Abstract
Denture stomatitis is the most prevalent form of oral candidiasis and the most frequent oral lesion in removable prosthesis wearers. It is characterized by an inflammatory response of the denture-bearing mucosa, especially the palatal mucosa, and its clinical signs include chronic edema and erythema, and papillary hyperplasia. Despite having a multifactorial etiology, its main etiological agent is the infection by Candida albicans. Given its high treatment failure rates, an in vivo model of denture stomatitis should be established to test alternative treatments. The aim of this study is to review the existing denture stomatitis models and to provide an overview of the main methodological differences between them. Over the last 40 years, different animal models were developed in order to study denture stomatitis etiopathogenesis and to assess novel therapies. Many approaches, including the use of antibiotics and immunosuppressors, have to be further investigated in order to establish which protocol is more appropriate and effective for the development of an animal model of denture stomatitis.
Introduction
Denture stomatitis is a chronic inflammatory condition characterized by edema and erythema of the mucosa underlying removable dental prostheses. 1 Despite having a complex pathogenesis, infection by Candida albicans is often considered its main etiological factor. 2 To date, there is no gold standard in the management of denture stomatitis, 3 and the high recurrence rate makes the investigation of new treatments a must. 1 However, prior to clinical studies, it is crucial to guarantee that any alternative treatment is safe and effective. 4
Alternative methods, such as invertebrate models5,6 and non-animal models,7,8 are ethically acceptable, have a good correlation to animal models, and do not require specialized facilities and personnel for maintenance.9,10 However, these models also have limitations. For instance, the morphological transition between yeasts and hyphae by C. albicans occurs at 37°C, and many invertebrate models do not withstand this temperature. 9 Also, comparisons between models are often restricted to a limited number of strains. 9 Although these alternative models can be useful as a screening tool to assess the virulence of mutant strains 5 and to evaluate new antifungal drugs, 7 animal models are still considered the gold standard for most fungal infections5,9,10 and remain fundamental for the advance of medical mycology. 11
Animal models have been used in biomedical research to improve scientific knowledge, 12 allowing the understanding of disease etiology, pathophysiology and progression 13 in a more complex environment than in vitro studies. 14 Pathogenesis studies in animals also contribute to the development and assessment of new drugs and therapies to treat and prevent diseases and infections, 15 overcoming the limitations of clinical trials with human subjects. 16 The premise for using laboratory animals for disease models should be based on their biological characteristics, which should be comparable to that of humans. 17
At first, primates were thought to be the ideal animal model for the study of Candida-associated denture stomatitis. 18 Theoretically, monkeys would serve as a good animal model because of their oral microbiota being similar to that of humans, their ability to wear and tolerate acrylic plates in the oral cavity and their susceptibility to Candida-related infections.19,20 However, these studies were no longer continued due to their high cost, difficult maintenance and non-reliable findings. 18
Considering the aforementioned disadvantages of monkey models, researchers started to use rats to induce denture stomatitis in order to study its pathogenicity and develop new treatments. Rats are easy to handle, have a low maintenance cost and have a sufficiently large oral cavity. 18 However, the fabrication of devices resembling denture prostheses is still necessary, 21 mainly because the palatal epithelium is covered by a hard and densely cornified keratin layer, which prevents the initial retention of Candida cells,22,23 and the exhaustion metabolites or low oxygen tension under the acrylic devices could induce hyphal formation and penetration. 21
The lack of reproducibility in animal research has been a topic of discussion in recent years, 24 so literature reviews regarding the existing models can be of great use by assembling and weighing all available data. 25 Some previous reviews aimed to describe and discuss animal models of oral candidiasis and/or denture stomatitis,18,26–28 but this is the first study to deeply explore the existing palatal device designs and highlight the main methodological differences and findings from the literature.
Literature review
This literature review used the PubMed, Scielo, Lilacs, Bireme ProQuest, Google Scholar and ScienceDirect databases. Studies that described the fabrication of palatal devices for rats and/or induced denture stomatitis in immunocompetent, immunocompetent under antibiotic therapy, or immunosuppressed animals were selected using the terms: palate, rat, candidosis. The keywords were used alone or in combination, covering the period from 1978 to 2021.
