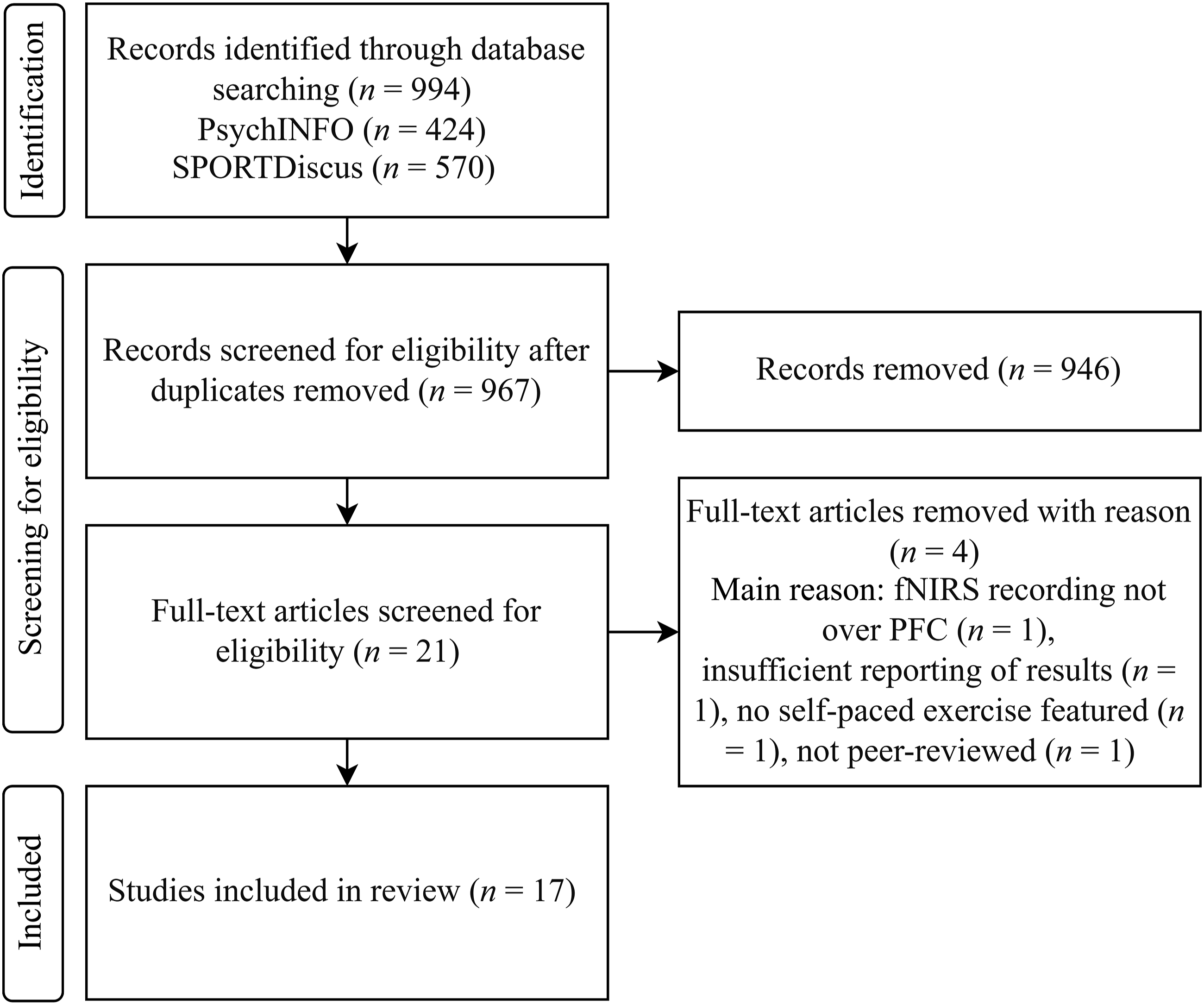
Data Extraction
Following the full-text review stage, we found 17 studies to meet our study criteria. We extracted the following information from these studies: study authorship and year of publication; sample characteristics, including sample size, gender, and proxies of cardiorespiratory fitness level; and key elements of study methodology and findings. Concerning fNIRS, we extracted the following information: brain region of interest (ROI); fNIRS recording methods, including methods of optode placement, source-detector separation distance and baseline recording protocols; and data processing and methods of analysis, including differential pathlength factor (DPF) determination, artifact removal methods and data analysis techniques.
