Abstract
Effective multimodal analgesia techniques are required when pigs are used as models in orthopedic human research. Regional anesthesia is a widely used technique to provide perioperative analgesia in animals undergoing orthopedic surgery. The brachial plexus (BP) block is indicated to desensitize the forelimb in many species but has not been yet described in pigs. The main aims of this study were to develop an ultrasound-guided axillary approach for the BP and to evaluate injectate spread and nerve staining in pig cadavers. Eight fresh F1 cross breed German Large White and German Landrace male pig cadavers were enrolled. Two cadavers were used for anatomical dissection of the axillary space and to determine the disposition of the BP. Six cadavers were used to perform a bilateral axillary ultrasound-guided approach for the BP, and after injecting 0.3 ml/kg of a solution of 2% lidocaine and new methylene blue (L-NMB), these were dissected to determine injectate spread and nerve staining. Upon dissection, the BP was observed in all the cases surrounded by the axillary sheath and in close proximity to the axillary artery. Ultrasonographic scanning and guidance for the approach to the BP was feasible in all cadavers and upon dissection, all the nerves forming the BP were stained in all their quadrants and in all the cases. In conclusion, the injection of 0.3 ml/kg of L-NMB through an ultrasound-guided axillary approach to the BP was feasible and adequate to entirely stain the BP in all pig cadavers. Further clinical studies are required to evaluate the effectiveness of this technique in live animals.
Introduction
Pigs are commonly used as models in numerous areas of human biomedical research. 1 , 2 They are considered optimal for translational investigations given the numerous pathophysiological similarities between species. 3 , 4 Experimental investigations related to orthopedic human research are commonly performed in pigs, 5 and in the face of major surgical procedures, effective multimodal analgesia regimens are required to maximize animal welfare. 6
Regional anesthesia is a widely used technique for effective perioperative pain management in animals, both in clinical and experimental settings. 7 It consists of the deposition of a local anesthetic solution around nerves to achieve desensitization of specific anatomic areas. The block of the brachial plexus (BP) is a commonly used technique in veterinary medicine to achieve motor, sensory and autonomic desensitization of the thoracic limb, specifically of the structures distal to the distal third of the humerus. 7 This technique has been shown to provide effective perioperative pain management in many animal species and for a variety of surgical procedures.8–12
Ultrasound-guidance is the state-of-the-art tool to perform regional anesthetic techniques. 13 It enables visualization of the relevant structures and of the distribution of injectate around the target, minimizing the risk of accidental intravascular injections. 14 However, the success of this method relies not only on highly specified technical skills for needle handling, but also on a thorough understanding of the anatomical structures involved. Several approaches for the BP block have been described in many species, but the ultrasound-guided technique through an axillary approach remains the mainstay for thoracic limb desensitization. 15 This technique, however, has not been yet described in pigs.
The aims of this study were (a) to describe the sonoanatomy of the axillary space, (b) to develop an ultrasound-guided axillary approach to the BP, and (c) to evaluate injectate spread and nerve staining after dye injection in pig cadavers.
Materials and methods
Animals
Eight fresh, 12-week old F1 cross breed German Large White and German Landrace male pig cadavers with a median weight of 38 kg (range 29–39 kg) and with no forelimb abnormalities were enrolled. All pigs were obtained from a non-related non-survival study which did not involve thoracoabdominal procedures and was approved by the ethics committee of the University of Veterinary Medicine Vienna; permit number: GZ68.205/0005V/3b/2018. The animals were housed and cared for on straw and in groups in such a way that the physiological and behavioral needs were adequately taken into account. Feeding took place ad libitum, clean drinking water was always available. Adequate activity material was also provided. Pigs were euthanized humanely by intravenous injection of T61 (Merck Sharp and Dohme, Kenilworth, NJ, USA) under general anesthesia. This study was designed to be carried out in two phases.
Phase I: Anatomical study
Two pig cadavers (37.5 and 39 kg) were studied. The specimens were positioned in dorsal recumbency and the left and right axillary regions were clipped. The skin of the axillary region was incised in a craniocaudal direction and reflected to expose the underlying structures. Then, the superficial and deep pectoralis muscles were cut and reflected, and a careful layer-by-layer dissection, including that of the axillary sheath, was performed in order to expose the BP and its related structures. The anatomical disposition of the nerves of the BP and its related structures was then determined.