Palatal acrylic devices for rats
Design, fabrication and fixation of palatal devices
Different designs and manufacturing techniques have been proposed to enable the use of intraoral devices in rodents for the subsequent induction of denture stomatitis. The first reported devices covered the entire hard palate and were constructed with cold-cured acrylic resin21,29 or heat-cured acrylic resin.30,31 To keep devices in position, researchers incorporated a metallic band around the maxillary incisors29–31 (Figure 1) or cemented them with cold-cure acrylic resin around the maxillary incisor teeth (Figure 2). 21 Another fixation method later described is the incorporation of stainless steel wires in the incisive region. 32 However, according to Hotta et al., 33 devices that cover the entire palate and are kept in position by metallic wires can be a source of great distress and cause weight loss.
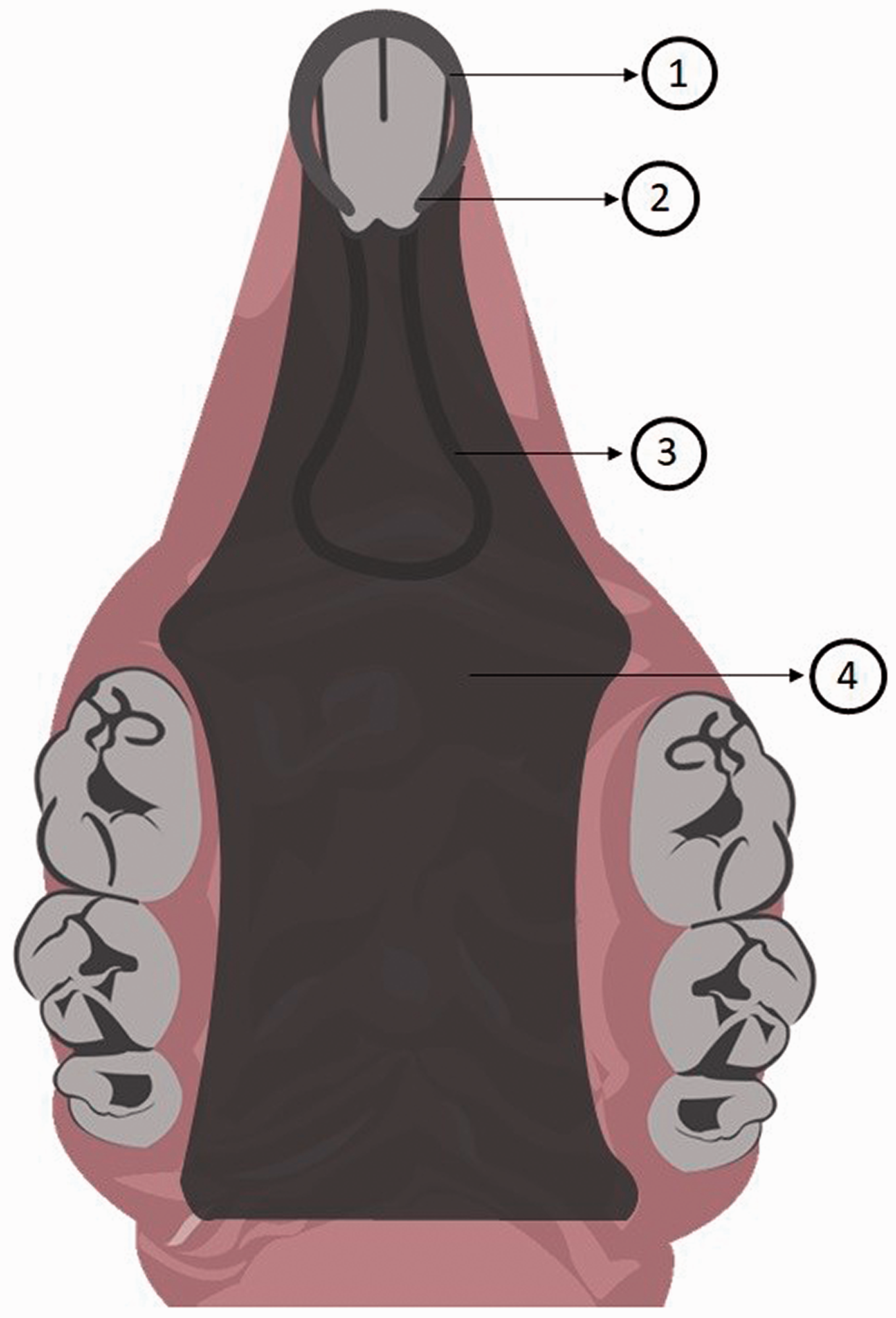
Schematic illustration of the palatal device developed by Olsen and Bondevik29 and Jennings and MacDonald.30,31 1: Stainless-steel band around the incisor teeth; 2: lateral notches were prepared in the incisors for device retention; 3: loop of a stainless-steel wire soldered to the acrylic resin; 4: acrylic device covering the entire hard palate.
