Abstract
Two healthy Landrace pigs anaesthetized with propofol suffered rapid onset of fatal sepsis. Clinical signs included severe arterial hypotension, loss of peripheral oxygenation, low end-tidal CO2, clinical onset of pulmonary oedema and cardiac dysfunction. Gross and histopathological examination revealed loss of vascular integrity with severe lung oedema and congestion, haemorrhages in several organs and fluid leakage into body cavities. Large numbers of Gram-negative bacteria, primarily Klebsiella sp., were present in the anaesthetic infusion containing propofol and were also cultured from internal organs of both pigs. The propofol was likely contaminated by bacteria after inappropriate handling and storage in the operating room. This report illustrates the potential for severe nosocomial infection when applying propofol in animals and humans and may serve as a reminder of the importance of strict aseptic practice in general, and specifically in the handling of this anaesthetic agent.
Introduction
Propofol is an intravenous (i.v.) anaesthetic agent commonly used in human and veterinary medicine. Due to its water insolubility, propofol is formulated as a lipid emulsion with soybean oil, glycerol, and egg lecithin. The formulation commonly used in human medicine contains no preservatives or antimicrobial additives and readily supports the growth of microorganisms.1,2 Contamination of propofol with Gram-negative bacteria may yield heavy loads of endotoxin or lipopolysaccharide (LPS), responsible for a range of adverse effects. 1 More than 20 outbreaks of iatrogenic infection caused by contaminated propofol have been reported in humans, some with fatal outcome.3 –6 In veterinary medicine, propofol use has been implicated in fatal sepsis in a dog. 7 To the best of our knowledge, this is the first report describing fatal septic shock due to contaminated propofol in pigs.
Case history
The pigs (Sus scrofa domesticus) were harboured in a research facility routinely using pigs for live-tissue training under the ethical approval of the Norwegian Food Safety Authority (FOTS no. 21341). The pigs were Landrace gilts of 58 kg (pig 1) and 64 kg (pig 2) purchased from a commercial Specific Pathogen Free pig farm. The pigs were acclimatized for seven days prior to anaesthesia and were fasted for 16 h. Hygienic measures including low animal density, substantial amounts of straw and hay, and regular cleaning of the pens ensured that the animals were dry and clean. The personnel handling the pigs wore gloves.
Pig 1 and pig 2 were anaesthetized three days apart. Both pigs were premedicated by intramuscular injection in the neck of a combination of 27 mg/kg ketamine (Ketador vet. 100 mg/ml, Richter Pharma AG, Austria) and 1 mg/kg midazolam (Midazolam 5 mg/ml, B. Braun, Germany). When sedated, the pigs were placed in dorsal recumbency on an operating table. A 20-gauge catheter (Venflon Pro, Becton Dickinson Infusion Therapy, Sweden) was placed in an auricular vein. Anaesthesia was induced by two (pig 1) or one (pig 2) bolus(es) of 8 mg of propofol (Propofol-Lipuro, 10 mg/ml, B. Braun, Germany). 8 This propofol product is commonly used in human medicine and contains no preservatives. Prior to intubation, both pigs were administered 1 mg atropine (Atropin 1 mg/ml, Takeda, Norway) i.v. to reduce saliva production and 200 mg ketamine i.v. to induce anaesthesia and analgesia. The pigs were intubated orotracheally, and anaesthesia maintained on 1 minimum alveolar anaesthetic concentration or 2 vol% sevoflurane (Sevofluran 100%, Baxter Medical AB, Sweden) in 30% oxygen in nitrogen along with simultaneous infusion of 160 mg/h propofol and 200 µg/h fentanyl (Fentanyl 50 µg/ml, Hameln, Germany) using a commercial infusion pump system (Braun Perfusor® Space, B. Braun, Germany). The pigs were positive pressure ventilated with a Dräger Primus Infinity respirator (Medsystems Ltd, UK) set to a tidal volume of 400–600 ml, a respiratory rate of 16–18 breaths/min, to keep the initial end-tidal carbon dioxide level within the range of 6.0 to 7.0 kPa. One external jugular vein and femoral artery of each pig was cannulated under aseptic conditions using a double-lumen and single-lumen catheter, respectively. The cannulation was performed with single-use, sterile kits (Certofix® Mono and Duo double lumen catheters, B. Braun, Germany) by ultrasound guided vascular access using the Seldinger technique. Hydration of the pigs was achieved with i.v. infusion of pre-heated (38°C) isotonic sodium chloride (Ringer Acetat Baxter Viaflo, Baxter Medical AB, Sweden) via the jugular vein. One litre of fluid was administered at a rate of 15 ml/kg per h, thereafter infusion was set to 8 ml/kg per h. Pig 1 and pig 2 received a total of 1000 and 3000 ml of fluid, respectively. The animals were kept warm using heating pads and monitored by electrocardiography (ECG), core temperatures, invasive arterial blood pressures, oxygen saturation and end-tidal carbon dioxide levels. In pig 1, ECG pads were applied after the onset of clinical signs.
