Abstract
The pathogenesis of Alzheimer’s disease (AD), a type of neurodegenerative disease characterized by learning and memory impairment, is often associated with pathological features, such as amyloid-beta (Aβ) accumulation and insulin resistance. The transgenic mouse, APPswePS1dE9 (APP/PS1), is one of the most commonly used animal models in pathogenesis studies of AD. The purpose of this study is to investigate the sex differences between APP/PS1 mice in the pathogenesis of AD. The impairment of glucose and insulin tolerance was found to develop earlier in male APP/PS1 mice than in females. Plasma insulin levels were significantly decreased in male APP/PS1 mice, while total cholesterol levels in male APP/PS1 mice were higher than those in females. Triglyceride levels in male mice in both the wild-type (WT) and APP/PS1 groups were higher than in their female littermates. Soluble and insoluble Aβ levels in female APP/PS1 mouse brains were higher than those in males. And the learning and memorizing abilities of female APP/PS1 mice were poorer than those of males. Our results concluded that there were sex differences in Aβ formation, pancreatic islet function and insulin sensitivity between male and female APP/PS1 mice during the pathogenesis of AD.
Alzheimer’s disease (AD) is a type of neurodegenerative disease, with clinical manifestations of memory and cognitive dysfunction, and abnormalities of personality and behavior. The three typical pathological characteristics of AD are: senile plaques, neurofibrillary tangles and extensive loss of neurons. Statistical data show that 5% of people above the age of 65 suffer from AD. The prevalence of AD increases with age, and by the age of 85, one in every three to four people is diagnosed with AD.1,2 Therefore, understanding the mechanism of AD pathogenesis is becoming more and more important and necessary.
The interaction of environmental and genetic factors results in a complicated etiology and pathogenesis with AD. Studies of the mechanism of AD pathogenesis remain controversial. Transgenic AD mouse models are widely used in current research into the disease. The APPswePS1dE9 (APP/PS1) mouse, of which the mouse/human amyloid precursor protein 695swe gene (amyloid precursor protein [APP], Mo/HuAPP695swe) and a variant of the human presenilin 1 (PS1-dE9 [PS1]) gene are knocked-in the genome, is one of the most commonly used transgenic mouse models of AD.
Both genes are involved in early onset AD. Amyloid plaques could be found in the brains of APP/PS1 mice at the age of 6–7 months; 2 meanwhile, impaired glucose tolerance and insulin resistance are accompanied by an increase of amyloid plaques. 3 Recent AD-related studies in APP/PS1 mice have used either solely male or female mice, or half of each gender.4–8 Although sample collecting time points and indicators of biochemical tests have differed, differences between male and female mice have not been compared. Male and female APP/PS1 mice have been used at the same time in some studies to compare the different amyloid deposition times, 9 while the differences of other biochemical indicators and behaviors that might have resulted from the deposition have not been compared. Epidemiological data indicated there were gender differences in mice of the same age during the pathogenesis of AD.10–12 Therefore the following should be addressed: are there any gender differences in the onset time or rate for AD in APP/PS1 mice? and what is the significance of the gender differences in the major indicators of AD, such as glucose tolerance, insulin resistance, amyloid-beta (Aβ) generation and the function of learning and memory during the pathogenesis of AD in the mouse model?
Our study investigated changes of body weight, the status of glucose and insulin tolerance, plasma levels of lipids and insulin, the Aβ levels in the brain, and spatial learning abilities and memory function of male and female APP/PS1 mice under a natural aging process with a regular diet. We found there were significant differences in the above indicators between the two genders of APP/PS1 mice in the pathogenesis of AD.
Animals
All experiments were performed in accordance with the guidelines on the care and use of animals for research purposes by the Institutional Animal Care and Use Committee of Sichuan University, and all the procedures were approved by the Institutional Animal Care and Use Committee of Sichuan University. The animals were maintained in an air-conditioned facility (temperature controlled at 25 ± 1℃) with a 12 h light/dark cycle with free access to water and standard rodent chow. C57BL/6J mice came from Jianyang Dashuo Animal Technology Co, Ltd (Sichuan, China). APP/PS1 mice on a C57BL/6J genetic background came from the Jackson Laboratory (Bar Harbor, ME, USA). APP/PS1 male mice were crossed with C57BL/6J female mice to produce APP/PS1+/− and WT littermate mice. The newborn mice were genotyped by polymerase chain reaction (PCR) analysis as described by the Jackson Laboratory. Six to 10 mice (of the same genotype and sex) with similar body weights were randomly allocated in one experimental group. A total of 32 mice were used in the behavior experiment, and the metabolic indicators were monitored throughout the research. The animals were killed by an overdose injection of 5% chloral hydrate solution.
Materials and methods
Body weight measurements
The body weight of each animal was measured and recorded using an electronic balance every week or every two weeks from the age of 16 weeks.
Metabolic measurements
Glucose tolerance tests (GTTs) and insulin tolerance tests (ITTs) were carried out as previously described. 13 Briefly, GTTs were performed on mice fasted for 16 h. The mice were injected intraperitoneally with glucose in 0.9% saline (2 g/kg). ITTs were performed on mice fasted for 4 h. The mice were injected intraperitoneally with human insulin in 0.9% saline (0.2 U/kg). Blood glucose levels (mmol/L) were measured from tail vein blood using the Sannuo blood glucose monitor (Sinocare Inc, Changsha, China) at indicated time points. Total plasma cholesterol and triglyceride levels were measured using a cholesterol kit and triglyceride kit, respectively (Xincheng Biotec, Sichuan, China) according to the manufacturers’ instructions. Plasma insulin concentrations were measured by enzyme-linked immunosorbent assays (ELISA; Shanghai Elisa Biotech Co, Ltd, Shanghai, China).