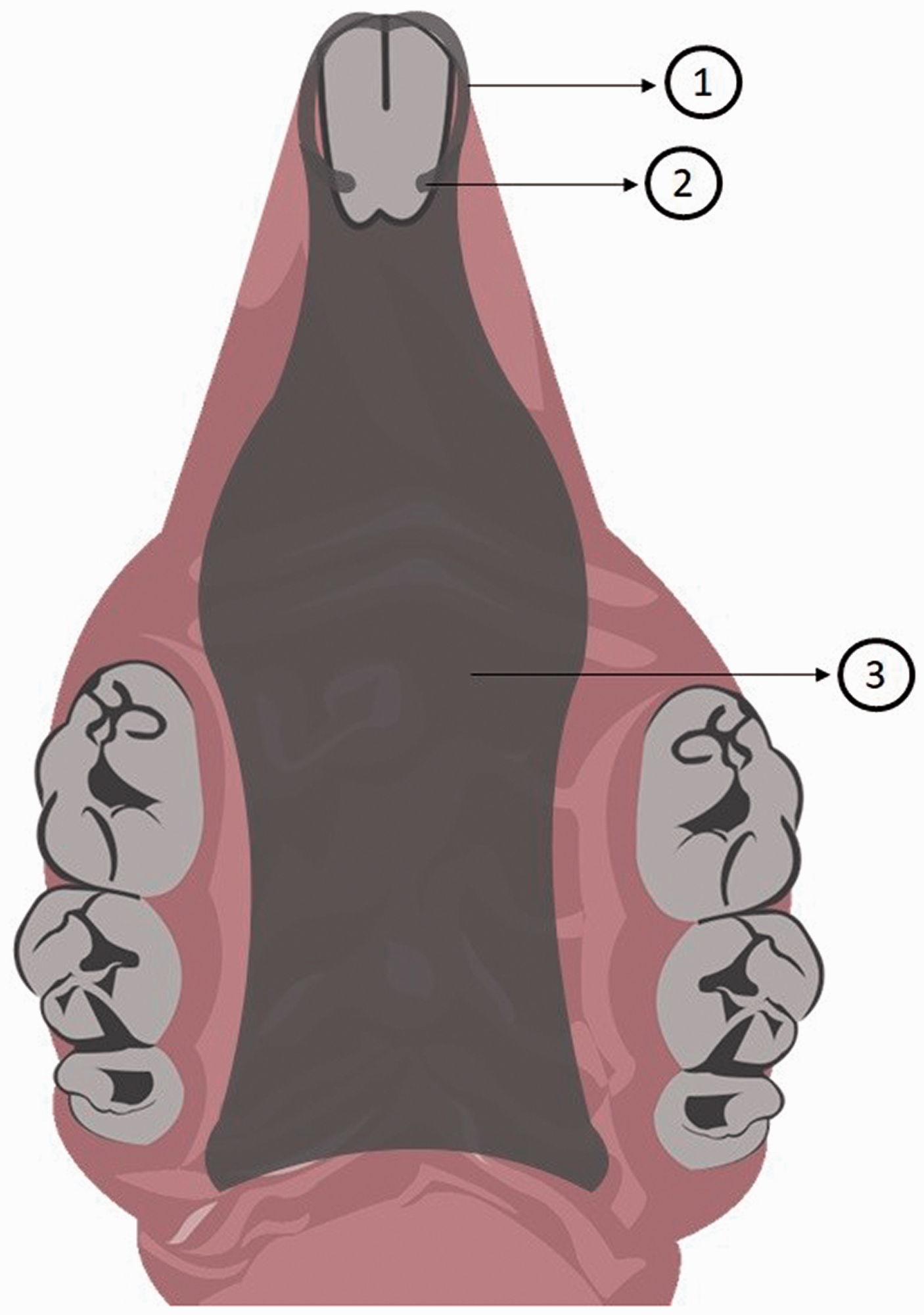
Schematic illustration of the palatal device developed by Shakir et al.21,41,47,49 1: The acrylic devices were cemented into position with cold-cure acrylic resin around the maxillary incisor teeth; 2: lateral notches were prepared in the incisors for device retention; 3: acrylic device covering the entire hard palate.
