Abstract
While over the past several decades mortality after pancreatic surgery has decreased to <5%, postoperative morbidity remains remarkably high, ranging from 15% to 65%. The development of a postoperative pancreatic fistula (POPF) is a significant contributor to morbidity in patients undergoing pancreatic surgery. POPF can lead to life-threatening conditions such as intra-abdominal abscess, uncontrolled hemorrhage, sepsis, and death. Rates of POPF have not significantly changed over time, despite the introduction of multiple technical and pharmacologic interventions aimed at their treatment and prevention. Unfortunately, there are few POPF experimental models that have been described in the literature and existing models are unable to reliably reproduce the clinical sequelae of POPF, limiting the development of new methods to prevent and treat POPF. Herein, we describe a new rat experimental model that reliably creates a POPF via transection of the common pancreatic duct.
Introduction
While mortality after pancreatic surgery has decreased to <5% over the past several decades, postoperative morbidity has remained remarkably high, ranging from 15% to 65%.1–3 Postoperative pancreatic fistula (POPF) is the defining complication of pancreatic surgery and a significant contributor to morbidity in patients undergoing pancreatectomy. 4 A POPF refers to leakage of pancreatic fluid after surgery secondary disruption of the exocrine pancreatic ducts. Enzymes within the leaked pancreatic fluid digest the surrounding tissues, resulting in complications. The International Study Group for Pancreatic Surgery classifies POPF into three categories based on severity and clinical impact for the patient. 5 Consequences of POPF vary from continued need for percutaneous drainage to abscess formation, hemorrhage, sepsis, multiorgan failure, and death. 6 Furthermore, POPF has been independently associated with a significant reduction in recurrence-free survival and long-term survival for patients with pancreatic cancer.7,8
Multiple interventions have been trialed to decrease the rate of POPF. These include different surgical techniques, application of topical hemostatic/occlusive agents, stenting of the pancreatic duct, and use of pharmacotherapy to reduce pancreatic secretions.9–15 The results of these interventions have been mixed, with none gaining broad clinical adoption.
In order to develop treatment strategies for POPF there is a need for POPF experimental models. Both porcine and canine models have been previously described.16,17 While these large animal models are useful, they pose challenges in terms of cost, regulatory hurdles, and ethical considerations. More recently a rat model has been described by Tanaka et al. 18 The rat pancreas anatomy offers some technical challenges due to size and anatomic variations but is a promising model (Figure 1). Tanaka et al. demonstrated that transecting the splenic duct resulted in a feasible short-term POPF model; however, rats rapidly healed without treatment, limiting the clinical utility of this model. Additional studies have also demonstrated rapid healing in rats as well as mild elevations in peritoneal amylase/lipase, indicating induction of a clinically insignificant POPF.19,20 These models that demonstrate excellent recovery without any intervention limit the ability to test new treatment strategies to prevent POPF.
