Abstract
Biostimulants are products that stimulate plant growth independently of the product's nutrient content. Efficacy testing is crucial given that many commercial products on the market have been shown to be ineffective. In the European Union, biostimulants fall under the Fertilizer Products Regulation, which stipulates that “the plant biostimulant shall have the effects that are claimed on the label for the plants specified therein.” We suggest that this definition of product efficacy is inadequate, and should be expanded to include both the mode of action and the quality of the product. The main types of biostimulants are non-microbial and microbial biostimulants, which we describe briefly. We then evaluate the European Committee for Standardisation (CEN) EN 17700-1 Standard that provides guidance on efficacy testing. This CEN Standard permits claims without field trials, lacks mandatory controls (e.g., sterile-carrier comparisons), and allows extrapolation from a limited set of specific crops to a large and diverse group of crops. Consequently, products can reach the market that are often ineffective, as demonstrated by numerous studies showing poor colonisation by microbial biostimulants, small and variable yield responses, and occasional phytotoxicity. We recommend the CEN standard be strengthened, incorporating lessons from the European and Mediterranean Plant Protection Organisation, FAO, and recent efficacy-testing proposals, and reconsidering CEN's role in standard-setting to ensure reliable, evidence-based products for farmers.
Keywords
Introduction
Farmers are faced with a plethora of products that promise to boost crop performance but that are not fertilisers or pesticides. These products are commonly grouped under the term “plant biostimulants”, although nomenclature varies regionally. For example, they are referred to as supplements in Canada, bio-inputs in Brazil, and biological agricultural products in Australia (Table 1). We note that the “bio” in biostimulants does not refer to the nature of the product (as also inorganic silicon and phosphite fertilisers are treated as such), but only to the nature of the target, to stimulate organisms (i.e., plants) as biological entities. In that sense, the term “plant biostimulant” is in fact a tautology.
The EU Fertiliser Products Regulation 2019/1009 (FPR, 2019) defines a plant biostimulant as “A product stimulating plant nutrition processes independently of the product's nutrient content with the sole aim of improving one or more of the following characteristics of the plant or the plant rhizosphere: (a) nutrient use efficiency, (b) tolerance to abiotic stress, (c) quality traits, (d) availability of confined nutrients in soil or rhizosphere.” The FPR (2019) was introduced to harmonise the definition of biostimulants across markets in Europe and to allow producers to affix a Conformité Européenne (CE) i trademark to their products; products sold without the CE trademark must instead comply with national regulations for each country.
The FPR (2019) distinguishes non-microbial and microbial biostimulants (Table 1). Currently, only N₂-fixing bacteria (both symbiotic Rhizobium bacteria and two genera of free-living N₂-fixing bacteria: Azotobacter and Azospirillum) and mycorrhizal fungi are included as microbial biostimulants under the regulation (FPR, 2019). By contrast, the regulations of other countries include all types of biostimulatory microorganisms (Table 1). Other microbial formulations, for example, Bacillus and Trichoderma spp. will likely be incorporated in the FPR (2019) in due course (Castiglione et al., 2021; EBIC, 2022), underscoring the need for a robust regulatory framework (O’Callaghan et al., 2022).
Definition and regulation of the efficacy of biostimulant products in selected regions of the world.