In both pigs, a similar sequence of events took place 10–20 min after intubation, including severe hypotension (e.g. a drop from normal arterial pressure of 119/83 to 18/14 for pig 2), lack of peripheral oxygenation (e.g. flattening and lapse of oxygen saturation measurements from the pig tail) and abrupt fall in in end tidal CO2 levels (e.g. in pig 2 from 5.9 kPa ≫2.1 kPa ≫ 1.1 kPa). Pale purple skin discoloration was observed in pig 1, while pig 2 showed transient red skin discoloration lasting a few minutes before turning pale. In pig 1, a thoracostomy into the left hemithorax allowed finger palpation of the heart, demonstrating weak contractility. ECG showed coarse ventricular fibrillation. Pig 2 did not initially suffer cardiac arrest as this was counteracted by administering adrenaline while starting chest compressions as described below.
Cardiopulmonary resuscitation (CPR) was initiated in both cases with manual compressions of the left part of thorax with the animals in right lateral position. Adrenalin (Adrenalin 1 mg/ml, Takeda, Norway) was administered i.v. in increasing concentration: 10–20 µg of adrenalin (10 µg/ml) was followed by 100–200 µg of adrenalin (100 µg/ml), and finally 1–2 mg of adrenalin (1 mg/ml). Even at the highest adrenalin concentrations, no effect and a transient effect were seen in pig 1 and pig 2, respectively. The latter brief effect was an increase in invasive arterial blood pressure from 14/10 ≫ 336/183 ≫ 187/116 ≫ 129/77 ≫ 37/29 and heart rate from 164 ≫ 209 ≫ 162 ≫ 130 ≫ 114, all aligned with the peak and half-life of blood concentration of adrenaline. Furthermore, in pig 2, a cardiac ultrasound scan showed serious heart failure with global hypokinesia of the left ventricle and bilateral scans of dorsal parts of the lungs revealed numerous, so-called B-lines (Figure 1), suggestive of pulmonary oedema. 