Abstract
Thiamine deficiency experimental models focus on using the pyrithiamine analog in male rodents, making the thiamine deficiency effects in females and the use of other thiamine antagonists, such as amprolium, unknown. We investigated the impact of thiamine deficiency with amprolium in the cerebral cortex and thalamus of male and female mice by evaluating the modulation of ERK1/2 phosphorylation. The animals were exposed for 20 days to thiamine-deficient chow with different doses of amprolium (20, 40, 60 and 80 mg/kg) and at different treatment periods (five, 10, 15 or 20 days) at a dose of 60 mg/kg. After treatments, ERK1/2 phosphorylation was analyzed by western blot. In male mice, we observed a progressive increase in ERK1/2 phosphorylation in both the cerebral cortex and thalamus in response to the dose of amprolium. In females, ERK1/2 phosphorylation did not progressively increase in response to the amprolium dosage. However, an increase in phosphorylation at the higher doses of 60 and 80 mg/kg was observed. We observed a more intense increase in ERK1/2 phosphorylation in males’ cerebral cortex and thalamus from 10 days onwards. In females, the ERK1/2 modulation profiles were similar. The results show that thiamine deficiency induction with amprolium is efficient, compatible with other recognized models that use pyrithiamine, showing changes in cell signaling in the nervous system. The study showed differences in response to thiamine deficiency with amprolium between male and female mice in relation to ERK1/2 phosphorylation and demonstrated that females respond positively to the method and can also be used as model animals.
Introduction
Thiamine has essential functions in the body and is involved in generating cellular energy. 1 Thiamine deficiency causes a series of complications in the nervous system associated with disorders in glucose catabolism and adenosine triphosphate generation related to oxidative stress, changes in neurotransmitters, inflammation and cell death. 2 Nervous tissue is characteristically affected by thiamine deficiency owing to its high metabolic and energetic rates. Thiamine deficiency can lead to neuroinflammation and neurodegeneration, the mechanisms of which are still poorly understood.2,3
Oxidative stress and inflammation are the main factors associated with neurodegeneration in thiamine deficiency. 3 Studies show that reactive oxygen species (ROS) and cytokines can cause changes in cell signaling pathways, resulting in several disorders with inflammatory characteristics. ROS, tumor necrosis factor and interleukins are related to the activation of mitogen-activated protein kinase (MAPK) pathways, 4 such as p38MAPK and extracellular signal-regulated kinases 1 and 2 (ERK1/2), suggesting the involvement of these signaling pathways in the pathogenesis of thiamine deficiency-induced neurodegeneration.5,6
Experimental thiamine deficiency models for in vivo studies in laboratory animals allow for in-depth investigation of the anatomical, physiological and cellular basis of the neurological disorders that occur in this deficiency. Two well-established experimental models for rodents are used to study thiamine deficiency.2,3 In the first method, thiamine deficiency is induced using vitamin B1-free diets for a period sufficient to deplete the body’s thiamine stores (3–4 weeks). In the second model, thiamine deficiency is generated more quickly, combining feeding with thiamine-free chow with the administration (intraperitoneal (i.p.) injections) of pyrithiamine (thiamine pyrophosphokinase inhibitor). In the latter model, pyrithiamine-induced thiamine deficiency, occurs a stereotyped progression of neurological and behavioral signs that have been mapped to changes in neuroanatomy and neurochemistry, mimicking the natural disease described for thiamine deficiency. 7
Some studies use other thiamin-analog antagonists, such as amprolium.8,9 These studies have addressed the pharmacological aspects of this substance; however, its effects have not been well-characterized yet. Nevertheless, studies have suggested that the in vivo action of amprolium in inducing thiamine deficiency may be better than that of the classic pyrithiamine model, 10 because of its capacity to block cellular uptake of the vitamin, not undergoing intracellular diphosphorylation and inhibiting thiamine diphosphorylation. Due to this feature, amprolium could be considered as the analogue that affects thiamine-dependent rather than thiamine diphosphate-dependent processes. 10 Recent relevant studies have demonstrated significant behavioral and metabolic disorders in thiamine-deficient mice with amprolium.11,12
Furthermore, there has recently been a significant movement to include both sexes in animal studies. 13 However, biological differences between males and females occur in several respects, which may restrict the use of both sexes in research. For example, different behavioral manifestations with sex-associated interference are well known.14,15 Hormonal variations associated with female reproductive phases interfere with several aspects of behavior. 16 Likewise, differences in antioxidant resistance and inflammation between males and females have been well characterized.17,18 Studies have demonstrated greater resistance to oxidative stress and neuroinflammation in females.19 –21 Nevertheless, depending on the study and experimental model used, the use of both sexes may be valid, thereby optimizing the use of laboratory animals in scientific research.
In this study, we aimed to improve our model of induction of dietary thiamine deficiency with amprolium, determine an efficient dose and sufficient exposure time to observe important changes, evaluate the effects of thiamine deficiency with amprolium in males and females and verify whether there is any interference in response to the experimental model. For this, we evaluated the modulation of ERK1/2 phosphorylation in the nervous tissue as a parameter, as it is an important signaling pathway for several cellular functions and is sensitive to several stress factors, including thiamine deficiency. 5
Methods
Animals
A total of 78 male and 78 female 50-day-old Swiss mice (Mus musculus) were provided by the Vivarium of the Universidade Federal do Tocantins (Araguaína, TO, Brazil). At weaning, the number of animals was randomly culled and housed by sex in open-top cages (3–4 per cage), maintained in an air-conditioned cabinet (23 ± 1°C; humidity 55 ± 5%;) on a 12 h light/dark cycle with tap water and food (AIN-93; PRAG Soluções Biociências, Jaú, SP, Brazil) available ad libitum. All mice were maintained in the institutional Experimental Pathology Laboratory under environmentally controlled conditions according to the Universidade Federal do Tocantins Ethics Committee on Animal Use (CEUA-UFT), following regulations of the National Council for Animal Experimentation Control (CONCEA) of the Ministry of Science, Technology, and Innovation, which follows international guidelines, including the EU Directive 2010/63. All treatments and euthanization by decapitation were performed according to the Ethics Code of Animal Use in Research and were approved by CEUA-UFT (process 23.101.001.708/2019-63). All efforts were made to minimize animal suffering and reduce the number of animals used. Euthanasia by decapitation is a necessary method for studying intracellular signaling pathways, as commonly used anesthetics interfere with the modulation of responses. 22 The sample size was chosen based on pilot studies and previous experience. 12 Nest building material was provided, and all cages contained enrichment such as a tunnel. The animals were always handled with sterile gloves, and the material for nests and bedding (autoclaved shavings) was monitored daily and changed every three days. The animals were monitored daily for welfare control, observing possible important changes in food and water consumption, as well as signs of pain, suffering and distress.
