Abstract
Avian species are used as model systems in research and have contributed to ground-breaking concepts in developmental biology, immunology, genetics, virology, cancer and cell biology. The chicken in particular is an important research model and an agricultural animal as a major contributor to animal protein resources for the global population. The development of genome editing methods, including CRISPR/Cas9, to mediate germline engineering of the avian genome will have important applications in biomedical, agricultural and biotechnological activities. Notably, these precise genome editing tools have the potential to enhance avian health and productivity by identifying and validating beneficial genetic variants in bird populations. Here, we present a concise description of the existing methods and current applications of the genome editing tools in bird species, focused on chickens, with attention on animal use and welfare issues for each of the techniques presented.
Introduction
Genome editing (GE) is a method for the rapid introduction of precise changes into an organism’s genome. GE tools consist of programmable site-specific nucleases, that is meganucleases, zinc finger nucleases, TALENs, and CRISPR/Cas9.1,2 All of these programmable site-specific nucleases or ‘genome editors’ can be used to efficiently create precise genetic changes, generated through double-stranded breaks (DSBs) at specific locations in the genome. The DSBs are repaired by two conserved cellular machinery pathways: the non-homologous end-joining (NHEJ) pathway or the rarer homology-directed repair (HDR) pathway. The NHEJ pathway is often used by the cell to repair DNA damage and can result in small genetic insertions or deletions (INDELs) at the DSB. The more accurate HDR pathway occurs in the presence of a sister chromatid or an exogenous DNA fragment containing a homologous region spanning the DSB which are used as templates to repair the DSB. 3 The HDR repair process has been exploited to generate defined small genetic deletions, insertions, single base-pair changes in a gene, and even the directed integration of large exogenous transgenes precisely into the host cellular genome. What is novel is that this technology enables the creation of site-specific genetic changes without leaving any other modifications in the genome (footprintless editing), and these changes are indistinguishable from naturally occurring genetic variants. The development of genome targeting through HDR has significantly improved ‘transgenic’ research, by creating both plant and animal models with more precise and defined manipulations of the genome.
Avians serve as an important source of animal protein, and as a research model for the study of developmental biology, immunology and infectious diseases. 4 The chicken is both the principal avian research model and an agricultural animal with a global population numbering over 60 billion animals annually. Commercial chicken flocks are the source of fertile eggs for many experiments and these eggs can also be for public food consumption. Specialised chicken research flocks are also maintained at research institutes in compliance with the Animals (Scientific Procedures) Act 1986 in the UK and these flocks have been selectively bred to have defined genetic resistances and susceptibilities to avian diseases, specific developmental genetic mutations, or have been genetically modified to carry exogenous transgenes. Many other bird species such as quail, duck and zebra finch are also used as standard laboratory research models. 5 The annual Statistics of Scientific Procedures on Living Animals Great Britain 2019 reported 0.13 million avians (8%) among all animal groups were used in experimental procedures, whilst only 0.1% of birds were used for the creation and breeding of genetically altered (GA) animals. 6
Avian flocks are susceptible to a plethora of bacterial and viral pathogens threatening their health and welfare, and directly threatening flock security and the global agro-economy.