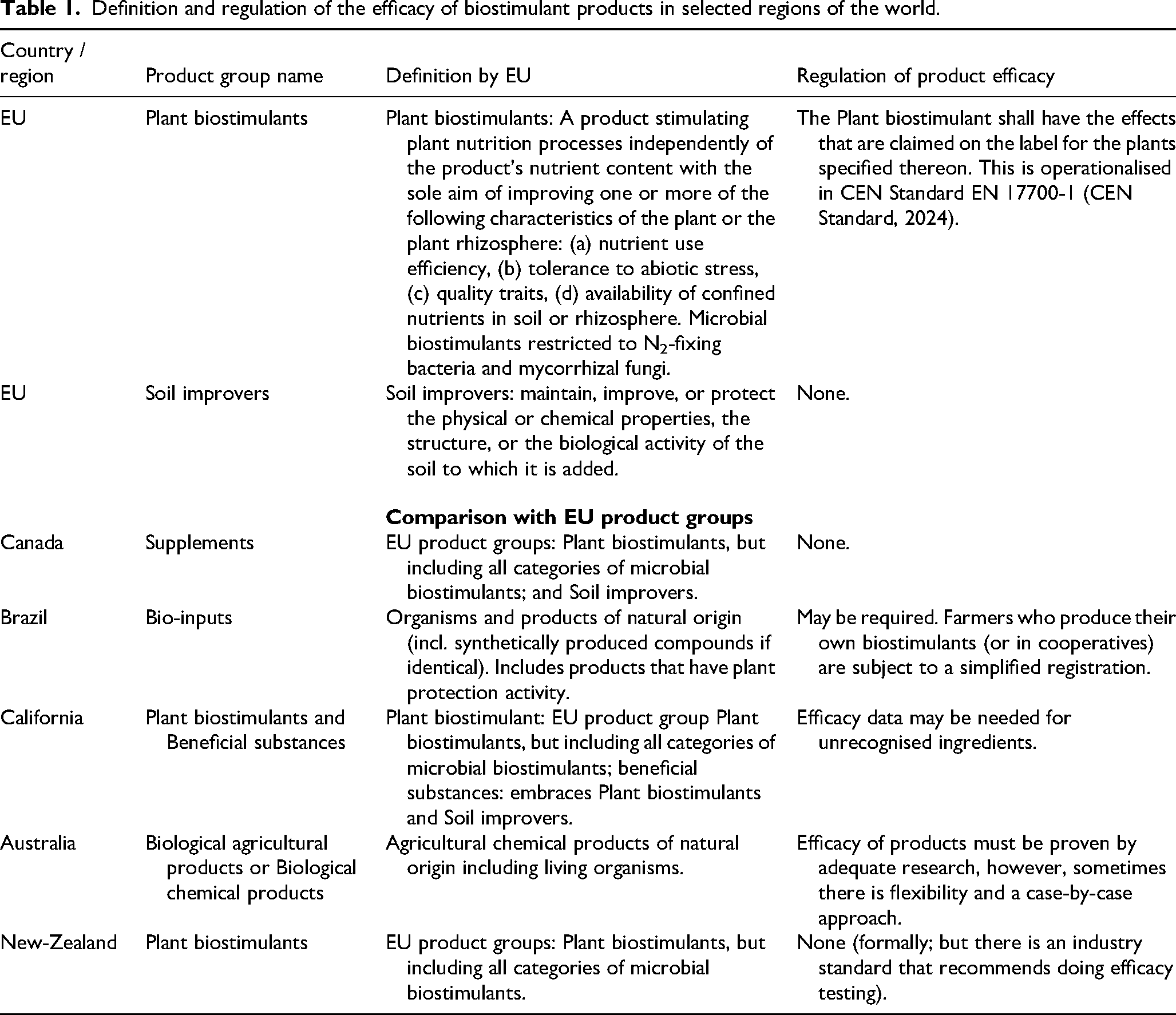
Registration of biostimulants generally requires that the product is safe for users, consumers, and the environment. In addition, the FPR (2019) stipulates that “the plant biostimulant shall have the effects that are claimed on the label for the plants specified therein,” implying that their efficacy must be demonstrable. While efficacy is also a prerequisite for registration in other regions, the stringency of the requirement varies (Table 1). In Brazil, for example, efficacy testing is minimal when farmers produce their own biostimulants (Goulet, 2023). Australia mandates efficacy testing for biostimulants, which are regulated as “biological agricultural products” ii on a case-by-case approach, acknowledging that “efficacy of products must be proven by adequate research, but flexibility may be applied.” In Canada, efficacy testing is not required for biostimulants as “supplements”. iii This regulatory divergence raises questions concerning the necessity of efficacy testing of biostimulants. Critics of mandatory efficacy testing argue that it slows innovation due to the time and cost involved, and that farmers can judge themselves whether products are effective. Proponents of efficacy testing argue that it reduces the number of ineffective products entering the market and thereby prevents farmers from wasting money.
