Abstract
Assessing and alleviating pain in animals involved in research is critically important. However, the effective implementation of pain management depends on the knowledge and attitudes of the personnel involved. Following a Federation of European Laboratory Animal Science Associations ‘Pain in Mice’ working group initiative, a questionnaire to survey current practices concerning analgesic use in laboratory mice was distributed to several professional groups in the field of laboratory animal science. Besides demographic data, attitudes to pain and analgesia and sources of information and advice on pain management were assessed. Data were gathered and analysed through an e-survey provider. Most respondents (N = 222) were from Europe (90%). Analgesics were administered to murine surgical models by 92% of respondents in most cases and by 66% to all mice undergoing surgery. Most respondents used multimodal analgesic regimens (69%). For non-surgical models, 34% of respondents provided analgesics. The most commonly administered classes of analgesics were opioids (mostly buprenorphine) and non-steroidal anti-inflammatory drugs (mostly meloxicam and carprofen). A wide range of dose rates of meloxicam and carprofen was reported. Local anaesthetics were also widely used in surgical models (mostly lidocaine). Pain assessment was undertaken by most respondents (98%). In conclusion, most respondents provided analgesics to mice undergoing surgery and used analgesics in some non-surgical models. A considerable variation in the dose range used and the timing of administration of analgesics likely reflects both a lack of data and variation in pain assessment methodologies.
Introduction
Pain in laboratory mice may occur when they are used in some types of biomedical research. The avoidance and alleviation of pain is both a regulatory and ethical requirement 1 and often relies on the use of analgesic agents. Effective use of analgesics requires careful selection of the analgesic regimen and evaluation of its efficacy, but this is often undertaken by non-specialised personnel. This staff may not always be appropriately trained or advised by veterinarians, 2 despite legislative requirements for training in pain management.
Surveys of analgesic use based on reviews of publications involving potentially painful procedures in laboratory species have indicated a relative underuse of analgesics, particularly in small rodents.3 –5 Several causes have been suggested as contributing to this apparent underuse of analgesics in laboratory species, including a lack of information on analgesic efficacy when used in different research models, a concern about the potential interactions and confounding effects of analgesics in particular models, an inability to recognise the intensity and duration of pain, and a consequent failure to implement effective pain relief.
Analgesic therapy should match both the level of pain and its duration. There has been a significant increase in published data, particularly over the last decade, that could enable more effective pain management. 6 New methods of pain assessment and pain control have been developed.7 –13 However, these more recent approaches may not be widely adopted.
As part of the activities of a Federation of European Laboratory Animal Science Associations (FELASA) working group on pain management in mice, we surveyed those involved in the use of mice in biomedical research. We aimed to improve our understanding of current practices and attitudes to pain management in mice to identify areas where improvements could be made and incorporated into current best practice recommendations.
Methods
A questionnaire to survey current attitudes and practices concerning analgesic use in laboratory mice was distributed to a number of professional groups (no animal research was conducted). A description of the questionnaire is shown in Appendix I in the Supplemental Material. Details of the questionnaire, which followed the CHERRIES guidelines, 14 are shown in Table 1.
Characteristics of the survey according to the CHERRIES guidelines for reporting. 14

Invitations to voluntarily participate (see Appendix I of the Supplemental Material) were sent via FELASA to its 21 member associations (https://felasa.eu/about-us/members) in April 2022. Additionally, the following professional associations were invited to distribute the survey to their members: ESLAV (European Society of Laboratory Animal Veterinarians), ECLAM (European College of Laboratory Animal Medicine) and IAT (Institute of Animal Technology). Survey details were also distributed through the CompMed email list maintained by the American Association of Laboratory Animals Science (AALAS). No invitations were explicitly sent to organisations representing academics or researcher workers. However, recipients were requested to forward the link to colleagues involved in in vivo research with mice.
The survey comprised four sections with a total of 54 questions: (a) a general section with questions on demographics and work environment; (b) a section aimed at evaluating attitudes to pain and analgesia in surgical models; (c) a section aimed at evaluating attitudes to pain and analgesia in non-surgical models; and (d) a concluding section evaluating sources of information and advice on pain management. Participants were asked to indicate their use of analgesia in mice in both surgical and non-surgical models, with details on the analgesic agents used and the treatment protocols followed. All responses were anonymous. Data were gathered through a professional e-survey provider, which included a data analysis function (Qualtrics XM, Provo, UT).
Statistical analysis
Data are expressed as absolute values and the percentage of valid responses. An analysis was performed using the Qualtrics XM Data & Analysis software (Crosstabs iQ option). The chi-square test between categorical variables was employed to compare data with a p-value of 0.05. When applicable, consistent response levels were grouped as appropriate (Europe or the rest of the world; younger or older than 40 years of age; laboratory animal science (LAS) personnel or scientists).
Results
Of the 442 responses received from 28 April to 8 August, 222 completed the questionnaire. Of the 220 incomplete responses, 57 had a progress level below 25%, and of these, 36 did not provide any demographic data (section 1). In addition, 56 responses were excluded, as they contained several identical responses received within a 12-hour period or were not completed. Only completed responses were considered for the analysis (N = 222). Since participants were not required to answer every question, the number of responses to each question is included in the results.
Demographics
Most respondents (n = 217) were from Europe (90%; European Union 60%, n = 134), the UK 17% (n = 36) and Switzerland 13% (n = 28), with those from the rest of the world accounting for 10% (n = 19) of responses. Considering the uneven distribution, no further analyses were performed comparing results between these geographic areas (Figure 1(a)). Most respondents (n = 216) were scientists (37%, n = 79), and the remaining respondents were personnel working with laboratory animals (LAS personnel 63% (n = 137); caretakers 6% (n = 13), technicians 18% (n = 38), welfare officers 11% (n = 23) and veterinarians 29% (n = 63)). From all respondents indicating their genre (n = 217), 68% (n = 147) were women, and 31% (n = 67) were men. Age ranges of respondents (n = 216) were <30 (13%, n = 28), 30–39 (31%, n = 67), 40–50 (34%, n = 73) and >50 years old (22%, n = 48). Thus, 44% (n = 95) were younger than 40 years of age, and 56% (n = 121) were aged 40 years or older (n = 216). Of these 217 respondents, some combined different jobs (n = 233; Figure 1(b)). Most were working in academia (61%) or funded research institutes (26%), and the remainder were in commercial organisations (Figure 1(c)).
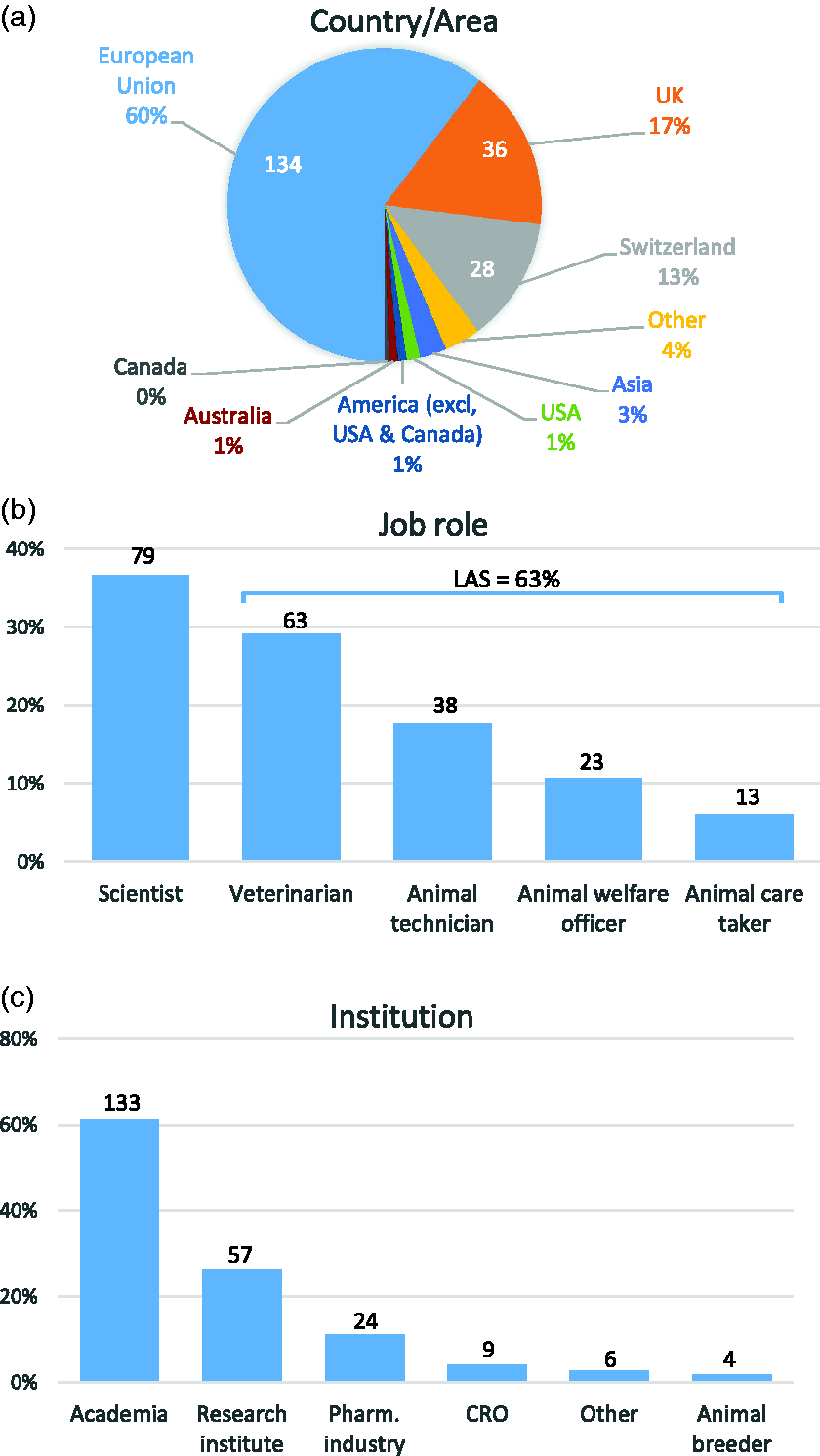
Demographic characteristics of surveyed respondents according to (a) their country or geographical area where they work (n = 217 responses), (b) their job role as scientists of laboratory animal science (LAS) personnel (n = 216 responses) and (c) the type of institution (n = 217 respondents; more than one response was allowed).