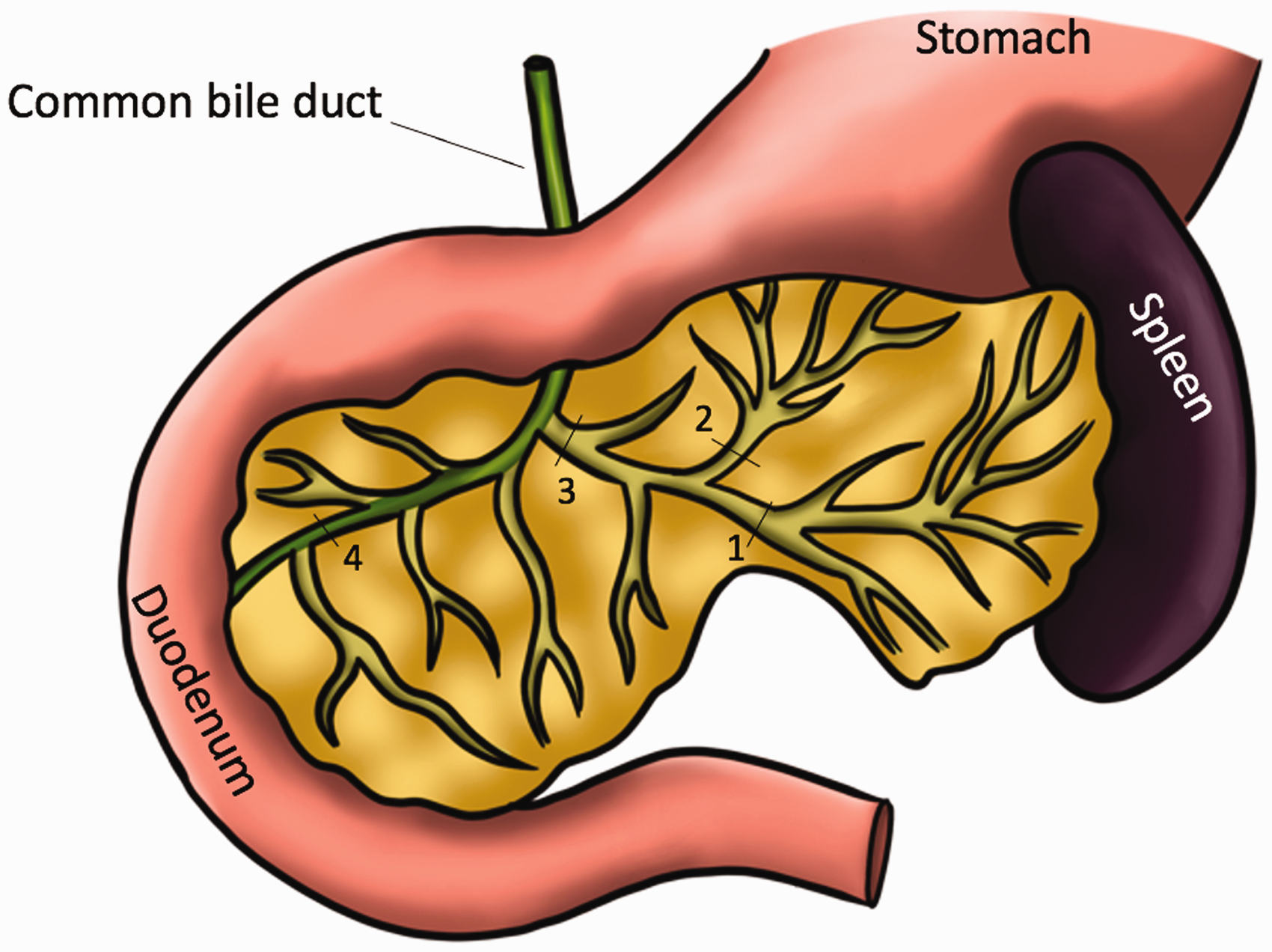
Illustration of the rat pancreas. Previous models have utilized transection of the splenic duct (1), gastric duct (2), or both the splenic and the gastric duct (3). In our model we propose transecting the common duct (4) in conjunction with ligation of the common bile duct.
In this study, we describe a new technique involving transection of the common pancreatic duct to produce a robust and reproducible POPF in a rat model that generates clinical, biochemical, and histological sequalae similar to that seen in humans.
Methods
This study was performed in accordance with the University of Washington Office of Animal Welfare guidelines for the humane use of animals, and all procedures were reviewed and approved by the Institutional Animal Care and Use Committee (Protocol # 4488-01).
Animals
Thirty-six female Crl:CD(SD); Hsd:SD; NTac:SD (Sprague–Dawley) rats with a mean weight of 236 g (IQR: 219–247 g) were purchased from Charles River Laboratories and housed in the University of Washington animal facility under standard conditions in Allentown Nexgen Rat 900 cages (https://www.allentowninc.com/rodent-housing/nexgen-rat-900/). Sprague–Dawley rats were chosen due to their reproducible hepatobiliary anatomy, adequate size, ease of care, and tolerance of surgical procedures. The vivarium light/dark cycles consisted of 06:00 h on, 20:00 h off. Temperature was maintained at 22°C. Humidity ranged from 37% to 50%. Bedding change occurred every 14 days and a full cage change every 28 days. As we were developing a novel model with unknown reproducibility, we were unable to perform power calculations a priori to determine appropriate sample size. We elected to use 36 rats in order to assess for reproducibility and perform basic statistical analyses. For establishment of this model, only female rats were used due to their docile nature. All rats were given unlimited access to water (Avidity recirculating water system) and rat chow (Labdiet PicoLab Rodent Diet 5053) and allowed seven days to acclimatize to the facility prior to any surgical procedure. Three animals required operation after only 48 h due to drug and vivarium resource availability.
Anesthesia and pain control
Buprenorphine SR 1 mg/kg (ZooPharm, Laramie, Wyoming, USA) and meloxicam 1 mg/kg (Patterson Veterinary Supply, Loveland, CO, USA) were injected subcutaneously 1 h prior to surgical procedure. Meloxicam (1 mg/kg) was also re-dosed every 24 h postoperatively for 48 h. Rats were placed in an induction chamber and 1–5% isoflurane administered until they were recumbent. Once induced the rats were removed from the chamber and positioned in a nose cone with 1–2% isoflurane administered to maintain anesthesia. Adequate depth of anesthesia is monitored by respiratory rate, corneal reflex, and response to toe pinch. Heat support and eye lubrication were provided.