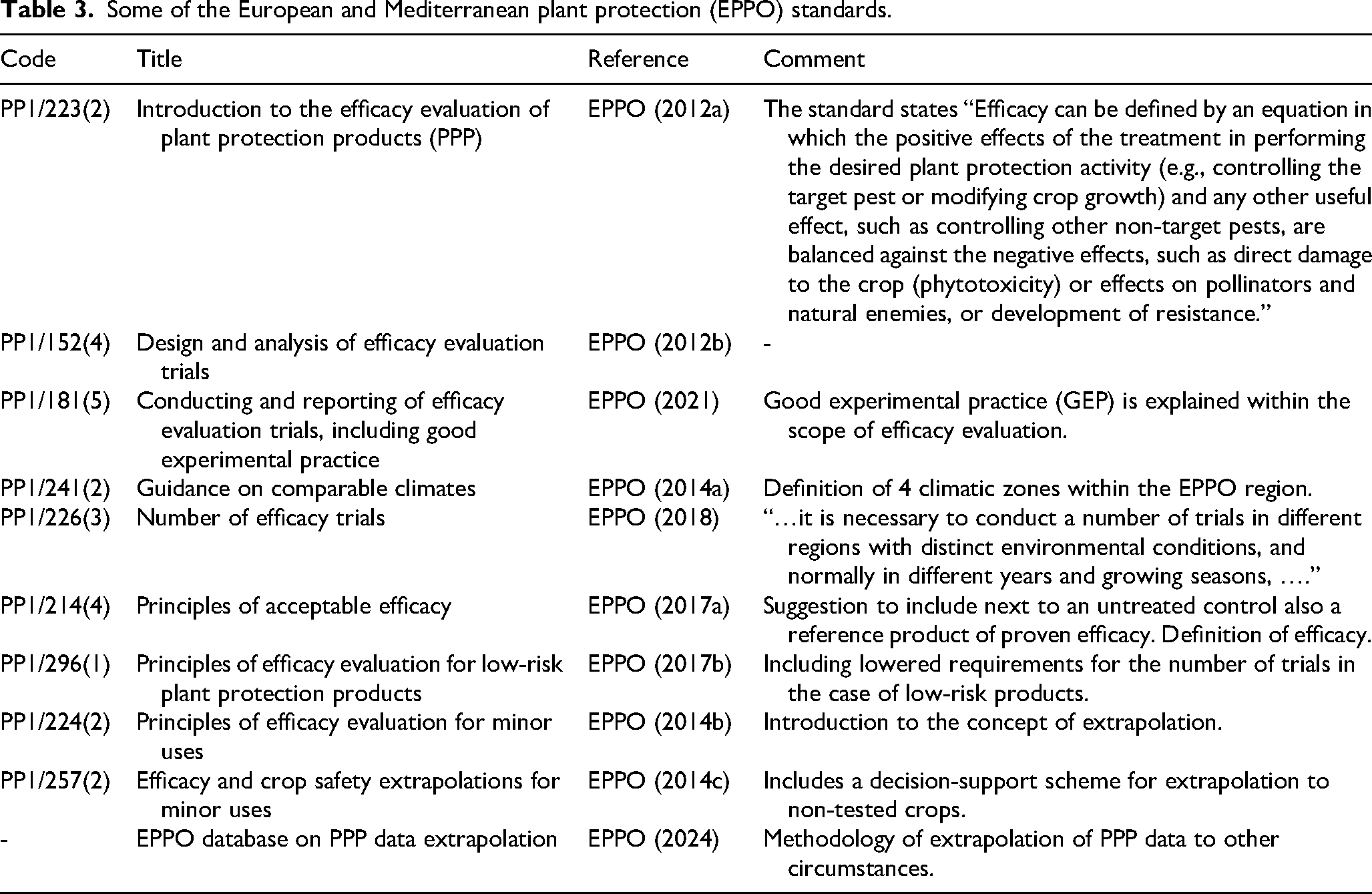
To address the lack of a harmonised efficacy framework in Europe, the European Commission tasked the European Committee for Standardisation (CEN) with developing standards for plant biostimulants. Technical Committee 455 (CEN/TC 455) produced the European Standard EN 17700-1 “Plant biostimulants – Claims – Part 1: General principles” (CEN Standard, 2024). Complementary parts of the standard cover sampling, analysis of human pathogens, assessment of abiotic contaminants, and labelling. iv These CEN Standard (2024) principles currently constitute the most detailed description of the requirements for demonstrating biostimulant efficacy, specifying experimental conditions, the minimum number of experiments required, and the necessary statistical handling of the data.
The aim of our paper is threefold:
First, we explore what “efficacy” means in relation to biostimulants;
Second, we review briefly the different types of biostimulants and demonstrate that the efficacy of many biostimulants is questionable, thereby justifying stricter efficacy regulation;
Third, although the CEN Standard (2024) represents the most comprehensive effort to date to regulate biostimulant efficacy, we argue that it falls short of guaranteeing that claimed effects are reliably delivered.
We conclude that the European Commission should not have assigned the task of developing the standard to CEN, as it primarily formulates a standard based on agreements between companies rather than one that serves the public domain.
What is meant by “efficacy” of biostimulants?
