Abstract
Objective
The objective of this work was to anatomically locate the urethral orifice in female minipigs and describe the use of video laryngoscopes in urethral catheterization.
Methods
Urethral catheterization guided by a video laryngoscope was attempted in 16 adult female Bama minipigs. The anatomical location of urethral orifices, operating time and complications (mucosal edema and bleeding in the vaginal vestibule, and the numbers of red blood cells (RBCs) and white blood cells (WBCs) in mid-stream urine samples) were recorded.
Results
The anatomical location of the urethral orifice: the depth of the urethral orifice in female Bama minipigs was 4.2 ± 1.2 cm; all the urethral orifices were covered by mucosal folds of the vaginal vestibule. In the supine position, the orifice of the urethra at 9–12 and 1–3 o’clock accounted for 6.25%, 6.25%, 18.75%, 50%, 12.5%, 6.25% and 6.25%, respectively. All animals were successfully catheterized and the operating time was 9.0 (6.0–12.8) min. Complications: no bleeding in the vaginal vestibule was observed; the incidence of mucosal edema was 12.5%, all of which were mild; of urine samples collected 1 h after catheterization, 12.5% were found to contain RBCs and no RBCs were detected 6 h after catheterization; no WBCs were detected 1 h or 6 h after catheterization.
Conclusions
The urethral orifice of female minipigs was located deep in the vagina at variable clock directions and was unexceptionally covered by mucosal folds. Applying a video laryngoscope in urethral catheterization allowed quick and accurate exposure of the urethral orifice and minimal operational injury in female minipigs.
Introduction
There is 84% sequence homology and a close resemblance in terms of anatomy, physiology and pathogenesis of diseases between humans and pigs. Therefore, as one of the most commonly utilized animal models, pigs are considered to be ‘an ideal non-clinical model system’1,2 frequently used for preclinical toxicology, 3 shock resuscitations, 4 metabolic diseases 5 and organ transplantation 6 studies. Indwelling urinary catheters are often required in pigs during experiments to monitor their pathophysiological conditions such as organ perfusion and metabolism via urine output and urinalysis. However, as the tip of the penis is cork-screw shaped in male minipigs and the urethral orifice in female minipigs is concealed among mucosal folds of the vaginal vestibule, it is difficult to perform urethral catheterization. 7 Therefore, invasive procedures like bladder puncture or urostomy are adopted to place a bladder catheter, which may affect the establishment of animal models and the evaluation of experimental effects. This study explored the use of video laryngoscope in urethral catheterization in female minipigs by analyzing the anatomical location of the urethral orifice in Bama minipigs.
Materials and methods
Animals
In total, 16 healthy female Bama minipigs, 6–8 months of age and weighing 35.9 ± 4.8 kg, sourced commercially from Beijing Keyu Animal Breeding Center, license number: SCXK (Beijing) 2017-0003, were used for this study. The study was approved by the Ethics Committee. All animals were acclimatized to the laboratory facilities for one week at a room temperature of 24–28°C and a humidity of 40–70%. Prior to anesthesia, food and drink were withheld for 12 h and 4 h respectively.
Anesthesia was induced with Zoletil 50 (Zoletil, Tieletamine and Zolazepam) at 5 mg/kg by intramuscular injection, 15 min prior to which subcutaneous injection of atropine sulfate at 0.1 mg/kg was administered. Upon induction of general anesthesia, one-half of the first dose of Zoletil 50 was given intravenously in the ear every 30–40 min to maintain anesthesia. A 7 mm diameter tracheal intubation was placed to ensure ongoing ventilation. The animal was breathing spontaneously without oxygen supplementation, and an oxygen probe was used to monitor blood oxygen saturation (SpO2), which was greater than 98% throughout the experiment in all animals. The animal was positioned supine and fixed to a V-shaped platform, on which there was a 38°C electric blanket to keep the animal warm.
All animals received no antibiotic treatment during the experiment. All pigs were awakened from anesthesia at the end of the experiment, and the tracheal intubation and the foley catheter were both removed.