Surgical procedure
General anesthetic was induced and maintained with 1–2% isoflurane delivered via an oxygen carrier at 0.8–1 l/min and anesthetic depth was monitored using respiratory rate (goal 35–60 breaths/min). During the procedure animals were warmed with a Kent Scientific far infrared warming pad and heat lamp postoperatively. The skin was prepared with three alternating scrubs of betadine and 70% ethanol and the entire procedure was carried out using sterile technique. A ventral transverse incision was performed. The duodenum and pancreas were identified and extracorporealized. Using gentle caudal traction on the duodenum the common bile duct was identified exiting the liver and was suture ligated with 5-0 prolene suture (Ethicon, Raritan, NJ, USA) (Figure 2(a) and (b)). Bile duct ligation in rats has been previously described and shown to be well tolerated. 21 Ventral traction was then applied to the duodenum allowing for easy visualization of the common pancreatic duct, which was sharply transected (Figure 2(c) and (d)). The duodenum and pancreas were returned to the abdomen. The abdominal incision was closed in two layers, interrupted 3-0 vicryl suture (Ethicon, Raritan, NJ, USA) for the fascia, and 9 mm wound clips for the skin. Our institutional experience is that clips provide more definitive skin closure relative to suture after laparotomy in these animals. Total procedure time was around 10–15 min per rat. Blood (0.25ml) was drawn from the tail artery in the standard fashion in order to have a baseline amylase/lipase level (n = 12). We did not perform sham surgeries as a control group as we did not think the benefit outweighed the suffering to the animals. Following surgery, the animals were examined twice daily and a modified scoring system for determining humane endpoints in short-term experiments focusing on physical appearance (0–3), unprovoked behavior (0–3), and response to external stimuli (0–3) was used to assess the rats’ welfare. Euthanasia was planned for any rat scoring >5 (see Appendix 1).
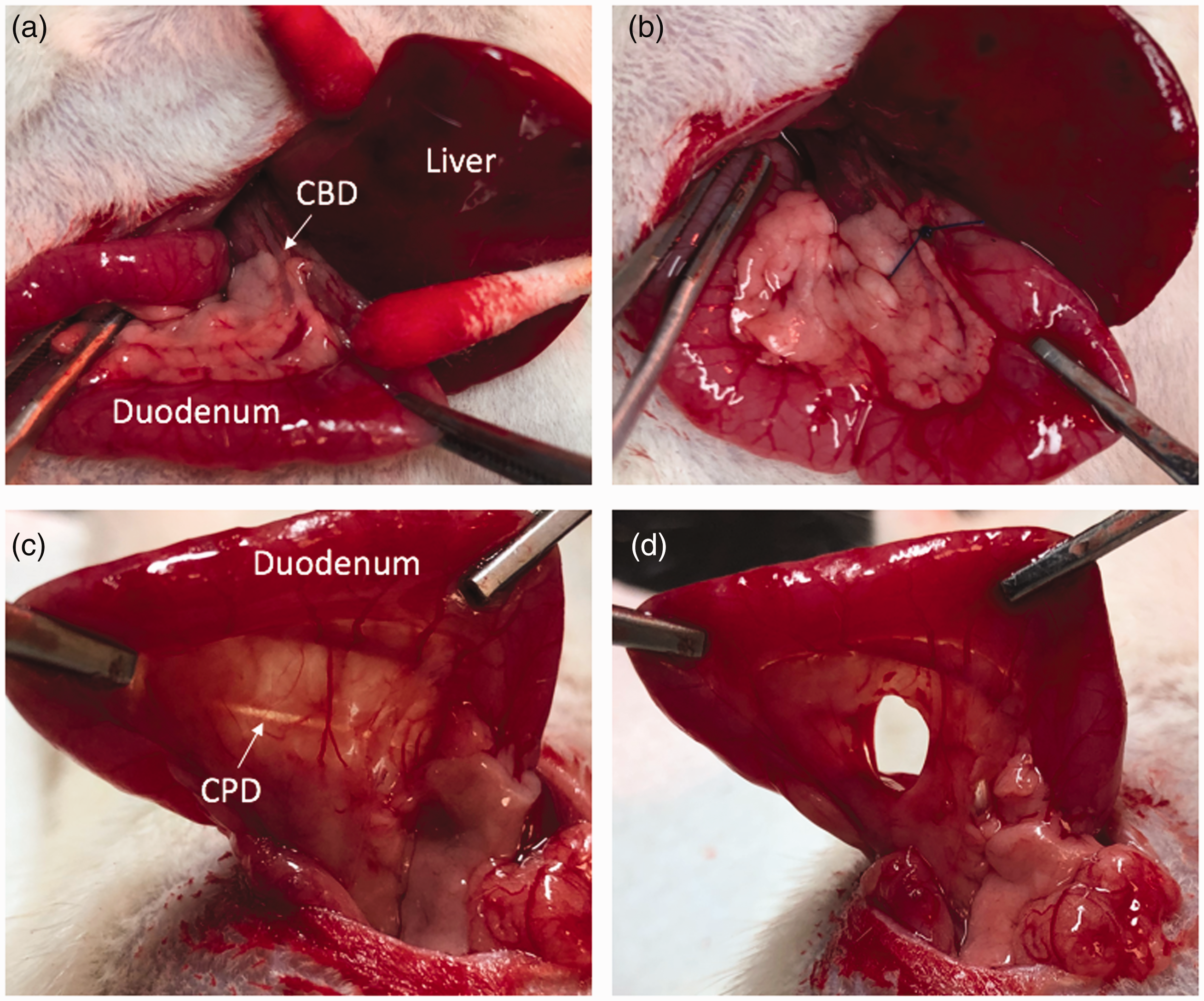
Steps to create the common pancreatic duct transection with bile duct ligation model. (a) The duodenum is extracorporealized though a transverse laparotomy incision and the common bile duct (CBD) is identified using gentle caudal traction on the duodenum. (b) The CBD is ligated with 5-0 prolene suture. (c) The common pancreatic duct (CPD) is identified with gentle ventral traction on the duodenum and (d) The CPD is sharply transected with an 11 blade.