Results and Discussion
Best Practice Recommendations for use of fNIRS for Cerebral Measurement and Number of Reviewed Studies that Adhered to these Recommendations.

Note. Adherence was inferred from sufficient reference to procedures in a full-text review. DPF = differential pathlength factor, EEG = electroencephalographic, fMRI = functional magnetic resonance imaging, fNIRS = functional near-infrared spectroscopy, HHb = deoxyhemoglobin, O2Hb = oxyhemoglobin, SNR = signal-to-noise ratio.
aRarely adopted approach in exercise-based research.
bJudgment is reserved as a subjective decision by researchers made in line with research design.
Positioning and Securing Optodes
Accurate and reliable positioning of optodes is crucial to allow for data recording from the ROI to make sound comparisons within and between participants. The gold standard for optode positioning is co-registration with magnetic resonance anatomical data (Orihuela-Espina et al., 2010). This is a costly procedure and therefore unlikely to be available to most investigators in sport and exercise science laboratory settings. Newer methods allow for the personalization of optode positioning by using measurements from a 3D digitizer (Fekete et al., 2011). This method has yet to become common practice, not least in sport science. The 10–20 electroencephalographic (EEG) system is typically used for optode positioning in sport and exercise research adopting fNIRS (Herold et al., 2018). Ten of the reviewed studies used the 10–20 EEG system for optode placement (Billaut et al., 2010; Bourdillon et al., 2014; Decroix et al., 2016, 2018; Liao et al., 2019; Pires et al., 2016; Robertson et al., 2016; Santos-Concejero et al., 2015; Wingfield et al., 2018, 2019). Seven studies made anatomical references to the PFC (i.e., forehead) but used no recognized system (Fan et al., 2013, 2018; Ferguson et al., 2018; Hamlin et al., 2010; Nielsen et al., 1999; Shannon et al., 2017; Shaw et al., 2020). Therefore, the reviewed studies partially reflect trends seen in the broader exercise-based literature.
Several methods were adopted by the reviewed studies to mitigate the impact of optode movement and to block extraneous light. These included securing optodes in some form of placeholder by black tape or adhesive discs (Billaut et al., 2010; Decroix et al., 2016, 2018; Ferguson et al., 2018; Hamlin et al., 2010; Nielsen et al., 1999; Pires et al., 2016; Robertson et al., 2016; Santos-Concejero et al., 2015; Shannon et al., 2017; Wingfield et al., 2018, 2019), and coverage of optodes with a dark headband or material (Billaut et al., 2010; Decroix et al., 2016, 2018; Robertson et al., 2016; Santos-Concejero et al., 2015; Shaw et al., 2020). These measures are important for ensuring good optode-skin coupling, limiting ambient light reaching detectors, reducing the prospect of optode movement artifacts and improving the wearer’s comfort (Orihuela-Espina et al., 2010).
Source-Detector Separation
Establishing the optimal source-detector separation is an important consideration. This is because a shorter distance results in signals originating from extra-cerebral structures and a longer distance (∼45 mm and above) results in reduced signal quality as little light reaches the detectors (Orihuela-Espina et al., 2010). The majority of exercise-based research has used a 30 mm distance (Herold et al., 2018). A variety of source-detector separations were adopted by the reviewed studies, ranging from 30 mm to 50 mm: 30 mm (Santos-Concejero et al., 2015), 35 mm (Shannon et al., 2017), 40 mm or 41 mm (Bourdillon et al., 2014; Decroix et al., 2016, 2018; Fan et al., 2013, 2018; Ferguson et al., 2018; Nielsen et al., 1999; Robertson et al., 2016; Wingfield et al., 2018, 2019), 45 mm (Billaut et al., 2010; Pires et al., 2016) and 50 mm (Hamlin et al., 2010). Therefore, all distances selected were around or just above distances commonly adopted in neuroscience more broadly and sport science research specifically. However, two studies did not report a source-detector separation distance (Liao et al., 2019; Shaw et al., 2020).