The FPR (2019) uses “efficacy” to imply that the product should have the desired effect on plant growth. Yet experience shows that for a biostimulant or microbial inoculant to have a consistent impact on plant growth, it must have a proven mode of action and product quality must be sufficient to achieve the desired effect. In the case of non-microbial biostimulants, quality implies that the product sold contains a sufficient concentration of the active ingredient. For microbial biostimulants, quality means that the product formulation should deliver the microorganism in sufficient numbers. In all cases, the products should be free of human and plant pathogens and abiotic contaminants. Thus, we conclude that the definition of product efficacy should be expanded to include both the mode of action and the quality of the product, two aspects that need different approaches to regulation as we discuss below.
Major types of biostimulants
As already discussed, the two types are non-microbial and microbial biostimulants (du Jardin, 2015; Table 2). We discuss each of these in turn. In practice, commercial products sold to farmers often contain mixtures of different biostimulants.
Some product groups of biostimulants, based on du Jardin (2015).

Referred to as “Botanicals” by du Jardin (2015).
Non-microbial biostimulants
A number of different product groups of non-microbial biostimulants can be distinguished (Table 2). The majority are organic compounds either extracted from soil, seaweed or plants, or created in the laboratory such as chitosan or protein hydrolysates. Some are inorganic in nature, largely diluted solutions of micronutrients such as zinc, manganese or silicon.
There is general agreement that both microbial and non-microbial biostimulants can have strong and significant plant-stimulating effects; however, their efficacy depends on many factors, including the plant species, fertilisation regime, soil conditions, climatic factors, and weather conditions (du Jardin, 2015; Pasković et al., 2024; Yuan and Dickinson, 2023; Li et al., 2022). This means that efficacy tests must account for these sources of variation. In an extensive review of applied research conducted in Croatia on various, mainly non-microbial biostimulants, Parađiković et al. (2019) concluded: “… the effects [of biostimulants] on plants may depend on dose, time of treatment, growth conditions, and plant species.” Cultivar-dependent results have also been reported (e.g., Golin et al., 2024; Zhang et al., 2025). Clément et al. (2023) compared ten commercially available biostimulants: at high salinity an effect range from severely detrimental to highly beneficial was observed.
Given the complex and variable nature of biostimulant performance, it is evident why conclusions about their efficacy are difficult to make. Often, if not usually, it is unknown how and where biostimulants can be applied so that they are effective. This is the reason why scientists very often conclude that more research is needed to understand variability in biostimulant efficacy. In a review on efficacy of biostimulants (here named biological amendment products), Abbott et al. (2018) concluded that insufficient adequate scientific research is done on efficacy of biostimulants, due to the wide variety of products that are on the market, and they warn that this could lead to proliferation of unsubstantiated assertions of efficacy. Thus, the current understanding of many non-microbial biostimulants suggests that their efficacy is inconsistent. This must be considered when evaluating product efficacy. The key question is: can farmers reliably trust producers’ claims for their specific cropping system, soil condition, climate, and growth conditions?
Microbial biostimulants
Arbuscular mycorrhizal fungi
The important role of arbuscular mycorrhizal fungi (AMF) as symbionts of the majority of higher plants is well-established; the symbiosis is key to extending the volume of soil explored to facilitate uptake of immobile nutrients such as phosphorus and zinc (Smith and Read, 2008). The majority of arable and fruit crops is heavily dependent on AMF. AMF species generally have a broad host spectrum so that a single AMF product can potentially be used for several crops. A few woody crops form other kinds of mycorrhizal symbioses (ectomycorrhiza, ericoid mycorrhiza). Agronomically important non-mycorrhizal plants are members of the Brassicaceae such as cabbage and mustard.
Crop responses to the application of AMF in the field are mixed at best. A lack of response to inoculation with AMF can be for three main reasons: a) the natural inoculum potential of AMF present in the soil is sufficient for adequate root colonisation or is competitively superior to the inoculum; b) the inoculum contains a strain of AMF equal to or less effective than the natural soil population; or c) the number of AMF propagules added through inoculation is too small to provide effective root colonisation (i.e., a problem of inoculant quality).