Morris water maze
The Morris water maze test was performed as previously described, 14 with adaptation (see Supplementary Information for the detailed experimental procedure online at http://lan.sagepub.com). Briefly, the pretrained mice were released from four pseudo-randomly assigned starting points in each phase. The same process was carried out as on the first day for each point with a resting period of 30 s intervals. The time the mice took to mount the platform was recorded. A probe session was performed on day 7. In the probe session, the hidden platform was removed and the mice were released from phase 3 (the most distal point) and allowed to swim freely for 60 s. The number of crossings of the area where the platform had been located during training was recorded.
Tissue sampling
Animals were killed by overdosing with chloral hydrate (10 g/kg). Blood was sampled from the fundus artery and blood serum was collected by centrifuging the clotted blood sample. The left brain hemisphere was snap-frozen in liquid nitrogen and stored at −70℃ for future biochemical analysis. The right brain hemisphere was fixed in 10% (w/v) neutral-buffered formalin (NBF) and stored as a cryosection at −20℃ in phosphate-buffered saline (PBS) with 50% (w/v) glycerol until use. The pancreas was fixed in 10% (w/v) NBF and embedded in paraffin wax until future use.
Quantification of brain Aβ peptide levels
The brain Aβ levels were analyzed by ELISA as described previously.
15
The frozen brain tissues were homogenized and sonicated in Tris-buffered saline (TBS) containing protease inhibitors (Sigma-Aldrich, St Louis, MO, USA). Twenty microliters of the homogenates were kept for total protein level analysis using bicinchoninic acid (BCA) kits (Beyotime Institute of Biotechnology, Jiangsu, China) according to the manufacturer’s instructions. Homogenates were centrifuged at 100,000×
Congo-red staining of the brain Aβ plaques
The compact Aβ plaque staining of the mouse brain was carried out as described previously, 15 with adaptation. The brain sections from cryosectioning were treated with a sodium chloride working solution (0.3% [w/v] NaCl in 80% [v/v] ethanol and 0.01% [w/v)] NaOH) at room temperature (RT) for 20 min, and then placed into a Congo-red working solution (containing saturated Congo-red in a NaCl working solution) for one hour, and dehydrated rapidly in absolute alcohol and then in xylene. The stained sections were mounted in neural gum and covered with a cover-slip for future observation. Images were collected at 40× magnification under a constant bulb temperature and exposure, with all images being acquired in the same session. The area of Congo-red-positive Aβ plaque was counted in each section for statistical analysis.
Immunofluorescence staining of pancreas
The paraffin imbedded pancreas sections were stained with a mouse monoclonal anti-insulin antibody (ab6995; Abcam, Shanghai, China). Sections were incubated overnight with primary antibodies at 4℃. DyLight 488-conjugated AffiniPure Goat Anti-Mouse secondary antibodies (Jackson ImmunoResearch, West Grove, PA, USA) were used. The sections were then stained with DAPI (5 µg/mL). Images were captured using an inverted fluorescence microscope (Nikon, Tokyo, Japan).
Statistics
GraphPad Prism version 5.00 for Windows (GraphPad Software, San Diego, CA, USA) was used for standard statistical analysis of the data. Data were presented as mean ± SEM. The results between groups were analyzed by unpaired t-test with Welch’s correction. P < 0.05 was considered to be statistically significant.
Results
Differences in the changes of body weight between growing male and female mice
To investigate the influences of the knocked-in APP gene (Mo/HuAPP695swe) and the presenilin 1 gene (PS1-dE9), on male and female C57BL/6J mice, the changes in body weight during the growing process of the mice were monitored. Although the body weights of female WT mice were lower than those of the male WT mice, the weight gain trends of the two genders of the WT mice were similar (Figure 1). Whereas, in the APP/PS1 mice, the weight gain trends of both genders were similar before the age of 30 weeks. From week 30, male APP/PS1 mice showed greater increases in their body weight than before, while in the female APP/PS1 mice, the weight gain rates were significantly higher than in their male littermates (Figure 1). Therefore, the male APP/PS1 and female mice showed differences in the times and extents of body weight gain.