7
Bacterial pathogens such as
GE of bird species
Mammalian assisted
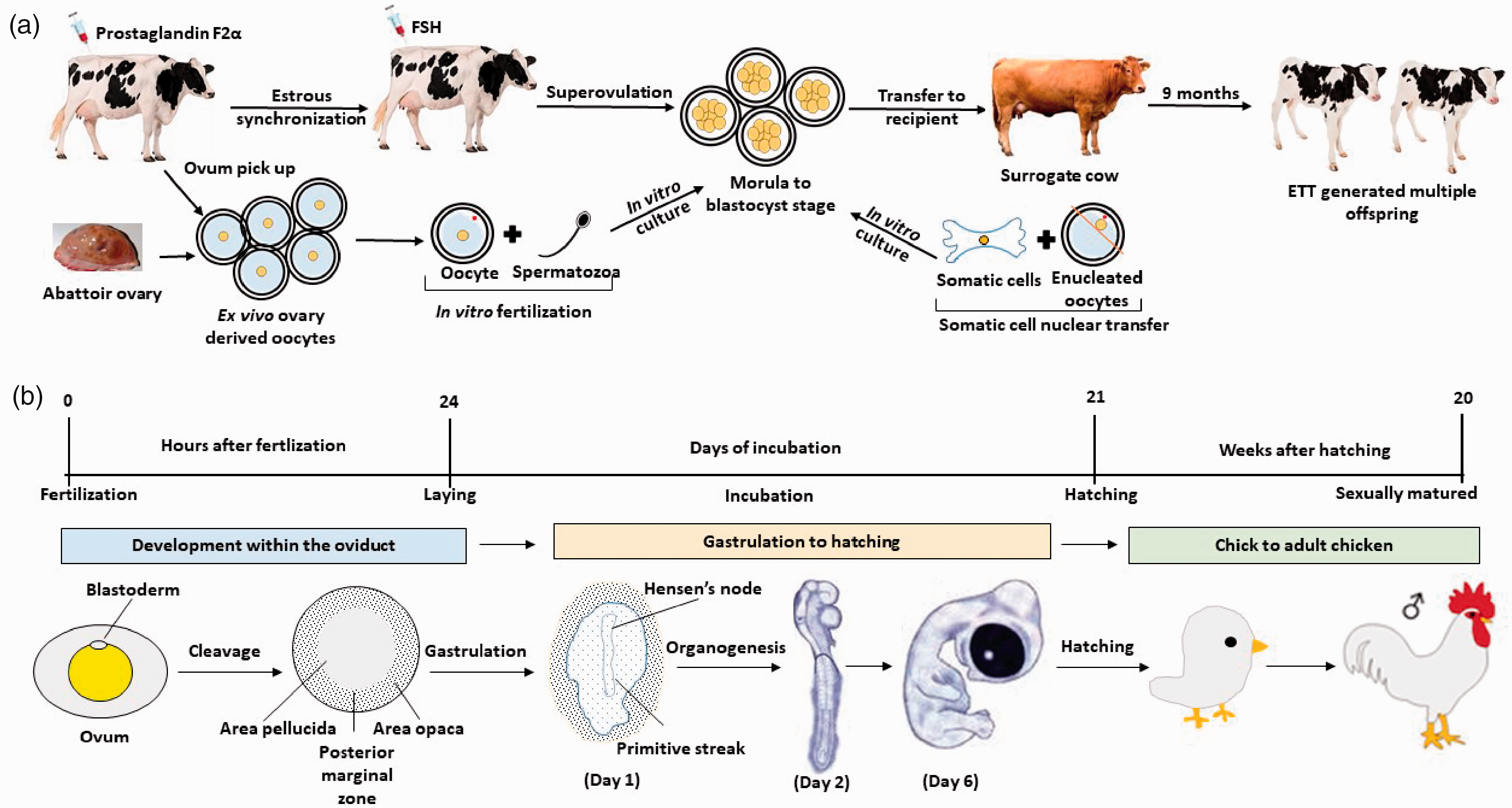
Schematic diagram of the reproductive physiology of bovine and chicken species. (a) Production of high genetic merit calves through the process of the synchronisation, superovulation, ovum pick up,
In contrast to mammalian species, avians have a unique reproductive physiology as well as distinctive structure of the ovum and pre-gastrulation stage embryo. In birds, the macrolecithal ovum is released from the ovary surrounded by a tough vitelline membrane, which serves as a protective layer around the deposited yolk. The ovulated ovum is rapidly fertilised with sperm that is stored by the female in sperm glands located in the oviduct. The single-cell zygote consists of a small pool of cytoplasm containing the fused pronuclei on the surface of the large yolk mass. The zygote takes approximately 24 h to pass through the oviduct region; the egg white is first added to enclose the yolk, followed by a shell membrane and a hard shell that are added during the final stages of passage through the oviduct. During this journey of the zygote from infundibulum to cloaca for laying of the egg, the single-cell zygote has undergone multiple rounds of cleavage and the pool of cytoplasm has developed into the blastoderm layer consisting of many tens of thousands of undifferentiated cells. Subsequently, the laid egg can be artificially or naturally incubated until hatching (Figure 1(b)). Thus, the relatively simple straightforward one-cell embryo microinjection procedures of generating genome edited mammalian embryos or SCNT cannot be easily replicated in birds as the early stage embryo is not accessible. Recent advances, however, have been made manipulating the early stage of ovum using intracytoplasmic sperm injection (ICSI) for fertilisation 24 and a surrogate shell for incubation. 25 However, this ICSI method has yet to be used for GE as few hatchlings were obtained from the ICSI embryos.