Apparatus and consumables
Video laryngoscope (TD-C-IV:85 mm × 11 mm, Zhejiang UE Medical, China); disposable laryngeal blades (TDC-3:108 × 23 mm, Zhejiang UE Medical, China); Hua Sheng H-III automatic urine analyzer (Beijing Hua Sheng Yuan Co., Ltd., China); disposable double-balloon silicone foley catheter (including a flexible guide wire, 12 Fr; Zhanjiang Star Enterprise Co. Ltd., China); disposable urine drainage bag (Beijing Kang Yu Medical Instrument Co., Ltd., China) and two Allis forceps.
Catheterization procedures
Two researchers who were familiar with the anatomy of the urogenital tract of minipigs, catheterization procedures and the use of video laryngoscope performed the catheterization cooperatively. The animal was placed on its back on a V-shaped platform with an appropriate support under the lumbar region, while its hind legs were bent and pressed to ventral sides (Figure 1(a)). Before catheterization, the mucosa and surrounding skin of the urogenital opening were sterilized with 1% benzalkonium bromide (Guangdong Heng Jian Pharmaceutical Co., Ltd., China) cotton balls and iodophor and then covered by a sterile surgical towel.
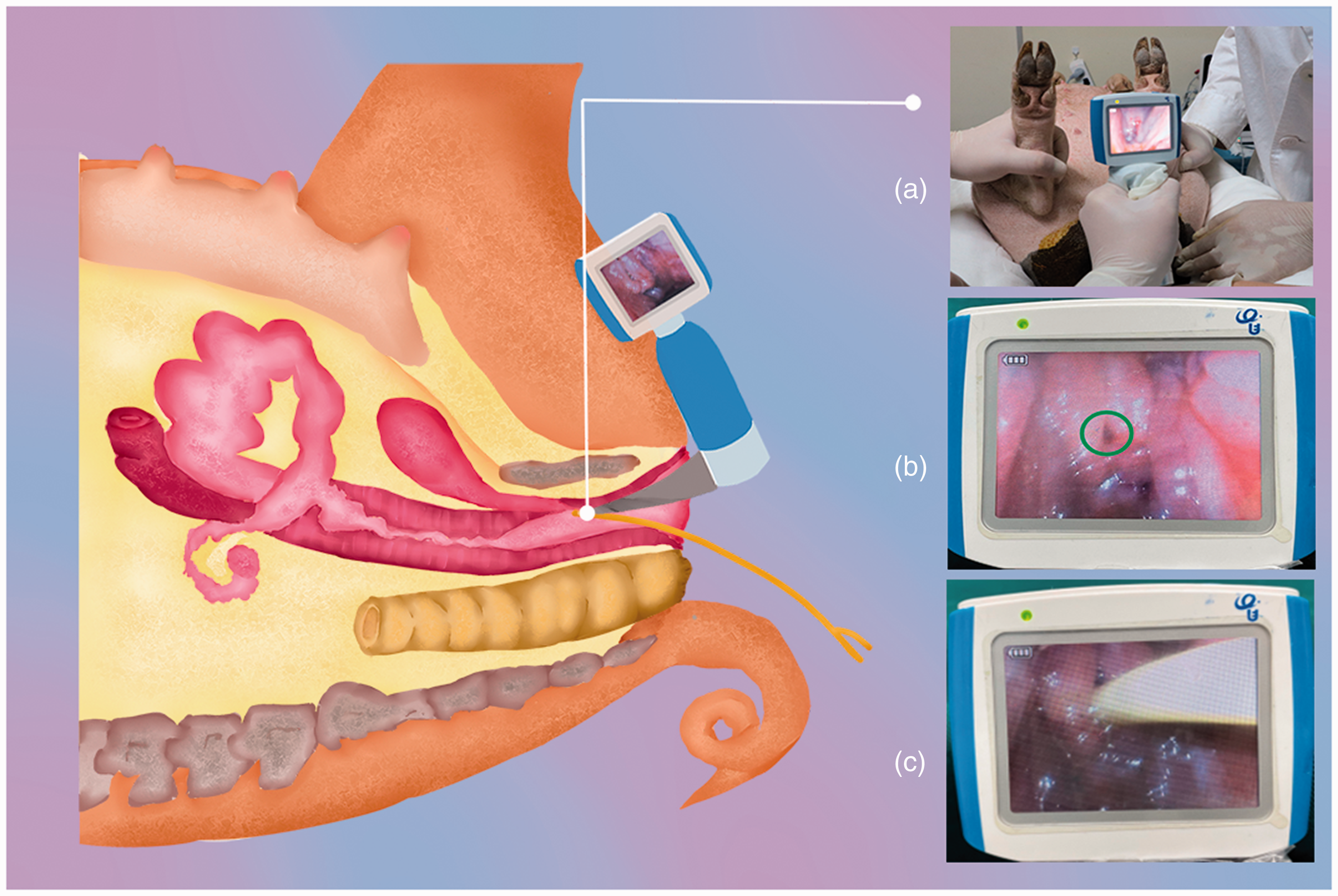
The procedure of video laryngoscope-guided urethral catheterization in female minipigs. (a) Searched for the urethral orifice; (b) the urethral orifice (green circle) was visualized; (c) the urinary catheter was inserted.