Murine models
The second and third parts of the survey focused on the attitudes to pain of personnel involved in surgical (n = 186) and non-surgical murine models (n = 159; Figure 2). Most respondents (n = 135, 61%) were involved in both types of models, 51 (23%) in surgical models only and 24 (11%) in non-surgical models only. Twelve (5%) respondents declared not being involved in either model.
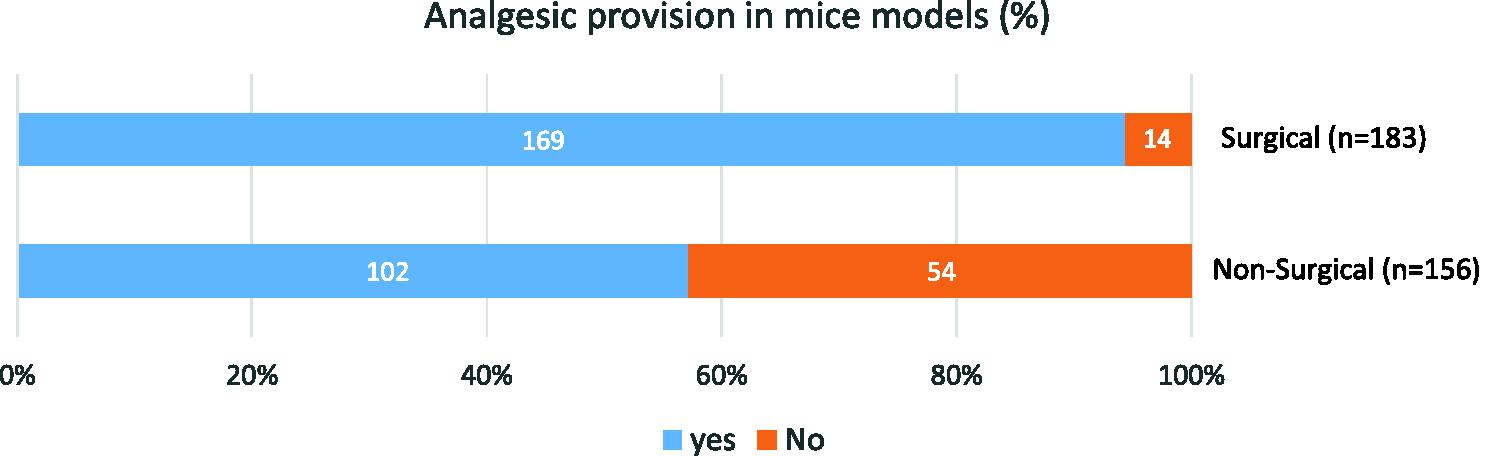
Percentage of surveyed respondents reporting the provision of analgesia to surgical (routinely) and non-surgical murine models. The number of respondents (N = 222) is indicated in parentheses for each type of model (surgical and non-surgical).
Pain management in surgical murine models
The most commonly conducted procedures involved superficial tissues (cutaneous or subcutaneous; 65%, n = 120), the brain (59%, n = 109) or abdominal procedures (53%, n = 98; Figure 3).
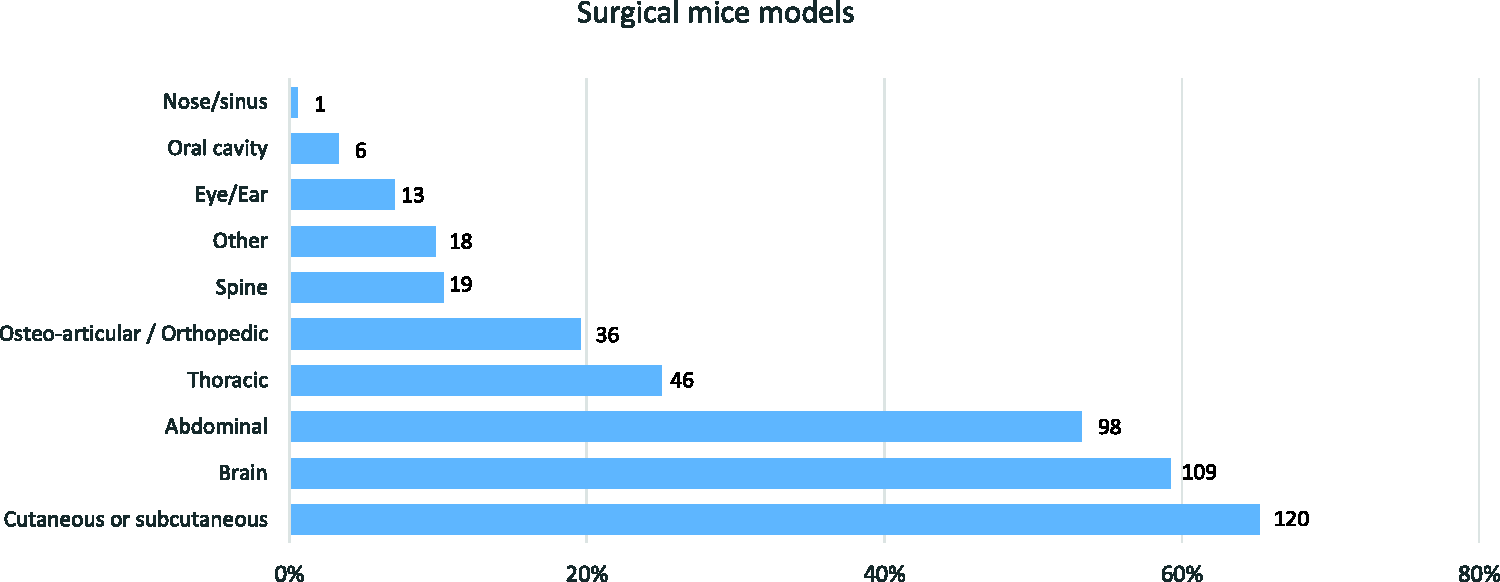
Most common types of surgeries performed in murine models by surveyed respondents (n = 184; more than one response was allowed).
Most respondents gave analgesics routinely (92%, n = 169), whereas 8% (n = 14) did not report routine use of analgesics perioperatively. There was no significant effect of job type (scientists or LAS personnel; p = 0.481), sex (p = 0.196) or age (p = 0.544). Of those reporting analgesic use routinely, 72% (n = 121) used multimodal analgesic protocols (defined in the questionnaire as ‘a combination of different analgesic drugs at the same time’), and 28% (n = 47) used only a single analgesic drug. Similarly, there were no differences between job type (p = 0.117), sex (p = 0.290) or age (p = 0.359) in the use of multimodal or single-agent analgesic protocols.
Opioids (82%, n = 148) and non-steroidal anti-inflammatory drugs (NSAIDs; 77%, n = 138) were the most frequently used analgesics, followed by local anaesthetics (63%, n = 114). Paracetamol was employed by 15% (n = 27) of respondents (n = 180) with little use of other analgesics (Figure 4).