Mouse and human embryonic stem cells (ES cells), derived from the blastocyst stage embryo, are useful cell lines as they can be genetically modified
Methods used for CRISPR/Cas9-mediated in-ovo GE in birds
In spite of the complex architecture of the avian zygote, researchers have developed several delivery methods to introduce genetic vectors into the early avian embryo and cells from the embryo. As CRISPR/Cas9 genome editors are proving highly efficient at modifying the target species genome, these vectors are revolutionising efforts to manipulate the genome of birds. Current delivery methods to avian embryos and gametes are shown in Figure 2.
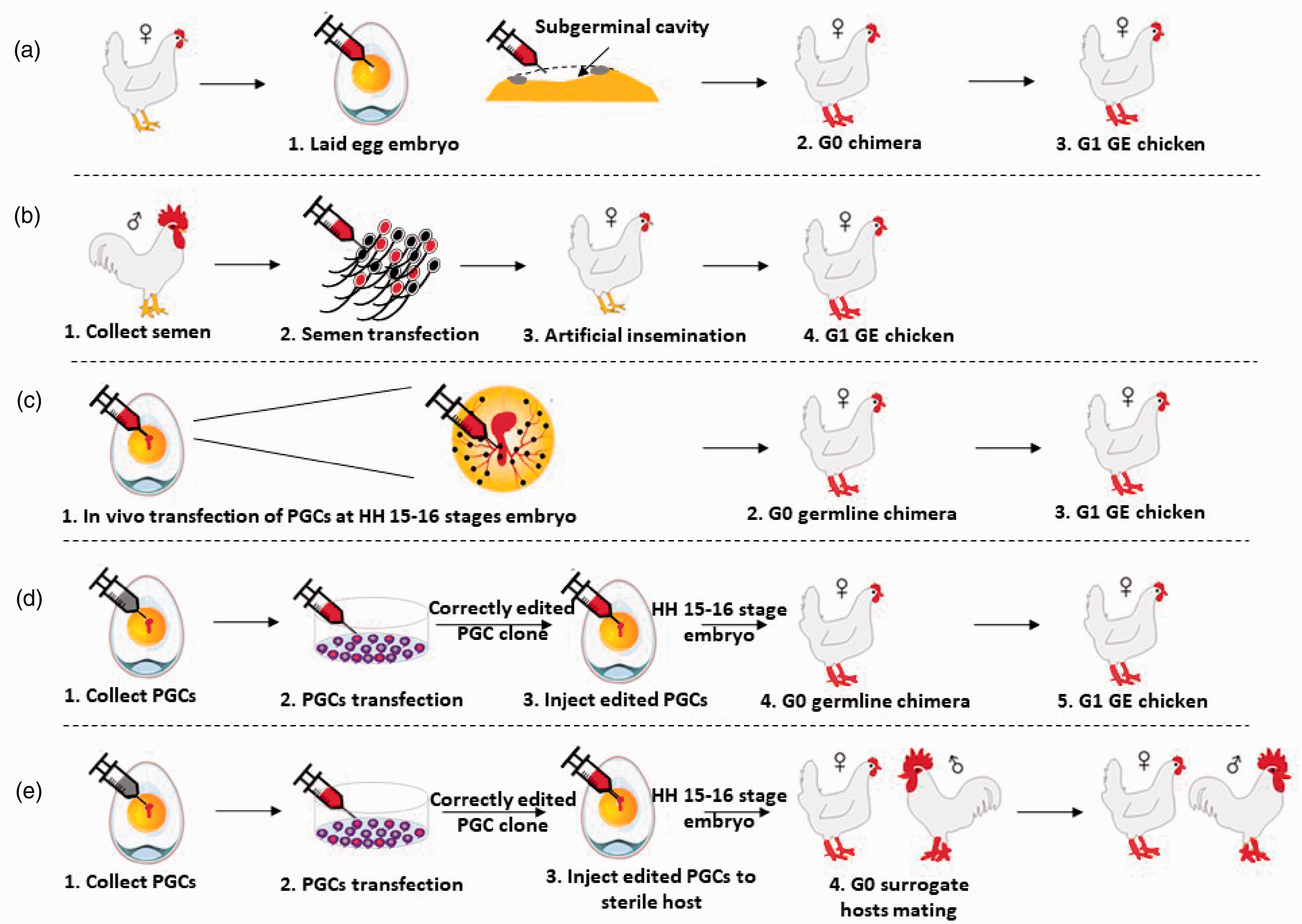
Schematic representation of different possible methods of establishing genome edited (GE) chicken. (a–e) the workflow of GE in the chicken via transduction with GE reagents to the embryonic blastoderm of a laid egg chicken embryo (a), sperm transfection for artificial insemination (b), primordial germ cell (PGC) transfection
Direct electroporation of embryos
The advantage of the avian model is that the developing avian embryo is directly accessible through a hole made in the surrounding egg shell, in a process called ‘windowing the egg’. The shell can be resealed and the egg re-incubated until the desired embryonic stage is reached or the embryo can be hatched. Direct
Blastoderm injection
Several research groups contributed the first genetically modified chickens using blastoderm injection of viral vectors. 32 A small cavity lies under the blastoderm, the ‘subgerminal cavity’, into which viral vectors can be introduced which will then transduce the overlying blastodermal cells and blastodermal germ cells (Figure 2(a)). The first genetically modified chicken was generated by the integration of the retroviral vector, known as avian leucosis virus (ALV), into the germline after blastoderm injection. 33 Later, the efficient generation of genetically modified chickens was reported using various replication-deficient viral vectors.34–36 Our laboratory and others showed an improved germline transmission of transgenes in chicken, quail and zebra finch using lentiviral-based vectors.36–39 Recently, adenoviruses were used to deliver a Cas9 transgene and a guide RNA directly to the quail blastoderm.40,41 Initially, authors targeted the quail melanophilin gene, resulting in grey plumage of homozygous GE quail offspring produced from the founder birds, whereas heterozygous and wild type quail exhibited dark brown plumage. 40 In 2020, the same laboratory targeted the quail myostatin gene, generated germline chimera and offspring. Homozygous myostatin edited quail showed significantly enhanced body weight and muscle mass. 41 In spite of the success of virus-mediated GE in quails and the feasibility of applying this delivery method of genome editors to other avian species, the efficiency of this virus-mediated method is low because of the low efficacy of transducing the blastodermal primordial germ cells after viral injection into the subgerminal cavity. In addition, increasing the size of lentiviral vectors to introduce Cas9 and the RNA guide leads to inefficient viral packaging that results in a low viral titer and reduced transduction efficacy. Similarly, windowing avian eggs for injection of virus followed by the subsequent hatching of chicks, will have varying survivability in different bird species.
