Abstract
Poorly designed preclinical studies may compromise human health due to erroneous conclusions regarding treatment effects in addition to contributing to experimental irreproducibility and wasted resources. Randomization is one of the crucial steps to enhance scientific rigor and is a commonly recognized bias-reducing instrument that increases the reliability and reproduction of studies involving animals (even with syngeneic animals). This procedure should be considered when planning a study and reported during data publication. In this context, this work aimed to highlight the importance of adopting quality measures in preclinical trials, with an emphasis on animal randomization. The ‘Mouse Randomization’ app was developed to help researchers estimate an adequate sample size to obtain significant statistical power, ensuring the ethical use of animals. This app is freely available on the internet to carry out animal randomization and calculate sample sizes for in vivo experiments. We believe that this brief discussion about animal randomization could raise awareness among researchers on how to improve the quality of preclinical research, increasing reproducibility and avoiding animal misuse.
Introduction
Preclinical studies’ irreproducibility is an important impediment to the translation between basic research and clinical therapy. The risks of bias can threaten the validity of preclinical research and may hinder its progress to the stage of clinical trials. Consequently, it is believed that by reducing the bias of animal studies, it is possible to contribute to the reduction in false-positive results and the failure of the use of some drugs in humans that work in animal models. The development of drugs to treat stroke is an excellent example of successful preclinical studies with subsequent failure in clinical trials. Thus, adopting quality standards may increase the effectiveness in selecting drugs, therapies, and treatments for clinical trials. 1