9 In pig 1, two electroshocks delivered by a Samaritan PAD 300 defibrillator (HeartSine Technologies, UK) had no effect and CPR was discontinued due to persistent asystole. Pig 2 was euthanized with 40 mg/kg pentobarbital (Exagon vet. 400 mg/ml, Richter Pharma AG, Austria). A total amount of 15–18 ml (150–180 mg) and 7–8 ml (70–80 mg) of propofol was administered to pig 1 and pig 2, respectively.
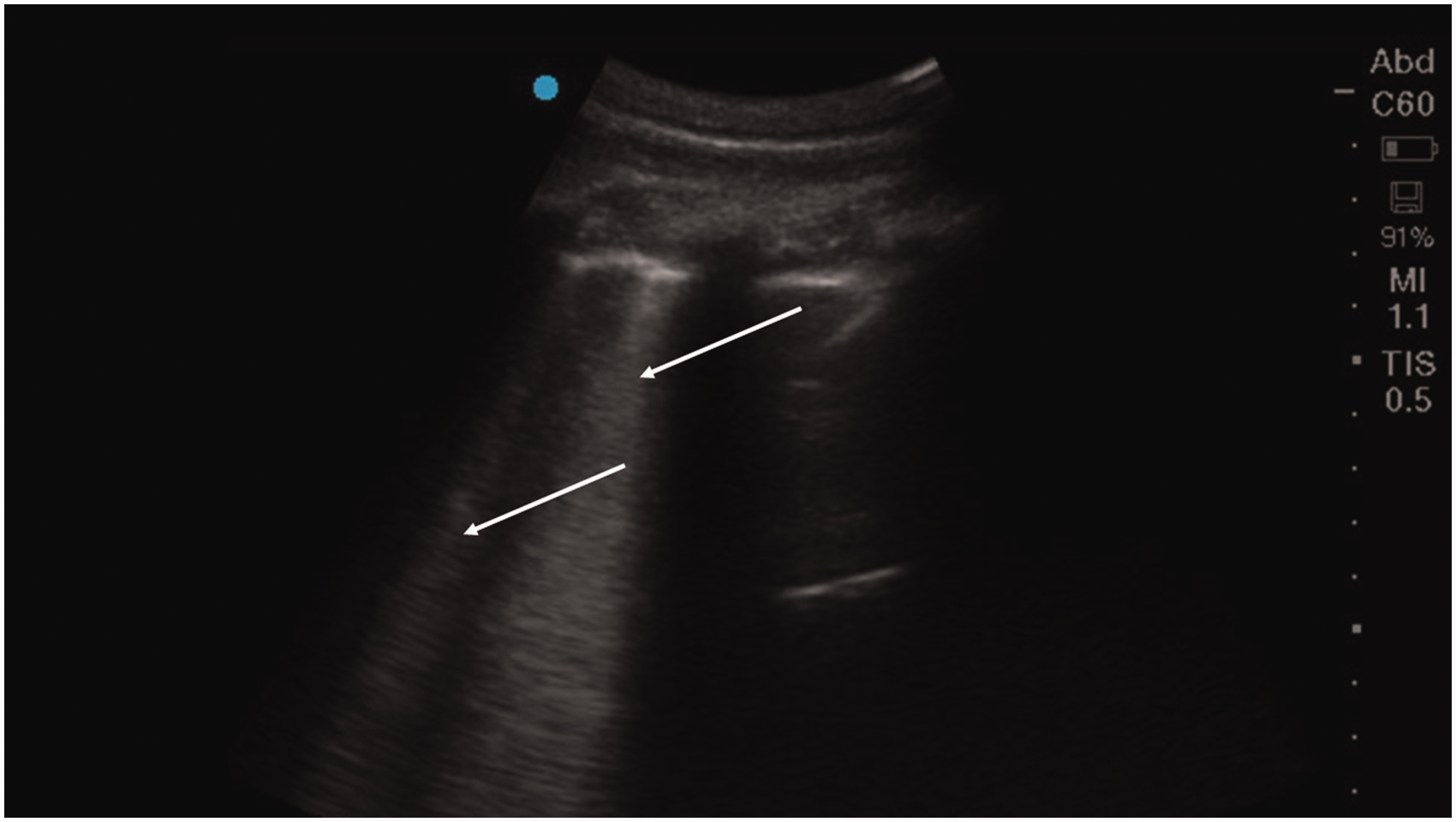
Ultrasound image of the lung of pig 2 with multiple B-lines (white arrows) extending from the pleural line to the bottom of the screen. This pattern is seen with pulmonary oedema indicating increased interstitial fluid.
Both pigs were necropsied, revealing similar findings. In pig 1, 5 ml and 10 ml of serous fluid was present in the thorax and pericardial sac, respectively. The abdomen contained 15 ml of blood-tinged serous fluid with fibrin strands. The liver and spleen were congested. The lungs were diffusely oedematous and congested (Figure 2, inset). Pig 2 had 35 ml and 170 ml of blood-tinged serous fluid in the thorax and abdomen, respectively. The pericardium contained 45 ml of serous fluid with fibrin strands. Large areas of the dorsal aspect of the lungs were congested and oedematous (Figure 2). Bordering this area, multifocal subpleural petechial haemorrhages were present. Subepicardial petechiae were present along the coronary arteries of the heart. The liver, spleen and kidneys were congested.