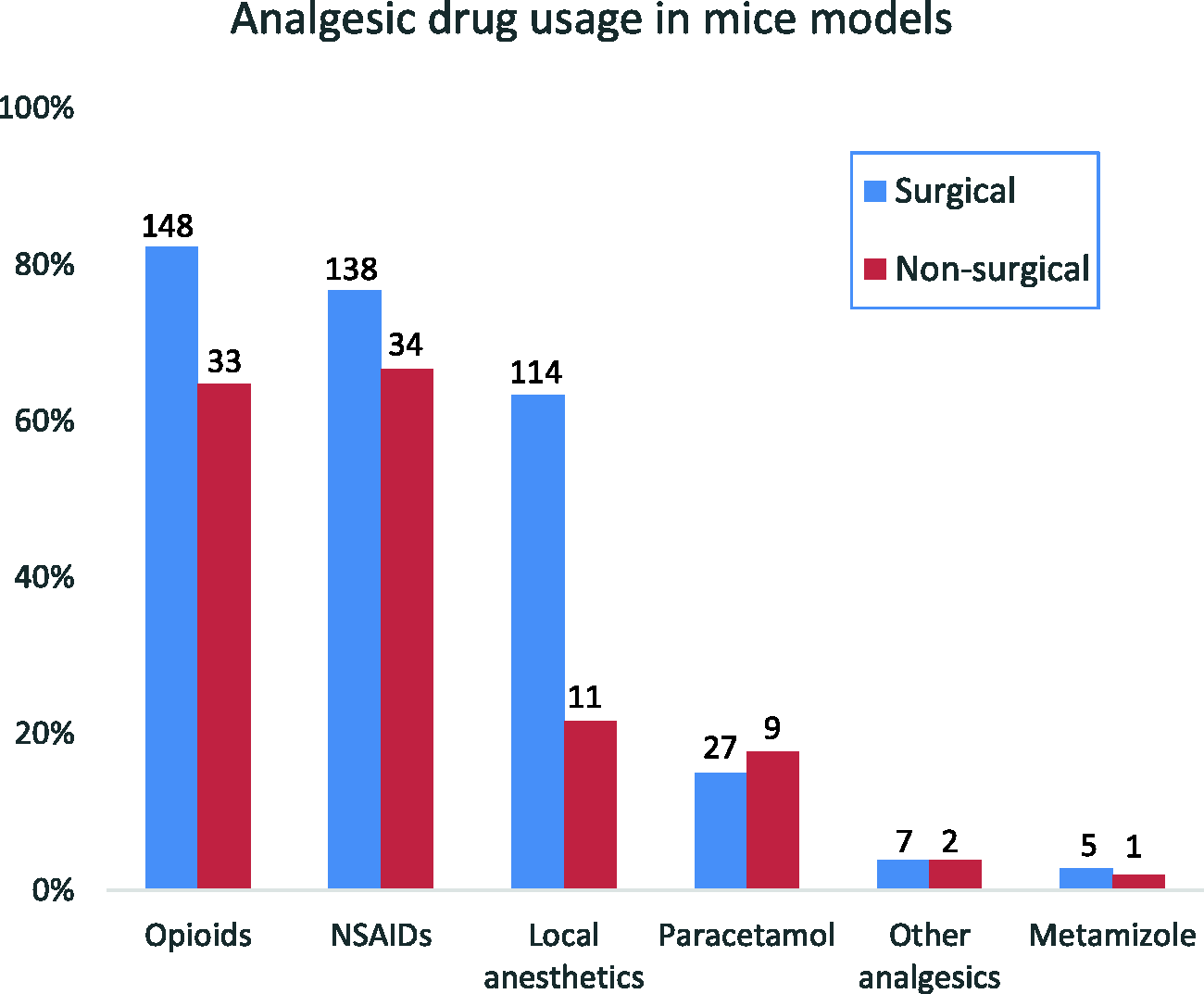
Use of analgesics in surgical (blue bars; n = 183; n = 180 indicate drugs used) and non-surgical (n = 156, n = 51 indicate drugs used) murine models reported by surveyed respondents. More than one response was allowed (see text for details).
Among respondents reporting specific opioid drugs (n = 145), buprenorphine was the most widely used analgesic (95%), with tramadol (9%) and other opioids (19%) being less widely used. Most reported doses (83%) for buprenorphine (n = 84) were in the range of 0.05–0.1 mg/kg, with 5% reporting the use of lower doses (lowest 0.01 mg/kg) and 12% of higher doses (up to 2 mg/kg). Buprenorphine (n = 138) was most frequently administered preoperatively (76%, n = 105) or intraoperatively (55%, n = 21), with only a few respondents administering the initial dose postoperatively (1.4%, n = 2). Administration of a second, additional dose of buprenorphine varied, with 21% (n = 28) of respondents (n = 136) redosing within 6 h, 32% (n = 43) within 6–8 h and 24% (n = 32) between 8 and 24 h. According to 24% (n = 33) of respondents, the mice required only a single dose of buprenorphine. Slow or sustained-release buprenorphine was used by only 12% (n = 17) of respondents (n = 137).
Among respondents reporting NSAID drug use (n = 134), carprofen (62%, n = 83) and meloxicam (61%, n = 82) were the most commonly used agents. Other NSAIDs were only used by 12% (n = 16) of respondents. From them, 21% (n = 28) reported using both carprofen and meloxicam. Most respondents (n = 82 and n = 80, respectively) administered carprofen (59%, n = 48) and meloxicam (63%, n = 80) before the start of surgery. Additional doses of carprofen and meloxicam were provided at varying time intervals, usually within 12–24 h or 24 h after the initial dose (Figure 5). From all respondents reporting doses for carprofen (n = 49), 51% were using a dose of 5 mg/kg, 10% were giving lower doses (lowest 0.5 mg/kg) and 39% were giving higher doses (up to 20 mg/kg). For meloxicam (n = 55), 47% were giving a dose of 5 mg/kg, 36% lower doses (lowest 0.2 mg/kg) and 16% higher doses (up to 20 mg/kg; Figure 6).
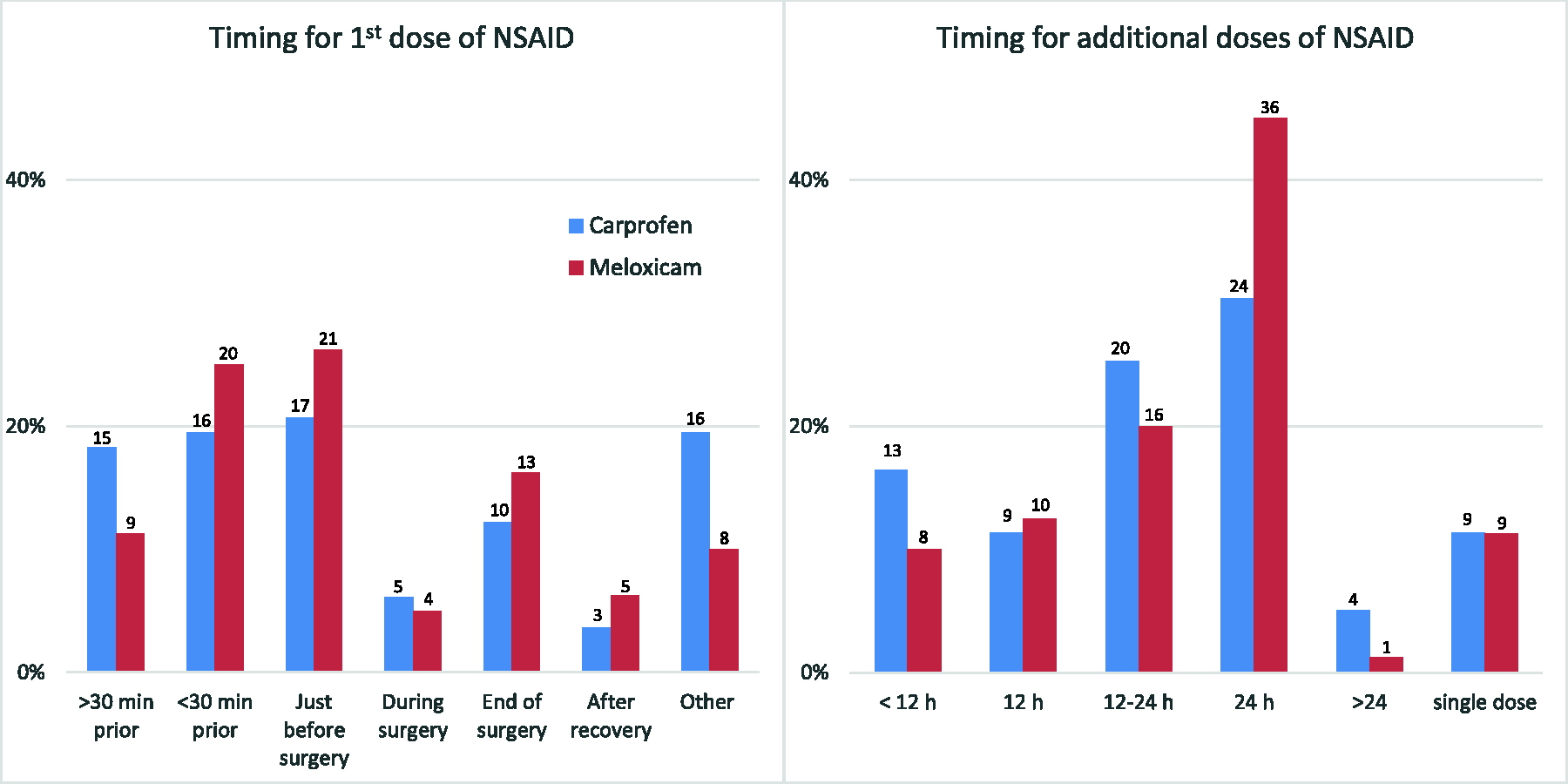
Timing of administration of the non-steroidal anti-inflammatory drugs (NSAIDs) carprofen and meloxicam in surgical murine models, first and additional doses, by surveyed respondents reporting NSAID drug use (carprofen, n = 82 and n = 79 responses, respectively; meloxicam, n = 80 responses, both). Bars indicate the percentage from all responses for every drug and graph, whereas figures indicate the number of responses.