Increases of body weight in wild-type (WT) and APPswePS1dE9 (APP/PS1) mice. The body weights of the animals were recorded from week 19. The increases of the body weight in females (F) were lower than males (M) in both WT and APP/PS1 mice. The rates of body weight gain in APP/PS1 mice were changed from week 30. The rates of the body weight gain in female APP/PS1 mice were higher than in the males. Data are expressed as mean ± SEM (n = 6–9).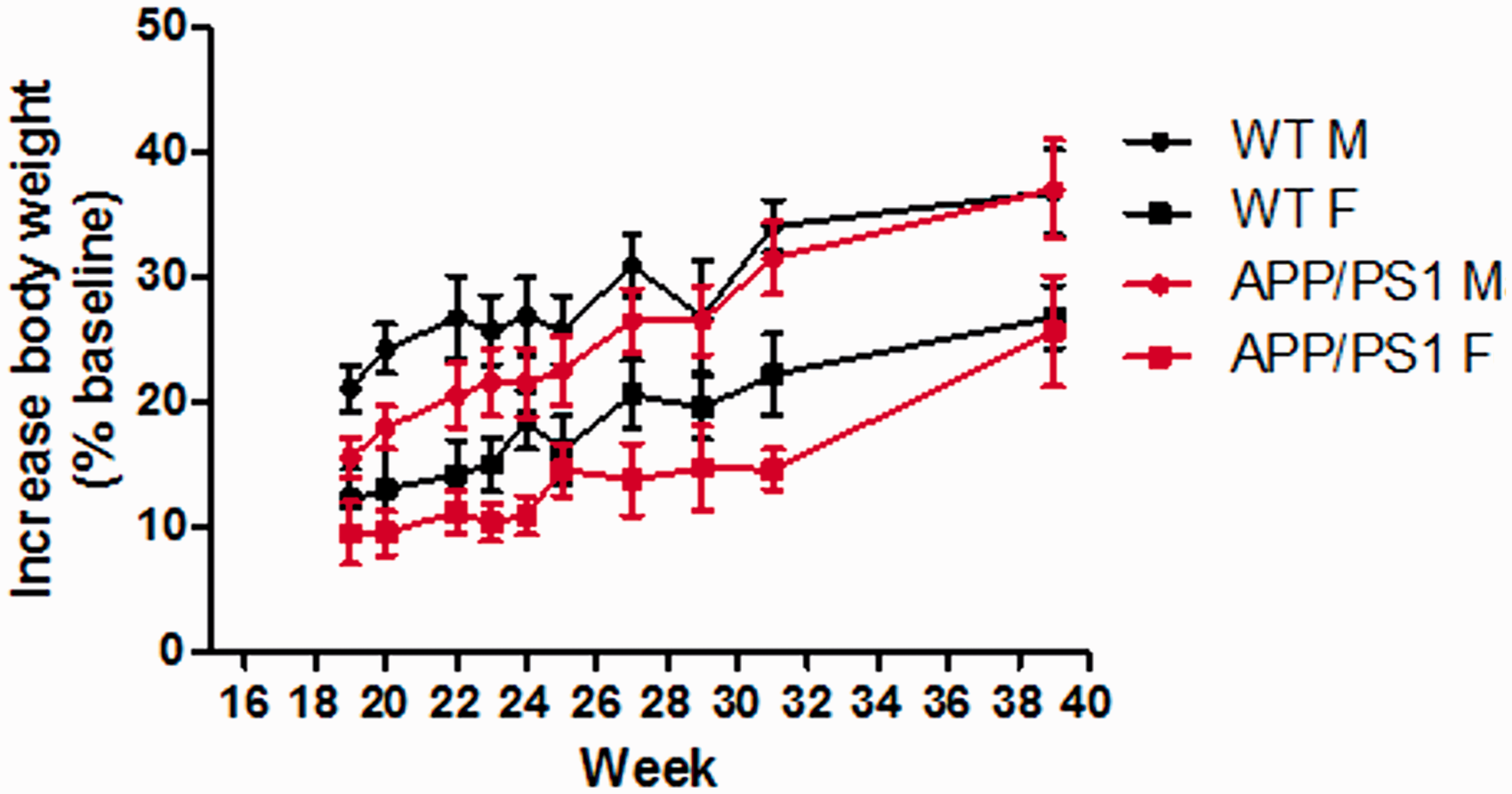
Impairment of glucose tolerance and insulin tolerance occurred earlier in male APP/PS1 mice than in female mice
Since impaired glucose and insulin tolerance were observed in the APP/PS1 mice, glucose and insulin tolerance were tested in the mice at the ages of six and nine months to see whether gender differences existed. At 6 months old, both the male and female WT mice exhibited normal glucose tolerance and there were no significant differences between the areas under the curve (AUCs) of the GTT curves of the two groups. Although there were no differences in the AUCs between the two genders in the APP/PS1 mice, the 6-month-old male APP/PS1 mice showed abnormal glucose tolerance. The delayed peak of GTT suggested a retardation of the glucose-stimulated insulin response (Figures 2a and b). The GTT of 9-month-old WT mice remained normal and there were no differences in the AUCs either. Impaired glucose tolerance was exacerbated in the male APP/PS1 mice compared with the normal glucose tolerance of the female mice (Figures 2c and d). Thus, male APP/PS1 mice displayed impaired glucose tolerance, but not the female mice, indicating a weakened islet β-cell function in the male mice. There was a gender difference in glucose tolerance in the APP/PS1 mice.