Reagents
The primary antibodies used were anti-phospho-ERK1/2 (Cat# M8159, RRID: AB_477245), anti-ERK1/2 (Cat# M5670, RRID: AB_477216) and anti-β-actin (Cat# A5441, RRID: AB_476744), and the secondary antibodies were rabbit anti-mouse IgG-horseradish peroxidase (HRP)-conjugated (Cat# A9044, RRID: AB_258431) and goat anti-rabbit IgG-HRP-conjugated (Cat# A9169, RRID: AB_258434) (Sigma-Aldrich, MO, USA). Acrylamide, Folin & Ciocalteu’s phenol reagent, 3,3′-diaminobenzidine tetrahydrochloride (DAB) and ammonium persulfate were purchased from Sigma-Aldrich. Amprolium was obtained from Santa Cruz Biotechnology (Dallas, TX, USA). Sodium dodecyl sulfate (SDS), N,N,N,N-tetramethylethylenediamine, Tween® 20, Tris and bovine serum albumin were purchased from Amresco (Solon, OH, USA). AIN-93M (standard) and AIN-93TD (thiamine-deficient) chow were purchased from PRAG Soluções Biociências (Jaú, SP, Brazil). Glycine was obtained from Uniscience (Osasco, SP, Brazil) and all other reagents were of the highest analytical grade.
Treatments
An in vivo model of dietary thiamine deficiency was used in this study, in conjunction with the administration of the thiamine antagonist amprolium. 12 Mouse categories (sex) were separated into 13 male and 13 female groups (n = 6 per group; eight treated groups and five control groups for each category) and subjected to two different treatment protocols to analyze the modulation of ERK1/2 phosphorylation in the central nervous system at different doses of amprolium (protocol 1) and at different exposure times (protocol 2) to thiamine deficiency with amprolium (Figure 1). The animals were randomly assigned to each treatment group and the treatments were carried out at the same time every day (14:00 h).
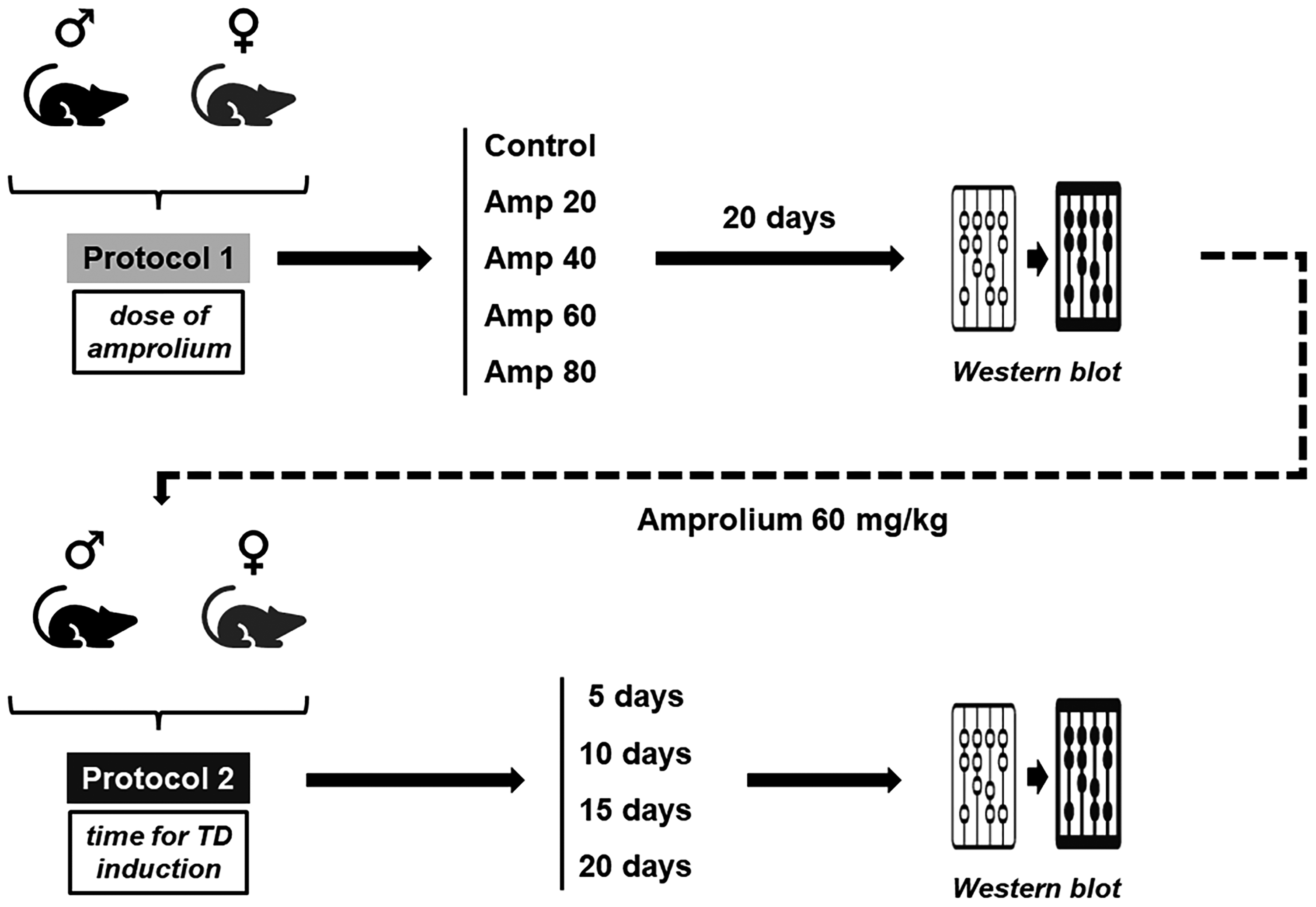
Experimental design of the dietary thiamine deficiency (TD) model with amprolium in male and female mice. In the first stage (protocol 1) the best dose of amprolium (Amp) for inducing thiamine deficiency was determined. The mice received different doses (20, 40, 60 or 80 mg/kg) for 20 days, when the modulation of extracellular signal-regulated kinases 1 and 2 (ERK1/2) phosphorylation was evaluated by western blot. In the second stage (protocol 2), the most suitable time for thiamine deficiency induction with amprolium was evaluated. The dose of amprolium that best modulated ERK1/2 (60 mg/kg) was used for five, 10, 15 or 20 days in both male and female mice. ERK1/2 modulation was assessed by western blot after each time. Control groups received AIN-93M standard chow and saline solution (0.9% NaCl, intraperitoneally (i.p.)) throughout the treatments; deficient groups received thiamine-free AIN-93TD chow and amprolium (i.p.).
In protocol 1, to evaluate different amprolium doses, male and female mice were treated for 20 days with daily i.p. injections of saline solution (NaCl 0.9%, control groups) or amprolium (diluted in saline solution) at doses of 20, 40, 60 and 80 mg/kg, totaling five groups per category. After the treatment period, the animals were euthanized and analyses were performed.