During the catheterization, one researcher retracted the vulva with two Allis forceps from the left and right sides while the other researcher held a video laryngoscope in the left hand and slowly advanced it into the vulvar vestibule along the vaginal wall under video visualization of the laryngoscope. After the vaginal opening was fully exposed, the blade was slowly withdrawn, and the laryngoscope was rotated from the 12 o’clock to the 9 o’clock position or from the 12 o’clock to the 3 o’clock position before being advanced to push aside the mucosal folds of the vaginal vestibule to look for the urethral orifice (Figure 1(b) and (c)). Once the urethral orifice was adequately visualized, a disposable sterile foley catheter, with its tip lubricated with sterile paraffin oil, was gently fed through the external urethral orifice for about 2–3 cm with the support of a guide wire. Then the guide wire was removed before the foley urinary catheter was further advanced for about 3–5 cm until the clear yellow urine was seen flowing. In the case of minimal or no drainage of urine, a 10 ml syringe was used to inject an appropriate amount of normal saline into the bladder to exclude blockage and ensure adequate liquids inside, and then to draw urine back so as to confirm the correct placement of the catheter.
The catheter was advanced for another 2–3 cm to make sure that the catheter balloon sat in the bladder, and then 5–10 ml normal saline was injected into the balloon to fix the urinary catheter, which was then connected to a urine drainage bag and secured with adhesive tape to the tail. Thus, the transvaginal catheterization was completed.
Observation indicators and methods
Observation of the anatomical location of urethral orifice. When the urethral orifice was visualized by the video laryngoscope, the distance of the urethral orifice to the mucocutaneous junction of the vulva was measured and was recorded as the depth of urethral orifice. The mucosal fold coverage over the urethral orifice was recorded and the ‘clock-ray method’ was adopted to observe the clock direction of the urethral orifice in Bama minipigs when they were positioned supine. The specific method was as follows: the Bama minipig was placed in the supine position while the coronal plane of the vaginal vestibule was divided into 12 equal parts like a clock face. The upper side, which was also the front of the vaginal vestibule (pointing to the abdomen) was set at 12 o’clock, and the lower side, which was also the back of the vaginal vestibule (pointing to the coccyx), was set at 6 o’clock, so the right side was set at 3 o’clock and the left side at 9 o’clock. The operating time and success rate of the urethral catheterization under the guidance of video laryngoscopes were recorded. Evaluation of postoperative complications. a) Mucosal edema and bleeding in the vaginal vestibule after catheterization were assessed. The classification of mucosal edema: none: mucosal morphology is normal; mild: the mucosa is slightly thickened, and rebounds quickly when pressed; moderate: the mucosa is noticeably thickened with shallow folds and rebounds slowly when pressed; severe: the mucosa is significantly thickened with no fold and looks tense and shiny. b) Urinalysis method: the catheter was opened before being clamped for 10 min and the drained mid-stream urine was collected for urine examination aseptically at 1 h and 6 h after catheterization. The red blood cells (RBCs) and white blood cells (WBCs) in urine samples were detected with an automatic urine analyzer to diagnose urethral bleeding (at least 10 RBCs per microliter in the midstream urine) and urinary tract infection (at least 25 WBCs per microliter in the mid-stream urine).