Phase II: Axillary ultrasound-guided approach for the BP
A total of six fresh pig cadavers were included in this phase. Ultrasonographic scans of the right and left axillary spaces were performed immediately after euthanasia and with the cadavers placed in dorsal recumbency (Figure 1). The area of interest was clipped and cleaned. A 13-6 MHz linear transducer (HFL38×, Sonosite Inc., WA, USA) attached to an ultrasound machine (MicroMaxx, Sonosite Inc., WA, USA) was used. Ultrasound gel was applied to facilitate acoustic coupling. The ultrasound depth was set to 6 cm and gain was adjusted to optimize the image. The transducer was placed in the axillary fossa, between the manubrium and the ipsilateral scapulohumeral joint, with a parasagittal orientation. The orientation marker of the probe was positioned cranially. Then, the transducer was tilted laterally and medially, trying to trace the projection of axillar artery and the nerves that form the BP. The dimensions of the BP and the distance skin–BP were measured using the ultrasound caliper. A 100 mm 21 gauge insulated needle (Sonoplex Stim Cannula, Pajunk Medical Produkte GmbH, Germany) prefilled with a 1:1 solution of new methylene blue (MethyleneBlue1%w/vaq.soln, AlfaAesar, ThermoFischerGmbH, Germany) and lidocaine (Xylanest purum 2%, Gebro Pharm GmbH, Austria) (L-NMB) was inserted using an in-plane approach. The needle was advanced under ultrasound-guidance through the pectoralis muscles in a caudo-dorsal direction towards the BP. The needle was advanced until its tip punctured the axillary sheath, which covered the neurovascular bundle. After aspiration to discard possible intravascular location of the tip of the needle, a test volume of 1 ml of L-NMB was injected to confirm adequate distribution inside the axillary sheath and around the neurovascular bundle. The remaining volume of 0.3 ml/kg was then divided into two equal aliquots and each injected around the target nerves, cranial and caudal to the axillary artery, respectively. The procedure was repeated in the contralateral limb. The chosen volume was based on previous publications. 