Data collection
Rats were euthanized by exposing them to gradually increasing concentrations of carbon dioxide, starting at 30% and ending at 70%. Total gas exposure was 10 min per our institutional policy, which was sufficient to ensure apparent clinical death. Euthanasia occurred on postoperative day 1 (n = 8), 2 (n = 20), or 3 (n = 8). Necropsies were performed on all rats. Blood (0.5 ml) and ascites (0.1 ml) were collected and checked for levels of amylase and lipase. All assays were performed by University of Washington CLIA‑licensed and CAP‑accredited Clinical Laboratories using a Beckman Coulter AU680 instrument. Volume of ascites was recorded. Necropsies were performed and the degree of saponification was recorded (n = 12) using a scoring system developed by our group termed the peritoneal saponification index (PSI). Saponification is the process by which fatty acids released during fat necrosis secondary to exposure to pancreatic enzymes react with calcium to form insoluble salts, changing involved tissues to a firm and yellow character. For calculating the PSI, the abdomen was divided into four quadrants and each quadrant was assigned a score of 0–3 points based on the total amount of clinically evident saponification for a possible maximum of 12 points (Figure 3(a) and (b)). A score of 0 signifies no saponification, 1 signifies saponification <4 mm, 2 signifies saponification 4–9 mm, and 3 signifies saponification >10 mm. Data was not collected from rats that died prior to euthanasia (n = 2) but necropsies were performed to determine cause of death.
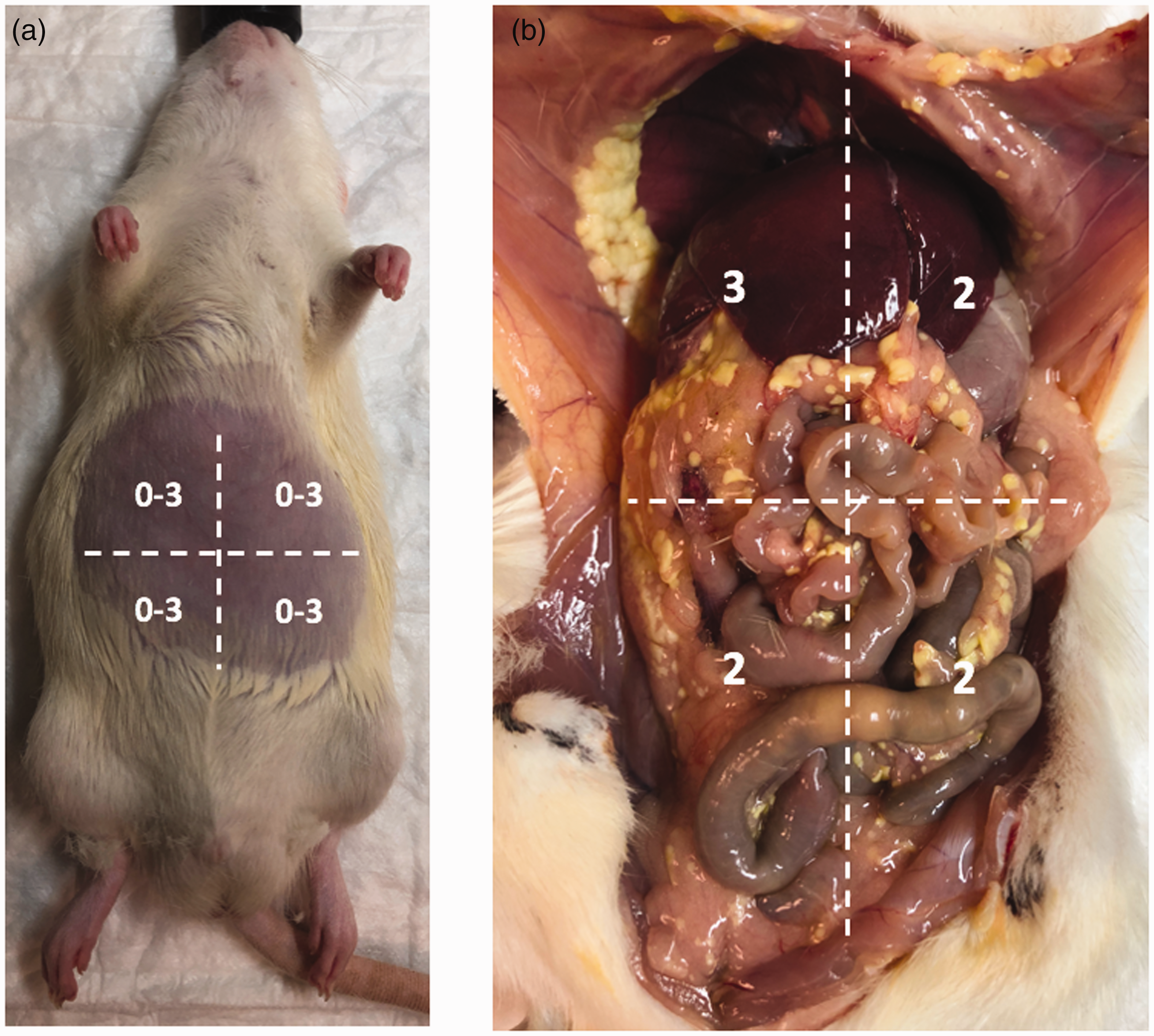
Peritoneal saponification index. (a) The rat abdomen is divided into four quadrants and each quadrant is assigned a score of 0–3 for a maximum score of 12. Zero signifies no saponification, 1 signifies saponification <2 mm, 2 signifies saponification 2–5 mm, and 3 signifies saponification >5 mm and (b) Example of a rat with a saponification score of 9.
Histology
Tissues from selected animals were collected and fixed in 10% phosphate-buffered formalin for 48 h. Fixed tissues were then embedded in paraffin, and using a microtome sectioned at 5 µm onto positively charged slides. Slides were stained with hematoxylin and eosin. All slides were examined by a pathologist.