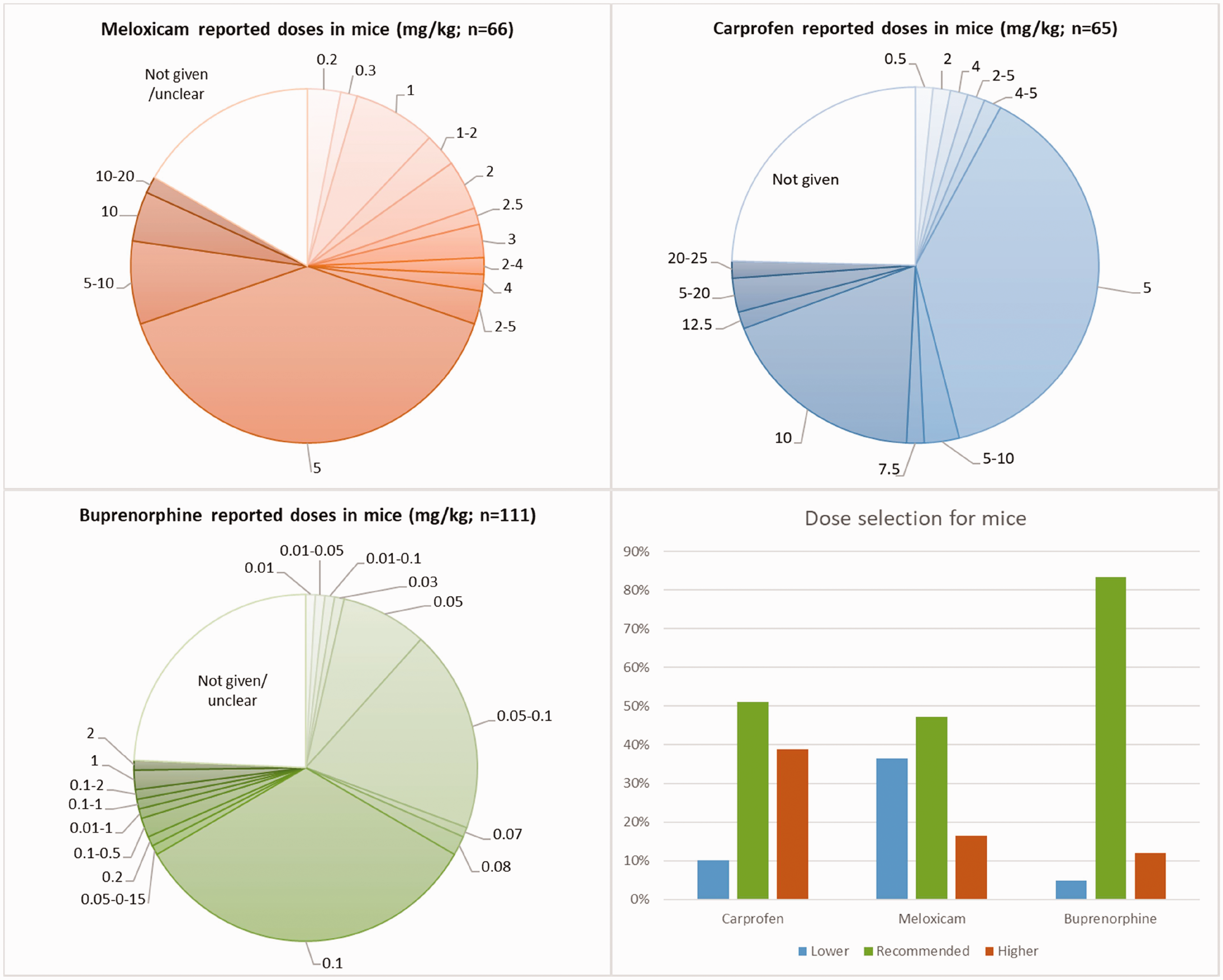
Reported doses of meloxicam and carprofen (mg/kg) by surveyed respondents, shown as percentages with the absolute number of respondents in parentheses. The recommended dose for the three drugs refers to that listed by Flecknell. 6 Thus, lower and higher doses indicate the percentage of respondents reporting the use of doses below or above those indicated in the previous reference, respectively.
Among respondents reporting the local anaesthetic used (n = 112), lidocaine was the single most used drug (69%, n = 77), although bupivacaine (26%, n = 29) or a combination of lidocaine and bupivacaine (24%, n = 27) were also used. Reported doses of lidocaine (n = 19) ranged between 2 and 10 mg/kg, although one respondent indicated a dose of 40–50 mg/kg.
Responses regarding the drugs’ doses and routes are shown in Table 2. From them, not all responses indicated the doses employed. Fewer responses were obtained when dealing with non-surgical models.
Analgesic drugs reported to be administered by respondents by different routes (SC, PO, IP, IV or other).
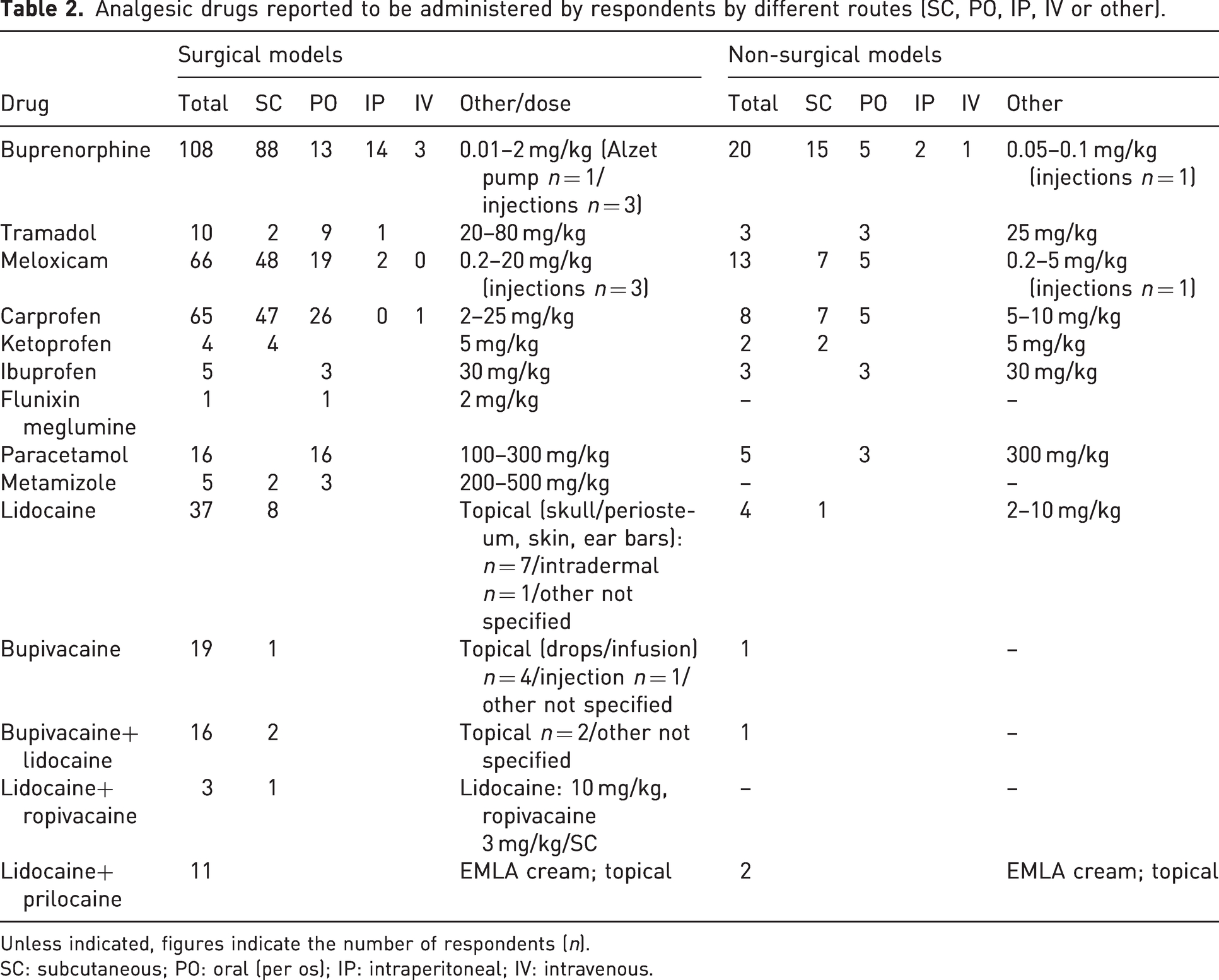
Unless indicated, figures indicate the number of respondents (n).