Baseline
Participant instruction, duration and environmental conditions are all important considerations when obtaining baseline data for accurate comparison of changes in experimental conditions from rest values. As Herold et al. (2018) highlighted, no general consensus exists concerning baseline duration. However, a 10–30 second baseline is suggested as a balanced consideration to remove the prospect of random physiological fluctuations for shorter baselines and the prospect of mind-wandering for longer baselines. Pertinently, on this latter point, mind wandering has been implicated in the medial PFC as measured by fNIRS (Durantin et al., 2015). Baseline procedures in the reviewed studies included both 60-second (Robertson et al., 2016; Wingfield et al., 2018, 2019) and 120-second durations (Billaut et al., 2010; Decroix et al., 2016, 2018; Hamlin et al., 2010; Pires et al., 2016; Santos-Concejero et al., 2015). The remaining studies used longer or undetermined baseline durations that often served to provide baseline data for several study measures, including fNIRS (Bourdillon et al., 2014; Fan et al., 2013, 2018; Ferguson et al., 2018; Liao et al., 2019; Nielsen et al., 1999; Shannon et al., 2017; Shaw et al., 2020). Concerning the broader base of exercise research, a 30 -second baseline was most commonly used, although several studies opted for longer durations (Herold et al., 2018). In addition, other techniques were also employed by reviewed studies to reduce movement artifacts and environmental stimuli as confounds during baseline recordings. These included removing auditory and visual distractions (Wingfield et al., 2018, 2019), asking participants to remain silent (Billaut et al., 2010; Decroix et al., 2016, 2018), to restrict bodily movement (Decroix et al., 2016, 2018; Pires et al., 2016) and to close their eyes (Billaut et al., 2010; Pires et al., 2016; Robertson et al., 2016; Santos-Concejero et al., 2015).
Given that posture-dependent differences in cerebral hemodyamics have been observed in the healthy adult brain (Tachtsidis et al., 2004), it is important to ensure that baseline procedures reflect the postural demands of the task. Therefore, for consistency across studies, most reviews recommend a seated position for baseline protocols (Herold et al., 2018). Promisingly, the reviewed studies appeared to use posture-appropriate baseline procedures that reflected the seated nature of most exercise tasks adopted (i.e., cycling). A noted exception is the use of a standing baseline in one study where the exercise task required treadmill running (Billaut et al., 2010). However, a seated baseline was conducted in two studies also using a running task (Santos-Concejero et al., 2015; Shaw et al., 2020).
Differential Pathlength Factor (DPF)
To derive quantitative concentration changes from measurements of light attenuation, the DPF must be known. Therefore, correct calculation of the DPF is vital to minimize the possibility of cross-talk (i.e., incorrect separation of chromospheres; Uludaǧ et al., 2004). Using constant DPF values is suboptimal given that subject-to-subject variability leads to systematic errors in fNIRS signal measurement (Strangman et al., 2002). Therefore, measurement of DPF values as a function of age is recommended (Duncan et al., 1996).
Six of the reviewed studies opted to use an age-dependent DPF (Billaut et al., 2010; Bourdillon et al., 2014; Decroix et al., 2016; Fan et al., 2013, 2018; Shannon et al., 2017), while eight studies opted to specify constant wavelength values (Ferguson et al., 2018; Hamlin et al., 2010; Nielsen et al., 1999; Pires et al., 2016; Robertson et al., 2016; Santos-Concejero et al., 2015; Wingfield et al., 2018, 2019). However, three studies did not report DPF values (Decroix et al., 2018; Liao et al., 2019; Shaw et al., 2020). The use of age-dependent DPF values appears to be rare in the broader field of exercise-based fNIRS work to date (Herold et al., 2018). Therefore, the reviewed studies did not fully follow this trend with a sizeable minority using an age-dependent formula.