In protocol 2, to assess the profile of ERK1/2 phosphorylation at different periods of thiamine deficiency with amprolium, groups of males and females were treated daily with saline (0.9% NaCl i.p., control groups) or amprolium (i.p.) at a dose of 60 mg/kg for five, 10, 15 or 20 days, totaling eight groups of treatments for each sex category (each treated group had its respective control). In this protocol, the dose used was determined by previous tests (protocol 1), using the dose associated with the most intense modulation of ERK1/2. Euthanasia and analyses of the different groups (control and deficient) were performed after each treatment period (five, 10, 15 or 20 days). The injected volumes in both protocols were adjusted to 0.1 ml/10 g of body weight.5,23
The control groups received AIN-93M standard chow (6.0 mg thiamine hydrochloride per kilogram of feed; PRAG Soluções Biociências) according to the American Institute of Nutrition 24 ad libitum throughout the treatments. The deficient groups received thiamine-deficient AIN-93TD chow (0.0 mg thiamine hydrochloride per kilogram of feed; PRAG Soluções Biociências) during periods of deficiency induction.5,6,12 The constituents of the thiamine-deficient diet were identical to those of the standard diet, except for a lack of vitamins.
Western blotting
The cerebral cortex and thalamus were dissected, mechanically homogenized in 300 μl of sample buffer (200 mM Tris, 40 mM EDTA, 4% SDS, pH 6.8) and immediately boiled for 5 min. A sample dilution solution (1:4 v/v; 40% glycerol, 50 mM Tris, and minimal bromophenol blue) and β-mercaptoethanol (at a final concentration of 5%) were added to each sample. The protein content was estimated at a wavelength of 750 nm, and the concentration was calculated using a standard curve of bovine serum albumin (BSA). 25 The samples (60 µg of total protein) were resolved by SDS–polyacrylamide gel electrophoresis (VERT-i10™ electrophoresis system, Loccus, Cotia, SP, Brazil) using 10% gels. 23 Proteins were transferred to nitrocellulose membranes (1.2 mA/cm2; 1.5 h) using a wet blotting apparatus (WEST-i10™, Loccus). 5 The membranes were blocked for 1 h with 2% BSA in Tris buffered saline (TBS; 10 mM Tris, 150 mM NaCl, pH 7.5). ERK1/2 total and phosphorylated forms, as well as β-actin, were detected by overnight incubation with specific antibodies diluted at 1:10,000 (P-ERK1/2 and β-actin) or 1:40,000 (T-ERK1/2) in TBS-T (10 mM Tris, 150 mM NaCl, 0.1% Tween® 20, pH 7.5) containing 2.5% BSA. Thereafter, the membranes were incubated with anti-rabbit or anti-mouse peroxidase-linked secondary antibody (1:5,000) for 1 h, and the reactions were developed using the DAB chromogen. 5 All blocking and incubation steps were followed by three washes (5 min) with TBS-T. The optical density (OD) of the bands was quantified using ImageJ version 1.52k software (RRID: SCR_003070; National Institute of Health, Bethesda, MD, USA). The phosphorylation level of ERK1/2 was determined as the ratio of the OD of the phosphorylated band to the OD of the total band, and the data were expressed as a percentage of the control (considered 100%).5,6,23 The values are presented as the mean ± standard deviation (SD). β-actin was used as a control for protein loading.
Statistical analysis
Data are expressed as mean ± SD and statistical significance was determined by analysis of variance, followed by Duncan’s post hoc test, when appropriate.6,23 Statistical significance was set at p ≤ 0.05. The data were processed using the STATISTICA ‘98 Edition software (RRID: SCR_014213, StatSoft, Tulsa, OK, USA).