Statistical methods
SPSS 22.0 statistical software was used for data processing and analysis. Normally distributed measurement data were reported as ( ± s) and count data were expressed as relative numbers.
Results
Animals
Bama minipigs weighed 35.9 ± 4.8 kg and their body length was 74.7 ± 4.0 cm (Table 1).
Experimental data for the catheterization of 16 Bama minipigs.
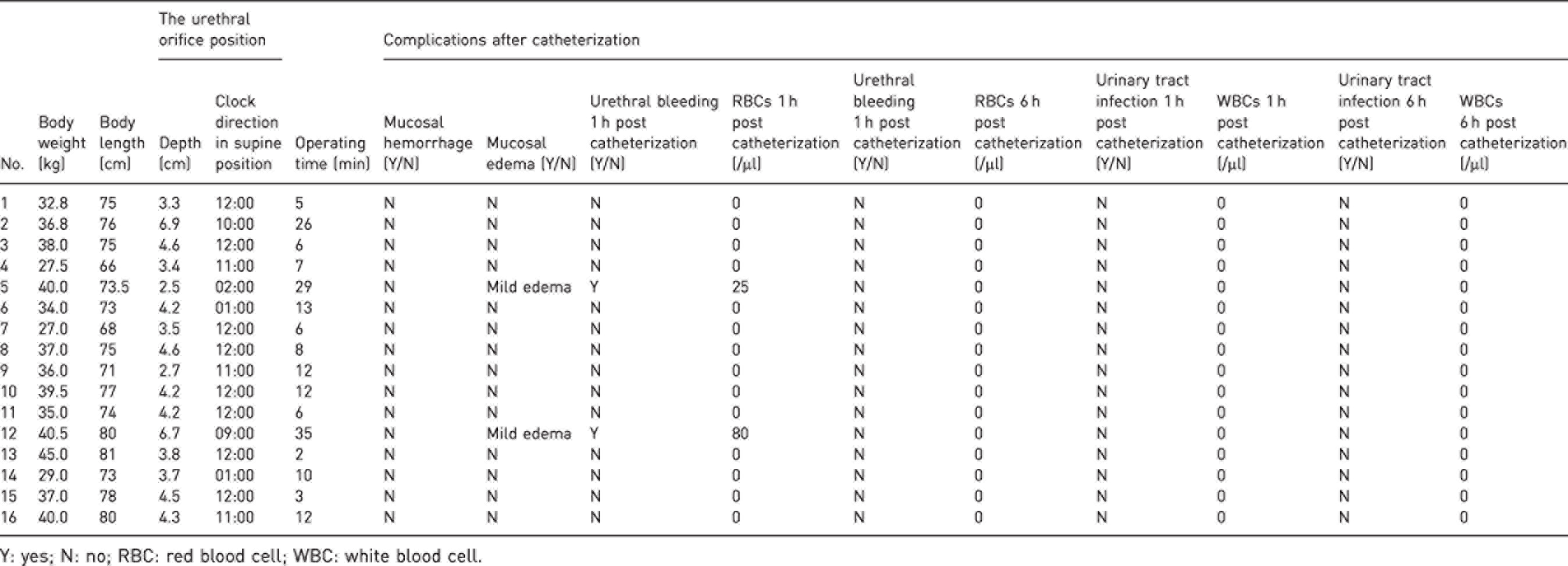
Y: yes; N: no; RBC: red blood cell; WBC: white blood cell.
Anatomical location of urethral orifices
All the urethral orifices of female Bama minipigs were covered by the mucosal folds of the vaginal vestibule, 93.75% of which were covered by vertical mucosal folds and 6.25% by longitudinal folds; the entry depth of the blade was 4.2 ± 1.2 cm when the urethral orifice was visualized in the screen of the video laryngoscope. In the supine position, the orifice of the urethra at 9–12 o’clock and 1–3 o’clock accounted for 6.25% (1/16), 6.25% (1/16), 18.75% (3/16), 50% (8/16), 12.5% (2/16), 6.25% (1/16) and 6.25% (1/16), respectively.