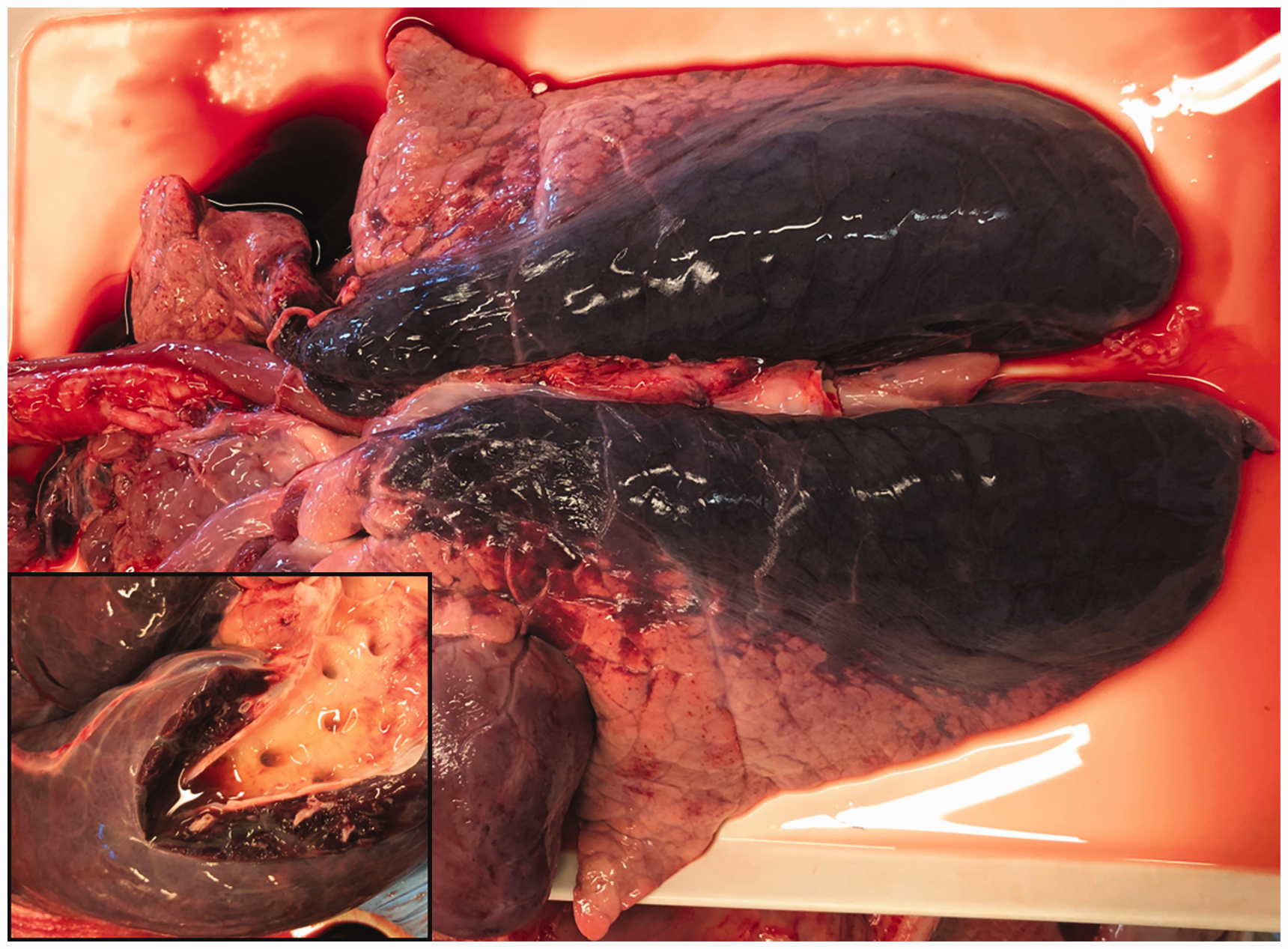
Macroscopic lung lesions. Main picture: lungs from pig 2 showing congestion and oedema in dorsal parts of the lungs. Inset: lungs from pig 1 showing diffuse oedema, congestion and free fluid in a main bronchus.
Tissues for histological examination were collected from the lungs of both pigs, and from pig 2 also from the kidney, heart and liver. Tissues were immersion fixed in 4% formaldehyde for several weeks and then dehydrated in graded ethanol and paraffin embedded. Paraffin sections (3 µm) were mounted on slides and stained with haematoxylin and eosin (H&E). The H&E sections were examined by light microscopy.
In the lungs of both pigs, a severe alveolar and interstitial oedema, haemorrhages and blood-filled alveolar septa were observed (Figure 3). Multifocally, an increased number of macrophages and fewer other inflammatory cells, including lymphocytes and neutrophils, and sloughed epithelium were present in the alveoli. In pig 1, there were few hyaline membranes and few neutrophils in septa. In organs from pig 2, the major finding was excessive congestion and haemorrhage. Multifocally in the kidney, there were mild degenerative changes of proximal tubules.