Results
Determination of the best dose of amprolium for induction of thiamine deficiency
Evaluating MAPK modulation as a cell change parameter, we observed a progressive increase in ERK1/2 phosphorylation in male mice, in both the cerebral cortex and the thalamus, in response to the dose of amprolium (Figure 2). In the cerebral cortex, the highest dose of amprolium (80 mg/kg) produced the greatest changes in ERK1/2 modulation, reaching increases of 125% (ERK1) and 110% (ERK2) compared with that in the control (ERK1, p = 0.00006; ERK2, p = 0.00003). Furthermore, in the cerebral cortex, there was a more progressive profile of increased phosphorylation, indicating differences in the intensities of ERK1/2 modulation between the two higher doses of 60 mg/kg and 80 mg/kg (ERK1, p = 0.020; ERK2, p = 0.013) (Figure 2(b)).
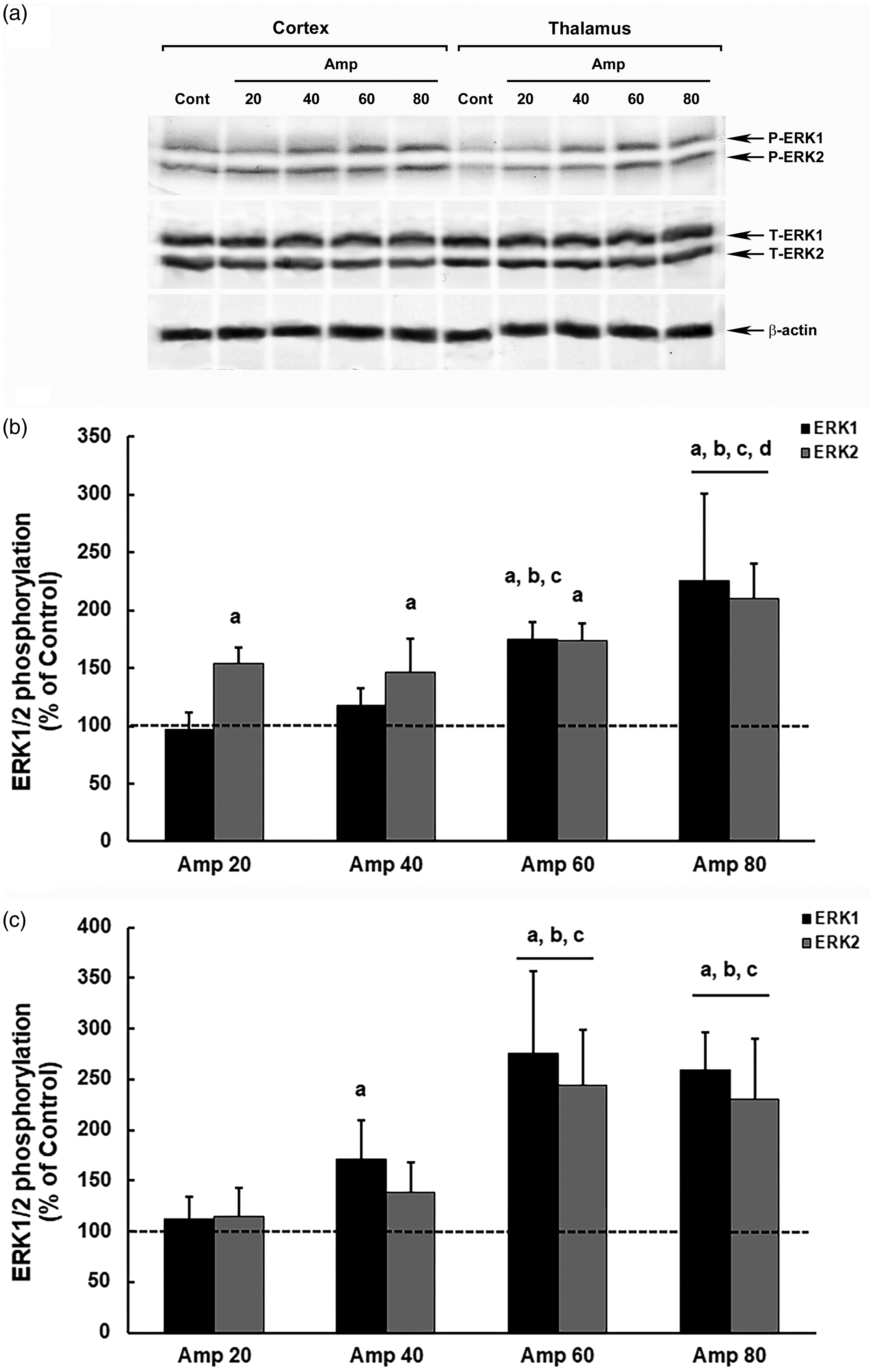
Modulation of ERK1/2 phosphorylation in the brain of model thiamine deficiency male mice that were subjected to different doses of amprolium (Amp). The panel shows representative immunoblotting (a) and quantification of ERK1/2 phosphorylation in the cerebral cortex (b) and thalamus (c) from deficient male mice treated for 20 days with AIN-93TD chow and amprolium (20, 40, 60 and 80 mg/kg) or AIN-93M chow and saline (NaCl 0.9%; control group). Total and phosphorylated forms of ERK1/2 and β-actin were detected by specific antibodies and the reaction was developed by 3,3′-diaminobenzidine tetrahydrochloride chromogen. The phosphorylation was determined as a ratio of the optical density (OD) of the phosphorylated band over the OD of the total band, and the data are expressed as a percentage of the control (considered as 100%). The values are presented as mean ± standard deviation derived from six independent replicates. The results were analyzed using analysis of variance followed by Duncan’s test. ‘A’ denotes p ≤ 0.05 compared with the control, ‘B’ p ≤ 0.05 compared with the Amp 20 group, ‘C’ p ≤ 0.05 compared with the Amp 40 group, and ‘D’ p ≤ 0.05 compared with the Amp 60 group.