Different experimental conditions were tested by Hotta et al. 33 in order to standardize an acrylic intraoral device that could be relined with temporary soft material (Figures 3 and 4). Optimal results were obtained from devices covering only the posterior palatal mucosa, which were obtained from individual palatal impressions and fixed with acrylic resin. These conditions provided a satisfactory retention, with few devices lost during the experiments. In contrast, Nett et al. 34 built devices without a previous palatal impression (Figure 5). Ill fitted devices can either be lost through the course of the experiment or make the palatal tissue more susceptible to fungal infestation, considering it would probably cause more local trauma. 31
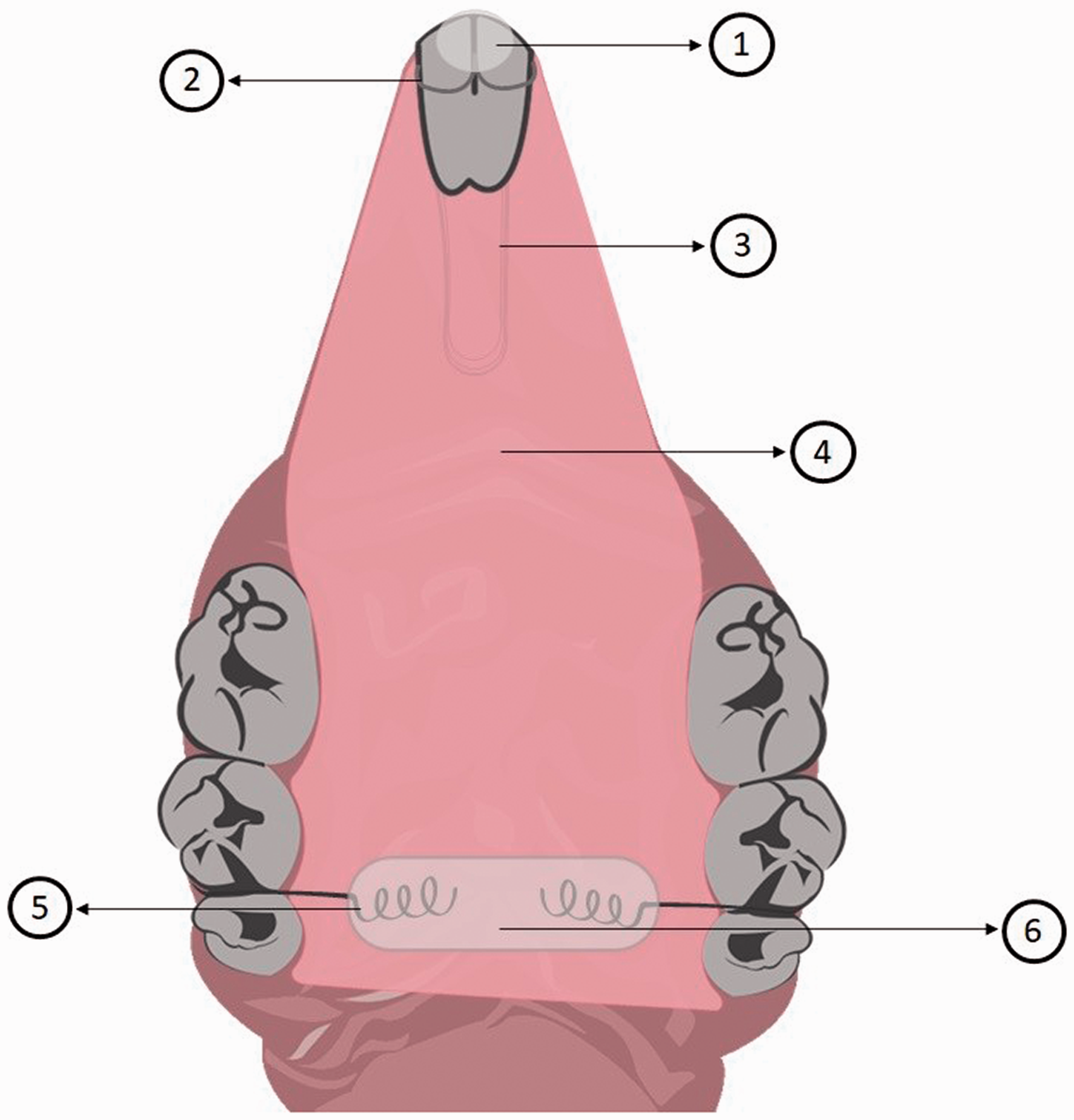
Schematic illustration of the palatal device developed by Hotta et al.33 1: Composite resin to protect the orthodontic wires tips; 2: four orthodontic wires were incorporated into the acrylic resin and were used to retain the device in the incisors; 3: loop of stainless-steel wire soldered to the acrylic resin; 4: acrylic device covering the entire hard palate; 5: orthodontic ligatures were incorporated into the acrylic resin and were used to retain the devices in the molars; 6: composite resin to protect the orthodontic wires tips.
