Abstract
The aim of this study was to compare the cardiorespiratory, arterial blood gas and antinociceptive effects of dexmedetomidine (D), dexmedetomidine–lidocaine (DL) or lidocaine (L) administered epidurally on conscious rabbits. Eight six-month-old male New Zealand rabbits were randomly distributed into three treatments: D (2.5 μg/kg); DL (2.5 μg/kg; 2 mg/kg); and L (2 mg/kg). The drugs were injected epidurally via a catheter. Cardiorespiratory, arterial blood gas and antinociceptive variables were recorded before administration, 5 and 10 min after drug administration, then every 10 min until the animals presented a positive response to nociceptive stimulation of perineal dermatomes. Two animals had permanent paralysis after DL treatment due to hemorrhage and congestion with neuron necrosis in spinal cord segments. There was a reduction in mean arterial pressure in treatment L at 5 and 10 min, compared with the baseline, and in treatment DL at 10–30 min. Increases in pH were observed in treatment D at 5 and 10 min, and in DL at all the times evaluated, compared with the baseline. No alterations were observed in other blood gas or electrolyte variables. Antinociceptive effects were evaluated in the perineal, sacral and lumbar regions, and were restricted to the perineal region following D and L treatment. The antinociceptive effects following DL were greater than D and L alone in all of the regions. L and D promotes short-term antinociceptive effects for up to 15 min and, when used in combination with D, increased the duration and extent of sensory block by up to 45 min.
Introduction
Epidural anesthesia is a regional anesthetic technique commonly performed in veterinary patients. 1 It has the primary aims of improving intra and postoperative analgesia and reducing the consumption of inhaled anesthetics, thereby consequently providing better hemodynamic stability. 1 This technique is conducted routinely using local anesthetics, such as lidocaine (L).
Dexmedetomidine (D) is an alpha 2 (α2)-agonist with high selectivity for α2-adrenoreceptors (α2:α1 ratio 1620:1). In humans, there is a wide range of uses for α2-agonists in epidural anesthesia, with the main advantages of extending sensory blockade, prolonging the duration of anesthesia and decreasing systemic absorption of local anesthetic. 2 In dogs, it promotes important analgesic and sedative effects with low cardiovascular depression compared with other a2-agonists. 3 In dogs and humans, epidural administration of D decreases the minimal alveolar concentration of isoflurane, 4 and in combination with local anesthetics, it reduces the consumption of analgesics during the postoperative period 2 and promotes better recovery from general anesthesia. 5 However, data on epidural administration of D for analgesia or antinociceptive effects among rabbits are scarce.
Therefore, the goal of this study was to comparatively analyze the cardiorespiratory, blood gases, electrolytes and antinociceptive effects of D, used alone or in combination with L, by the epidural injection in conscious rabbits. Our hypothesis was that the dexmedetomidine–lidocaine (DL) combination would promote better and longer-lasting antinociceptive effects than either drug used alone, with minimal cardiorespiratory and arterial blood gas changes.
Materials and methods
This research was conducted under the approval of the Ethics Committee on Animal Use of the University of Franca (#8820030418). All procedures were conducted in compliance with the ethical principles of good practice of animal experimentation.
Animals
A total of eight six-month-old male New Zealand rabbits (from ANILAB – Animais de Laboratório Criação e Comércio Ltda, Paulínia, SP, Brazil), weighing 3.1 ± 0.1 kg, were selected for the study. The microbiological monitoring and status was performed according to Federation of European Laboratory Animal Science Associations recommendations. 6 All rabbits were free from ecto- (Sarcoptes scabei var. cuniculi) and endoparasites (Toxoplasma gondii and Eimeria spp.). They received commercial 4-mm pelleted feed (Presence Nutrição Animal, São Paulo, SP, Brazil) specific for this species, along with Tifton-grass hay and mains drinking water, untreated, ad libitum. The animals were housed in the University of Franca vivarium in individual galvanized wire cages (80 × 50 × 30 cm), and were maintained in an open system. Room temperature (22 ± 2°C) and humidity (63 ± 3%) were regulated. The temperature, humidity and husbandry during the experiment were the same. The artificial light (fluorescent lighting of 300 Lux) and dark cycle was 12/12 hours. The airflow (40 m3 per hour) in the room was constant, using an exhaust system. The animals were conditioned for 30 days, which consisted of transferring them to the place of the experiment, slight physical restraint and keeping them in the anesthesia induction box for 30 min. Animal health was assessed via laboratory tests (blood cell count, kidney and liver biochemistry function) and clinical evaluation. The experiment was performed between 09:00 a.m. and 05:00 p.m. using the methods described in the following.