In the thalamus, the modulation of ERK1/2 was more intense, reaching an increase of 175% (ERK1) and 144% (ERK2) at a dose of 60 mg/kg of amprolium, compared with those in the control (ERK1, p = 0.00005; ERK2, p = 0.00004). However, there was no difference in the increases in ERK1/2 phosphorylation between the two higher doses of 60 mg/kg and 80 mg/kg (ERK1, p = 0.609; ERK2, p = 0.540) (Figure 2(c)).
In contrast, we did not observe a progressive response in the modulation of ERK1/2 in relation to the increase in the amprolium dose in female mice, showing an abrupt increase in phosphorylation at higher doses of 60 mg/kg and 80 mg/kg (Figure 3). In the cortex, ERK1 showed increases of 103% and 92% at doses of 60 mg/kg (p = 0.00006) and 80 mg/kg (p = 0.00007), respectively, in relation to that in the control, and ERK2 increased by 112% and 118%, respectively, compared with those in the control at the two highest doses (60 mg/kg, p = 0.0001; 80 mg/kg, p = 0.00007; Figure 3(b)).
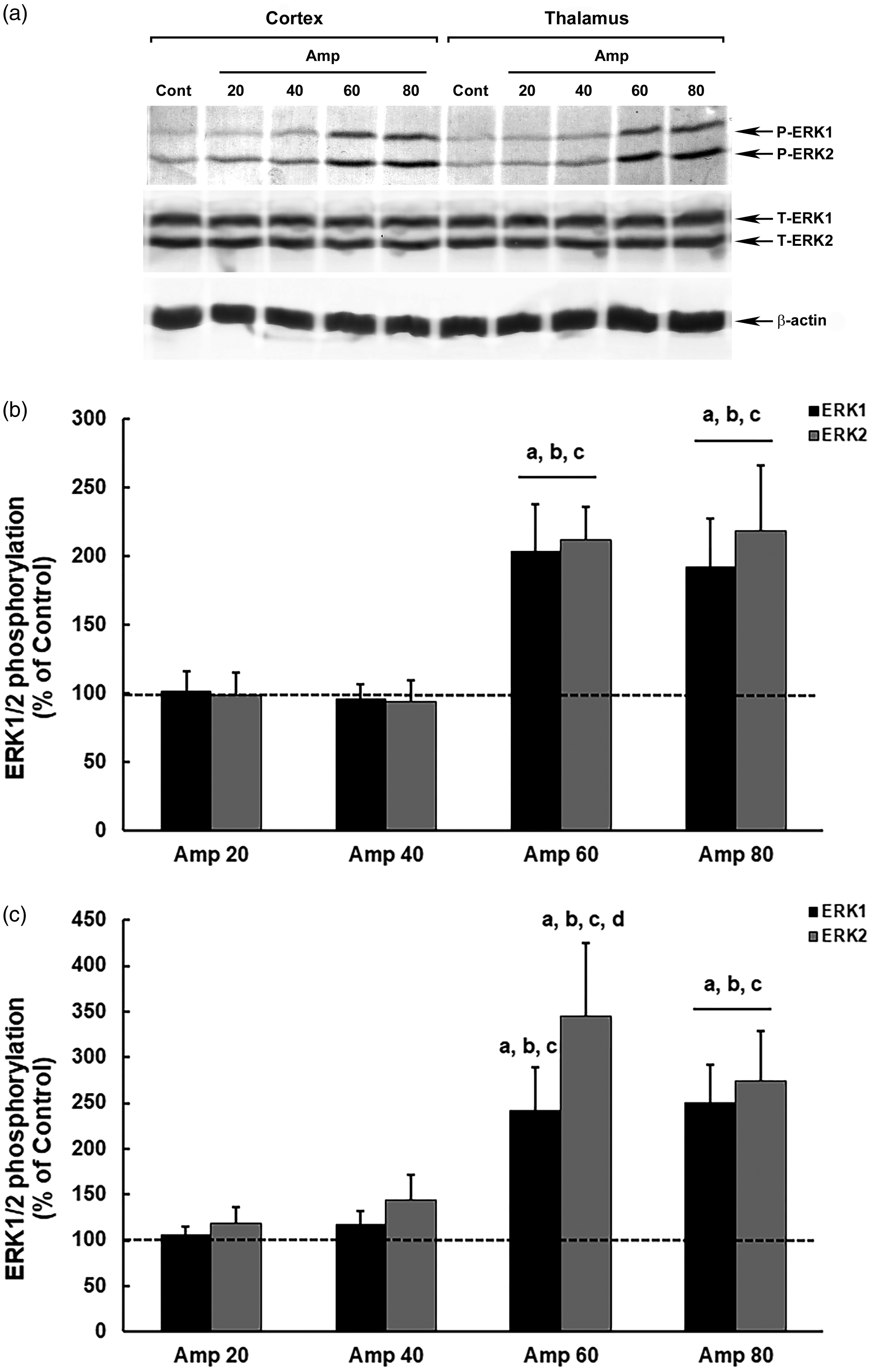
Modulation of ERK1/2 phosphorylation in the brain of model thiamine deficiency female mice that were subjected to different doses of amprolium (Amp). The panel shows representative immunoblotting (a) and quantification of ERK1/2 phosphorylation in the cerebral cortex (b) and thalamus (c) from deficient female mice treated for 20 days with AIN-93TD chow and amprolium (20, 40, 60 and 80 mg/kg), or AIN-93M chow and saline (NaCl 0,9%; control group). Total and phosphorylated forms of ERK1/2 and β-actin were detected by specific antibodies, and the reaction was developed by 3,3′-diaminobenzidine tetrahydrochloride chromogen. The phosphorylation was determined as a ratio of the optical density (OD) of the phosphorylated band over the OD of the total band. The data are expressed as a percentage of the control (considered 100%). The values are presented as mean ± standard deviation derived from six independent replicates. The results were analyzed using analysis of variance followed by Duncan’s test. ‘A’ denotes p ≤ 0.05 compared with the control, ‘B’ p ≤ 0.05 compared with the Amp 20 group, ‘C’ p ≤ 0.05 compared with the Amp 40 group, and ‘D’ p ≤ 0.05 compared with the Amp 80 group.