Abilities of glucose and insulin tolerance in wild-type (WT) and APPswePS1dE9 (APP/PS1) mice. (a) Glucose tolerance test (GTT; 2 g/kg) in WT and APP/PS1 mice at the age of six months (n = 6–9). (b) Area under the curve (AUC) of GTT in WT and APP/PS1 mice at the age of six months. (c) GTT (2 k/kg) in WT and in WT and APP/PS1 mice at the age of nine months. (d) AUC of GTT in WT and APP/PS1 mice at the age of nine months. (e) Insulin tolerance test (ITT; 0.2 U/kg) in WT and APP/PS1 mice at the age of six months (n = 6–9). (f) AUC of ITT in WT and APP/PS1 mice at the age of six months. (g) ITT (0.2 U/kg) in WT and in WT and APP/PS1 mice at the age of nine months. (h) AUC of ITT in WT and APP/PS1 mice at the age of nine months. Data are expressed as mean ± SEM. *P < 0.05, **P < 0.01 (increased blood glucose level: APP/PS1 male versus APP/PS1 female mice).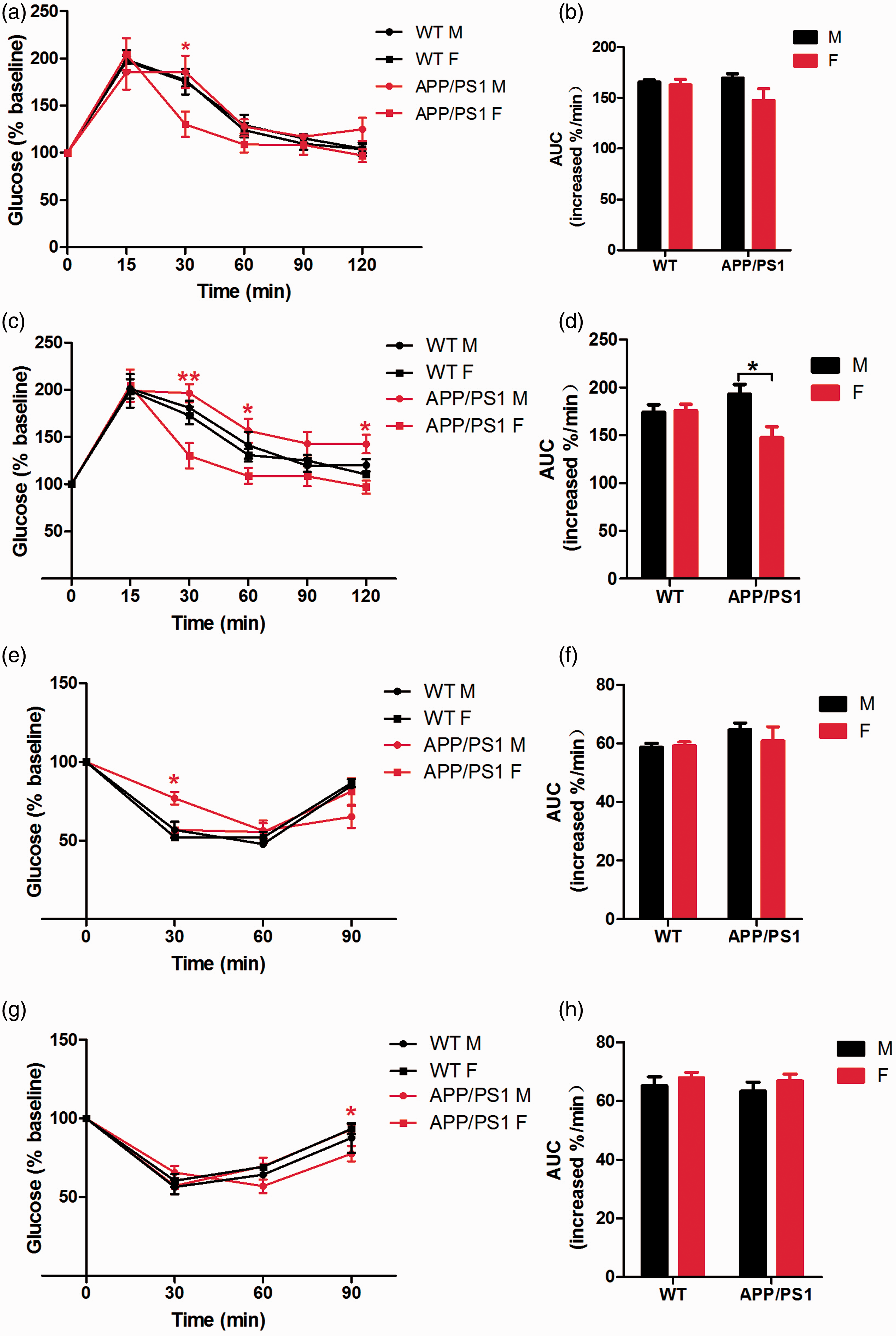
The insulin tolerance status of the animals was also examined. Male APP/PS1 mice showed an abnormal shape of the ITT curve at the ages of 6 and 9 months, while there were no differences in the AUCs of the two genders. The results showed that the insulin-stimulated decrease of the blood glucose level was delayed in the male APP/PS1 mice, indicating an occurrence of abnormal insulin signaling and signs of insulin resistance (Figures 2e–h).
Gender differences of insulin and lipid metabolism levels in APP/PS1 mice
The impaired glucose and insulin tolerance indicated attenuated insulin secretion or insulin signaling in the APP/PS1 mice. Consequently, lipid metabolism was also affected. Therefore, we examined insulin expression in the pancreas via immunofluorescence and measured the plasma insulin, total cholesterol, and triglyceride levels in all the groups. The male APP/PS1 mice showed a significant decrease in plasma insulin levels (Figure 3a). The immunofluorescence results were consistent with the plasma insulin levels, such that the pancreatic insulin signals in the male APP/PS1 mice were lower than in the rest of the groups (Figure 3b). However, their total cholesterol and triglyceride levels were significantly higher than those of the females (Figures 3c and d). The above results indicated a possible delayed and decreased insulin secretion in the male APP/PS1 mice, which led to the lipid metabolism disorder and elevated blood lipid levels. In the meantime, the WT male mice exhibited increased triglyceride levels compared with their female littermates, while no differences in the total cholesterol level were observed.