Studies on field application of AMF inocula indicated beneficial effects for potato (Hijri, 2016), with a 10% yield increase and a benefit : cost ratio around 6, and cereals (Zhang et al., 2019) with a yield increase of 16%. The latter study also noted that almost 25% of the studies did not show a positive effect. Both analyses were based on studies where researchers provided the mycorrhizal inoculum. Recent reports on commercial AMF inocula provide a bleak picture. Salomon et al. (2022) investigated 28 commercial AMF products. In 21 out of 25 inoculants tested in the laboratory, application of the commercial inoculum did not result in mycorrhizal root colonisation, and in 20 out of 25 cases no increase in plant growth was observed. Subsequent studies confirmed the great concerns over product quality. Koziol et al. (2024) tested 15 commercial mycorrhizal inocula and noted that in pot experiments eight did not result in mycorrhizal root colonisation and, even more strikingly, a positive plant growth response was only noted with one inoculum. It was therefore not surprising that there was no significant difference between the treatment with live inoculum and autoclaved inoculum. In addition, five out of these 15 inocula contained the plant pathogen Olpidium and four of these had detrimental effects on plant performance. A subsequent study by Koziol et al. (2025) noted a crop yield increase in response to commercial inocula (+19%) which was much smaller than the yield increase in response to inocula derived from the laboratory (+38%). The authors also noted that the beneficial effect of commercial inocula was overestimated because in 6% of the cases complete crop failure occurred. Considering that about half of these commercial inocula had a positive impact on plant performance, despite zero or negligible mycorrhizal root colonisation, one wonders to what extent the fungi were responsible for plant growth stimulation. Again, inoculum purity was an issue, and around 20% of the inocula contained Olpidium. Product labelling is a further issue of concern. Vahter et al. (2023) noted a considerable discrepancy between the AMF species claimed to be present and the actual species occurring in these products.
N2-fixing bacterial inoculants
With respect to microbial inoculants containing N2-fixing bacteria, a clear distinction must be made between rhizobial inoculants that fix N2 in symbiosis with legumes, and inoculants of free-living or endophytic N2-fixing bacteria that are claimed to support N2-fixation in cereals and other non-legumes.
There is a long history of research that demonstrates the efficacy of rhizobial inoculants; the largest market for which is soyabean. Rhizobia is a generic term for a group of bacteria that can colonise root nodules on plants belonging to the legume family (Fabaceae). The most common rhizobial genera are Rhizobium and Bradyrhizobium, although bacteria belonging to at least eight genera can nodulate legumes, and the diversity of rhizobia continues to increase as the symbioses of wild species are studied (Martínez-Romero et al., 2024). There is a high degree of specificity between legume species and their rhizobia in both the ability to form nodules and in the effectiveness in N2-fixation (Giller, 2001). This means that different inoculant products are needed for each legume species. Although “cross-inoculation groups” can be identified where single rhizobium strains can nodulate multiple legume species, the effectiveness in N2-fixation cannot be guaranteed.
A variety of formulations of rhizobial inoculants are available (Deaker et al., 2016). The most common type is a solid inoculum on a carrier of finely-ground peat, although granular, liquid, and freeze-dried products are also sold. When a solid carrier such as peat or clay is used, it first needs to be sterilised to reduce the number of contaminants (Deaker et al., 2016). This is often achieved through pasteurisation although this rarely achieves complete sterilisation. Gamma sterilisation can produce a completely sterile carrier. When sterilisation is incomplete this tends to reduce the effective shelf-life of the inoculant as contaminants gradually overgrow the inoculant strain.
Clear quality standards need to be met for rhizobial inoculants to be registered and sold in Australia (Deaker et al., 2016) and Brazil (de Souza et al., 2019), but not in Europe. Quality standards specify a numerical standard for the number of live rhizobia in the inoculant, in Australia for peat inoculants this is >1 × 109 cells per gram when produced and >1 × 108 cells per gram at the expiry date claimed for the shelf-life (Deaker et al., 2016). The quality standards also often specify the maximum titre of contaminant bacteria allowed, generally <1 × 106 cells per gram.
By contrast with rhizobium inoculants for legumes, the claims made that microbial inoculants can satisfy a large proportion of the nitrogen requirement of cereals and other non-legumes do not stand up to scrutiny (Giller et al., 2025). The two categories of free-living N2-fixing bacteria currently included in the FPR (2019), Azospirillum and Azotobacter, have been shown to stimulate crop growth through production of plant hormones such as indole acetic acid (IAA) but not through N2-fixation (Giller and Merckx, 2003; Giller et al., 2025). Nevertheless, companies make wild claims that a wide range of inoculant bacteria can enter non-legume plants through stomata, colonise all plant cells, and fix N2 from the atmosphere at no energy cost to the plant, without providing evidence to support these claims. Providing such evidence is compulsory under the FPR (2019). This regulation stipulates, as mentioned above, that “the plant biostimulant shall have the effects that are claimed on the label for the plants specified therein.” A range of different commercial products said to contain N2-fixing bacteria were tested on maize, spring wheat, sugar beet, and oilseed rape across ten states in the North-Central Region of the USA by a network of land-grant universities (Franzen et al., 2023). Significant increases in crop yields were observed in only two out of 61 trials. Field tests of 14 different commercial biological products conducted by the University of Delaware found no significant increases in maize yields (Adkins and Miller, 2023). Thus, independent field tests do not support the claims made by the product manufacturers. Nevertheless, such microbial inoculants are sold and used on millions of hectares in North America and are aggressively marketed in Europe.