Data on irreproducibility in preclinical trials deserve attention and should awaken researchers’ critical thinking about the ethical use of animals in experiments. Following the steps detailed in Figures 1 and 2, before and after decision-making on using experimental animals in preclinical trials is important. It is not uncommon to observe in vivo data obtained from a small number of animals, for example, five or six animals per group. In some cases, these experiments are conducted in a single day, and, in this sense, there is still a possibility that the observed effect occurs by chance, given the non repetitive nature of the experiment, which does not allow the effect pattern to be confirmed. In addition, the low number of animals may still be insufficient to achieve statistical power (which will be discussed below). Although it may seem more ethically appropriate to use a small number of animals, if the experiment is not well conducted (in terms of reproducibility and sufficient power), it may result in waste, which ends up violating the principle of ‘Reduction’.
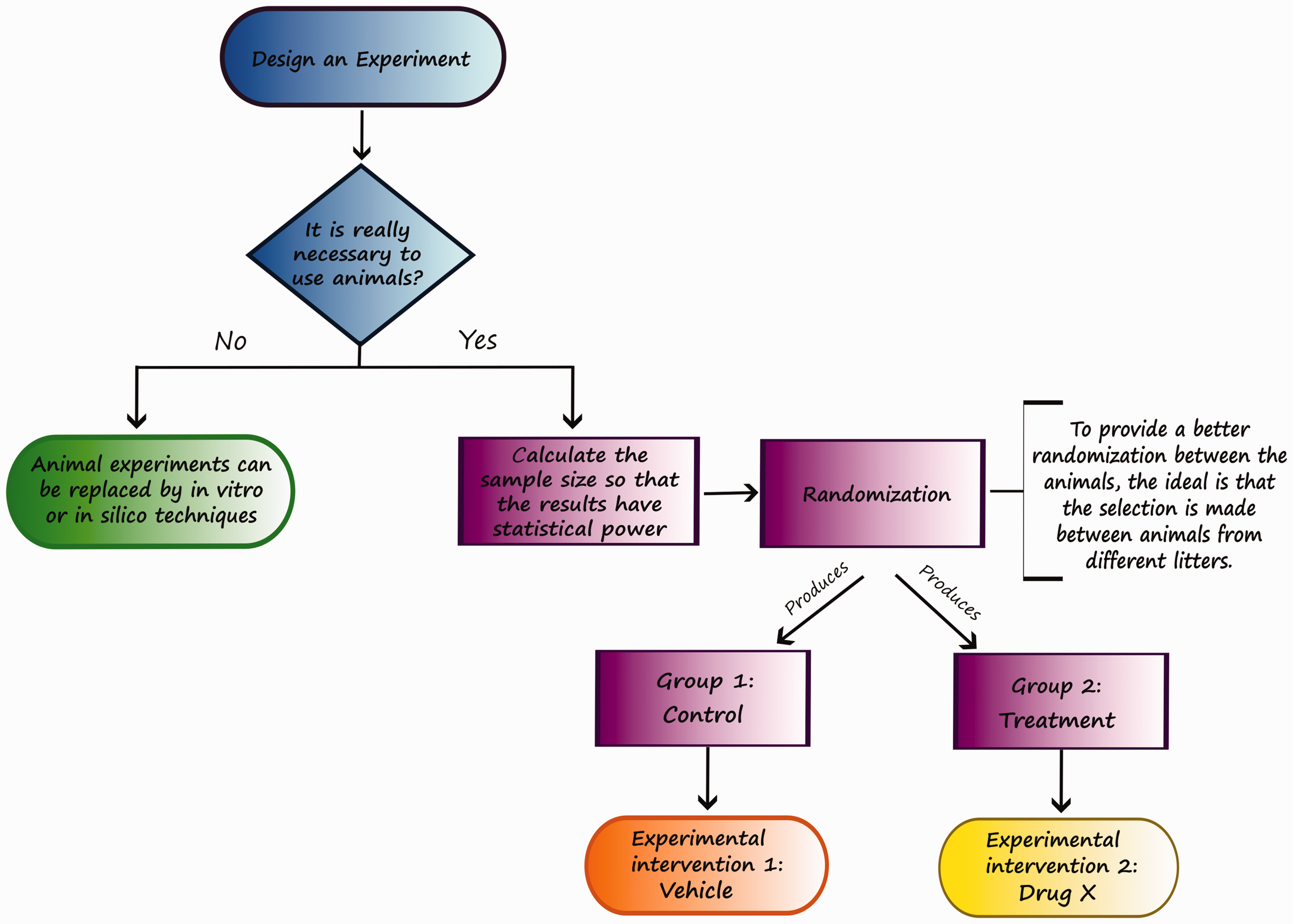
Critical steps to avoid bias in animal testing. First, it must be determined whether animals are necessary for the experiment. If animals are not needed, the experiments should be performed in silico or in vitro. However, if they are required, it is important to calculate the appropriate sample size to obtain statistical power and then perform the randomization of the animals with a random number generator to assign animals to the test and control groups. Pay attention to the importance of using animals obtained from different litters, thus avoiding potential biases. Finally, ethical and accurate collection and analysis of data for publication in scientific journals should be clear and transparent.
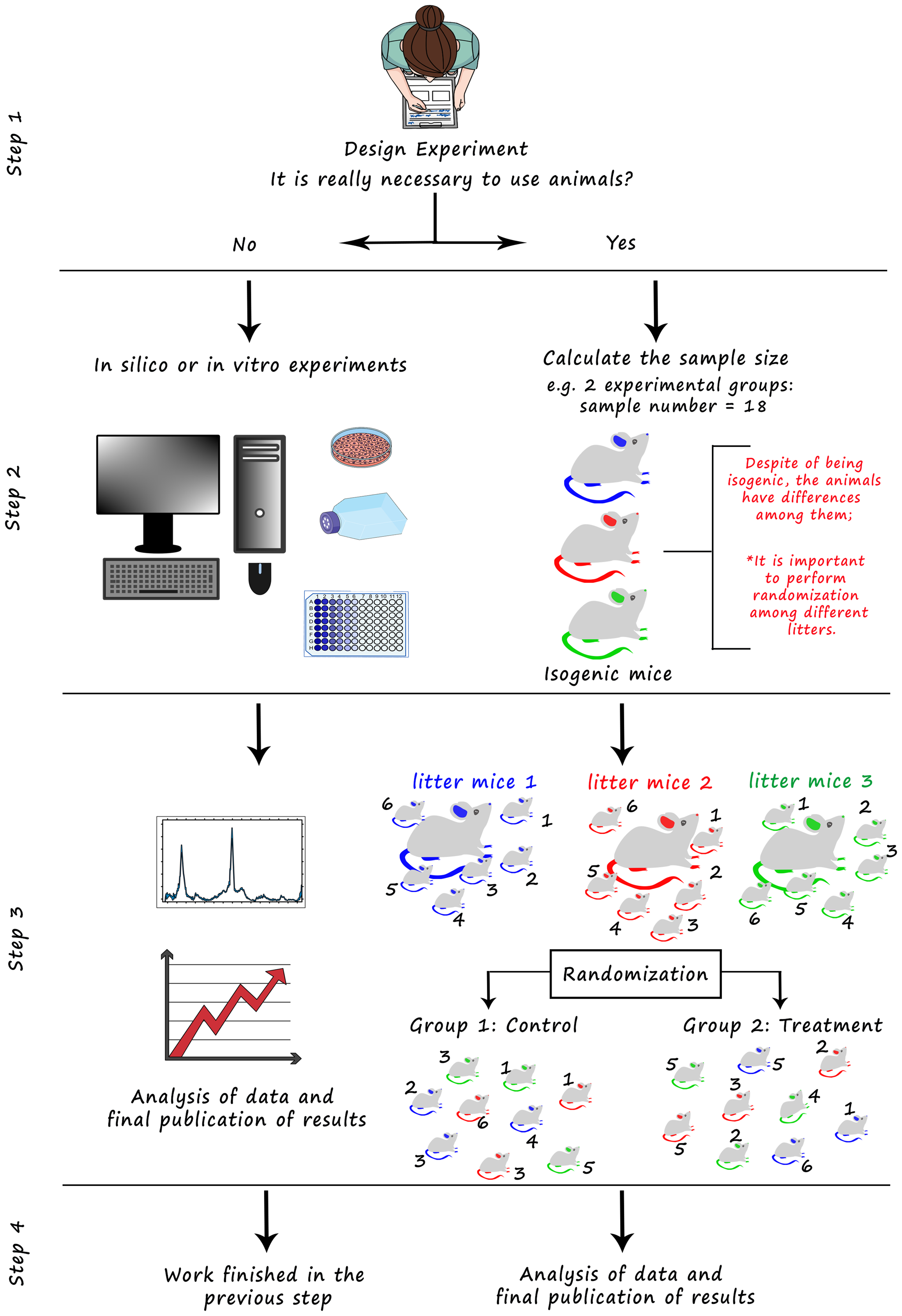
Pictorial flowchart illustrating the decision-making process when carrying out in vivo research and highlighting the importance of randomization in the reproducibility of scientific research as mentioned in Figure 1. Initially, the researcher needs to evaluate the need to use experimental animals in the study (step 1), remembering that it is always ideal to minimize and reduce the use of animals in scientific research. When there is no need for the use of