Experimental design
The animals and treatments were randomly distributed by means of a draw, with a minimum one-week interval between treatments, in a prospective crossover study. The treatments were designated D (2.5 μg/kg; Dexdomitor, Zoetis do Brazil), DL (D, 2.5 μg/kg plus L, 2 mg/kg; Xylestesin, Cristália Indústria de Produtos Químicos e Farmacológicos Ltda) and L (2 mg/kg).
Epidural catheter implantation
For anesthesia induction, each rabbit was placed in a plastic box (area 127 cm2) in which isoflurane Inspired fraction (Fi): 3.5%; Isoforine, Cristália) in 100% oxygen delivery at 4 L/min. After absence of the palpebral reflex had been verified, the rabbits were removed from the box and placed in sternal decubitus. The lumbosacral region was clipped and the skin was prepared aseptically using alcoholic chlorhexidine (2 and 70%, respectively). The skin area around the puncture site was desensitized using local anesthetic, consisting of 0.5 mL of L, 5 min before the puncture (Xylestesin, Cristália). Subsequently, this area was covered with a fenestrated drape and a Tuohy needle (18G × 3.5″ – BD, Becton Dickinson Indústrias Cirúrgicas S.A.) was introduced at an angle of approximately 30°. This needle was a guide for introduction of an epidural catheter (20G × 36″ – BD), which was placed 5 cm cranially into epidural space. During the entire procedure, the animals received isoflurane via a facemask (flow: 2 L per minute and Isoflurane Fi: 1.5%). The correct catheter position was confirmed by filling the catheter with 0.16 mL iodinated contrast (Iopamiron 300, Patheon Italy) and, subsequently, radiography. The epidural injection of drugs performed using this catheter took 1 min. The final volume of the drugs was 0.2 mL/kg, and this was adjusted whenever necessary using a 0.9% sodium chloride (NaCl) solution. The isoflurane was discontinued and the rabbit allowed recovery from anesthesia before epidural injection.
The cardiopulmonary variables, blood gas and electrolytes were monitored and data were evaluated at the following times: zero or baseline (animals completely recovered from anesthesia, with catheter insertion and before drugs injection); 5 min after administration of drugs; 10 min after drug administration; and every 10 min thereafter until recovery of sensitivity of dermatomes in the perineal region. Antinociception was tested at the same time using a hypodermic needle. The evaluators were blinded to the protocol used. The data collection was performed in the order described below.
Cardiopulmonary variables
Heart rate (HR) was evaluated via transthoracic auscultation of cardiac cycles using a stethoscope in the region of the fourth left intercostal space for 1 min. The mean arterial pressure (MAP) was evaluated using a percutaneous catheter (22GX1" – BD) that was inserted into median auricular artery (right or left). This was connected to a non-compliant tubing filled with heparin solution (50 UI/mL) and was connected to the pressure transducer system of a multiparameter monitor (DX2022 – Dixtal Biomédica Indústria e Comércio Ltda). The transducer was leveled at heart height (xiphoid process). The transducer was calibrated using a mercury column, prior to the evaluation. Respiratory rate (fR) was assessed by observing the movements of the thorax for 1 min. Peripheral hemoglobin oxygen saturation (O2SP) was evaluated using the pulse oximeter of the multiparameter monitor, with a sensor positioned in glabrous, colorless skin of the ear or lip.
Blood gases and electrolytes analyses
Before the experiment started, the hair over the right or left auricular artery region was clipped and the skin was prepared aseptically using alcoholic chlorhexidine. The skin area around the puncture site in the ear was desensitized using anesthetic cream, 14 min before the puncture (EMLA Cream, AstraZeneca do Brazil Ltda). Arterial blood samples were collected to obtain values for pH, partial pressure of carbon dioxide (pCO2), partial pressure of oxygen (pO2), arterial oxygen saturation (O2sat), bicarbonate (HCO3–), base excess (BE), sodium (Na+), potassium (K+), chloride (Cl–) and ionized calcium (iCa2+). Arterial blood collection was conducted via the same catheter for direct arterial pressure using the three-syringe technique. The blood volume of each sample was 0.3 mL, and these samples were collected using a disposable syringe containing heparin. The needles were sealed with rubber bungs, thus avoiding contact between the blood and the air of the environment. The samples were immediately processed using a pH and blood gas analyzer, with correction for body temperature (Cobas b 121 Roche).
Antinociceptive evaluation
The antinociceptive evaluation was conducted by stimulating dermatomes located in the perineal, sacral and lumbar regions using the pinprick method with hypodermic needles (25 × 8 mm). The stimulation was started in the perineal region, followed by sacral and lumbar in areas pre-defined each region and points (Figure 1). Presence of tremors, limb movements or attempts to change position were considered to be positive responses, while absence of these manifestations was considered to be a negative response. When the animals presented a positive response, both stimulation and data collection were stopped.