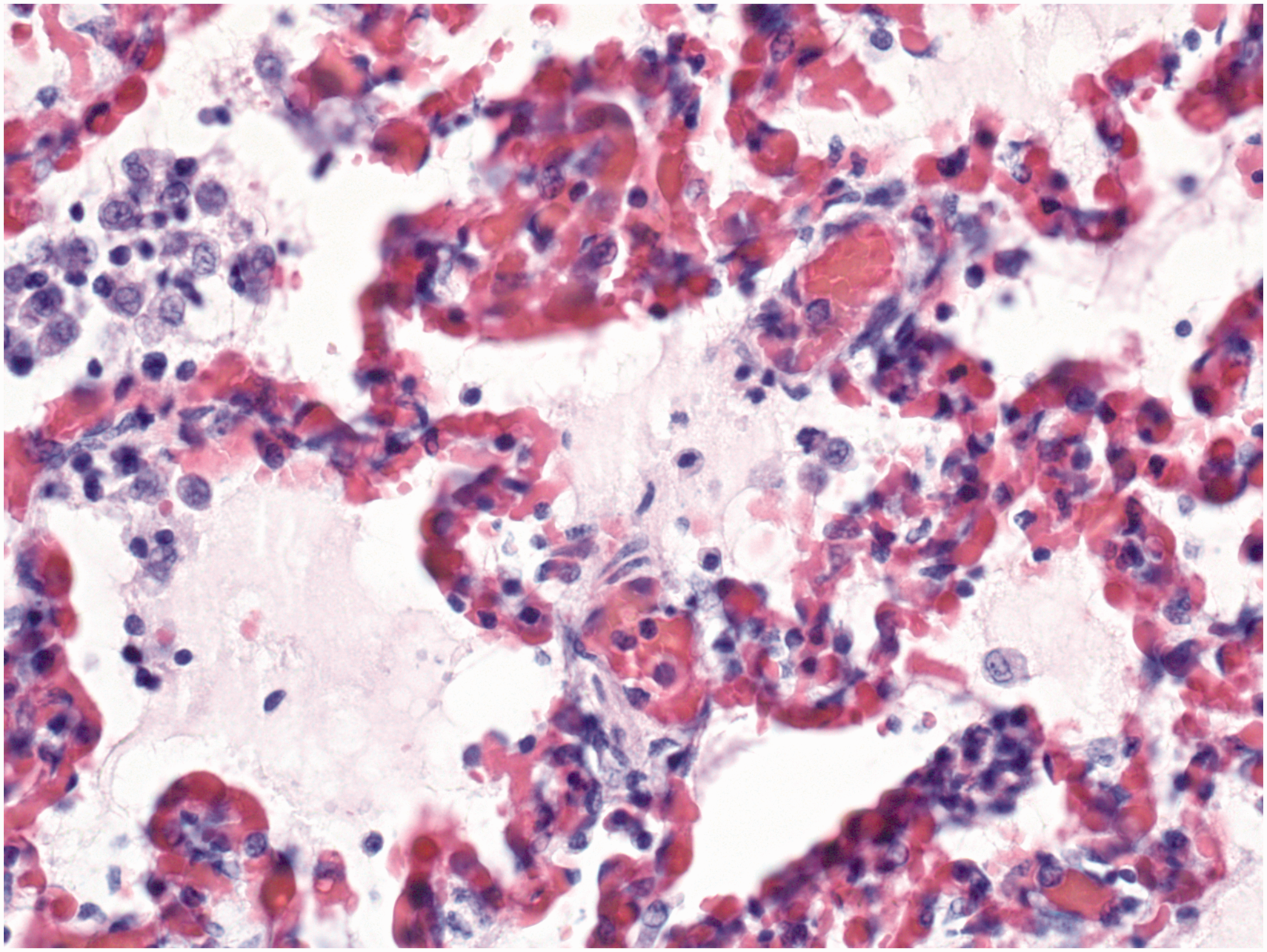
Histology picture of lung, pig 2, showing alveolar oedema and blood-filled alveolar septa. An increased number of macrophages and fewer lymphocytes and neutrophils are present in the alveoli. Haematoxylin and eosin, 40x.
Bacteriological culture from internal organs and propofol syringes was performed on Difco Columbia blood agar base (Becton, Dickinson and Company, France) with 5% sheep blood, incubated at 37°C overnight in 5% CO2. Preliminary bacteriological identification was performed by Gram staining, subculturing on Brolacin agar (C.L.E.D. agar) (Merck, Germany) and basic biochemical testing, that is, oxidase, catalase and indole (BBL DrySlide Indole, Becton, Dickinson and Company, France). Selected bacterial isolates were sent to an external laboratory for identification by matrix assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF). From both pigs, slight to moderate bacterial growth was detected in the lungs and liver, and from pig 1 also the spleen. Heavy bacterial growth was observed from all syringes (Figure 4). Isolates from both pigs and propofol syringes were identified as either species Klebsiella oxytoca (two isolates) or genus Klebsiella (two isolates). Enterobacter cloacae was recovered from one syringe.