animals, research can proceed with in vitro or in silico experiments. If the use of animals is required and justified, it will be necessary to calculate the appropriate sample size to reduce the number of animals to the minimum and to ensure the study is appropriately powered (step 2). Next, it is necessary to randomize the animals among the different study groups. The first step is to assign identifying numbers to each mouse and then perform randomization, that is, a ‘draw’ that will generate random numbers and allocate mice to study groups. This randomization can be performed by programs and applications, such as the one developed in this work, many of which are available on the internet and free of charge. Remembering that an important step for randomization is to take the total number of animals from different litters to reduce bias (steps 2 and 3). Finally, perform open and transparent analysis and publication of the results obtained (step 4).
To resolve all the problems surrounding the issues of failure in preclinical studies related to reproducibility, the National Institutes of Health, journal editors, funding agencies, and scientific leaders have proposed guidelines to improve the rigor and reproducibility of preclinical research. 2 Among the first standards adopted were randomization, blinding, sample-size estimation, littermate control, and data handling. 2
Randomization or random allocation is a technique that chooses individuals for treatment and control groups by chance without preference (e.g., weight) to avoid bias. 2 This allows researchers to account for known and unknown factors that may affect results, which will be similarly distributed among the treatment and control groups, 3 allowing statistical inferences about the treatment effect. 4
Despite the recommendation of randomization in preclinical studies, 5 its use has increased little. Several researchers still believe that syngeneic mouse models are genetically identical. Thus, the experiment would not need randomization. Nevertheless, this idea is not correct, as even monozygotic twins are different. 6 In this context, it is vital to implement randomization.