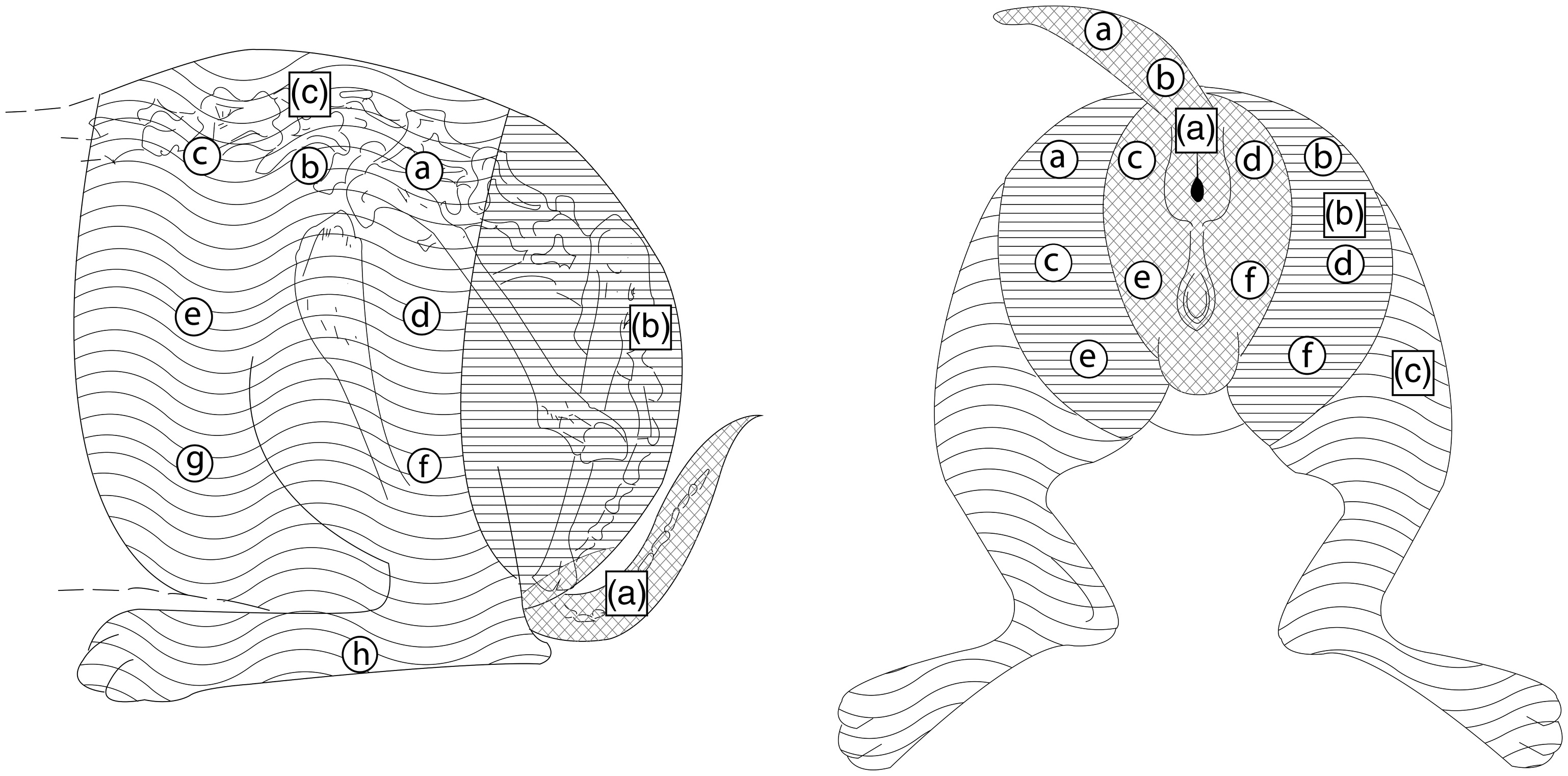
Schematic representation of the anatomical areas subjected to pinprick stimulation: perineal (a), sacral (b) and lumbar (c) areas. Letters a–h each represent one point of nociceptive stimulation, which was performed in the alphabetical sequence.
Statistical analysis
The sample power calculations were performed using HR and MAP in the pilot study; n = 8 was enough to generate power analyses of 80% (
Results
The mean time (in minutes) to place epidural catheter was 43.2 (SD = 7.6) in D treatment, 44.6 (SD = 6.1) in DL treatment and 45.6 (SD = 8.9) in L treatment. There were no significant differences among the treatments (p > .05). The animals did not present disease signs during the experiment.