Insulin secretion and lipid levels in wild-type (WT) and APPswePS1dE9 (APP/PS1) mice at the age of nine months. (a) Quantification of plasma insulin (INS) levels by enzyme-linked immunosorbent assay (ELISA) (n = 6–9). (b) Immunofluorescence of insulin secreted in islets of pancreas. (c) Quantification of total plasma cholesterol levels (n = 6–9). (d) Quantification of plasma triglyceride levels (n = 6–9). Data are expressed as mean ± SEM. *P < 0.05, **P < 0.01.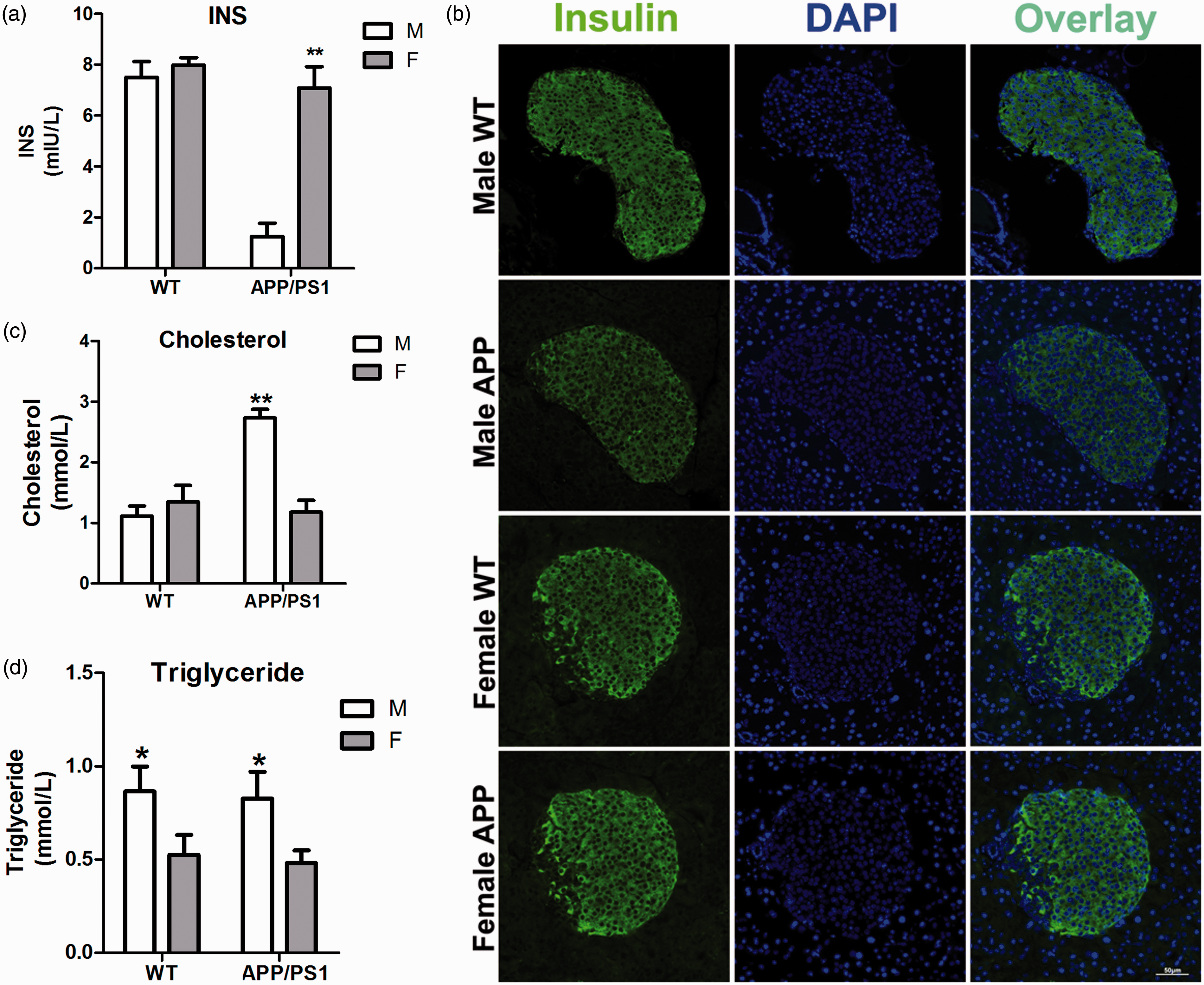
Gender differences of the brain Aβ and amyloid plaque levels in APP/PS1 mice
In addition to insulin resistance, APP/PS1 mice also exhibit distinct pathological features in the brain, such as extracellular deposition of amyloid plaques, cellular neurofibrillary tangles, loss of cholinergic neurons, and progressive attenuation of cognitive function. Aβ plays a key role in neurotoxicity and in the development of AD.16,17 Among various forms of Aβ, oligomers are the most toxic, leading to neuronal death, neurodegeneration, and synaptic transmission dysfunction.17–20 To explore the influence of Aβ in the cognitive function of 9-month-old APP/PS1 mice, we examined the brain Aβ levels of the mice in the forms of TBS soluble Aβ (TBS–Aβ, representing the soluble Aβ), SDS soluble Aβ (SDS–Aβ, representing Aβ oligomers), and FA soluble Aβ (FA–Aβ, representing Aβ plaques), and the amounts of Aβ40 and Aβ42 in the three solvents. The Aβ levels in the WT mice were significantly lower than in the APP/PS1 mice; while in both genotypes, the Aβ levels in female mice were higher than in the males (Figure 4a). Moreover, the amounts of Aβ40 and Aβ42 plaques in the female mice were significantly higher than in the males (Figure 4b). The ELISA results were consistent with the results of Congo-red staining (indicating the Aβ plaques), in which the female APP/PS1 mouse brain samples exhibited a lot more positive plaques than in their male littermates (Figure. 4c). There were no visible positive plaques in the WT mouse brain samples (data not shown).