Artifacts
There are two main sources of fNIRS signal noise: (i) physiological oscillations (e.g., due to heart rate beats) and (ii) movement-related artifacts (e.g., signal changes evoked by bodily movements; Pinti et al., 2019). Pertinently, these sources of noise are of particular importance to exercise-based research. Filtering methods are typically adopted to reduce the contribution of noise to task-evoked hemodynamic response, therefore improving the signal-to-noise ratio. In addition, several software packages are now available that allow for signal processing through algorithmic checks and visual assessment (for an overview, see Orihuela-Espina et al., 2010). Despite commonplace efforts to improve the signal-to-noise ratio in exercise-based research (Herold et al., 2018), only one study referred to taking these extra measures by using a low-bandpass filter (Pires et al., 2016).
Data Analysis
To account for individual variability, a baseline correction to fNIRS data should be performed prior to analysis. Studies often perform ANOVA’s to inferentially analyze mean values, however, the use of median values may be preferable as they are less susceptible to outliers for datasets with small samples (Herold et al., 2018). This is particularly pertinent for sport science research where small sample sizes are commonplace (Bernards et al., 2017). Using both O2Hb and HHb provides different strengths for analysis and interpretation. As Orihuela-Espina et al. (2010) summarize, O2Hb is more sensitive to cerebral blood flow, but HHb may better reflect oxygen supply and demand. Many studies also commonly report the tissue saturation index (TOI), which is calculated as the proportion of O2Hb relative to tHb concentration. Therefore, the TOI provides a relative balance between oxygen delivery and utilization (Tisdall et al., 2009).
Where appropriate, all studies performed a baseline correction of data to express concentration change from baseline. Mean values were used by 14 studies to perform an ANOVA or non-parametric equivalent (Billaut et al., 2010; Bourdillon et al., 2014; Decroix et al., 2016, 2018; Fan et al., 2013; Ferguson et al., 2018; Liao et al., 2019; Nielsen et al., 1999; Pires et al., 2016; Robertson et al., 2016; Santos-Concejero et al., 2015; Shannon et al., 2017; Wingfield et al., 2018, 2019). However, other analyses were performed, including mixed-effects models (Fan et al., 2018; Hamlin et al., 2010), correlations (Bourdillon et al., 2014; Wingfield et al., 2018, 2019) and linear regression analysis (Hamlin et al., 2010). This eclectic mix of inferential testing perhaps reflects the ongoing debate concerning the best practice for fNIRS data analysis. For instance, the inadequate removal of noise can lead to violations of assumptions for common statistical tests (Huppert, 2016). Only two studies did not report findings for at least O2Hb and HHb (Decroix et al., 2018; Ferguson et al., 2018). However, these studies, alongside other reviewed work, did report findings for TOI (Decroix et al., 2018; Ferguson et al., 2018; Hamlin et al., 2010; Liao et al., 2019; Robertson et al., 2016; Santos-Concejero et al., 2015). Therefore, the data analysis techniques of reviewed studies broadly reflect the predominant use of ANOVA’s to analyze at least O2Hb in exercise-based research using fNIRS (Herold et al., 2018).
Other Considerations
Exercise Task and Protocol
A cycling-based exercise task was used by 13 studies (Bourdillon et al., 2014; Decroix et al., 2016, 2018; Fan et al., 2013, 2018; Ferguson et al., 2018; Hamlin et al., 2010; Liao et al., 2019; Pires et al., 2016; Robertson et al., 2016; Shaw et al., 2020; Wingfield et al., 2018, 2019). However, rowing (Nielsen et al., 1999) and running (Billaut et al., 2010; Santos-Concejero et al., 2015; Shannon et al., 2017) tasks were also used. The completion of a set-distance TT effort was required of participants in 13 studies (Billaut et al., 2010; Bourdillon et al., 2014; Fan et al., 2013, 2018; Ferguson et al., 2018; Hamlin et al., 2010; Liao et al., 2019; Pires et al., 2016; Santos-Concejero et al., 2015; Shannon et al., 2017; Shaw et al., 2020; Wingfield et al., 2018, 2019), while four studies demanded a set-time effort (i.e., 20-minute TT; Decroix et al., 2018; Decroix et al., 2016; Nielsen et al., 1999; Robertson et al., 2016). Many studies use stationary cycling exercise for fNIRS-based research as it is comparatively easy to maintain a consistent posture despite high workloads (Perrey & Besson, 2018), although direct comparison across exercise modes is lacking. Nonetheless, direct comparison between supine and recumbent cycling revealed little difference in PFC oxygenation as measured by fNIRS (Faulkner et al., 2016).