Plant growth promoting rhizobacteria (PGPR)
Another category of microbial biostimulants is formed by the plant growth promoting rhizobacteria (PGPR). Some of the free-living N2-fixing bacteria, including Azotobacter and Azospirillum which are mentioned in the FPR (2019), have been shown to stimulate crop growth through production of plant hormones such as indole acetic acid (Kennedy, 2003; Giller and Merckx, 2003), and can be considered to be PGPRs. Some strains of Pseudomonas fluorescens have been shown to produce iron-chelating siderophores which contribute to their ability to promote plant growth (Kloepper et al., 1980). A further mechanism by which inoculant bacteria might enhance plant growth is through making soil phosphorus more available for plant uptake – the so-called phosphate solubilising bacteria – although the significance of this mechanism is contested (Barrow, 2025).
Apparently, there is a gap between the claims that companies make for the products they sell to farmers and their reliability based on scientific evidence. Because the newly released CEN Standard (2024) provides the most comprehensive guidance on efficacy testing, we examine its provisions in greater detail in the following section.
Our concerns regarding the CEN standard
Below we outline our principal concerns regarding the CEN Standard (2024), which specifies the requirements for demonstrating the efficacy of biostimulants as defined by the FPR (2019).
Experimental issues
According to the CEN Standard (2024), efficacy claims for a biostimulant can be made even if no field trials have been conducted, and, in the case of soil-amended biostimulants, even without testing on plants. Both practices are undesirable, as farmers need assurance that the biostimulant works effectively on their crops under practical conditions.
The CEN Standard (2024) specifies no requirements about controls in efficacy experiments. A control with the formulation only must be included when testing microbial biostimulants (O’Callaghan et al., 2022). For instance, arbuscular mycorrhizal fungal inoculum is sometimes coated on bentonite, which can affect soil physical properties like water retention which could promote plant growth, so checking only for nutrient content is inadequate. In order to exclude any effects not due to addition of the microorganism, a control with the autoclaved or sterilised inoculum should be mandatory. In the case trials are done under controlled conditions, the CEN Standard does not describe the soil or growing medium to be used, which should be the same (unsterilised) soil or growing medium as used in practice.
Efficacy experiments are allowed to be conducted by the applicant company (CEN Standard, 2024). This is undesirable because such companies can be strongly biased in reporting effectiveness. Rather, experiments should be conducted by independent, certified research institutions. Experiments to be used for product registration should be entered into a database before they are conducted to ensure that all results, positive or negative, are made public (Nosek et al., 2018). By providing a time-certified document in the public domain that states the research questions, the hypotheses to be tested, the way(s) in which data are gathered, analysed, and reported, one considerably reduces (unintended) risks associated with, for example, reframing of hypotheses after results are known (postdicting rather than predicting), selective reporting of results or even non-reporting of non-significant or negative results, and p-hacking (data manipulation until at least one significant result is obtained).
Crop grouping and minimum number of trials
The CEN Standard (2024) allows efficacy to be claimed for one of three large crop groups (broadacre crops, woody perennials, and other crops; listed in Appendix 1) after reporting efficacy for only two of the crops in that group. For example, if successful trials are conducted only on two broadacre crops (such as wheat and rye), the CEN Standard would allow an efficacy claim for all crops within that group – even for crops like potato and sugar beet, which may respond differently. Both the broadacre and the other crops categories contain crops that do or do not form functional arbuscular mycorrhizal symbioses (e.g., the Brassicaceae) and even for the plants that do, there is a wide range in mycorrhizal benefit, depending on plant traits like root diameter and root hair length and abundance (Hetrick, 1991). The category for the woody perennials includes two species (chestnut and hazelnut) that form ectomycorrhizal and two species (blueberry and cranberry) that form ericoid mycorrhizal symbiosis. Under the current testing scheme for crop categories, testing of the AMF product on only two species suffices. However, we can safely predict that adding ectomycorrhizal or ericoid mycorrhizal fungal inoculum will fail for all other listed woody perennials that associate with arbuscular mycorrhizal fungi. Likewise, using AMF inoculum will fail for ectomycorrhizal and ericoid mycorrhizal plants.