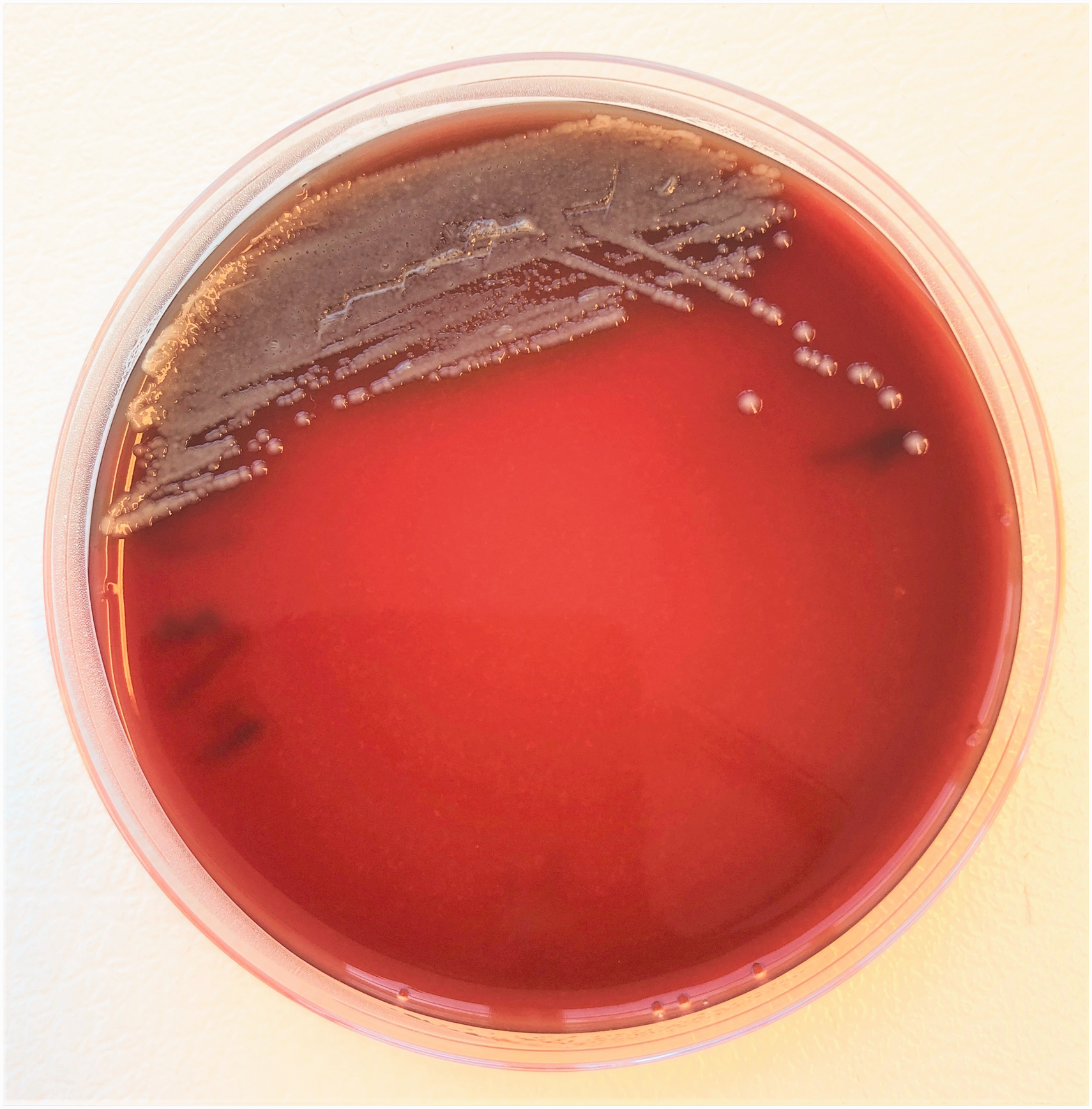
Growth of Enterobacteriaceae after inoculation of about 10 µl propofol from a syringe on blood agar.