Statistical analysis
Data was analyzed using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA) and statistical significance was determined using Student’s t-test. Statistical significance was determined by a p-value <0.05.
Results
Bile duct ligation with common pancreatic duct transection was performed in 36 rats. There were no intraoperative complications from the bile duct ligation and none of the rats was found to have bile peritonitis at necropsy. Similarly, there were no intraoperative complications from the common pancreatic duct transection. All rats appeared well immediately following recovery from the procedure; however, two rats were found to have decreased spontaneous activity and increased respiratory rate within the first 24 h (6%). Both rats were electively euthanized and found to have large dilated stomachs and signs of aspiration on necropsy. The remaining rats were found to have a mean score of 2.25 (interquartile range (IQR): 1–3) on the modified scoring system for determining humane endpoints.
After common bile duct ligation and pancreatic duct transection, peritoneal amylase and lipase collected postoperatively were significant elevated (Figure 3(a) and (b)). Median volume of peritoneal ascites was 0.25 ml (IQR: 0.002–1.00 ml). Mean peritoneal amylase was elevated to 210,000 U/l (IQR: 18,000–351,000 U/l), 171,000 U/l (IQR: 31,000–280,000 U/l), and 103,000 U/l (IQR: 41,000–126,000 U/l) on postoperative day 1, 2, and 3, respectively. Similarly, mean peritoneal lipase was elevated to 42,000 U/l (IQR: 3000–62,000 U/l), 63,000 U/l (IQR: 7000–79,000 U/l), and 65,000 U/l (IQR: 28,000–61,000) on postoperative day 1, 2, and 3, respectively (Figure 4(a)). The difference in amylase and lipase between the three timepoints was not statistically significant (Figure 4(b)). Serum amylase and lipase measured at time of euthanasia showed a corresponding increase with values of 16,000 U/l (IQR: 4000–22,000 U/l) and 4000 U/l (IQR: 500–4000 U/l), respectively. Three of 34 samples submitted for analysis returned as “quantity not sufficient.”