Similarly, it would be possible to claim efficacy of symbiotic N2-fixing biostimulants for non-N2-fixing crops such as potato or sugar beet if efficacy has been shown for two legume crops (there are in fact only nine N2-fixing legumes in the broadacre category). Given the high degree of specificity between rhizobial strains and legumes, the use of crop categories makes no sense: the inoculant should be tested on the target crop for which it is sold. Ultimately, for microbial inoculants we need a crop classification system that links crop properties to specific microbial mechanisms. As developing this will take time, an interim measure could be to use a more natural crop grouping as handled by the Australian Pesticides and Veterinary Medicines v . EPPO (2014b) provides guidelines on how to pursue efficacy testing in minor crops (Table 3).
While three soil pH and four soil type classes are distinguished in the CEN Standard (2024), efficacy can be claimed even if trials have not been conducted on different types of soil. These factors will likely affect the efficacy of many soil-amended biostimulants. This will also be the case for soil organic matter content, which we propose to include (e.g., soil organic matter classes <2%, 2–5% and >5%). For microbial inoculants, the background soil fertility is a further factor to be considered because excess nitrogen frequently reduces the likelihood of crop response to N2-fixing rhizobia and excess phosphorus generally reduces arbuscular mycorrhizal benefit. Another issue is that climate zones have not been considered in the case of field trials. Furthermore, for horticultural crops cultivated on growing media, testing should be conducted on a type of substrate that is representative for the practical cultivation of the crop under investigation.
Data recording
The data to be provided are not precisely described in the CEN Standard (2024) and are too limited. For example, the CEN Standard states that phytotoxicity has to be recorded in the trials. In addition, the standard should require information on the dose at which phytotoxicity is apparent, and for biostimulants applied above-ground, effects on adjacent crops should also be given.
Statistics
According to the CEN Standard (2024), unrealistically lax P values of 0.10 (for experiments in controlled environments) and 0.15 (for field experiments) are sufficient to claim efficacy. Rather, P values of respectively 0.05 and 0.10 should in our opinion be the maximum acceptable (Sokal and Rohlf, 2011).
In the CEN Standard, a biostimulant is declared effective if there is a significant effect compared to the control. Since statistical significance can be influenced by the number of replicates, effect size is also essential (Nakagawa and Cuthill, 2007; Sullivan and Feinn, 2012). In their proposals for efficacy testing of biostimulants and biostimulant-like products (“enhanced efficiency fertilisers”) Neuhoff et al. (2024) and Lyons et al. (2025) advocated including effect size. However, mandating a minimum effect size may be too difficult, since effects are often small. We suggest that, next to statistical significance, the range of observed effect sizes should be reported on the product label.
Absence of plant protection effects
FPR (2019) and the CEN Standard (2024) do not prescribe methods for demonstrating the absence of a plant-protective effect. Yet, if a product claims to have such an effect, it is classified as a plant-protection product (PPP) and must comply with the corresponding regulatory framework. In recent years, for many biostimulants their plant-protection claims have been withdrawn, precisely to avoid triggering the far more stringent PPP registration process. This issue is already relevant considering the increased attention for non-nutritional benefits of the arbuscular mycorrhizal symbiosis (Delavaux et al., 2017) and it will become increasingly salient as microbial biostimulants based on Trichoderma and Bacillus spp. are incorporated into the FPR. Moreover, even non-microbial biostimulants, such as algal extracts or humic substances, may have plant-protection activity (Agarwal et al., 2021; Faccin and Di Piero, 2022). Consequently, distinguishing between biostimulants and PPPs will be challenging. Brazil's regulatory approach reflects this complexity by permitting biostimulants to exhibit plant-protective effects under certain conditions.
Absence of routine quality testing
Under the current regulations there is no requirement for spot-testing of products to check whether they consistently meet the criteria stated on their labels. Once a product has been registered, it can be sold without any further checks on quality. Independent testing is particularly important for microbial inoculants, which can deteriorate in quality during handling and transport if conditions along the supply chain are not ideal. Microbial inoculants are likely to lose efficacy over time because contaminants may increase in abundance, outcompeting the intended inoculum. A system of routine testing of microbial inoculant quality and quantity by an independent body of accredited institutions is required to ensure consistency of the quality of products being sold.