SC: subcutaneous; PO: oral (per os); IP: intraperitoneal; IV: intravenous.
Most participants (n = 182) either provided postoperative analgesia for three days (26%, n = 47) or administered only a single dose of analgesic (23%, n = 42). Fewer respondents gave analgesics for one (7%, n = 13), two (11%, n = 20) or three or more days (12%, n = 21).
Supportive care involved cage enrichment (77%, n = 144), environmental enrichment (77%, n = 141) and fluid and nutritional support (72%, n = 133). Most respondents using cage enrichment also reported environmental enrichment (n = 116), although further details on the type of enrichment were not requested. Only 4% (n = 8) of respondents (n = 183) indicated that no supportive care was provided.
Most respondents (55%, n = 118 out of 216) stated that they always administered analgesics to surgical murine models. The main reason stated for withholding analgesics was the potential interference with the research outcomes (19%, n = 42). Other reasons for not administering analgesics were that mice did not show signs of pain after surgery (9%, n = 19) or that analgesic administration would be too stressful for the mice (5%, n = 10). Two respondents (1%) withheld analgesic therapy because they considered recovery was better without analgesic use (Figure 7).
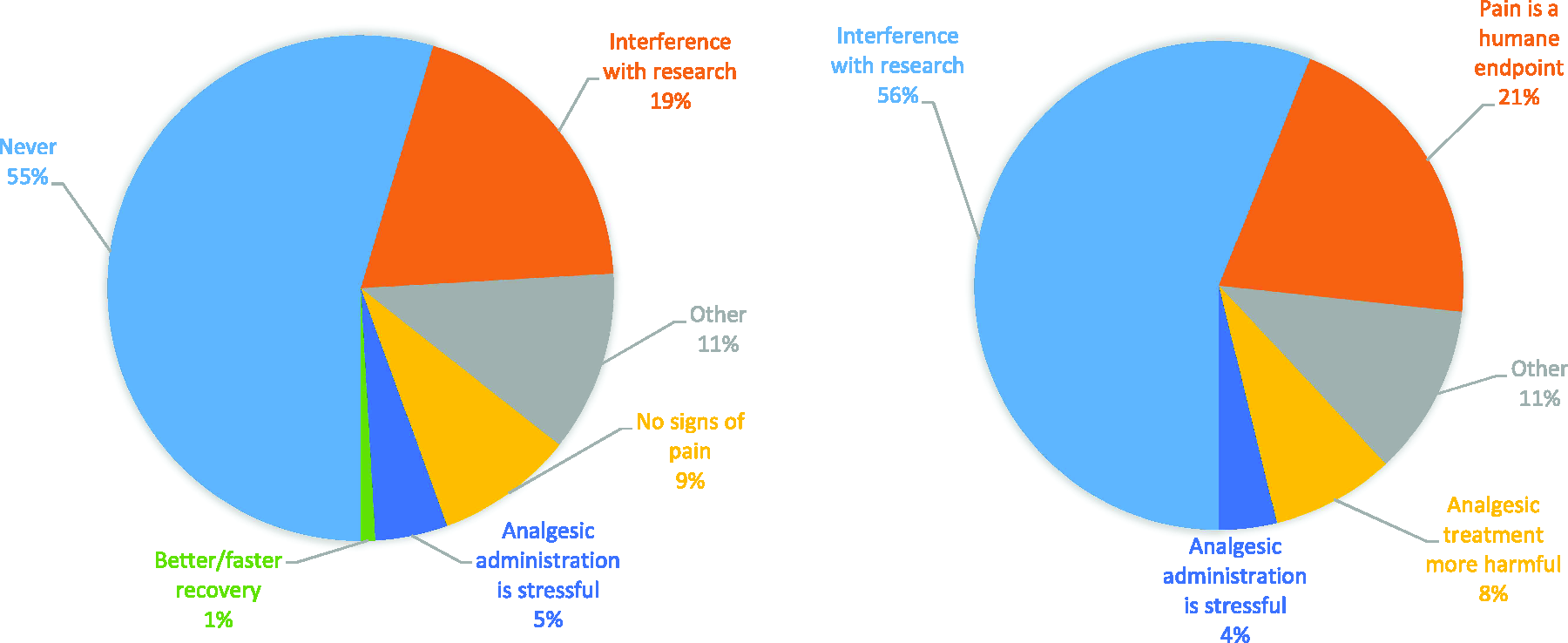
Reasons for withholding analgesic use among surveyed respondents for surgical (left, responses n = 216, respondents n = 179) and non-surgical (right, responses n = 200, respondents n = 141) murine models. More than one response was allowed.
Pain management in non-surgical murine models
The most commonly reported non-surgical models are shown in Figure 8 (n = 156). Of 222 responses, 159 (72%) respondents were involved in non-surgical models. Only 54 (35%) reported analgesic provision (Figure 2), with significant (p < 0.01) differences between LAS personnel (43%) and scientists (17%). Analgesic provision was higher for LAS personnel (72%) compared to scientists (40%; p = 0.006), with no differences related to the sex (p = 0.062) or age (p = 0.299) of respondents. From 51 respondents reporting analgesic type use, NSAIDs (67%, n = 34) and opioids (65%, n = 33) were the most commonly used analgesics. Local anaesthetics (22%, n = 11) and paracetamol (18%, n = 9) were less widely used (Figure 4). The use of multimodal analgesia in non-surgical models was reported by 28% (n = 14) of the respondents.
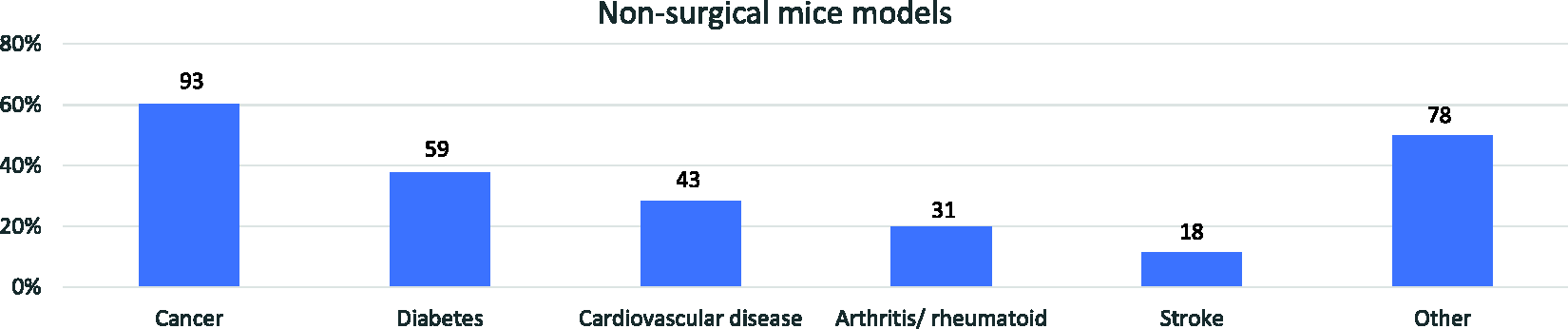
Most common non-surgical murine models reported use by surveyed respondents (n = 156). More than one response was allowed.