Embryonic vascular system injection
A method for producing both genetically modified chicken and quail is by the direct injection of Tol2 transposon vectors into the vascular system of young embryos to target the germ cells during their migration through the circulation (Figure 2(c)).42,43 Transposons are self-integrating DNA vectors that can carry a transgenic cargo within the vector. Serralbo et al. succeeded in producing many fluorescent reporter lines of transgenic quail using this technique.
44
A limitation of this technique is that the transmission rate (frequency of producing GE offspring) is
In vitro propagated avian PGCs
As stated above, germline-competent ES cells have not been isolated in avian species, which can be attributed to the early segregation of the germ cell lineage from the somatic cell lineage in birds. As is true for all vertebrate species, PGCs of birds are the progenitors or precursor cells of the sperm and oocytes. In freshly laid chicken eggs, as mentioned above, there are approximately 50 PGCs located within the centre of the blastoderm which contains approximately 40,000–60,000 cells. 46 Subsequently, PGCs migrate to the anterior germinal crescent from where 100–200 PGCs enter the embryonic circulatory system at 48–60 h of incubation before migration to the forming gonad. PGCs will undergo sexual differentiation into spermatogonial stem cells in the male, producing spermatozoa. In females, PGCs undergo meiosis and will form mature oocytes upon sexual maturity. 47 In striking contrast to mammals, PGCs from chicken embryos can be isolated and cultured indefinitely, while keeping their commitment to the germline and germline competency. 48 Chicken PGCs are extracted from the vascular system at early developmental stages and placed in culture. 49 A defined serum-free culture medium for propagating chicken PGCs has been developed by optimising the signalling pathways necessary for avian germ cell self-renewal. 50
The long-term culture of PGCs does not compromise their ability to colonise the gonad when injected into the vascular system of surrogate host embryos and form functional gametes and offspring when the surrogate hosts are subsequently bred. This has been a turning point for the generation of GE chicken models through GE of culture PGCs (Figure 2(d) and (e)). 51 Numerous reports based on using PGCs for the generation of GE chickens have been published including homologous recombination, 52 TALENs,53,54 and CRISPR/Cas9 system.55,56 Among the genome editors, CRISPR/Cas9 has been successfully used in PGCs in developing functional genetic models by generating immunoglobulin heavy chain transgenic chicken, 55 ovomucoid KO chicken 56 and myostatin KO chicken. 57
Using cultured PGCs, a demonstration of GE for avian disease resistance has been achieved. Acidic nuclear phosphoprotein 32 family member A (ANP32A) was recently identified as a cellular host protein in birds required for avian influenza virus (IAV) polymerase adaptation and activity.