16 All cadavers were dissected immediately after injection, and the distribution pattern of injectate evaluated. The presence of L-NMB staining the nerves of the BP around its entire circumference for a length of more than one centimeter, was considered successful. 17 Additionally, presence of dye in the pleural cavity as well as vascular damage or intraneural staining were evaluated.
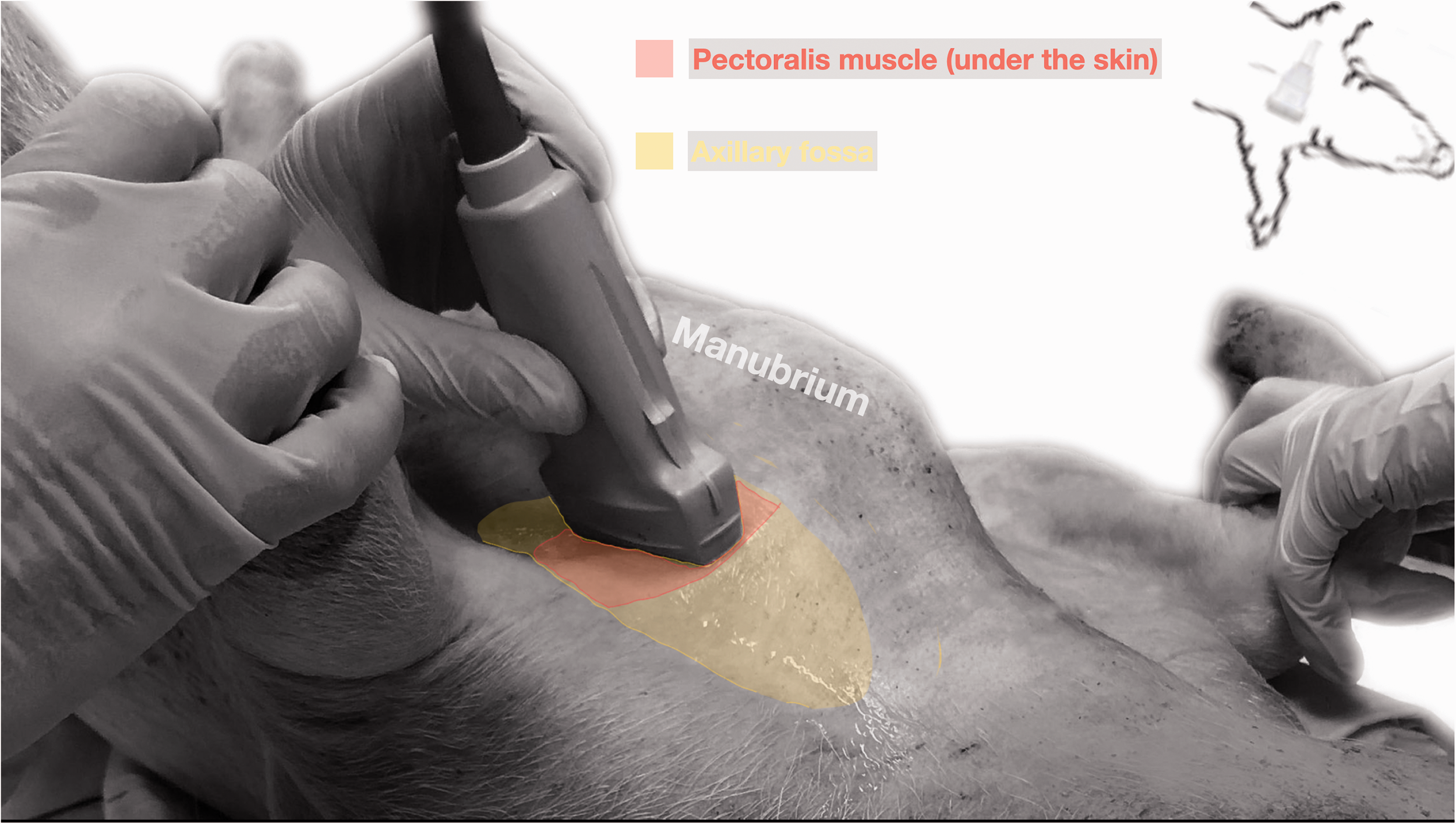
Pig cadaver positioned in dorsal recumbency for an ultrasound guided brachial plexus block using axillary approach.
Results
A total of 16 thoracic limbs belonging to eight pig cadavers were enrolled in this study.
Phase I: Anatomical study
In the four axillary spaces dissected from the two cadavers, the BP was observed originating from the ventral roots of the C5, C6, C7, C8 and T1 spinal nerves and emerging from the ventral border of the scalenus muscle. At the level of the axillary fossa, the nerves forming the BP were observed surrounded by the deep thick axillary sheath. After dissecting the medial aspect of the axillary sheath, a neurovascular bundle was observed composed of the BP and its related vascular structures. In all the cases, the nerves were observed in close proximity to the axillary artery (Figure 2). The BP was observed limited cranially by the brachiocephalicus muscle, medially by the serratus ventralis muscle, laterally by the subscapularis muscle, and ventrally by the pectoralis muscles. The median (range) width of the neurovascular bundle (BP + axillary artery and vein) was 3.7 (3.5–3.9) cm. The median (range) length of the nerves running in the axillary fossa was 5.1 (4.8–5.2) cm.