Brain amyloid-beta (Aβ) levels in nine-month-old wild-type (WT) and APPswePS1dE9 (APP/PS1) mice. (a) Enzyme-linked immunosorbent assay (ELISA) of Aβ in Tris-buffered saline (TBS), sodium dodecyl sulfate (SDS), and formic acid (FA) fractions of brain homogenates. Total Aβ level was added from the TBS, SDS, and FA fraction ELISA results (n = 6–9). (b) ELISA of Aβ40 and Aβ42 in TBS, SDS, and FA fractions of brain homogenates (n = 6–9). (c) Congo-red staining and the density of the plaques for the APP/PS1 mouse brain frozen sections (n = 3). Data are expressed as mean ± SEM. *P < 0.05, **P < 0.01.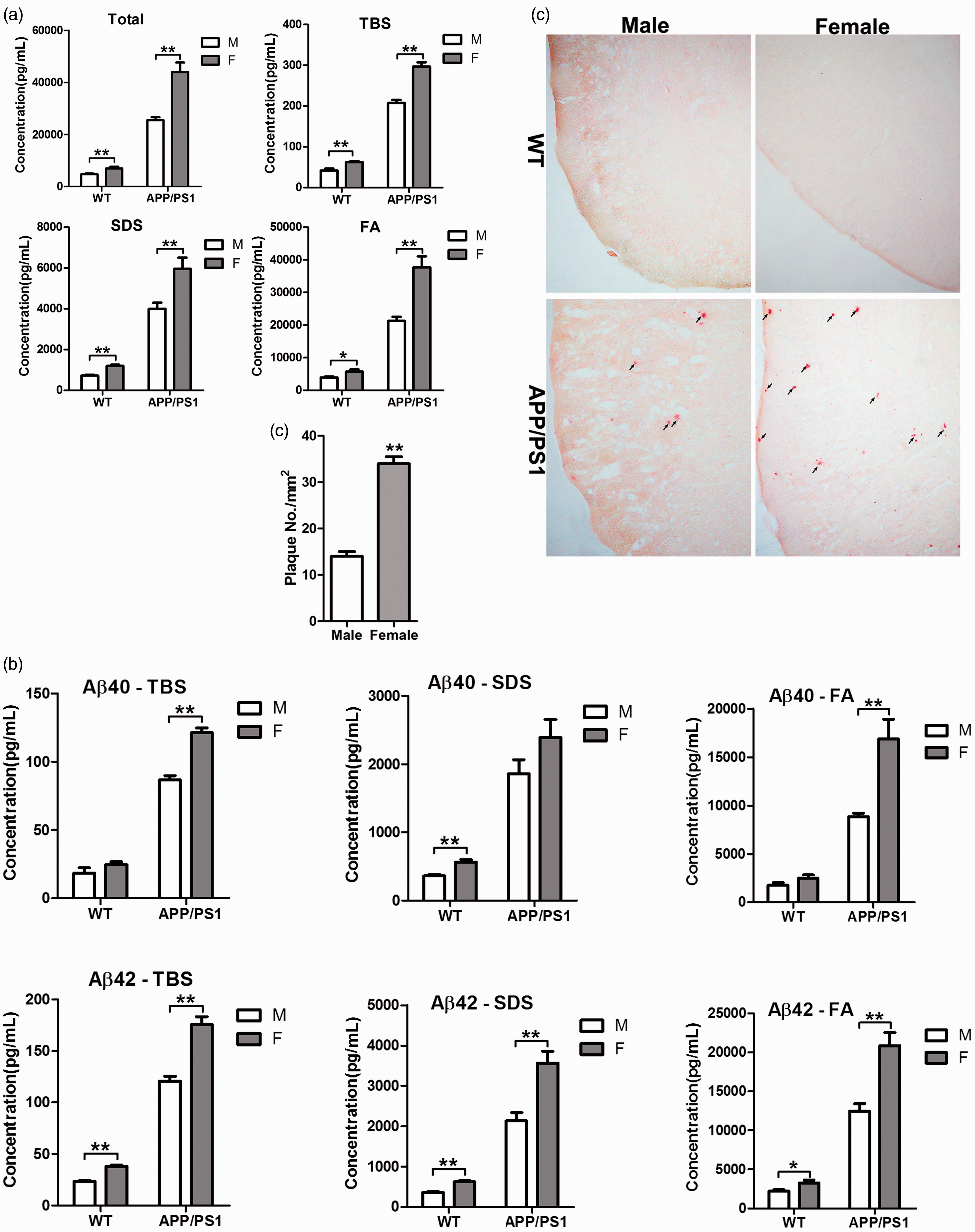
Cognitive function of female APP/PS1 mice was attenuated earlier than in the males
Since APP and the presenilin 1 enzyme genes are knocked-in in the APP/PS1 mouse, it exhibits an accelerated generation of amyloid proteins in the brain, affecting its cognitive ability.21,22
The Morris water maze test was used to investigate gender differences of APP/PS1 mice with regard to cognitive ability. Escape latency and the numbers of times of finding platforms and in crossing the platforms were monitored. The results showed that 6-month-old APP/PS1 mice had a longer escape latency compared with their WT littermates, and there were no differences between the male and female animals in both genotypes (Figure 5a). However, the male APP/PS1 mice crossed the platform more times than the females did. The animals were retested in their learning and memorizing abilities at 9 months old. The male APP/PS1 mice showed shorter escape latency than the females did (Figure 5b). The results indicated that, with an increase of age, the re-learning and memorizing abilities of the female APP/PS1 mice were attenuated earlier than were the males.