Success rate and operating time of the catheterization
All the catheterizations guided by the video laryngoscope were successful and the success rate was 100% accordingly. The median operating time was 9.0 (6.0–12.8) min.
Postoperative complications of the catheterization
No bleeding was found in the vaginal vestibule after catheterization; the incidence of mucosal edema was 12.5% (2/16), all of which were mild. Of urine samples collected 1 h after catheterization, 12.5% were found to contain RBCs (2/16, the detected numbers were 25/µl and 80/µl respectively) and no RBCs was detected in urine samples collected 6 h after catheterization; no WBCs was detected in urine samples collected 1 h or 6 h after catheterization.
Discussion
With the rise of the animal protection movement and the promotion of the ‘3Rs’ principle (Reduce, Replace, Refine), the International Organization for Animal Protection has imposed stricter restrictions on the use of non-human primates and dogs, which are gradually being replaced by the laboratory pigs. Take Canada as an example: while the use of rodents has dropped by 25%, the use of laboratory pigs has increased by 55%. It has become the most used large animal model in biomedical research and medical operation training, and demonstrated important and practical values in life science studies regarding, for example, cardiovascular diseases, urinary system diseases, nutritional and metabolic diseases, burn shock, oral diseases and pharmacology, toxicology3 –6,8
Catheterization is an indispensable procedure in laboratory pigs for biomedical research, which can effectively prevent urine retention during general anesthesia. There are some other reasons for catheterization: 1) serial urine sampling and urinalysis to evaluate the metabolism of the animal during the experiment; 2) measurement of urine output, as a sensitive variable the blood volume and renal perfusion; 3) measurement of intra-abdominal pressure; 4) bladder irrigation and drainage; 5) intravesical drug administration.
In biomedical research on pigs, researchers usually performed bladder puncture or urostomy to place a bladder catheter in male minipigs due to the corkscrew shaped penis. Though there are no similar anatomical obstacles in female minipigs and the urethral catheterization is theoretically feasible, invasive bladder puncture or urostomy has been adopted instead of non-invasive transurethral catheterization because of the difficulty and the prolonged time consumed in finding the urethral orifice, which is concealed among mucosal folds of the vaginal vestibule. 9 However, invasive procedures can disrupt the continuity of the bladder, increase the risk of infection, invalidate the simulation of the physiological state of the urinary system and affect the establishment of the animal model. In addition, bladder wall hemorrhage caused by puncture or urostomy may affect the accuracy of urinalysis, and fluid leaking out of abdominal incision may easily result in intra-abdominal effusions and thus affect the functions of abdominal organs.
Researchers have successively explored the use of nasal endoscopes, vaginal speculum and ordinary pediatric laryngoscopes for urethral catheterization in female pigs,7,9 –11 but urethral catheterization was still difficult and time-consuming. Musk et al. reported the use of a vaginal speculum for transurethral catheterization in female laboratory pigs, but the procedure was unexpectedly complicated and took up to 60 min in some animals. 9 Equipment such as a vaginal speculum or an ordinary laryngoscope that we had tried to use resulted in blocking the external light source, hindering us from finding the urethral orifice hidden in mucosal folds of the vaginal vestibule. Also, excessive traction and repeated attempts could lead to mucosal edema and bleeding in the vaginal vestibular, which increased the challenge of catheterization. In a word, the urethral catheterization with ordinary equipment was time-consuming, requiring generous time allocation to the procedure, so the efficiency of catheterization was unsatisfactory. In addition, since there was a shallow diverticulum on the ventral floor of the urethral orifice, as described in the literature, 9 the catheter might be accidentally inserted into the diverticulum if the urethral orifice failed to be exposed adequately, which emphasized the importance of adequate visualization of the urethral orifice in increasing the efficiency of urethral catheterization.