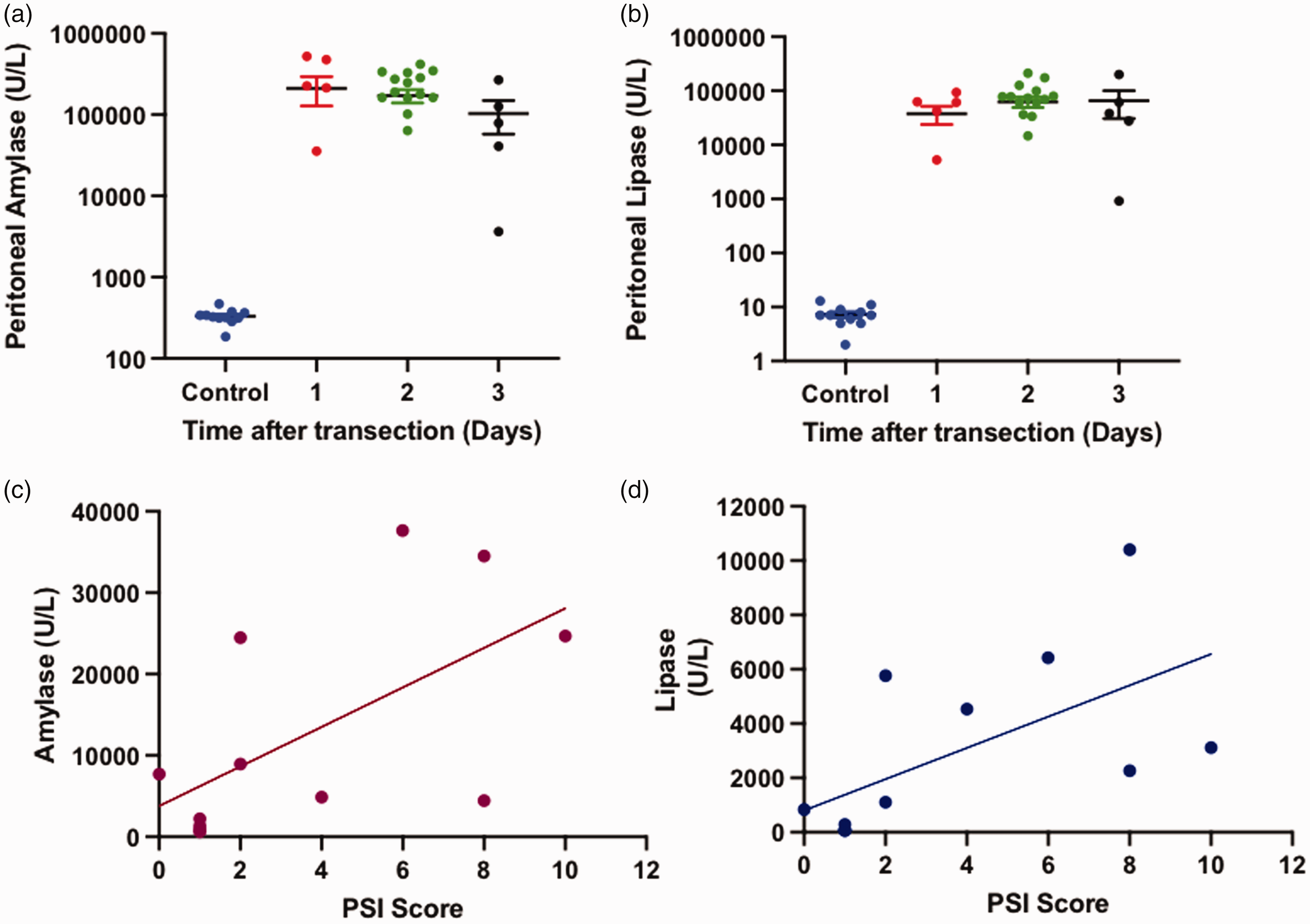
(a) and (b) Peritoneal amylase and lipase levels postoperative day compared with normal rat serum amylase and lipase. The difference in amylase and lipase between the three timepoints was not statistically significant. (c) and (d) Correlation of amylase and lipase with peritoneal saponification index (PSI) score.
Rats with elevated peritoneal amylase/lipase demonstrated a robust inflammatory response with gross saponification visible throughout the peritoneum but variable volume of ascites. Using the PSI scoring system, the inflammatory response was quantified and demonstrated to correlate with the level of amylase/lipase (Figure 4(c) and (d)). Histopathological examination of the head of the pancreas and duodenum demonstrated findings consistent with a POPF but not pancreatitis. The pancreatic tissue appeared normal (Figure 5(a)), whereas the peritoneum overlying the pancreas and duodenum demonstrated inflammatory changes (Figure 5(b) and (c)). There was also omental fat necrosis, which corroborates the gross saponification observed at necropsy (Figure 5(d)).