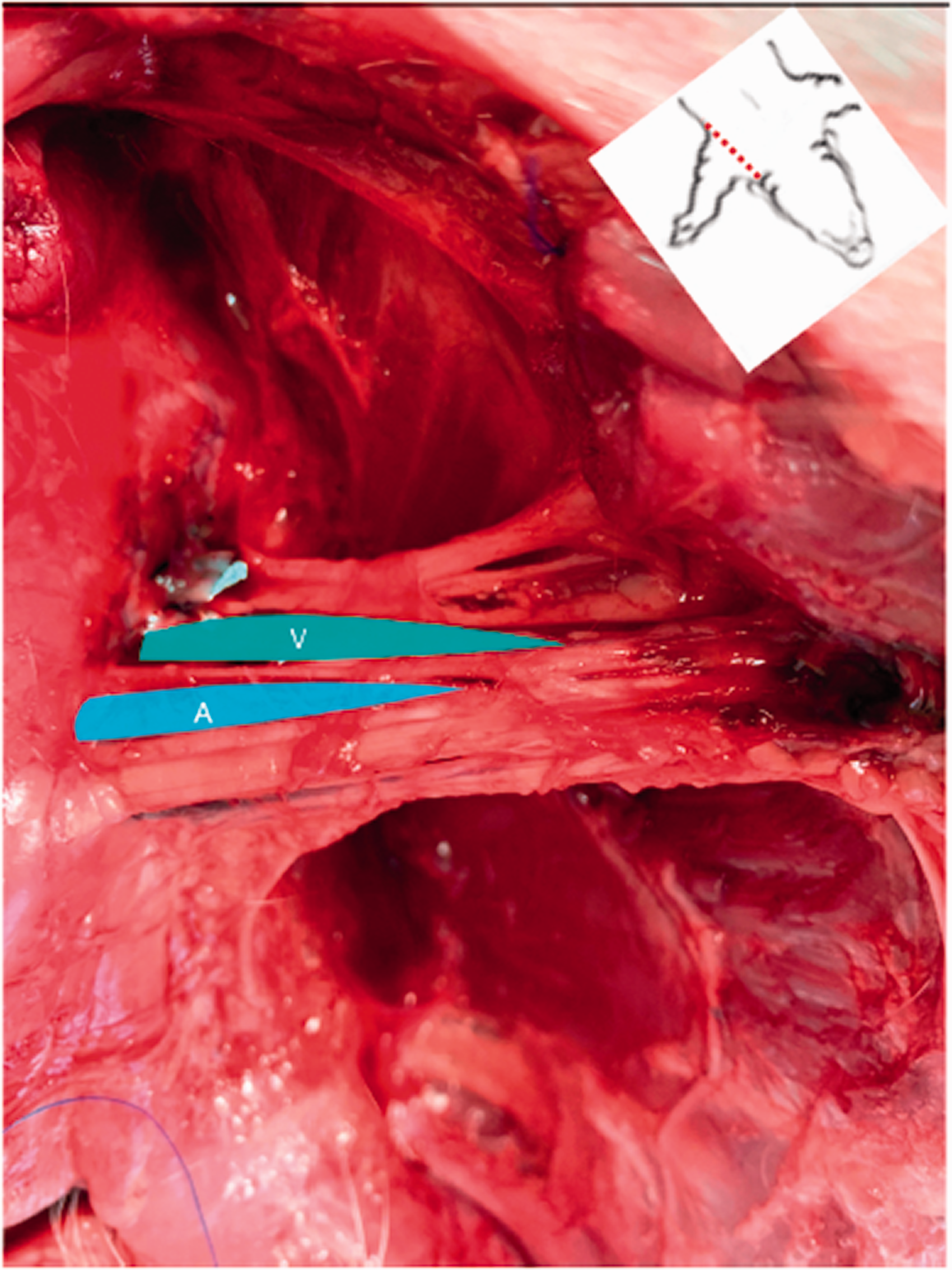
Dissection of the axillary space showing the axillary nerves and vessels. The pectoral muscles have been removed. A, axillary artery; V, axillary vein.
Phase II: Axillary ultrasound-guided approach for the BP
Ultrasound scanning of the right and left axillary spaces allowed identification of the BP in all the pig cadavers. The axillary artery could be identified in all cadavers as an anechoic round structure surrounded by the nerves of the BP. The axillary vein was identified by applying gentle pressure on the ultrasound probe, and observed caudal to the axillary artery. The axillary sheath was identified as a hyperechoic linear structure surrounding the neurovascular bundle. The target nerves appeared as anechoic or hypoechoic structures and were round in shape. The bellies of the superficial and deep pectoralis muscles could be identified ventrally to the neurovascular bundle (Figure 3). The median (range) cranio-caudal and dorso-ventral dimensions of the BP were 3.1 (2.9–3.3) cm and 2.4 (2.3–2.5) cm, respectively. The median (range) skin–BP distance was 4 (3.8–4.2) cm. In all the cases, adequate visualization of the needle’s shaft during advancement was obtained and the distribution of the injectate was observed within the neurovascular bundle in real-time. Upon dissection, all the nerves forming the BP were stained in all their quadrants and in all the cases. The median (range) craniocaudal and dorsoventral extension of spread was 8.4 (7.9–8.8) cm and 6.1 (5.8–6.2) cm, respectively. A small amount of dye was observed inside the pectoralis muscle in correspondence with the needle’s pathway. There was no evidence of intrapleural spread, vascular damage or intraneural staining in none of the cadavers.