Concerning protocol design, many of the recommendations are inappropriate to apply to the reviewed literature as they relate to neurocognitive domains (e.g., weighing the pros and cons of event-related vs. block designs; Chee et al., 2003). In exercise-based research, most studies have opted to use a central tendency value calculated over distinct time periods (i.e., windows or epochs; Herold et al., 2018). This approach was adopted across most reviewed studies, with only two studies preferring to simply analyze average values for the entirety of the exercise task (Nielsen et al., 1999; Shannon et al., 2017). This may be due to the comparatively short nature of the tasks used in these studies, namely a 6-minute rowing effort and a 3 km treadmill TT.
Region of Interest (ROI)
Ten studies opted to place fNIRS optodes over the dorsolateral PFC, corresponding to Fp1-F3 and Fp2-F4 regions according to the international EEG 10-20 system (Billaut et al., 2010; Bourdillon et al., 2014; Decroix et al., 2016, 2018; Liao et al., 2019; Pires et al., 2016; Robertson et al., 2016; Santos-Concejero et al., 2015; Wingfield et al., 2018, 2019). Three studies did not reference a particular brain ROI but did provide anatomical measurements for determining optode placement (Ferguson et al., 2018; Shannon et al., 2017; Shaw et al., 2020), while four further studies did not provide such measurements (Fan et al., 2013, 2018; Hamlin et al., 2010; Nielsen et al., 1999). It should be noted that research objectives may dictate the necessity to highlight a ROI. For the seven studies not using the 10-20 system, it appears that the assessment of Cox is the dependent variable, with no specific hypotheses concerning a ROI stated. However, Cox during exercise appears region-dependent. For instance, deception regarding the expected length of exercise related to changes in oxygenation between brain areas involved in the central executive network and default mode network (Radel et al., 2017).
The dorsolateral PFC is implicated in the ability to represent and maintain the attentional demands of a given task in working memory (MacDonald et al., 2000). Therefore, the dorsolateral PFC has become a ROI for the psychology of endurance performance, given its plausible role in the ability to integrate external (e.g., competitor behavior) and internal (e.g., perceived exertion) stimuli to aid pacing (Hyland-Monks et al., 2018). Moreover, according to the RAH model previously outlined (Dietrich & Audiffren, 2011), during high-intensity exercise above the ventilatory threshold, lateral areas of the PFC are compromised to preserve more vital brain regions. This leads to a compromise of cognitive control and therefore top-down self-regulation of endurance performance. As a result, fNIRS-based research has investigated the use of self-regulatory interventions to mitigate such effects, including the use of imagery (Tempest & Parfitt, 2013).
Findings of Reviewed Studies
Overview of Reviewed Studies.