58
Long et al. used CRISPR/Cas9 to knock out the ANP32A in chicken PGCs, demonstrated that fibroblasts derived from GE PGCs did not support either mammalian or avian influenza virus polymerase activity during
Sperm mediated GE
Cooper et al. demonstrated that spermatozoa can be directly transfected and targeted with CRISPR/Cas9 GE tools. 62 Subsequently, the GE sperm can be used for artificial insemination. This process is called sperm assisted gene editing (STAGE) and has been used to make GE chickens in a single generation (Figure 2(b)). However, the drawbacks of this technique are the genetic deletions in the offspring did not closely correspond to the cleavage site of the CRISPR/Cas9 in the genome and the rate of genetic modification in the offspring from the founders was from 0–26%. 62
The above results show that genetic modification of chicken and quail is highly advanced. A comprehensive list of genetically modified chicken and quail available in different research laboratories are reported in other reviews.44,63
Use of avian sterile hosts to address the principles of the 3Rs
A key challenge for GE of bird species is to efficiently produce GE offspring from the mosaic founder or surrogate hosts without breeding hundreds of non-GE offspring. Until now, the most successful method of generating genome edited chickens is by injecting exogenous edited PGCs intravenously into stage 15–16+ HH (Hamburger Hamilton) surrogate embryo in windowed eggs. However, the edited donor PGCs and endogenous PGCs compete with each other to form functional gametes. Thus, there is a greater opportunity for the endogenous PGCs to transmit its genome to the subsequent generation of offspring than the donor PGCs. To expedite the germline transmission from the donor edited PGCs, endogenous PGCs can be eliminated by busulfan and γ-irradiation, both of which can cause death and health problems for the treated host bird.65,66 However, GE can be used to create gene knockouts to eliminate the competition from endogenous PGCs by rendering the host bird devoid of sperm or eggs. Two sterile chicken host models have been produced: a TALEN-mediated knockout of the germ cell determinant,
The future for GE in other avian species
The injection of GE reagents directly into the bloodstream of embryonated eggs through a window made in the egg shell can theoretically be applied to all bird species. However, this technique is inefficient for the generation of transgenic and GE offspring (0.3–1%) and the hatch rate of the windowed injected eggs will differ drastically between bird species.68,69 Transmission rates of the genetic modification using this technique are currently low in chicken and multiple generations will be needed to generate pure offspring containing a homozygous GE allele. These methods will prove more difficult when applied to other avian model species such as zebra finch, ducks and turkeys. It would be extremely beneficial to be able to propagate the PGCs
Conclusions
The chicken has been a mainstay of vertebrate embryology research for many decades because their embryos come conveniently packaged in eggs and offer easy accessibility of developmental stages for experimental manipulations. 11 Many fundamental questions yet have to answer in developmental biology around neural development, organogenesis and patterning of the embryo. The advancement of the GE tools in chicken in combination with live-cell imaging and single-cell transcriptomics in both embryos and adult chicken has the potential of exploring these pertinent questions. Transferring these tools to other bird species would facilitate research efforts in many laboratory settings.
The chicken is an invaluable model for studying basic immunology and provides seminal contributions to fundamental immunological principles such as graft-versus-host responses and the key role of lymphocytes in adaptive immunity. 76 Infectious disease outbreaks in poultry is a persistent threat to the poultry sector as well as concerns of zoonotic transmission. GE tools can help to investigate disease-resistance in birds and also heat tolerance to combat climate impact on poultry production. In summary, the ongoing improvements in genome modification tools with avian embryo manipulation and PGC culture continues to refine the production of GE avian species with positive outputs on the 3R principles.
Footnotes
Author contributions
Conceptualisation, SKP and MJM; writing – original draft preparation, SKP; writing – review and editing, SKP and MJM; visualisation, SKP; supervision, MJM; funding acquisition, MJM.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