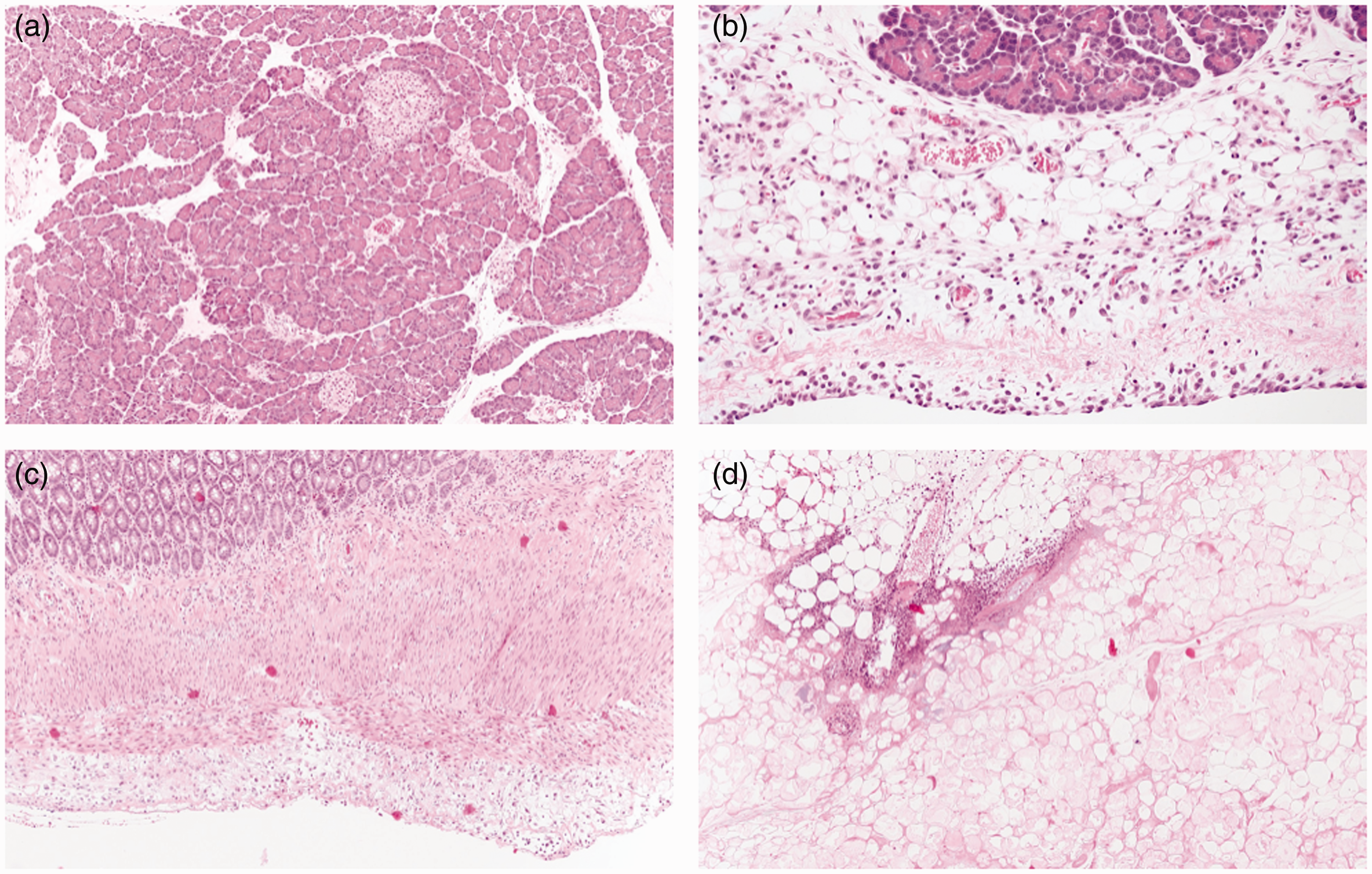
Histopathological examination of rat tissues obtained at necropsy. (a) Normal pancreas without any signs of inflammation (100x). (b) Pancreas with overlying peripancreatic fat inflammation consistent with postoperative pancreatic fistula and peritoneal mesothelial reaction (200x). (c) Duodenum with serositis (100x) and (d) Omentum with evidence of fat necrosis (100x).
Discussion
Postoperative pancreatic fistula is a significant contributor to morbidity and mortality after pancreatic surgery. Several studies have attempted to decrease the incidence of POPF by improving surgical techniques, utilizing topical hemostatic/occlusive agents, stenting the pancreatic duct, and using pharmacotherapy to reduce pancreatic secretions.9–15 However, no one intervention has been shown to be reliably efficacious, and progress has been hindered by the lack of reliable POPF experimental models. In this study, we describe the development of a novel rat model for POPF through transection of the common pancreatic duct after ligation of the common bile duct to prevent biliary peritonitis. This technique results in robust and reproducible pancreatic leak with acceptable morbidity and mortality. Additionally, we describe a new objective scoring system, the PSI, which quantifies the degree of intraabdominal saponification resulting from POPF and can be used as a relevant clinical endpoint for future experimental studies.