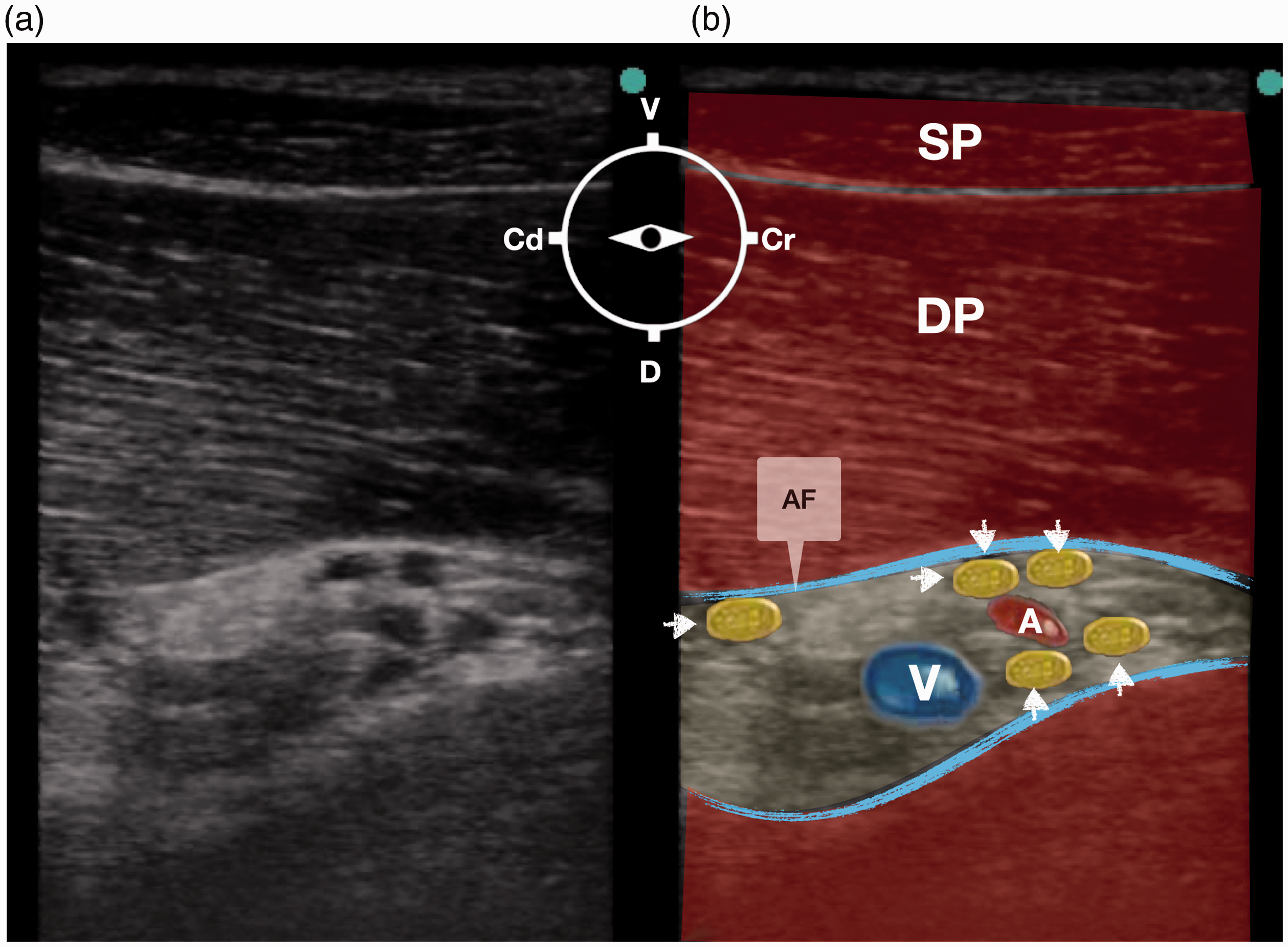
Ultrasound image showing the axillary space at a depth of 6 cm: (a) sonoanatomy and (b) revised ultrasound anatomy. A linear transducer was placed in a parasagittal position with a ventrodorsal orientation. Arrows indicates the brachial plexus; A, axillary artery; AF, axillary fascia; V, axillary vein; DP, deep pectoral muscle; SP, superficial pectoral muscle, Cr, cranial; Cd, caudal; D, dorsal, V, ventral.