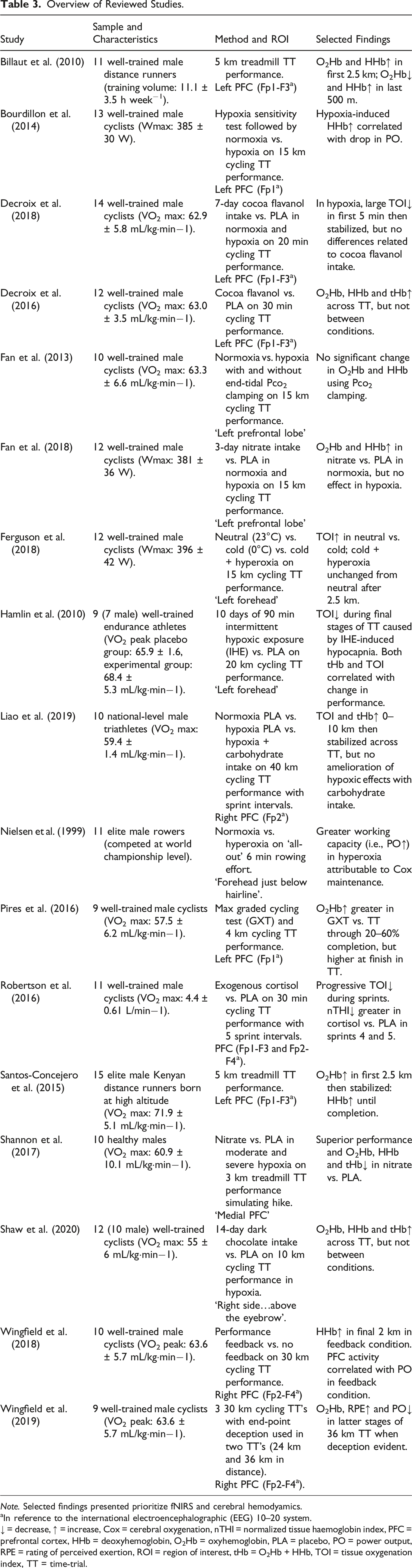
Note. Selected findings presented prioritize fNIRS and cerebral hemodyamics.
aIn reference to the international electroencephalographic (EEG) 10–20 system.
↓ = decrease, ↑ = increase, Cox = cerebral oxygenation, nTHI = normalized tissue haemoglobin index, PFC = prefrontal cortex, HHb = deoxyhemoglobin, O2Hb = oxyhemoglobin, PLA = placebo, PO = power output, RPE = rating of perceived exertion, ROI = region of interest, tHb = O2Hb + HHb, TOI = tissue oxygenation index, TT = time-trial.
Beyond the more exploratory work above, research has also explored the cerebral hemodynamic response to endurance performance with experimental manipulations. For instance, Pires et al. (2016) examined the influence of two VO2max-matched exercise modes on PFC oxygenation in trained cyclists. PFC O2Hb was higher between 20-60% completion during an incremental test compared to a 4 km TT, but O2Hb was higher in the TT at 100% completion. Despite these PFC perturbations, activity in the primary motor cortex was maintained throughout the duration of both exercise bouts. This suggests that there is a preferential preservation of the primary motor cortex during strenuous exercise. Moreover, it supports the notion of PFC and motor cortex integration in exercise regulation. However, reviewed studies most commonly investigated changes in oxygen availability to bodily tissues in order to simulate altitude change. Nielsen et al. (1999) measured hemodynamic response over the forehead of elite rowers completing an all-out 6-minute rowing effort, with inspired oxygen fraction at .21 (i.e., normoxia) versus .30 (i.e., hyperoxia). A greater work capacity (i.e., increased power output) and Cox were observed in hyperoxia, despite no difference in muscle oxygenation across the conditions. The findings suggest that an elevated inspiratory oxygen fraction increased exercise performance related to maintained Cox, rather than to any effect on the working muscles. In addition, Bourdillon and colleagues found hypoxia-induced increases in HHb correlated with decreases in power output during a 15 km TT among trained cyclists (Bourdillon et al., 2014). Taken together, these findings suggest that brain oxygenation may play a limiting role in aerobic exercise capacity. If this notion is correct, there is potential for interventions aimed at improving Cox during exercise and ultimately as a result, exercise capacity.