Cardiopulmonary variables
To standardize the data collection with antinociceptive effects, the cardiopulmonary variables, blood gases and electrolytes parameters are presented until the times of 10, 70 and 30 min of evaluation, for treatments D, DL and L, respectively.
A reduction in MAP was observed in treatment DL, compared with the baseline, at 5 (p < .05), 10, 20 and 30 min (p < .01). In treatment L, the MAP mean values at 5 min (p < .01) and 10 min (p < .05) were lower than those at the baseline (p < .05). There were no differences in MAP following treatment D (p > .05) (Table 1).
Mean ± standard deviation of heart rate (HR), mean arterial pressure (MAP), respiratory rate (fR) and peripheral hemoglobin oxygen saturation (O2SP) in conscious rabbits (n = 6) submitted to epidural anesthesia with dexmedetomidine (D, 2.5 μg/kg), dexmedetomidine plus lidocaine (DL, D + 2 mg/kg) or lidocaine (L, 2 mg/kg) at different time points.
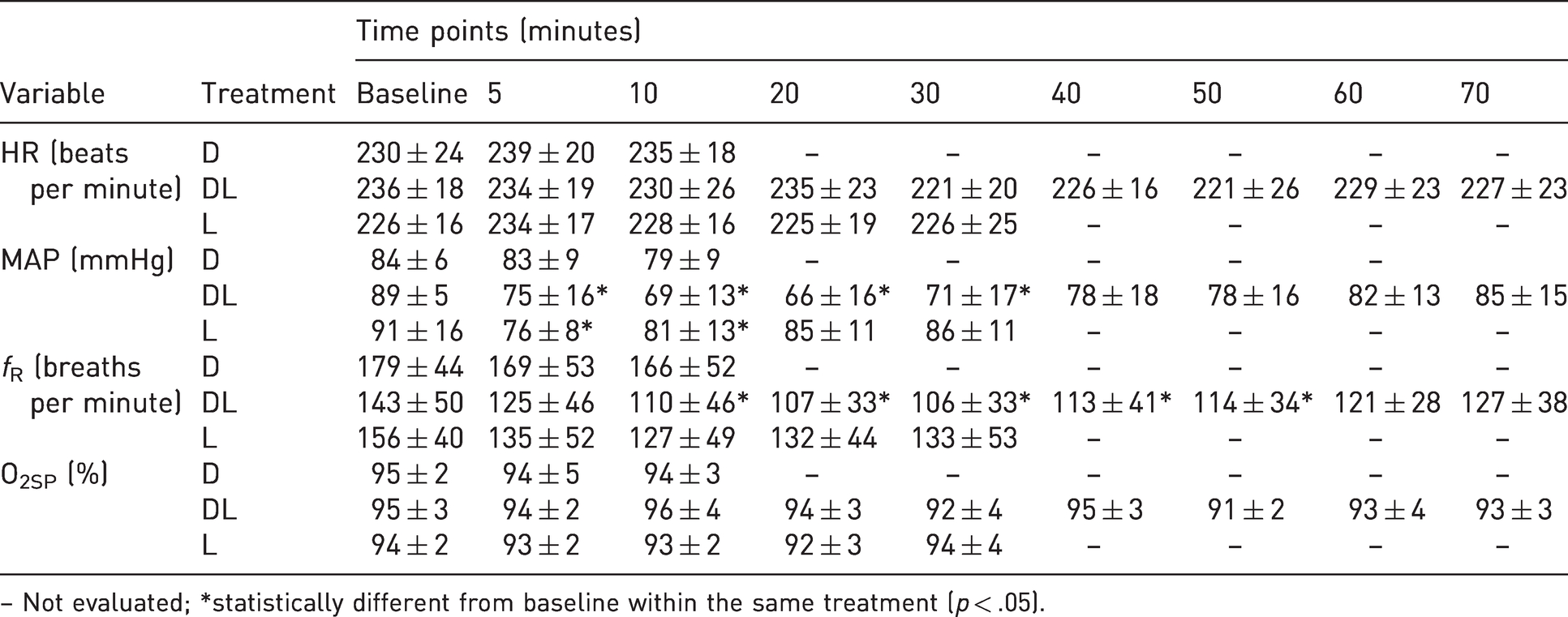
– Not evaluated; *statistically different from baseline within the same treatment (p < .05).
The respiratory rate values were lower at 10–30 min (p < .01) and 40–50 min (p < .05) compared with baseline in the treatment DL (Table 1).