The video laryngoscope provided a feasible solution for the efficiency improvement of urethral catheterization in female laboratory minipigs. With its cold-light system that ensured a bright view and the real-time imaging system that provided high-definition images for observing the vaginal vestibule structure, the video laryngoscope not only improved the efficiency of exposing the urethral orifice, but also assisted the transurethral insertion of the catheter. The study found that most (93.75%) urethral orifices in Bama minipigs were covered by vertical mucosal folds of the vaginal vestibule, while only one urethral orifice was covered by longitudinal folds, resulting in significantly more time spent on the urethral catheterization of this minipig than others.
According to previous studies, the urethral orifice in the female minipig was located in the mucosal folds of the vaginal vestibule approximately one-third to one-half 7 of the distance to the cervix. The deep location of the urethral orifice was an important reason for the difficulty of urethral catheterization. Musk et al. found that the urethral orifice was one-half of the distance to the cervix when they dissected the urogenital tract of large white pigs. 9 However, because it was more difficult to expose the cervix, the practical value of exploring the distance from the urethra to the cervix was limited for guiding urethral catheterization.
In this study, the distance of the urethral orifice to the mucocutaneous junction of the vulva was measured, and the average distance was about 4 cm. The result has practical value in helping researchers to identify the urethral orifice more quickly and accurately.
The study found that the clock direction of the urethral orifice in female minipigs was variable and was at 10–12 o’clock and 12–2 o’clock directions in the supine position in most cases with a probability as high as 95%. The video laryngoscope was advanced while turned left and right to search for the urethral orifice. With the rotatable shaft connecting the screen and the camera, angles between them could be adjusted flexibly to avoid blind spots and facilitate the blade to push aside vaginal folds at different clock directions to visualize the urethral orifice. Compared with nasal endoscopy and vaginal speculum, it brought about minimal damage to the urogenital tract mucosa because the vagina opening was no longer required to be retracted excessively to expose the urethral orifice.
The video laryngoscope is a common and available device in tracheal intubation, which has been used clinically for many years. It is also widely used in both in-hospital and out-of-hospital settings.
In this study, the video laryngoscope improved the efficiency of exposing the urethral orifice as it significantly shortened the operating time of catheterization and reduced the incidence of postoperative complications such as mucosal edema and bleeding in the vagina, comparing with previous studies. In addition, the disposable laryngeal blade of 108 mm in length was long enough to expose urethral orifices and its smooth blunt tip allowed little irritation to animals and minimal injury to the vaginal mucosal. The disposable laryngeal blade is cheap and can also prevent cross-contamination.
The following aspects should be noted during video laryngoscope-guided urethral catheterization: 1) operators should be familiar with the anatomical structure of the urogenital tract of female minipigs and correctly identify the urethral orifice and vaginal orifice as well as their anatomical relationship; 2) general anesthesia and adequate sedation should be given during the operation; 3) proper positioning is critical to visualizing the urethral orifice, which could be achieved by adjusting the angle of the pelvis with an appropriate support under the lumbar region and bending the hind legs while pressing them to the ventral sides to reverse the urethral orifice tending to outwards from fall inwards; 4) inserting a guide wire in the catheter helped better control the catheter to smoothly pass through the narrow part of the urethra and thus increase the success rate of catheterization. The guide wire should be appropriately flexible otherwise it might scratch the urethra.
Conclusion
The urethral orifice of female minipigs was not readily visualized since it was located deep in the vagina at variable clock directions and was without exceptional covered by mucosal folds. Applying a video laryngoscope in urethral catheterization allowed quick and accurate exposure of the urethral orifice and minimal operational injury, which was believed to be an effective approach for urethral catheterization in female minipigs.
Footnotes
Acknowledgements
We hereby express our thanks to Yuezeng Niu, MA, and to Xinyi Cai, MA, for his support in polishing the article.
Data availability statement
All data generated or used during the study appear in the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Major Program of Military Logistics Research Plan (grant number ALB18J001), the 13th Five-year Plan for Key Discipline Construction Project of PLA (grant number A350109), the Program of Beijing Municipal Natural Science Foundation (grant number 7204311) and the Program of National Natural Science Foundation of China (grant number 82072169).