Application window for biostimulants
Microbial and non-microbial biostimulants do not work under all conditions. Plant responses to such products are said to be idiosyncratic or context dependent. Both are descriptive terms, and such terms do not help farmers to decide what, when, where, how, and on what crop to apply a certain product. It is therefore reasonable to expect from producers a form of specification of the context (environment, soil, plant species and genotype, management) that allows minimal prediction whether it is likely that a product will succeed or fail, i.e., will enhance the performance of the crop or not. For microbial inoculants of which the mode of action is well-known, specification of that context can yield a number of general principles. For inoculation with arbuscular mycorrhizal fungi the context includes soil fertility, especially phosphate availability, the extent to which the soil will be regularly disturbed, and the use of fungicides. If these conditions are unfavourable, application of commercial AMF inoculum is very unlikely to succeed. Next to these unfavourable factors, the quality, quantity and competitive ability of indigenous inoculum should be added, although these properties may be much more difficult to assess. For application with rhizobia, we should refrain from excessive use of nitrogen fertiliser and ameliorate aluminium toxicity and phosphate deficiency (Giller et al., 2016). For phosphate-solubilising fungi it is evident that their mode of action (production of organic acids) makes their use ineffective under acid conditions where phosphate is fixed to iron(hydr-)oxides and other positively charged mineral surfaces (Barrow, 2025). In the case of many other biostimulants, where the mode of action is still unknown, it is hardly possible to delineate this application window except in very general terms, which are probably too general for farmers to serve as a basis for decision-making. Li et al. (2022) introduced one relevant factor. Their regression of response rate as a function of soil organic carbon implies no effect in soils with more than 3% organic carbon. It is therefore not surprising that most experiments were executed in soils that were (very) low in soil organic carbon. As a consequence, research bias may be introduced, a set of experiments executed under limited environmental conditions that make generalisation very hard at best. This specific form of research bias, next to publication bias, likely contributes to the many positive reports on the positive effects of biostimulants on agricultural production. The observation of Li et al. (2022) that there is a very shallow relationship between relative application dose and crop response implies that the mode of action is hardly dose-responsive. Consequently, the amounts that should be applied can be far less than is currently recommended, allowing considerable savings by reduction of the doses applied. From such observations it seems unavoidable to conclude that knowledge of the mode of action of a certain product is imperative to define the context under which a product could be effective.
Conclusion
While many biostimulants can enhance plant growth, their efficacy is highly variable and often poorly substantiated. On the basis of a meta-analysis, Li et al. (2022) claimed an average crop yield increase of 17.9% through application of plant biostimulants. In our opinion, this is an unlikely, high value, probably increased by research bias (executing experiments under specific conditions while suggesting context-independency of such outcomes) and publication bias towards positive effects and/or by the lack of proper controls. Such findings underscore the need for stricter, harmonised testing regimes. Li et al. (2026) already called for regulatory clarity concerning biostimulants to distinguish them from pesticides in order “to foster innovation and enable the deployment of biostimulants”. While we agree on the need for regulatory clarity, we caution that the current marketing of biostimulants that fail to deliver the benefits they claim requires the designation and implementation of robust regulatory control.
The current CEN Standard (2024) offers the most detailed guidance, yet it remains insufficient—it permits efficacy claims without field trials, lacks mandatory controls, and allows extrapolation from a few crops to entire crop groups. It is hard to understand why the CEN Standard has not learned from the experience on efficacy testing of EPPO (Table 3), FAO (2006), and others (e.g., Neuhoff et al., 2024; Lyons et al., 2025). Currently, the CEN committee is mainly comprised of members representing companies who sell biostimulant products. It is clear that the current consensus-driven process does not adequately protect farmers from ineffective or mis-labelled biostimulant products. We recommend that obligatory efficacy testing of biostimulants by accredited institutions rather than only by the suppliers themselves is in the interest of farmers. To this end, the European Commission should appoint an independent committee to oversee the regulation of biostimulants. The documents on the CEN website to which we refer are behind a paywall. In line with the spirit of our article, we strongly recommend that these documents be made publicly available.
Some of the European and Mediterranean plant protection (EPPO) standards.
We conclude that the current CEN Standard (2024) is insufficient to ensure that biostimulants that enter the market consistently deliver the effects claimed. The FPR (2019) was erected to regulate the biostimulant market and to prevent the sale of all kinds of miracle products. For farmers, it is at best difficult to decide which products will have the desired effect which emphasises the need for more stringent testing and more robust regulations.
Footnotes
Acknowledgments
We are grateful for a critical review of the submitted manuscript, which helped us to improve the original submission.
Ethical approval and informed consent statements
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