The model described above has several key advantages over currently available models. Division of a minor pancreatic duct by Tanaka et al. 18 was facilitated by portal venous injection of indocyanine green, which is technically cumbersome and extends the length of the surgical procedure by at least 30 min in order to allow time for biliary excretion. 20 Ascitic amylase and lipase levels begin decreasing immediately after the first postoperative day and normalize spontaneously without intervention, in contrast to POPF seen after pancreatectomy in humans. Additionally, median ascitic amylase and lipase levels in the minor duct model are 2- and 3-log-fold lower than levels induced by our new model, limiting the leak’s clinical impact as well as its durability. Our model induces a robust and clinically significant leak that persists for at least three days, generating very similar intraabdominal findings as seen in human patients with POPF. These markedly increased peritoneal enzyme levels compared with baseline may increase the statistical power of future studies, potentially allowing for the use of fewer animals to test future therapies.
Other models seeking to reproduce the pathophysiology after distal pancreatectomy utilize transection of the splenic duct with removal of the distal pancreatic tissue as well as splenectomy without the use of indocyanine green.20,22 This model requires the investigator to take care to ligate the splenic vessels while ensuring that the splenic duct of the pancreas is not also ligated, which is technically challenging, particularly without the aid of a microscope. Kuscher et al. used this model but performed the distal pancreatectomy and splenectomy irrespective of ductal anatomy. 20 This procedure did not result in consistent peritoneal ascites and they performed peritoneal lavage in order to collect sufficient peritoneal fluid for analysis. There was an elevation in peritoneal levels of amylase and lipase to postoperative day 4, after which the levels spontaneously returned to normal, recapitulating the limitations of the Tanaka model.
In order to create more robust POPF we sought to create a model that involved transection of the common pancreatic duct. However, as demonstrated by Tanaka et al. and our own experience, transection of the common pancreatic duct alone was non-survivable secondary to bile peritonitis. In order to prevent this complication we simultaneously ligated the common bile duct as has been previously described in non-POPF models. 21 Bile duct ligation in the rat is a well described component of hepatic injury models and there is no evidence that short-term biliary obstruction (1–3 days) would alter the physiology of or systemic response to POPF. Of note, only female rates were used in these experiments due to their relatively docile nature and ease of handling. There is no available evidence to suggest that the physiologic response to POPF or rates of POPF development would differ between male and female rats; however, this could be validated in future studies.
Transection of the common pancreatic duct with ligation of the common bile duct was technically reproducible and yielded a robust biochemical, histopathological, and clinical response in the rats. Total anesthesia time was less than 10–12 min per animal, while procedure time was generally less than 7 min. Similar to prior studies, we found the volume of peritoneal ascites to be highly variable; however, peritoneal amylase and lipase were consistently elevated and several log-fold higher when compared with models that transected the minor pancreatic ducts.14,20 On necropsy the rats demonstrated a strong inflammatory response with saponification throughout the abdomen. The degree of saponification correlated with amylase and lipase levels but was independent of the volume of ascites. We thus developed a peritoneal saponification index score which we found to be a fast and reliable way to assess and quantify the macroscopic reaction of the abdominal cavity to POPF as well as its clinical impact. Finally, microscopic examination of the tissues confirmed the inflammatory response secondary to the POPF without any evidence of pancreatitis.
Overall, this model reproducibly recreates the pathophysiology of a POPF biochemically, microscopically, and grossly. We propose that this model will be of particular use as a short-term model in the study and development of topical agents that prevent POPF by acting as a sealant, suppression of pancreatic secretions, or a combination of the two. While this model shows promise there are some limitations: 1) while PSI or peritoneal amylase/lipase levels are consistent, variable peritoneal ascites volume makes biochemical evaluation difficult in some animals; 2) while there was no histologic evidence of pancreatitis, the effect of sealing the ductal leak, and causing temporary pancreatic duct obstruction, is not known.; 3) the long-term effects of bile duct ligation in the setting of pancreatic leak are not known and may potentially affect the rat’s response to yet-developed therapies.
In conclusion we describe the development of a new experimental rat model for POPF that leads to robust and reproducible elevations in peritoneal pancreatic enzyme levels after transection of the common pancreatic duct and ligation of the common bile duct. We also propose a new clinical scoring system (PSI) for standardization of the assessment of intraabdominal inflammation secondary to POPF as an endpoint for assessing efficacy of future interventions. This reliable small-animal model should facilitate further study of innovative techniques to reduce POPF.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. Funding supporting this research: 1. Swim Across America Young Investigator Award, 2. Americas Hepatopancreatobiliary Association Research Development Award, 3. Philanthropic support from the Nowoj Family, Mortenson Construction, and Beach Family.