Among respondents using opioids (n = 31), buprenorphine was the most widely used analgesic (90%, n = 28), with limited use of other opioids (tramadol: 16% (n = 5); other opioids: 13% (n = 4)). Among respondents using NSAIDs (n = 31), meloxicam (74%, n = 23) and carprofen (42%, n = 13) were the most widely used agents, with other NSAIDs being used by only 13% (n = 4) of respondents. Only 11 out of 156 respondents using analgesics reported local anaesthetic use (lidocaine: n = 6; bupivacaine: n = 1) or a combination of lidocaine and bupivacaine (n = 2).
Reasons for withholding the use of analgesics (n = 141 respondents) were the potential interference with research outcomes (48%, n = 67), pain being considered a humane end point (41%, n = 58) and the higher potential harm (21%, n = 29) and stress (6%, n = 8) of analgesic treatment in mice experiencing only mild pain. Other reasons (27%, n = 38) were mainly related to non-painful models where no pain was observed or expected (Figure 7).
Pain assessment
Pain was assessed by most respondents (98%, n = 218), with 1.4% (n = 3) considering assessment not necessary and 0.5% (n = 1) never assessing pain. Methods of assessment are shown in Figure 9. Duration of pain assessment (n = 215) usually involved the analgesic treatment period (13%, n = 27) or longer – for example, at least until the day after ending the analgesic treatment period (43%, n = 93; both 56%; Figure 9). Under ‘other’ (n = 62), most answers fell under one of the described categories.
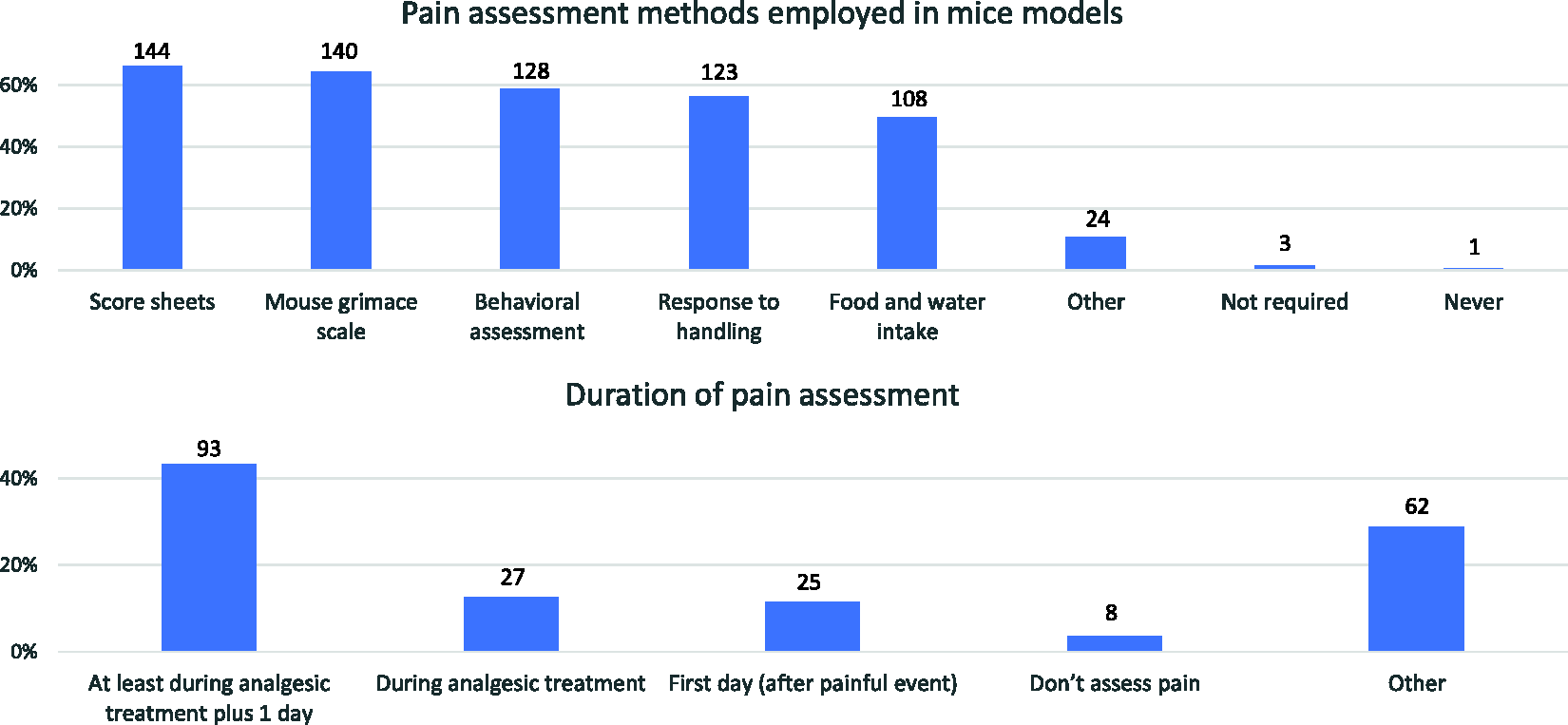
Pain assessment methods employed in murine models (top; n = 218; various responses allowed) and duration of pain assessment (bottom; n = 215) provided by surveyed respondents.
Most respondents assessed pain at least once daily (86%, n = 214). Monitoring frequency was higher on the first day following a painful event (25%, n = 53) or was performed twice a day (23%, n = 50) or more frequently (4%, n = 8). When pain was, or might be, expected, mice were assessed twice a day (45%, n = 95) or daily (42%, n = 89; both 87%), with a few respondents assessing pain every two to three days (2%, n = 4) or weekly (1%, n = 2). To ensure that analgesic therapy was effective, most respondents (n = 200) assessed pain before and after analgesic administration (50%, n = 99), whereas fewer respondents assessed pain only after administering the analgesic (14%, n = 28) at fixed time points between administrations (14%, n = 28) or before administering the analgesic (10%, n = 20). Most respondents (n = 212) never modified their analgesic protocol based on their assessments (dose or frequency, 61%, n = 129), with 39% (n = 83) doing so. Finally, no side effects from analgesic treatment were noted by 73% (n = 152) of respondents (n = 209), with 27% (n = 57) observing side effects.
Resources and consultation
Respondents used a variety of sources of information when providing analgesia (n = 218; Figure 10). Consultation on pain management involved advice from veterinarians (84%, n = 183), animal welfare officers (37%, n = 80) or senior colleagues (32%, n = 69)
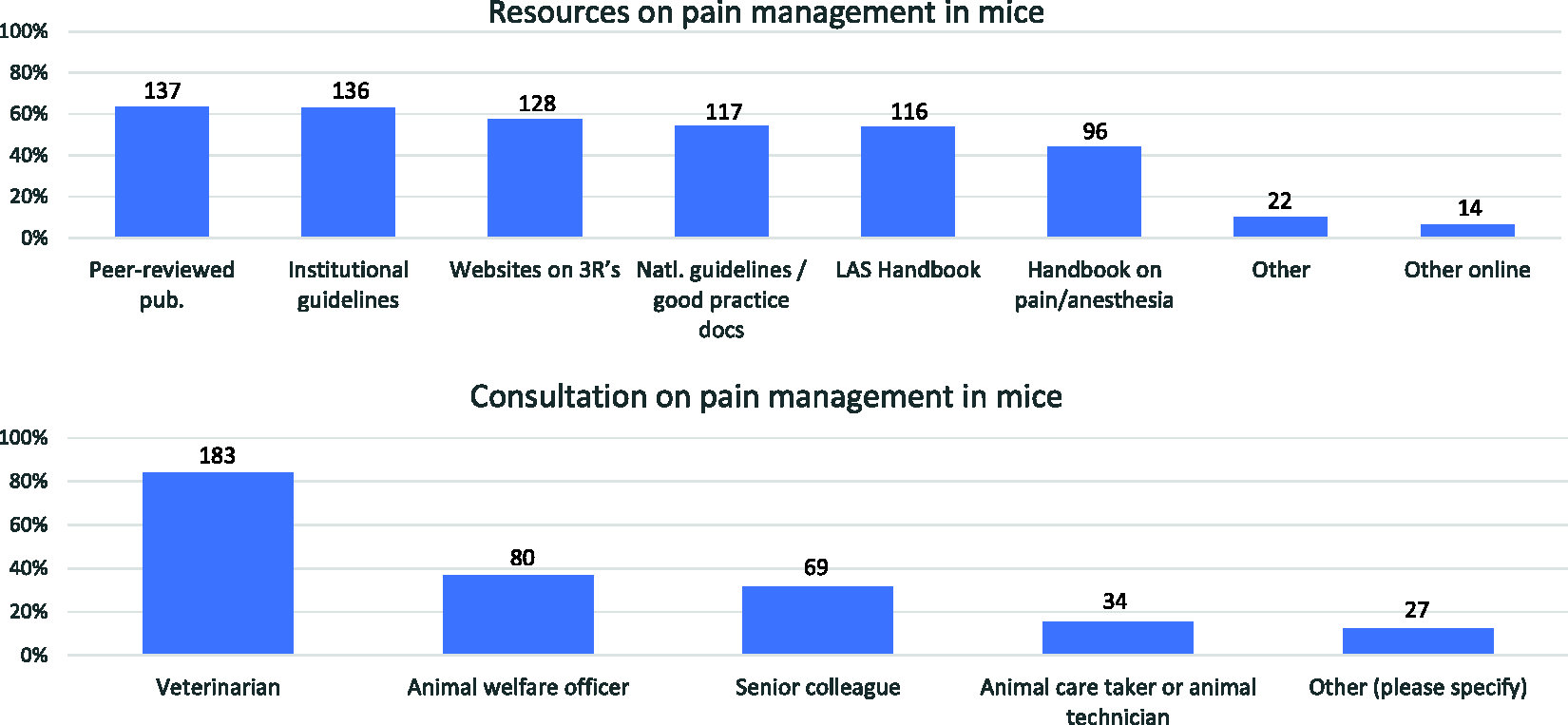
Resources (top; n = 216) and consultation (bottom; n = 218) on pain management in mice reported by respondents. More than one response was allowed.