No significant alterations were observed between treatments for any of cardiopulmonary variables (p > .05).
Blood gases analysis and electrolytes
Increases in pH were observed in treatment D (p < .05) at 5 and 10 min, and in treatment DL at all the times evaluated (p < .01), compared with the baseline. No significant alterations were observed in the comparative analysis among treatments (Table 2).
Mean ± standard deviation of pH, arterial partial pressure of carbon dioxide (pCO2), arterial partial pressure of oxygen (pO2), arterial oxygen saturation (O2SAT), bicarbonate (HCO3–), base excess (BE) in conscious rabbits (n = 6) submitted to epidural anesthesia with dexmedetomidine (D, 2.5 µg/kg), dexmedetomidine plus lidocaine (DL, D + 2 mg/kg) or lidocaine (L, 2 mg/kg) at different time points.
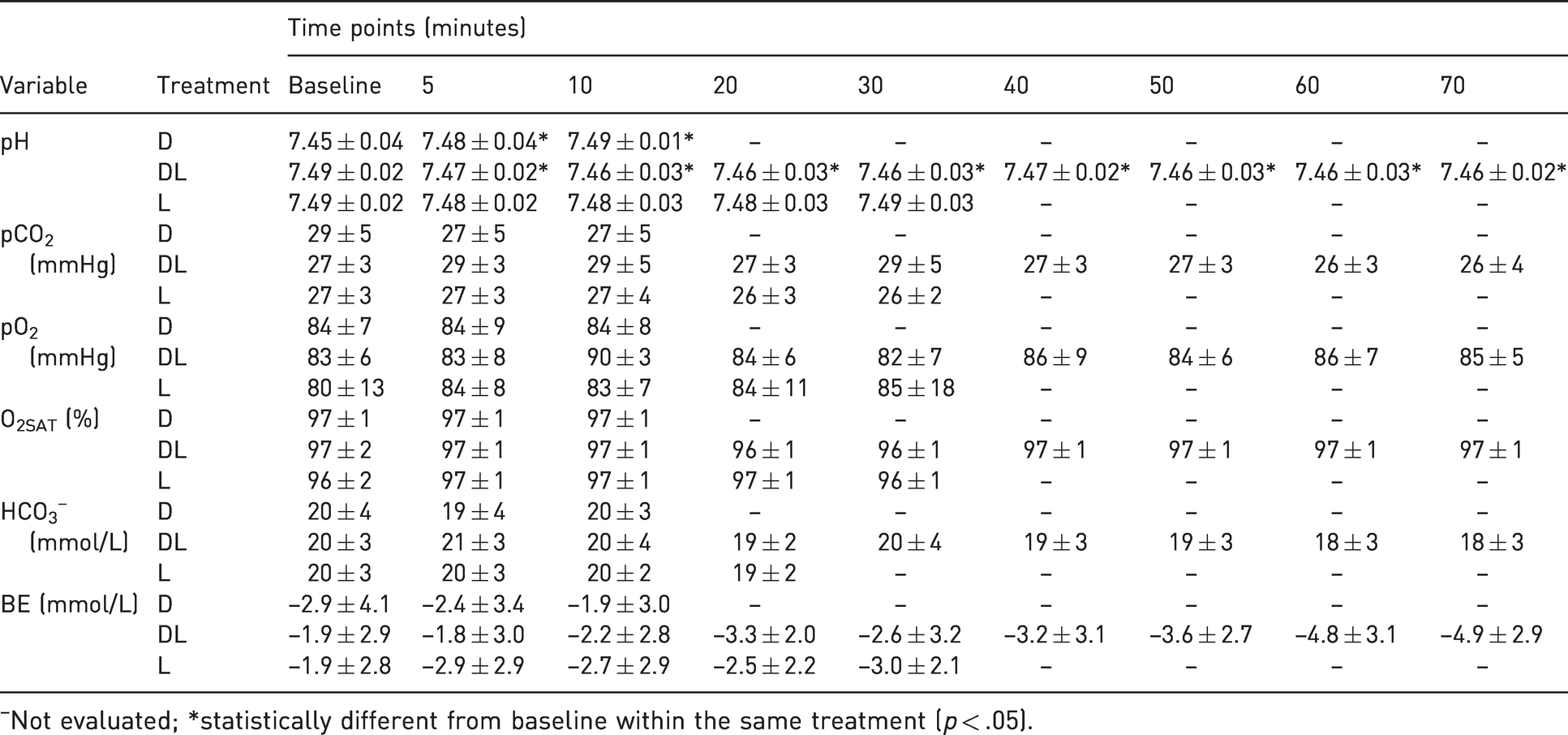
Not evaluated; *statistically different from baseline within the same treatment (p < .05).