Cognitive abilities of wild-type (WT) and APPswePS1dE9 (APP/PS1) mice. (a) Water maze tests for mice at the age of six months (n = 6–9). The mean annulus crossings between APP/PS1 male and female mice were significantly different (*P < 0.05). (b) Water maze tests for mice at the age of nine months (n = 6–9). The escape latencies in sections 1 and 3 between APP/PS1 male and female mice were significantly different (**P < 0.01), while the mean annulus crossings between WT male and female mice were significantly different (*P < 0.05). Data are expressed as mean ± SEM.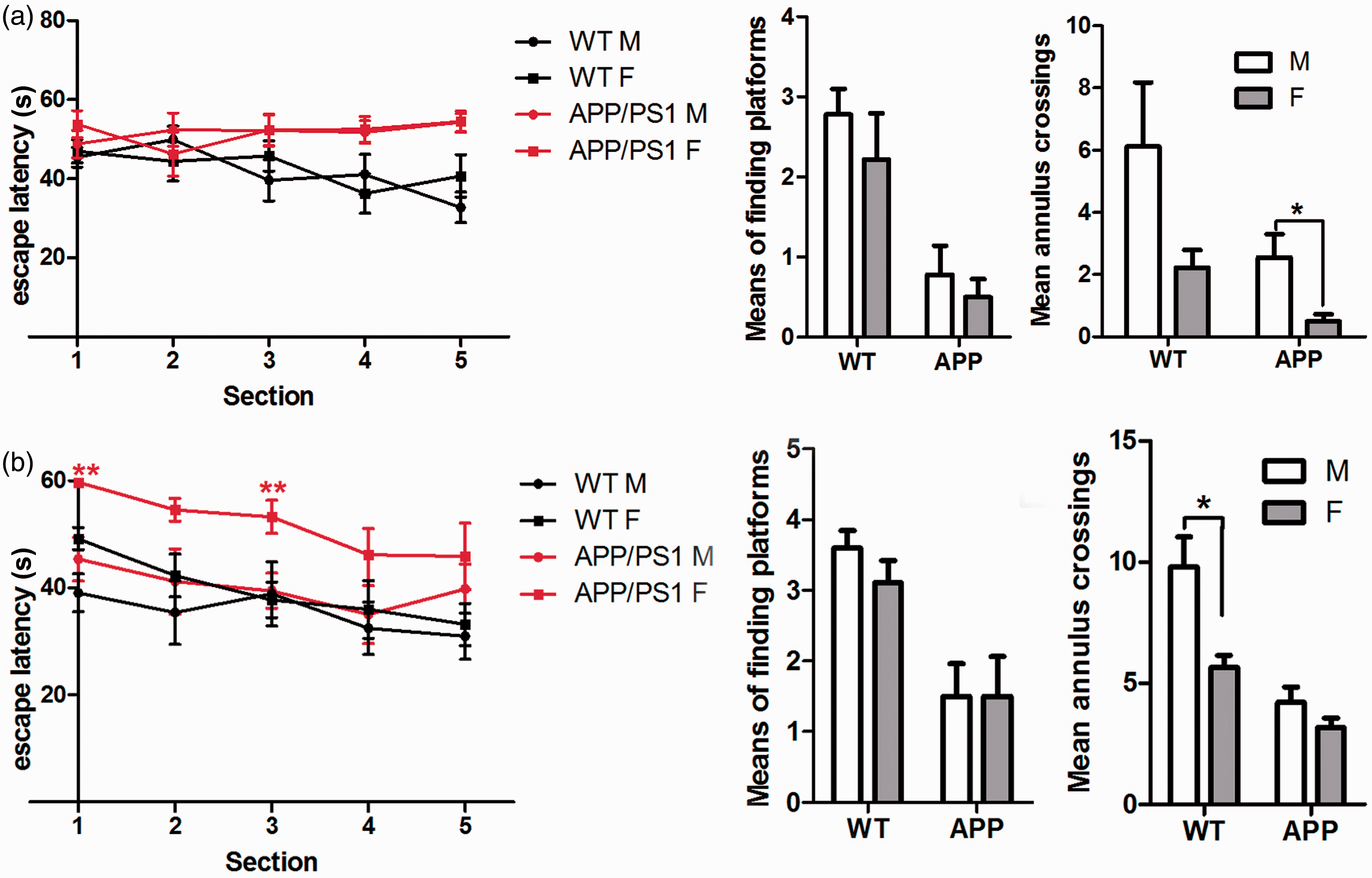
Discussion
The APP and PS1 genes are knocked-in the genomes of APP/PS1 mice simultaneously. As the mouse grows, Aβ accumulates and deposits into amyloid plaques (senile plaques) along with an increased production of the protein in the brain. Studies have shown that starting from four months of age, the production of brain Aβ increase in APP/PS1 mice, compared with their WT littermates; few deposits are found at six months, and the amyloid plaques are formed significantly at nine months. Meanwhile, APP/PS1 mice exhibit decreased or even complete loss of learning and memory abilities.3,9,23,24
In addition to increased Aβ production with age, APP/PS1 mice also show insulin resistance, with impaired glucose and insulin tolerance; therefore, AD is also known as type-3 diabetes.22,25 Our results showed that there were gender differences in the GTT and ITT curves in the APP/PS1 mice. The GTT curves of six-month-old male APP/PS1 mice indicated delayed glucose-stimulated insulin secretion compared with the same-age female mice, suggesting an attenuated islet β-cell function. At nine months, the APP/PS1 male showed significant impaired glucose tolerance, and the AUCs of GTTs of the two genders of the APP/PS1 mice were significantly different, indicating progressive deterioration of the islet β-cells. Meanwhile, the six-month-old male APP/PS1 mice already showed abnormal insulin tolerance, demonstrating the pathological characteristic of delayed insulin signaling and insulin resistance. These findings suggested that gender differences existed in the changes of glucose and insulin tolerance of the APP/PS1 mice during their growth along with the generation of Aβ. The male APP/PS1 mice were more vulnerable to abnormal glucose and insulin tolerance, manifested as an insulin resistance characteristic, such as delayed glucose-stimulated insulin release, and slow response to insulin signaling. Furthermore, we also demonstrated a reduced insulin secretion in the nine-month-old male APP/PS1 mice. Past studies have shown that, along with the production of excessive Aβ, early onset of insulin resistance and elevated insulin levels can be observed in APP/PS1 mice. Similar symptoms also occur in the early stages of AD patients.3,26 However, inflammatory factors and long-term high levels of blood glucose would produce oxidative stress to induce the apoptosis of islet β-cells,27,28 whereas females are protected from β-cell apoptosis and hyperglycemia due to the protective effect of estrogen on β-cells.29–32 Our results were consistent with these reports, that male APP/PS1 mice were more vulnerable to attenuated glucose and insulin tolerance. The reduced insulin levels in the nine-month-old male APP/PS1 mice might be related to β-cell apoptosis in the late stage of insulin resistance. Thus, gender differences are significant in the onset of insulin resistance in APP/PS1 mice.