In the thalamus, the increase in ERK phosphorylation in females was more intense, similar to that in male mice, reaching an approximately 160% increase in ERK1 at doses of 60 mg/kg (p = 0.00006) and 80 mg/kg (p = 0.00003) and in ERK2 at doses of 80 mg/kg (p = 0.00007), in relation to that in the control, and up to a 245% increase in ERK2 phosphorylation at a dose of 60 mg/kg, compared with that in the control (p = 0.00003) (Figure 3(c)).
Determination of the most suitable time for thiamine deficiency induction with amprolium
After induction of thiamine deficiency for 20 days to determine the dose of amprolium that best demonstrated ERK modulation in both male and female mice, we evaluated the treatment time required for deficient animals to exhibit a significant increase in ERK1/2 phosphorylation in the cerebral cortex and thalamus. Therefore, we used a dose of 60 mg/kg, considering that the phosphorylation modulation responses were evident in both sexes and brain structures (Figures 2 and 3).
In male mice (Figure 4), we observed increased phosphorylation of ERK1/2 in the cerebral cortex at all time points of thiamine deficiency treatment with amprolium. However, ERK1/2 modulation was markedly more intense when amprolium treatments reached 15 days, with increases in phosphorylation of up to 189% (ERK1) and 201% (ERK2) compared with those in the controls (ERK1 and ERK2, p = 0.00003) (Figure 4(b)).
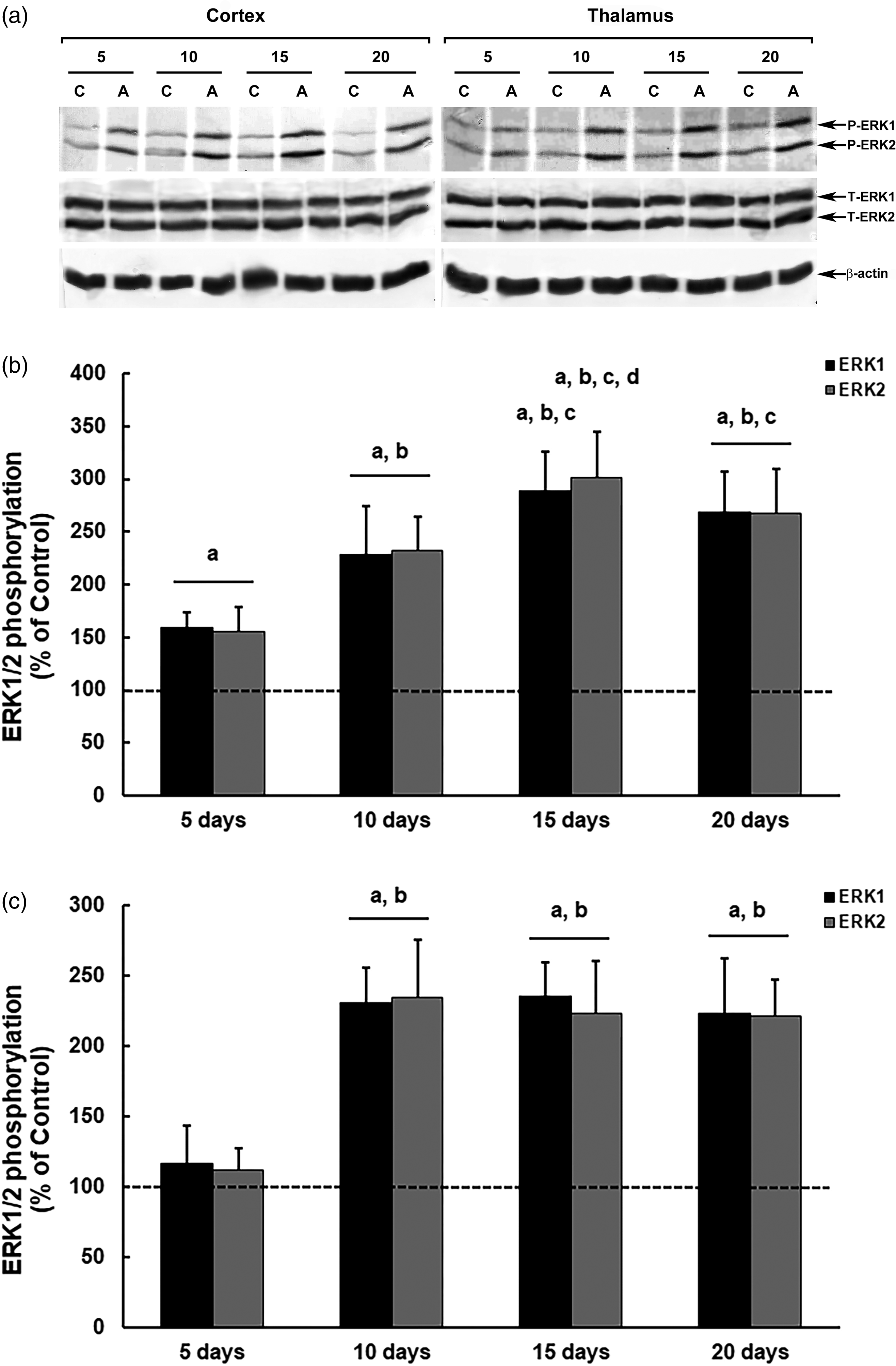
Modulation of ERK1/2 phosphorylation in the brain of male mice submitted to a thiamine deficiency model with amprolium (Amp) at different treatment periods. The panel shows representative immunoblotting (a) and quantification of ERK1/2 phosphorylation in the cerebral cortex (b) and thalamus (c) from deficient mice treated for five, 10, 15 or 20 days with AIN-93TD chow and amprolium 60 mg/kg, or AIN-93M chow and saline (NaCl 0,9%; control groups). Total and phosphorylated forms of ERK1/2 and β-actin were detected by specific antibodies, and the reaction was developed by 3,3′-diaminobenzidine tetrahydrochloride chromogen. The phosphorylation was determined as a ratio of the optical density (OD) of the phosphorylated band over the OD of the total band, and the data are expressed as a percentage of the control (considered as 100%). The values are presented as mean ± standard deviation derived from six independent replicates. The results were analyzed using analysis of variance followed by Duncan’s test. ‘A’ denotes p ≤ 0.05 compared with the control, ‘B’ p ≤ 0.05 compared with the group treated for five days, ‘C’ p ≤ 0.05 compared with the group treated for 10 days, and ‘D’ p ≤ 0.05 compared with the group treated for 20 days.