No differences in serum concentrations of the Na+, Cl–, K+ and iCa2+ ions were observed, in comparison with the baseline and among the treatments (p > .05) (Table 3).
Mean ± standard deviation of sodium (Na+), potassium (K+), chloride (Cl–) and ionized calcium (iCa2+) in conscious rabbits (n = 6) submitted to epidural anesthesia with dexmedetomidine (D, 2.5 μg/kg), dexmedetomidine plus lidocaine (DL, D + 2 mg/kg) or lidocaine (L, 2 mg/kg) at different time points.
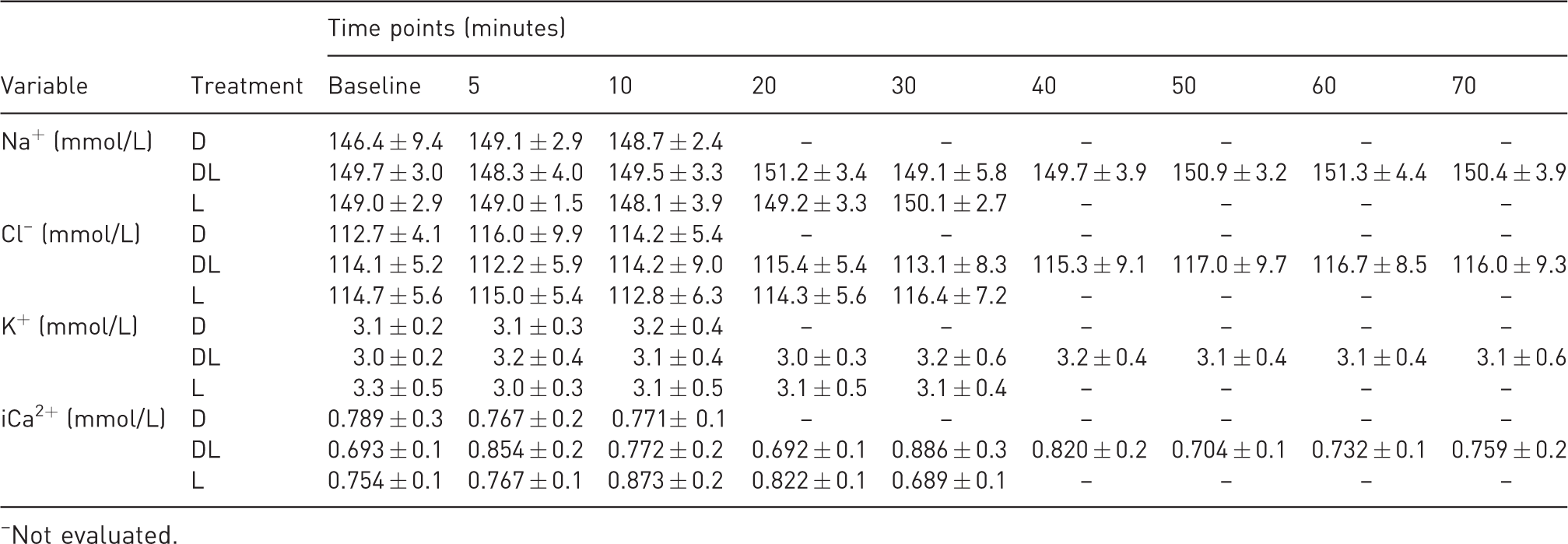
Not evaluated.
Antinociceptive effects
Two animals were excluded from the study because they presented permanent paralysis of the pelvic limbs: one during the second treatment and the other during the third treatment; both were in the DL treatment group. These rabbits underwent euthanasia and necropsy. In animal 1, macroscopic description of the spinal cord of lumbar segment (L1–L3) presenting a greenish color with areas of malacia; subdural space presenting clots. Microscopic description of the spinal cord: 1) cervical segment: slight hemorrhage and congestion with moderate Wallerian degeneration, neuron necrosis and gitter cells in the spinal canal. 2) Thoracic segment: moderate Wallerian degeneration; gitter cells in the spinal canal and slight lymphoplasmacytic infiltrate. 3) Lumbar segment: moderate Wallerian degeneration and infiltrate of degenerated heterophils in gray matter and meninges. Moderate liquefactive necrosis, and hyperemia. Slight hemorrhage in white matter with prominent infiltrate of gitter cells. In animal 2: macroscopic description of the spinal cord: nothing worth noting. Microscopic description of the spinal cord: (a) cervical segment: nothing worth noting. (b) Thoracic segment: multifocal areas of hemorrhage and Wallerian degeneration. (c) Lumbar segment: hemorrhage area close to the central canal, associated with malacia and small number of gitter cells in the L7 region. (d) Sacral segment: slight lymphoplasmacytic infiltrate.