Impaired insulin signaling usually leads to an abnormal lipid metabolism. Our results showed elevated levels of cholesterol in the nine-month-old male APP/PS1 mice compared with the female APP/PS1 mice, while there were no differences between male and female WT mice; furthermore, the cholesterol levels in the male APP/PS1 mice were significantly higher than in the WT males. Our findings were consistent with other studies of total plasma cholesterol levels in AD mouse models, indicating that the process of AD pathogenesis may cause changes in plasma lipid levels. 9 On the other hand, both the male WT and APP/PS1 mice had elevated levels of plasma triglyceride compared with their female littermates, respectively; however, no differences were observed between WT and APP/PS1 mice of the same gender. This was consistent with some epidemiological studies in humans and rodents, in which triglyceride levels in males are higher than in females.9,33–35 Our results indicated that reduced insulin levels and attenuated insulin signaling in male APP/PS1 mice might lead to lipid metabolism disorders in these mice. There were significant differences in lipid metabolism between the two genders of the APP/PS1 mice.
In addition to the insulin resistance feature, APP/PS1 mice also exhibit abnormal Aβ production and deposition of amyloid plaques in the brain, leading to neuronal tau hyperphosphorylation, neurofibrillary tangles, synaptic dysfunction, neuron death, and learning and memory loss during development.24,36,37 The brain soluble oligomer, and deposited Aβ levels, especially levels of Aβ42, in the female mice were higher than in the males. This was consistent with the Congo-red staining of amyloid plaques, in which more positive plaques were observed in the female APP/PS1 mice than in the males. Extracellular deposits of Aβ plaques have been recognized as a cause of neuronal death and have subsequently led to impaired cognitive function;16,17 meanwhile, impaired brain insulin signaling has attracted more attention recently from AD researchers. 38 Furthermore, cholesterol also has a complicated relationship with the pathogenesis of AD. Dietary hypercholesterolemia aggravates the amyloid burden in animal models, 39 whereas pharmacological reduction of cholesterol reduces the amyloid level. 40 However, it remains controversial whether the accumulation of extracellular Aβ leads to impaired brain insulin signaling and elevated total plasma cholesterol levels or the reverse. Our water maze test results showed that six-month-old APP/PS1 mice already had prolonged escape latency compared with WT mice, while there were no differences between male and female mice in both groups. However, the fact that the male APP/PS1 mice crossed platforms more times than the females did indicate that the memorizing ability of the male APP/PS1 mice was slightly better than that of the females. When the mice were retrained in the water maze test at nine months, the escape latency of the male APP/PS1 mice was significantly less than that of the females, suggesting that the learning and memorizing abilities of the male APP/PS1 mice were better than those of the females. Taking the normal glucose and insulin tolerance and plasma lipid levels of the female APP/PS1 mice into account, the impaired learning and memorizing abilities of the female APP/PS1 mice might be related to amyloid plaques in the brain. Therefore, the influence on cognitive function of extracellular Aβ deposits might be more powerful than that of impaired insulin signaling and elevated plasma lipid levels.
Conclusions
Our experiments have demonstrated gender differences in APP/PS1 mice in GTTs, ITTs, plasma lipid levels, brain Aβ levels, the deposition of amyloid plaques, and cognitive abilities. Our findings provide reference data for researchers doing AD-related studies using APP/PS1 mice. One should take gender differences in animals into account in either mechanism research or drug design.
Footnotes
Acknowledgements
We thank Professor Xinfu Zhou (University of South Australia) for his help in data analyses of this paper; Professor Yanjiang Wang (Daping Hospital, Third Military Medical University) for his support in providing the APP/PS1 mice and data analyses of this paper; Professor Tinghua Wang (Sichuan University) and Professor Jianguo Qi (Sichuan University) for their technical and equipment support. We thank Mr Xiang Zheng (Sichuan University) for his great help in the histological techniques.
XL, WW and YF carried out the experiments, JZ, CMF, and GZY advised on experimental procedures and statistical techniques. GZY and YF wrote the paper. GZY and XZ conceived the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant (Grant No: 2012JY0014) of the Science & Technology, Department of Sichuan Province, the Applied Basic Research Project in Sichuan Province (Grant No: 2014JY0118), and Capacity Promotion Project of the National Natural Fund (Grant No: 0040105401092).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