In the thalamus, we observed changes in the modulation of ERK1/2 only after the 10th day of treatment (Figure 4(c)). However, in animals treated for thiamine deficiency with amprolium during the 10, 15 or 20 days, the increases in ERK1/2 phosphorylation were similar to each other (∼130%), evidencing the formation of a plateau in the ERK activity after 10 days of thiamine deficiency (10 days, ERK1, p = 0.00003; ERK2, p = 0.00002; 15 days, ERK1 and ERK2, p = 0.00003; 20 days, ERK1 and ERK2, p = 0.00006; in relation to the controls).
In females, the ERK1/2 modulation profile in thiamine-deficient mice with amprolium was similar in both structures and at different treatment times (Figure 5). We observed an increase in ERK phosphorylation at all analyzed periods, with more intense increases in phosphorylation from the 10th day onwards, in the cerebral cortex (10 days, ERK1, p = 0.00002; ERK2, p = 0.00003; 15 days, ERK1 and ERK2, p = 0.00003; 20 days, ERK1 and ERK2, p = 0.00006; in relation to the controls) and thalamus (10 days, ERK1, p = 0.00002; ERK2, p = 0.00003; 15 days, ERK1 and ERK2, p = 0.00005; 20 days, ERK1, p = 0.00005; ERK2, p = 0.00003; in relation to the controls), with levels ∼115% higher compared with those in the controls (Figure 5(b) and (c)).
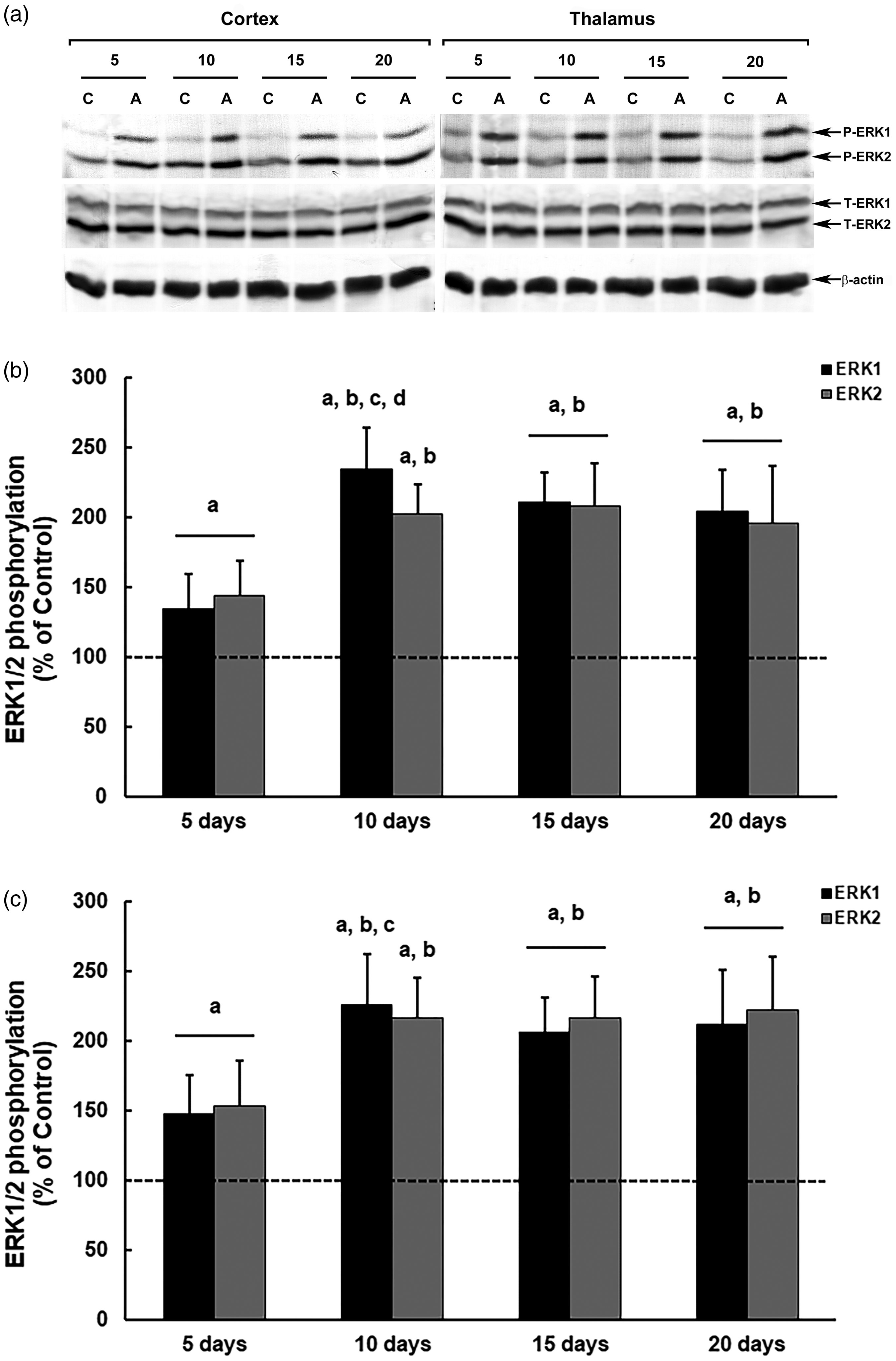
Modulation of ERK1/2 phosphorylation in the brain of female mice submitted to a thiamine deficiency model with amprolium at different treatment periods. The panel shows representative immunoblotting (a) and quantification of ERK1/2 phosphorylation in the cerebral cortex (b) and thalamus (c) from deficient mice treated for five, 10, 15 or 20 days with AIN-93TD chow and amprolium 60 mg/kg, or AIN-93M chow and saline (NaCl 0,9%; control groups). Total and phosphorylated forms of ERK1/2 and β-actin were detected by specific antibodies and the reaction was developed by 3,3′-diaminobenzidine tetrahydrochloride chromogen. The phosphorylation was determined as a ratio of the optical density (OD) of the phosphorylated band over the OD of the total band. The data are expressed as a percentage of the control (considered 100%). The values are presented as mean ± standard deviation derived from six independent replicates. The results were analyzed using analysis of variance followed by Duncan’s test. ‘A’ denotes p ≤ 0.05 compared with the control, ‘B’ p ≤ 0.05 compared with the group treated for 5 d, ‘C’ p ≤ 0.05 compared with the group treated for 15 days, and ‘D’ p ≤ 0.05 compared with the group treated for 20 days.
Discussion
Several researchers have investigated thiamine deficiency and its effect through animal experimentation, where damage can be observed, mainly in the brain. 3 The experimental models used included various thiamine-deficient diets, with or without thiamine antagonists, such as pyrithiamine. 2 The association of an inadequate diet with antagonists becomes interesting, as it accelerates the process and appearance of neurological signs in the animals, which can be observed from the 10th day of thiamine deficiency induction when pyrithiamine injections are used. 2 However, in this study, we chose to use i.p. administration of amprolium, an easier to access thiamine analog. Furthermore, little is known about the use of amprolium in rodent models of thiamine deficiency. Unlike pyrithiamine, amprolium acts mainly extracellularly, competitively inhibiting the transport of thiamine into the cell and, thus, generating cellular deficits even when nutrients are still present in the body. 26