Further research has investigated environmental effects and ergogenic aids on exercise performance in normoxia, hypoxia and hyperoxia. Hamlin et al. (2010) asked trained endurance athletes to complete 10 days of intermittent hypoxic exposure (IHE) training prior to performing a 20 km TT. During the final stages of the TT, IHE-induced elevations in muscle O2Hb and reductions in cerebral TOI were observed, with no subsequent effect on performance. The reduction in oxygenation may relate to cerebral vasoconstriction through reduced CO2 levels (i.e., hypocapnia) due to IHE. This is supported by moderate-to-strong relationships observed between TOI, tHb and performance. Therefore, the lack of improvement may relate to a ‘balancing out’ of beneficial adaptations to muscle, but not brain. Fan et al. (2013) examined the effect of clamping partial pressure of end-tidal CO2 on cerebral perfusion and performance during a 15 km TT by trained cyclists in severe hypoxia. Clamping normalized PFC oxygen delivery to normoxic values. However, clamping did not result in a performance improvement in hypoxia. Therefore, the authors refute the hypothesis that cerebral deoxygenation caused by hypoxia-induced hypocapnia reduces exercise performance. Following on, Ferguson et al. (2018) assessed the impact of hyperoxia in ameliorating the negative impact of hypothermia on 15 km TT performance in trained cyclists. Under conditions of hypothermia, Hyperoxia restored performance levels to that seen at neutral temperatures. Although cerebral TOI was higher at neutral temperatures compared to cold conditions, TOI levels were similar between neutral and cold plus hyperoxic conditions after the 2.5 km point. The findings suggest that the restoration of performance via hyperoxia is related to oxygen availability.
Research has attempted to explore the possibility of ‘boosting’ Cox to aid cognition and endurance performance via cocoa flavonoids, which possess nitric oxide-induced vasodilatory properties (Socci et al., 2017). Decroix et al. (2016) tested the impact of cocoa flavanols on 30-minute TT performance in trained cyclists. Although PFC oxygenation increased at baseline in the experimental group, this was not reflected in superior Stroop task performance pre-exercise. Cocoa flavanol intake did not significantly influence the exercise-induced hemodynamic response, which likely overruled any smaller detectable impact of cocoa flavanols on Cox during exercise that was observed at rest. Extending this research, Decroix et al. (2018) investigated 7-day cocoa flavanol intake on 20-minute TT performance in both normoxia and hypoxia among trained cyclists. Although cocoa flavanol intake raised PFC oxygenation levels at rest, no differences were observed in TT performance or oxygenation levels during exercise. Most recently, Shaw et al. (2020) used trained cyclists to assess the impact of 14-day dark chocolate (high in flavonoids) intake on 10 km TT performance at simulated altitude. Although some metabolic benefits to dark chocolate intake were observed, no benefits to PFC oxygenation or performance were found.
Dietary nitrate has been shown to restore metabolic function and muscular endurance performance to normoxic levels at simulated altitude (Vanhatalo et al., 2011), but the effects on Cox during whole-body, endurance performance remain unclear. Fan et al. (2018) tasked trained cyclists with completing a 15 km TT in normoxic and hypoxic conditions after 3 days of dietary nitrate intake. Dietary nitrate selectively improved PFC oxygenation during exercise in normoxia, but these effects were not extended to hypoxia. This lack of change in hypoxia may relate to compensatory mechanisms to protect the oxygen supply to vital homeostatic control centers. A further study investigated nitrate-rich beetroot supplementation on 3 km treadmill TT (set at 10% gradient) performance in both moderate and severe hypoxia (Shannon et al., 2017). PFC O2Hb, HHb and tHb were lower in the beetroot condition, which may reflect differential PFC perfusion related to superior performance via beetroot intake. This may also indicate a lower cerebral blood volume or oxygen extraction rate. Cerebral deoxygenation is hypothesized to compromise EF and play a limiting factor in exercise tolerance (Subudhi et al., 2009).
Aside from flavonoid- and nitrate-rich interventions, others have tested the impact of carbohydrate and exogenous cortisol on exercise performance and PFC hemodyamics. Liao et al. (2019) examined the potential for carbohydrate intake to ameliorate the effects of hypoxia on 40 km TT performance in competitive triathletes. Carbohydrate intake failed to ameliorate hypoxia-induced deleterious effects on TT performance and PFC hemodyamics. Furthermore, as exogenous cortisol has been shown to increase risk-taking behavior (Putman et al., 2010), Robertson et al. (2016) wanted to explore the impact of exogenous cortisol on trained cyclists’ performance in a 30-minute TT featuring five 30-second sprint intervals. A greater reduction in normalized tissue haemoglobin index (nTHI; O2Hb + HHb expressed as change from baseline in a.u.) was observed in the latter sprint intervals in the cortisol condition, indicating greater PFC desaturation and blood volume. However, no differences in TT performance were found across conditions.