In treatment D, the antinociceptive effects were restricted to the perineal region, and were not significantly different from treatment L (p < .05). The antinociceptive effects observed in treatment DL were longer than those in treatments D and L, in the perineal, sacral and lumbar regions (p < .05) (Table 4).
Mean (in minutes) ± standard deviation of antinociceptive effects in the perineal, sacral or lumbar dermatomes in conscious rabbits (n = 6) submitted to epidural anesthesia with dexmedetomidine (D, 2.5 μg/kg), dexmedetomidine plus lidocaine (DL, D + 2 mg/kg) or lidocaine (L, 2 mg/kg).
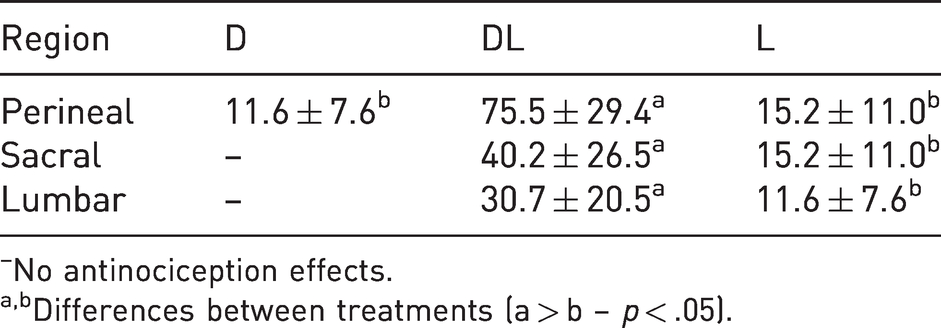
No antinociception effects.
Differences between treatments (a > b – p < .05).
Discussion
The results from the present study showed that a 2 mg/kg dose of L promoted a short period of antinociceptive effect, while D in combination with L increased the duration of antinociceptive time and the extent of blocking, but without promoting any cardiorespiratory depression compared to L or D alone.
For the domestic species, the dose of L for epidural administration ranged from 3.8 to 6 mg/kg.1,7 However, in domestic rabbits the recommended dose is 2 mg/kg, 8 with a final volume of up to 0.2 mL/kg. 9 In our study, the rabbits treated with L alone presented short analgesic effects, similar to effects previously related.10,11 Tavakoli et al. 10 observed a short duration of analgesia (mean of 23.6 min) among rabbits that were treated with epidural L at doses ≥9 mg/kg, administered epidurally. Doherty et al. 11 also observed motor blockage <29 min using the same species, route and drug. These findings may be related to the rapid absorption of L to the systemic circulation, since significant concentrations are detected in plasma, 5 min after epidural administration. 11
In dogs, the recommended dose for D administered intramuscularly, with antinociceptive effects, is 10 μg/kg. 12 However, if administered epidurally, it is approximately five times more potent. 13 Therefore, we chose to use a dose of 2.5 μg/kg, based on a pilot study and on our practical experience.
In conscious dogs, the cardiovascular depressive effects of D are dose-dependent, 3 usually causing reduction in HR or even bradycardia, with maintenance of MAP values. 12 In the present study, no cardiovascular alterations with clinical significance were observed, and the reduction in MAP among the animals treated with DL or L was less than 15%, in relation to the baseline values. These findings could be due to the low doses of D, together with its slow distribution into the systemic circulation.
The interpretation of alterations to blood gases values in rabbits presents limitations because these values present large oscillations in conscious animals. 14 In the present study, alterations were only observed in relation to pH in treatments D and DL, and these did not have any clinical significance. The animals evaluated in this study were conditioned to the environment and to the researchers involved. However, from analysis on the baseline values of respiratory rate, flaws in this conditioning may have been present, which could have influenced the blood gas results observed.
There are descriptions of various analgesic doses of D administered epidurally. In humans, this drug used in combination with several local anesthetics at doses ranging from 0.5 to 1.5 μg/kg has been shown to have an important analgesic effect. 2 Campagnol et al. 4 administered three different doses of D epidurally in dogs (1.5, 3 and 6 μg/kg), and reported that there was a significant dose-dependent reduction in the minimal alveolar concentration of isoflurane. Souza et al. 15 observed that female cats that underwent ovariosalpingohysterectomy under anesthesia with isoflurane and epidural L plus D (4 μg/kg) presented noticeably lower consumption of volatile anesthetic and improvement in the quality of recovery. However, Pohl et al. 16 observed that the analgesic effects of D (2 μg/kg) plus L administered epidurally in bitches that underwent ovariosalpingohysterectomy do not promote sufficient analgesia for surgical procedure requiring rescue analgesia.