Studies on thiamine deficiency in laboratory rodents have shown that the use of amprolium together with a thiamine-deficient diet is feasible, showing manifestations of behavioral and metabolic changes characteristic of thiamine deficiency11,12 in a reduced exposure time. 12 However, despite unprecedented evidence of amprolium’s effectiveness in inducing thiamine deficiency in rodents, an adequate dose and the minimum time necessary for the animals to exhibit notable neurological alterations have not yet been defined. In this study, we used the modulation of ERK1/2 phosphorylation in brain structures critically affected by thiamine deficiency in rodents, according to previous studies.2,3,5 We observed that within the parameters evaluated, the dose and time sufficient to produce neural alterations was 60 mg/kg and 10 days of treatment, evidenced mainly in the thalamus, the brain region considered most sensitive to thiamine deficiency. 2 Interestingly, the modulation of the ERK1/2 signaling pathways associated with thiamine deficiency with amprolium chronologically approached the neurological changes that occur in models using the classic method with pyrithiamine (10 days of treatment),2,3,5,6 but without showing severe manifestations (loss of consciousness, convulsions, and coma) and fatalities, as in the pyrithiamine model. Furthermore, and very importantly, our study showed that female mice could also be used to investigate neural alterations in thiamine deficiency with amprolium.
Mice are commonly used for in vivo experimental studies. Experiments investigating the neurological defects caused by thiamine deficiency are no exception to this, where predominantly male animals are used. Thus, it is necessary to analyze the effects of thiamine deficiency induction on the nervous tissue of both sexes and investigate whether differences could occur, whether this may make the use of females unfeasible or if the use of males is just a paradigm. The aspects considered most critical for the use of females in studies are related to behavior14,15 and oxidative stress,17,18 probably related to hormonal variations during the reproductive cycle 16 and the higher antioxidant resistance when compared with males,19 –21 respectively. These factors did not prevent the detection of neural alterations in females in our amprolium thiamine deficiency model. However, it is notable that females showed more evident manifestations of alteration in ERK modulation when they were more markedly deficient (higher doses), possibly reflecting greater antioxidant resistance, mainly because oxidative stress is considered the primary mechanism associated with neurodegenerative processes in thiamine deficiency 2 and is directly related to the activation of MAPKs. 4 Additionally, it is still unclear whether the difference depends on sexual diversity or difference in endocrine milieu. Therefore, it is necessary to determine the reproductive stage in the sexual cycle in females, as well as comparison between orchiectomized and intact males, to identify the substance of the sex difference.
In this study, it was possible to improve the model of induction of dietary thiamine deficiency associated with amprolium administration, developed by our group, by determining the most efficient dose (60 mg/kg) and exposure time (10 days) to observe important neural alterations in mice, such as the modulation profile of ERK1/2 MAPK signaling pathways. Notably, both female and male mice presented cellular alterations, indicating that both sexes can be used in experimental models of thiamine deficiency induction with amprolium.
Footnotes
Acknowledgments
Author contributions
All listed authors meet the authorship requirements. MPS, CASC, and FMC conceived and designed the experiments. MPS, FWBL, AGSM, JPN, and FMC performed the experiments. MPS, CASC, and FMC performed the experiments and wrote the manuscript. All the authors have read and approved the manuscript.
Data availability statement
Data may be made available upon request.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