Discussion
This study describes the anatomy of the axillary space and proposes an ultrasound-guided axillary approach to reach the BP in F1 cross breed German Large White and German Landrace male pig cadavers. The injection of 0.3 ml/kg of L-NMB solution was sufficient to stain all the nerves of the BP in all the studied thoracic limbs. Using this volume (0.3ml/kg) of lidocaine as a local anesthetic, the dose of lidocaine would remain in the clinical range (6 mg/kg). The positioning of the animals in dorsal recumbency, the parasagittal placement of the ultrasound probe in the axillary space and the in-plane advancement of the needle under ultrasound visualization appeared to be adequate to identify the BP and related structures and to perform the injection in pig cadavers.
The block of the BP through an axillary approach is indicated to desensitize the thoracic limb distal to the mid humerus and is used in a variety of painful conditions in many animal species. 9 , 12 , 15 All the nerves of the BP were observed in close proximity to the axillary artery and therefore, the sonographic identification of the latter could potentially be a useful reference for the execution of the block through an axillary approach. As demonstrated in this study, the injection of an adequate volume of the L-NMB solution in relation to the axillary artery seemed to be adequate to obtain maximal staining success rate, which is in agreement with studies performed in humans, 18 dogs 16 and cats. 19 Although the existence of internal septa between the components of the BP has been documented in many species 18 , 20 , 21 the results of this study showed no indication of their presence in the pigs enrolled.
It has been proposed that three nodes of Ranvier must be exposed to local anesthetic in sufficient concentration, corresponding to 3–4 mm, to block nerve impulse conduction. 17 In this study, a minimal length of 1 cm around the circumference of the nerve was considered a requirement to classify the nerve as stained. The injection of a volume of 0.3 ml/kg of L-NMB appeared to be sufficient to stain all the nerves of the BP in all the studied pigs. However, if the same volume is to be injected in live animals, care should be taken not to exceed the maximal dose of lidocaine for this species. Furthermore, the volume injected was slightly larger than that commonly employed in other species when performing BP injections 8 , 19 and promoted nerve exposure larger than one centimeter. Therefore, further anatomical and clinical studies are required to determine the minimal effective volume for performing a BP block through an axillary approach in pigs.
The block of the BP has been associated with several complications in animals such as vascular puncture, 19 , 22 intrathoracic puncture 23 and nerve damage. 24 This study was performed in cadavers and, therefore, their anatomy and lack of functionality may have not entirely reflected the conditions of the living, which could play a role in the occurrence of these adverse events. However, no signs of pleural, vascular or neural puncture were observed in the cadavers of this study. Further studies in live animals are warranted to determine the likelihood of these complications in pigs.
This study has several limitations. First, only pig cadavers ranging from 29–39 kg were included. Performing this approach in pigs with different body weights would be more representative of the porcine population. However, the weight range used is in agreement with the weight most commonly used in human translational studies. 5 Second, no comparison of injected volumes was performed and therefore, further cadaveric and clinical studies are required to determine the minimal effective volume to stain and block the BP. Finally, this was a cadaveric study and the evaluated spread in these specimens may have not entirely reflected spread of dye in the living.
In conclusion, an ultrasound-guided axillary approach to the BP allowed identification of the target nerves and related structures in F1 cross breed German Large White and German Landrace male pig cadavers. Needle handling towards the target was feasible and the injection of 0.3 ml/kg of L-NMB was adequate to entirely stain the BP in all pig cadavers. Further clinical studies are required to evaluate the effectiveness of this technique in live animals.
Footnotes
Acknowledgements
The authors would like to thank Priv.-Doz. Dr Ulrike Auer and Ms Anja Angerer for their assistance.
Declaration of conflicting of interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Data are available upon request (
Ethical statement
Pigs were humanely euthanized for reasons unrelated to this study, which did not involve thoracoabdominal procedures and was approved by the ethics committee of the University of Veterinary Medicine Vienna; permit number: GZ68.205/0005V/3b/2018.
Author contributions
RT conceived the experiment, performed all injections and dissections, analyzed data and contributed to writing of the manuscript; NV contributed to data analysis and to the writing of manuscript; IC data collection and interpretation, revised the manuscript; CK contributed to anatomical dissections and revised the manuscript; PEO contributed to the study design, data analysis and to the writing of manuscript.