Based on previous studies,4,15 and in our experience, we are hypothesizing that D could potentiate the antinociceptive effects of epidural L. Three mechanisms for the analgesic effects of D have been proposed. The first of these relates to specific activation of α2-adrenoreceptors in the dorsal horn of the spinal cord through inhibition of reception of excitatory neurotransmitters in the presynaptic neurons of afferent nociceptive fibers. 17 The second of these involves specific blocking of nerve fibers and neuron hyperpolarization. 18 The third and most plausible mechanism consists of local vasoconstriction through binding of D to α2A subtypes of the adrenoreceptors, thus hampering its absorption into the systemic circulation, improving its analgesic effects and increasing the effects of local anesthetics. 19
D promotes an important sedative effect in dogs. In this regard, there might be misinterpretation between sedative and antinociceptive effects. 12 In the present study, the rabbits showed no signals of sedation (no behavioral changes). Although it is unlikely that the antinociceptive effects observed could have been mistaken for sedation, this possibility cannot be ruled out, given that D used alone also showed antinociceptive effects.
One of the limitations of the present study concerns the methodology that was used to evaluate the antinociceptive effects. In order to evaluate these effects, the stimulation needs to be constant in intensity and time, thereby limiting the pinprick method. 19 In addition, repeated perforation of the skin may lead to local hypersensitivity, thus compromising the interpretation of the results. Despite the potential limitations, the needle prick technique was chosen as being one that is easily performed and relatively reproducible in other studies.20,21
The results from the present study showed acute spinal injury in 25% of the rabbits treated with D in combination with L. A possible explanation for this injury can be associated with the commercial presentation of the D. Cardoso et al. 22 observed that a toxic effect was present after epidural administration of a dose of D (10 μg/kg) in rabbits, and that important alteration to the meninges occurred, with adherences and lymphoplasmacytic infiltration in the pia mater and arachnoid mater, and in their blood vessels. These authors highlighted that induction of these lesions may be a factor that limits administration of this substance epidurally. No other studies have reported any alterations in other species.4,13,15 However, the formulations used in these studies were different (Precedex, Hospira). The product used by these authors was preservative-free, and a 0.9% NaCl solution was added to control pH (4.0–7.5).
D used in this study (Dexdormitor) is a commercialized product that contains two preservatives derived from methylparaben (methyl parahydroxybenzoate and propyl parahydroxybenzoate) and presents pH levels ranging from 4.0 to 6.0. Gürün et al. 23 evaluated the effects of intrathecal administration of methylparaben in conscious rats and sheep, and did not observe any clinical or histological signs in the spinal cord that might indicate toxicity. Although neurotoxicity was not investigated in the present study, we cannot discharge a possibility that medullar toxicity induced by preservatives caused the spinal injury observed. Odette and Smith 5 administered D epidurally, in the same formulation as in the present study, in combination with bupivacaine, in 20 dogs that underwent orthopedic surgery, and did not observe any acute or chronic toxicity. The difference in the pH of D in relation to the spinal canal was associated with local vasoconstriction and induction of toxicity. 24 On the other hand, Cardoso et al. 22 attributed these effects to the meningeal reaction to the drug itself.
The necropsy findings from one of the animals were compatible with the characteristics of acute inflammatory lesions, and this may have been related to the technique used. Rabbits present anatomical differences in relation to dogs and cats, with different numbers of lumbar vertebrae, including among individuals of the same species, such that their spinal cord extends as far as the second sacral vertebra. 25 To our knowledge, this is the first report of using epidural study in conscious rabbits; however, we cannot exclude the possibility that repeated puncture might have increased or even induced the spinal cord trauma observed.
In conclusion, the results from the present study suggest that when administered epidurally, L in combination with D promotes longer regional anesthesia, compared with either drug alone, without promoting cardiorespiratory depression. However, the occurrence of spinal cord injuries in two animals may represent a limitation on the use of this product via this administration route. Nonetheless, it can be suggested that new studies should be conducted to differentiate the possibility of injury according to the technique used and to verify the applicability of the protocol for conducting clinical procedures.
Footnotes
Acknowledgements
The authors thank Ana Márcia Zago, Rodrigo Aparecido de Souza and the Design Department of the University of Franca for their assistance in creating the figure, and Professor Daniel Kan Honsho for use of the experimental area.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was conducted during a scholarship (PROSUP/CAPES) supported by CAPES – Brazilian Federal Agency for Support and Evaluation of Graduate Education within the Ministry of Education of Brazil (Financial code – 001).