We must consider that there may be several random variables (or random effects) that cannot be accounted for and need to be considered during experimental design and data analysis. 7 For example, behavioral differences could be caused by circadian rhythm differences in data collected from the morning to the afternoon; members of one mouse litter may be more similar to each other than mice of different litters. Thus, the ideal situation would be to start the randomization process at the animal breeding centers, that is, the total number of animals used in the study should be, preferably, taken from different litters, as illustrated in step 3 of Figure 2. 8 Random effects can appear when measurements are made by different people, at different times, or even with a reagent that deteriorates over time. These effects should be considered during statistical analysis or even at the design stage. 9 Certain characteristics cannot be known, and in this scenario, using the randomization technique is the only way to control their potential influence. However, this effect is limited when the sample size is low, which is the main limitation of many preclinical studies. 10
A randomization system involves generating random numbers for each subject or condition of treatment. 11 There are currently several computer programs and websites available that can quickly generate random numbers, facilitating the randomization process for researchers and students. Table 1 lists some examples of online randomization software and programs. However, it is possible to carry out these same tasks using tools from the Excel and LibreOffice programs (not mentioned in Table 1).
Online software for randomization

The first three programs listed in Table 1 are more generalist and generate only a simply randomized classification of the number that represents the total analyzed sample. The Experimental Design Assistant (EDA) and RandoMice are more detailed programs. The EDA requires a login, but it is free and fully accessible online, while RandoMice needs to be downloaded.
To contribute to the quality of preclinical in vivo studies, our group has developed a fast and practical randomization program entitled ‘Mouse Randomization’ that was developed using scripts in Python and JavaScript languages. The program was developed as an easy-to-use tool to help researchers during animal randomization and allocation steps. Researchers can enter the number of animals that will be used, and the number of groups, and the application divides them appropriately. However, this requires the animals to be numbered sequentially. Notably, each time the program runs, it organizes the animals in a random way, different from the previous runs.
A sample-size checking tool was also developed for this app. This tool is as important as the randomization tool, as it can estimate the number of animals needed to obtain a reliable result with the correct representation of animals. In other words, it is necessary to carry out in vivo tests with an adequate sample of animals for the tests to have sufficient statistical power, thus avoiding type II (β) errors.12,13 An insufficient number of animals (e.g., too few animals tested in a single day of the experiment) can lead to false results and may not reflect the true biological effect, especially if the effect size is small.12,14 Aiming to help researchers calculate an appropriate sample size, this tool suggests the number of animals for the experiment based upon the parameters of effect size, alpha, and power, suggesting values of 0.8, 5%, and 80% as standards, respectively.12,13 However, the user is free to input the values they would like to work with and input the number of groups to which the animals will be assigned.
Conclusion
Animal models used in preclinical studies are an important source for discovering new drugs or treatment applications. However, there are inherent limitations to models that can undermine the results when extended to humans, particularly if they are biased in some way. Poorly designed preclinical studies are a source of risk to human health since they may lead to incorrect conclusions regarding treatment effectiveness. Thus, it is necessary to adopt crucial measures for improving the quality of preclinical research, such as randomization (to avoid selection bias), blinding (to minimize performance, detection or attrition bias) and appropriate sample-size estimation (to ensure adequate statistical power and reasonable use of animals). In this work, we approach randomization as one of the primary steps for the success of preclinical studies. Consequently, we have developed an application able to randomize animals into different groups, including a control group. This application also has a tool to estimate the sample size to obtain a given statistical power. We hope that this auxiliary tool will facilitate the appropriate experimental design for animal models, minimize bias, increase experimental reproducibility, and ultimately, decrease animal misuse.
Footnotes
Data availability statement
The datasets generated during the current study are available from the corresponding author on reasonable request via e-mail
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics statement
Our study did not require ethical board approval because it did not contain human or animal trials.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Oswaldo Cruz Foundation (Fiocruz), Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (Faperj – grant E-26/010.002422/2019), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq – grant 309718/2022-8).