Discussion
Klebsiella sp. were obtained in pure culture from several organs of both pigs, demonstrating systemic infection. This, together with the clinical and pathological findings, indicates septic shock. Sepsis is defined as a life-threatening organ dysfunction caused by a dysregulated host response to infection, 10 while septic shock is a subset of sepsis with higher risk of mortality due to severe circulatory, pulmonary, cellular and metabolic dysfunction. 11 Both pigs suffered from a severe pulmonary oedema and congestion. Sepsis is a common mediator of acute lung injury (ALI) and its most severe manifestation acute respiratory distress syndrome (ARDS), leading to acute onset of impaired oxygenation and ultimately acute respiratory failure. 12 A disruption of the alveolar barrier function leading to fluid leak and pulmonary oedema is a hallmark of ALI and ARDS. 13 ALI may lead to right ventricular dysfunction of the heart due to increased pulmonary vascular resistance. 14 Sepsis may also cause direct myocardial depression leading to systolic and diastolic dysfunction of both the left and right sides of the heart.15,16 The cardiac scan showed this clearly in pig 2, with pronounced global hypokinesia of the heart combined with multiple B-lines from both lungs suggestive of interstitial and alveolar fluid accumulation. Furthermore, generalized fluid accumulation in other tissues, as observed during necropsy, is a common feature of sepsis. 17 The vascular leakage is caused by endothelial dysfunction and may contribute to organ failure. 18
The propofol was heavily contaminated by Gram-negative bacteria, and therefore likely also contained large amounts of endotoxin. Endotoxaemia may account for many of the observed clinical and pathological findings, as previously described in experimental LPS infusion studies.19,20 We cannot, however, exclude that other toxins may have been partly responsible for the progression of clinical signs. Strains of both K. oxytoca and E. cloacae have been shown to produce toxins.21,22 In vitro cytotoxin production by K. oxytoca is enhanced in soy-based media, 23 which is interesting considering that propofol is formulated in soybean oil. K. oxytoca is a human opportunistic pathogen displaying a global increase in multi-drug resistance and is regarded as a potential major threat to human health. 21 In addition to the direct impact on the experimental animals, the potential impact of bacterial contamination on people handling the animals, the propofol, or a contaminated environment should not be overlooked.
Two bacterial isolates were identified to only the genus Klebsiella by MALDI-TOF. Using this method, a score of ≥2.0 and a substantial difference between score value of best and second-best database match is needed to identify an isolate to the species level. 24 K. oxytoca was the best match (scores >2.25) for both isolates. The difference between the first and second-best match, Raoultella ornitholytica, was, however, insufficient to discriminate between the two species. Due to the close relatedness of K. oxytoca and R. ornitholytica, differentiation by MALDI-TOF has proven difficult. 24
Klebsiella spp. and E. cloacae are ubiquitous in environments such as water and soil and are common inhabitants of the mammalian intestinal tract.25,26 The pigs used for live-tissue training were not routinely cleaned before the procedure and their skin may have been contaminated by faecal matter. Simultaneous handling of the pigs and administration of propofol may have led to faecal cross-contamination of a propofol vial which was subsequently used for filling multiple syringes. The propofol vials were opened and kept at room temperature until empty. Used propofol syringes were not immediately discarded but re-filled with propofol as needed. Subsequent storage of syringes at room temperature, potentially for several days, constituted excellent conditions for bacterial growth and endotoxin development in more than one syringe. The propofol vial suspected of being contaminated had already been discarded and hence not available for culturing.
This case report may serve as a reminder of the importance of applying strict aseptic standards in the operating room in general, and specifically in the handling of propofol. Careful review of product data sheets is of utmost importance; according to this and to other published literature, propofol should be aseptically drawn into sterile syringes or infusion sets immediately after the opening of a vial, and administration should start directly thereafter.8,27 A strict aseptic technique should be applied throughout the period of administration, and the propofol contained in one syringe or vial is for single patient use only. Any remains in syringes or vials should be discarded after 6 or 12 h, respectively.8,27 Propofol formulations containing antimicrobial additives such as benzyl alcohol or EDTA have been developed in order to reduce the risk of nosocomial infection. 28 Some factors may, however, limit their usefulness, such as use only for short-time anaesthesia in veterinary patients or the unavailability of such products in some countries.
Footnotes
Data availability statement
Original data is available upon request by e-mail to
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