Among respondents involved in surgical models, when a specific analgesic drug or class could not be administered to mice, the most common sources for advice on alternatives (n = 165; Figure 10) were veterinarians (76%, n = 125) and scientific publications (67%, n = 111). Other sources were reference texts or books (23%, n = 38), senior researcher advice (21%, n = 34) and animal technician advice (16%, n = 26).
Among respondents involved in non-surgical models, when a specific analgesic drug or class could not be administered to mice, the most common sources for advice on alternatives (n = 131) were veterinarians (79%, n = 103) and scientific publications (51%, n = 67). Other sources were senior researcher advice (21%, n = 28) and animal technician advice (16%, n = 21).
Discussion
Demographics
The present survey data provides updated information on current pain management practices of laboratory animal science personnel and scientists working with murine models where pain might occur. The survey was distributed primarily through the constituent associations of FELASA and their electronic mailing lists. Although the survey was not distributed directly to scientists, many respondents identified themselves as scientists (34%). Most respondents were from Europe, and therefore the results mainly indicate the attitudes of personnel from European countries toward pain management in mice. More than 66% of scientist respondents were female – a higher percentage than the proportion of women scientists in Europe (41%). 1 However, this might reflect a different proportion of female scientists involved in in vivo research. 15 Alternatively, it may reflect a greater concern for murine welfare among female scientists. A similar bias towards analgesic use was reported in surveys of analgesic use by clinical veterinarians,15 –17 and women have been reported to be more concerned than men about the suffering of research animals. 18
Pain management in mice
Surgical models involve pain, and the degree of pain is likely to vary, depending on the degree of surgical trauma. Analgesic protocols should therefore be designed to match the likely pain severity. Most surgeries involved cutaneous or subcutaneous tissues (65%) and are likely to have resulted in mild to moderate pain, depending on the surgical model. However, other surgical models used by respondents can be expected to cause moderate to severe pain unless effective analgesia is provided. Ideally, all mice undergoing surgery should be provided with pain relief. Among respondents, 92% stated that they administered analgesics to mice undergoing surgery, and 66% always administered analgesics. However, analgesia was withheld in some circumstances (8%). When non-surgical models are considered, analgesic provision was reduced to one-third, with relevant differences between LAS personnel and scientists, with the latter group administering analgesics much less frequently (83% vs 17%). Based on the responses received, these differences in analgesic use are likely to result from an underestimation of the degree of pain produced by non-surgical pain models,19,20 difficulties in pain assessment in non-surgical models or a greater concern regarding potential confounding effects of analgesic use.
Analgesic treatment was most often provided for the first 24 h post procedure (30%) or three days post procedure (26%). Only 21% of respondents varied the duration of analgesic administration depending upon the type of procedure. Given the potential severity of several surgical models, the adequacy of providing pain relief for only 24 h should be confirmed by effective pain assessment. Similarly, provision of analgesics for three days may represent over-treatment of minor surgical procedures, and this could be determined by pain assessment. Although the use of pain assessment was widely reported, it was not possible to determine how effective these assessments would have been (see below).
Analgesic drugs
Pain management relies primarily on the administration of analgesic agents. However, other factors may be considered, such as refinement of the procedure to reduce the amount of pain (e.g. reduced surgical trauma) or environmental manipulations to promote a positive affective state (enrichment, distractors, social caging, etc.). 21 Interestingly, most respondents used cage and environmental enrichment and nutritional support for surgical murine models, suggesting they were aware of the relevance of these measures in pain management. 6 However, it remains unclear whether these environmental enrichments were also provided routinely.
The use of analgesics in surgical models did not differ between scientists and LAS personnel, suggesting that both groups are aware of the importance of pain management. Most respondents were already administering multimodal analgesia, likely the most effective means of providing analgesia to animals suffering from moderate to severe pain. However, 24% of respondents gave only a single analgesic drug, which might suggest the need for additional education and training in analgesic use. 6 It is also essential that those with oversight of project and protocol approval should ensure an appropriate analgesic plan has been described and implemented.
Buprenorphine was the single most widely used analgesic in surgical models, with other opioids being employed rarely. This opioid is considered an effective analgesic for perioperative pain, 6 although there are concerns regarding the dose and timing of administration to alleviate pain effectively.22 –24 The most commonly reported doses were in the range of 0.05–0.1 mg/kg, but doses up to 2 mg/kg were also reported. The timing of additional doses of buprenorphine varied widely. Suggested timing currently ranges from 3 to 6 h,7,13,25,26 but these rely on assumptions about the effective plasma concentration (based on data in people). Data are also lacking on the influence of the prolonged receptor binding of buprenorphine on its duration of action. Only 21% of respondents gave buprenorphine every 6 h, and most administered the drug every 6–8 h (32%), which might be suboptimal. Up to 24% of respondents gave this drug between 8 and 24 h following the previous dose, which is likely to result in a period of inadequate analgesia. Determining the most appropriate dose of analgesic and the timing of additional doses requires careful pain assessment. The use of long-acting formulations of analgesics, 27 such as slow-release buprenorphine, can provide more prolonged periods of analgesia. However, pain assessment is still required to determine that an effective dose of analgesic has been administered. Only 12% of respondents reported using slow-release buprenorphine, most probably because this product is only available in the USA.
Presumed effective buprenorphine plasma concentrations are achieved some 20–30 min after its administration, 28 and half of the respondents administered this analgesic before the start of surgery. So, it is likely the full analgesic effect would be reached in the immediate postoperative period. However, to provide an antinociceptive intraoperative effect (‘pre-emptive’ or ‘preventive’ analgesia), buprenorphine should be administered before the induction of anaesthesia. Interestingly, one-third of respondents gave buprenorphine more than 30 min before surgery, and overall, one-half gave this drug before anaesthesia, suggesting they are aware of the pharmacokinetics of buprenorphine.
The use of NSAIDs was slightly less common than the use of opioids in surgical models (overall use of 77%). NSAIDs can be used effectively to control postoperative pain, primarily because of their anti-inflammatory effects and the consequent reduction in peripheral sensitisation following tissue injury. A particular advantage of NSAIDs is their relatively long duration of action (12–24 h), which facilitates the provision of continued analgesia, since the administration of opioids (e.g. standard buprenorphine formulations) four or more times per day has practical limitations. Meloxicam and carprofen were the most commonly used agents (60%), with other NSAIDs being much less widely used. As with buprenorphine, the dose range used varied. The most frequently reported doses for carprofen and meloxicam were 5 mg/kg, although they ranged considerably from 0.5 to 25 mg/kg (carprofen) and 0.2 to 20 mg/kg (meloxicam). Although high doses of carprofen (up to 20 mg/kg) can be used safely in mice, a similar dose of meloxicam may cause gastric toxicity. 29 The range of dose rates used probably reflects the uncertainty of an appropriate dose range in mice. In a study using the Mouse Grimace Scale to assess the efficacy of pain relief, doses of carprofen 20–25 mg/kg were required to produce a detectable analgesic effect. 30 Other studies have shown that relatively high doses of NSAIDs are required in mice,31 –33 although they may be associated with adverse effects in some mouse strains.33,34 The efficacy of carprofen as a postoperative analgesic in mice has also been questioned. 35 As mentioned earlier, given the wide variation in analgesic requirements between different strains of rodents, the only means of ensuring that an appropriate analgesic regimen is selected for a specific group of mice is by assessing pain in those animals and adjusting the dose and analgesic agents used as required.