The nascency of the intervention-based research presented above offers an opportunity to learn from and build on this work moving forward. Firstly, it is suggested that testing at a more ecologically valid genuine high altitude or in hypobaric hypoxia may elicit different physiological responses than normobaric hypoxia (Shannon et al., 2017; Shaw et al., 2020). However, both normobaric hypoxia and hypobaric hypoxia have been observed to induce broadly similar responses to terrestrial altitude, and therefore may be considered acceptable surrogates (Woods et al., 2017). Following on, it is also important to consider gender differences in response to interventions. For instance, Fan et al. (2018) refer to unpublished data showing an enhanced cerebrovascular response to nitrate intake in male, but not female participants. Likewise, one should consider fitness status. A meta-analysis found a small ergogenic effect of nitrate intake on exercise performance in non-athletes, but this was not extended to athlete populations (Campos et al., 2018). Lastly, although the scope of this review is limited to the PFC, it is feasible that interventions elicit more global changes in the brain during exercise. For instance, flavonoids have been observed to increase cerebral blood flow in several areas of the brain, including the anterior cingulate cortex (Lamport et al., 2015). Interestingly, functional adaptations to the anterior cingulate cortex through aerobic training is theorized to be a possible mechanism for enhanced resistance to mental fatigue effects on exercise performance (Martin et al., 2016).
There is a rich history of work that highlights how feedback deception or removal can influence subjective experience (e.g., perceived exertion), exercise economy and endurance performance (Jones et al., 2013). Wingfield et al. (2018) examined the influence of performance and distance feedback blinding on 30 km TT performance by trained cyclists. When feedback was provided, power output was higher in the final 2 km of the TT and was strongly correlated with HHb. These findings suggest some neural influence of the PFC over pacing by top-down regulation of physiological reserve in anticipation of an end-spurt. Complementing this research, Wingfield et al. (2019) investigated the performance of trained cyclists across three ‘30 km’ TT’s. However, feedback was withheld from participants and two trials were 24 km and 36 km in length. Rating of perceived exertion was significantly higher in the latter half of the 36 km versus 24 km TT. Although not significant, O2Hb was higher at 70% completion in the 36 km versus 24 km TT. The results point to the PFC increasing perceived exertion to reduce the risk of catastrophic failure by driving down perceived exertion once end-point deception was detected. Therefore, while performers may have a pre-planned pacing strategy in mind, subconscious processes may also help govern physiological resources to avoid premature fatigue (Micklewright et al., 2017).
Conclusion
Promisingly, the small but growing body of literature reviewed here largely adopted some, but not all of the best practice guidelines for fNIRS use more widely and for exercise-based research specifically. However, there were several important guidelines that not all studies adopted, or at least had omitted from reporting in their method sections. As advances in sport performance are predicated on ‘marginal gains’, methodological rigor when using fNIRS technology is important to ensure sensitivity to detect meaningful changes in cortical activity. Following on, the body of literature presented here suggests that some degree of homeostatic disturbance (i.e., cerebral deoxygenation) is tolerated under high workload, but cerebral oxygen availability is a limiting factor on aerobic exercise performance. Furthermore, the PFC would appear to play a key role in integrating performance feedback to govern the self-regulation of endurance performance. Although no benefits were observed for flavonoids, nitrate-rich interventions showed some benefits to exercise performance and PFC perfusion. However, future work that addresses the limitations of the research presented here may provide a more conclusive picture. Finally, studies reviewed predominantly used trained and elite-level male athletes, with only four participants having been female. Therefore, more research is needed to ascertain the effects of both fitness status and sex on cerebral perfusion during self-paced exercise.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