The reported time of administration of NSAIDs was either before surgery (66%) or at the end of surgery. Preoperative administration is likely to provide more effective analgesia by reducing the inflammatory response and consequent peripheral sensitisation caused by surgery. Although preventing the increased potential of side effects of NSAIDs intraoperatively (reduced perfusion and damage to the kidneys or the bowel), 36 postoperative administration is likely to be less effective, particularly since the onset of action of both carprofen and meloxicam requires 15–30 min following injection. Subsequent doses were mostly given at 24 h or earlier, although up to 5% of respondents administered the NSAIDs at longer intervals, which may not be adequate.
Nearly 70% of respondents used the local anaesthetic lidocaine, with the longer-acting agent bupivacaine being used less often (by 26% of respondents). Depending on the concentration and dose administered, lidocaine is effective for less than 90 min, providing only a short period of analgesia. A more extended period of analgesia can be provided by the use of bupivacaine. 6 The use of a combination of both agents is based on the assumption that lidocaine will provide rapid-onset effects, albeit of short duration, whereas bupivacaine has a slower onset of action but longer duration. A remarkable finding was the reporting of a very high dose of lidocaine (5- to 10-fold the recommended dose) by one respondent, likely to induce toxicity.
Compared to surgical models, the use of NSAIDs and opioids in non-surgical models was only slightly lower (67% and 65%, respectively), but local anaesthetics were much less frequently used (22%), almost certainly because local anaesthetics can be used most effectively during surgical procedures. This relatively high reported use of analgesics does not indicate that respondents were using these agents in all non-surgical models but rather that they have used them in at least one model. Again, buprenorphine was the most used opioid (90%), followed, to a much lesser extent, by tramadol (16%). When NSAIDs are considered, meloxicam was used twice more frequently than carprofen. This may be related to the availability of a palatable oral preparation of meloxicam that may be more suitable for prolonged administration in non-surgical models.
Although concerns related to side-effects were not given as a reason to withhold analgesics, some respondents reported having observed adverse effects of analgesics. These included the recognised effects of opioids – prolonged recovery, respiratory depression and pica – but also the opposite effects of excitement (n = 4) and sedation or apathy (n = 8) in mice. 1 Only one respondent reported decreased live birth rate after embryo transfer, although it is not supported by controlled studies of the effects of analgesic treatment after this procedure. 37 The risk of undesirable side-effects reinforces the need to provide analgesics at appropriate doses and for an appropriate time period to control pain while avoiding unnecessarily high dose rates or prolonged periods of treatment. This can be achieved by implementing effective pain assessment protocols.
Previous surveys of analgesic use in mice have suggested lower use of analgesics. However, these surveys were based on the assessment of published papers3,4 and on protocols submitted to a regulatory authority. 5 So, although the higher reported rate of analgesic use in our survey could represent a greater use of these agents, the differing methodologies make such a comparison difficult to interpret with confidence.
Pain assessment
The provision of pain assessment may be considered as high and may reflect an increased awareness and knowledge of the 3Rs promoted by mandatory training. 38 Nearly all respondents (98%) reported assessing pain in mice. Thus, almost all personnel using laboratory animals were aware of the need for evaluating pain, but our survey did not ask respondents to indicate how many animals were actually assessed within a procedure. Pain assessment using score sheets was the most frequently reported method (66%). However, other behaviourally based methods, with poor specificity to assess pain in mice, were also reported (behavioural, response to handling, food and water consumption: 49%–55%). This suggests the best tools available for measuring pain, such as the Mouse Grimace Scale, may not be being implemented widely. Although clinical assessments and the use of scoresheets are valuable in assessing the overall clinical state of animals, they may not necessarily include specific indicators of pain, such as the behavioural responses seen after abdominal surgery, 32 or more generally applicable measures, such as the Mouse Grimace Scale. 8
The duration of pain assessment was most frequently reported as extending throughout the period of analgesic treatment and beyond, and this should be considered good practice. The frequency of pain assessment required depends on factors such as the severity and speed of onset of pain and the likely duration of action of the analgesics used. Frequent monitoring is needed to limit any pain and distress to an absolute minimum. 2 Most respondents (82%) indicated that pain assessments were performed daily or more frequently (>80%). However, only 50% checked the efficacy of their analgesic regimen. This may indicate that the results of the pain assessments were simply noted rather than being used as a tool to monitor the effectiveness of analgesics and to revise the analgesic protocol as necessary. Most respondents never modified their analgesic plan (61%). Broadening the involvement of clinical care staff in the monitoring of mice has been suggested as a means of improving the frequency and duration of pain assessment. 38
Resources and consultation
Respondents used various resources on pain management, primarily peer-reviewed publications and institutional and national guidelines as well as websites and specialised handbooks. However, when consultation on pain management was required, veterinarians were the most frequently used advisers (>80%), supporting the regulatory requirement for the availability of veterinary advice in all EU member states.
Limitations
The survey was distributed to all FELASA member associations. However, the nationality of respondents was not evenly distributed across Europe. The survey was provided in English, and this may have limited the response rate from countries where staff are less fluent in this language. Distribution of a similar survey in the main languages of different states may provide greater insight into national variations in pain management. The data may also be biased, since those responding may be more likely to be concerned about pain management in mice. Those who do not use analgesics or conduct pain assessments may not have completed the questionnaire because they have little interest in the topic. Finally, the total number of recipients could not be clearly defined. FELASA sent the survey to all its affiliated associations, but the total number of recipients was unknown. ECLAM and ESLAV also distributed the survey, and it is likely that all members of these two associations are also involved in a FELASA association. Finally, a message was sent to COMPMED, but the response rate from relevant countries outside Europe (USA, Canada and Japan, among others) was low. Furthermore, the survey was not restricted to LAS personnel, likely largely increasing the potential target population. Therefore, an estimation of the error margin of the sample cannot be provided.
The survey provides an overview of current practices in pain management and pain assessment, but more detailed assessments of the measures employed in specific models would be of value. This is particularly relevant for the non-surgical models where a wide range of models are involved, with pain and its intensity being also highly variable or unknown, which makes the overall outcome data unable to support any specific guidance to a better and refined pain assessment and management. Similarly, a more detailed evaluation of the effectiveness of the pain assessment tools used would be of benefit.
In conclusion, the present survey indicates that most LAS personnel and scientists who responded to the survey provided analgesics to mice undergoing surgery and used analgesics in some non-surgical models to relieve pain in mice. A very limited range of analgesics was used, notably the opioid buprenorphine and two NSAIDs, meloxicam and carprofen. The use of local anaesthetics was widespread, but the agent used, lidocaine, will provide a relatively short duration of analgesia, which will be of most value intraoperatively. A multimodal approach to pain management was often adopted, and this is more likely to provide effective analgesia than regimens using a single agent.
The wide variation in the dose rates used and variations in the route and timing of administration of analgesics likely reflects both a lack of data in this area and variation in pain assessment methodologies. The most common routes of administration of the most frequently used analgesic drugs (buprenorphine, meloxicam and carprofen) were the subcutaneous and oral routes. For the latter route and when provided in drinking water, the amount consumed, environmental conditions and the stability of the drug should be carefully monitored to ensure accurate and consistent dosing,39,40 as summarised elsewhere. 6 Although the use of pain assessment methods was common, further evaluation is needed to determine whether the methods used are effective.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772241300779 - Supplemental material for Current practices of pain assessment and analgesic use in laboratory mice: A 2022 FELASA Working Group survey
Supplemental material, sj-pdf-1-lan-10.1177_00236772241300779 for Current practices of pain assessment and analgesic use in laboratory mice: A 2022 FELASA Working Group survey by Ignacio A Gomez de Segura, Saskia Seeldrayers and Paul Flecknell in Laboratory Animals
Supplemental Material
sj-xlsx-2-lan-10.1177_00236772241300779 - Supplemental material for Current practices of pain assessment and analgesic use in laboratory mice: A 2022 FELASA Working Group survey
Supplemental material, sj-xlsx-2-lan-10.1177_00236772241300779 for Current practices of pain assessment and analgesic use in laboratory mice: A 2022 FELASA Working Group survey by Ignacio A Gomez de Segura, Saskia Seeldrayers and Paul Flecknell in Laboratory Animals
Footnotes
Acknowledgements
Thanks to the ‘Pain in Mice’ FELASA Working group (Alessandra Bergadano, Delphine Bouard, Paulin Jirkof, Anastasia Tsingotjidou and Josipa Vlainic) for helping in the survey design, and to FELASA for creating and supporting the initiative.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Open Access has been funded by FELASA.
Supplemental Material
Supplemental material for this article is available online.
Note
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