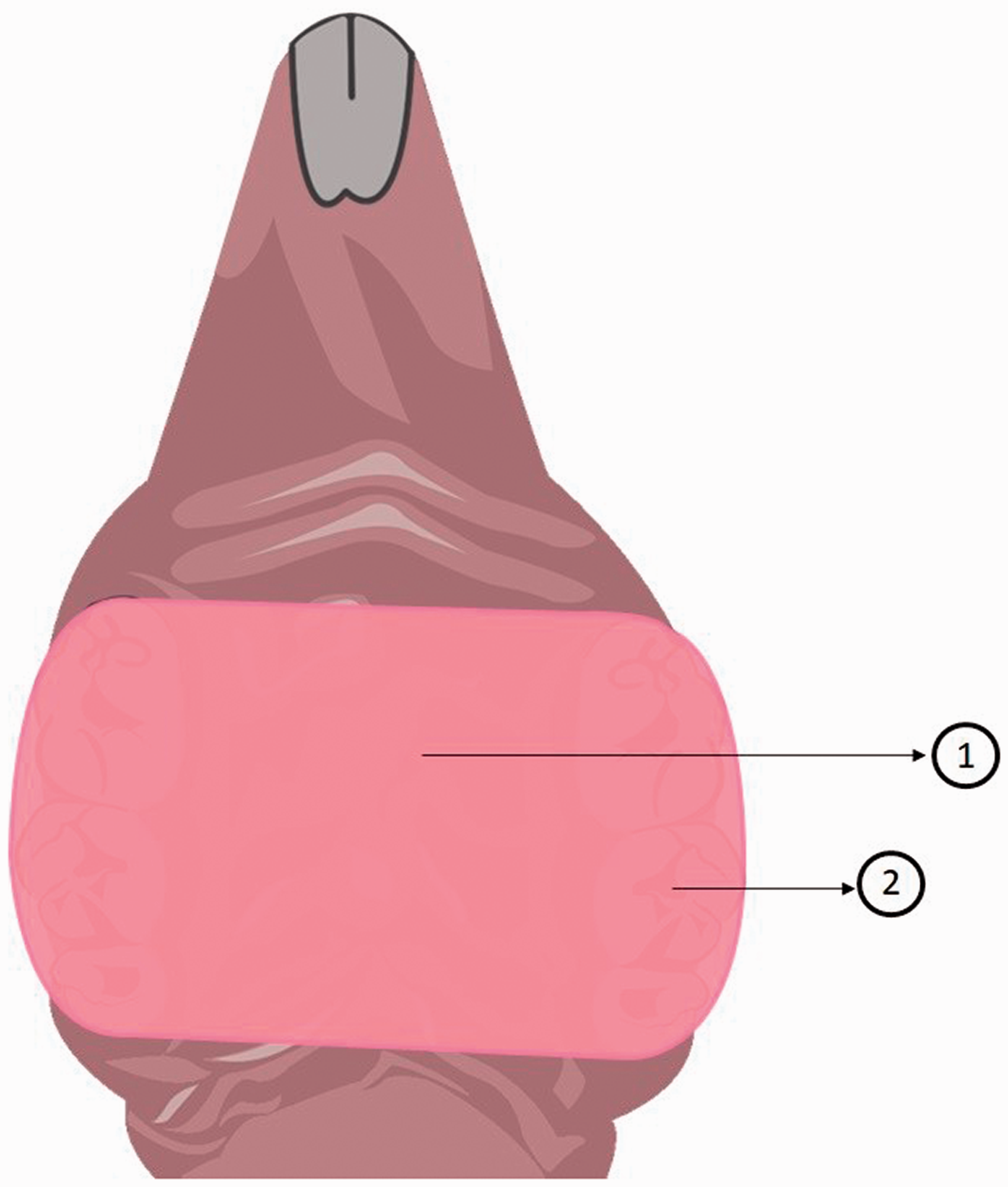
Schematic illustration of the palatal device developed by Hotta et al.33 1: Acrylic device covering only the posterior portion of the hard palate; 2: the devices were cemented into position with an autopolymerizing acrylic resin, which was inserted in the molar region bilaterally.
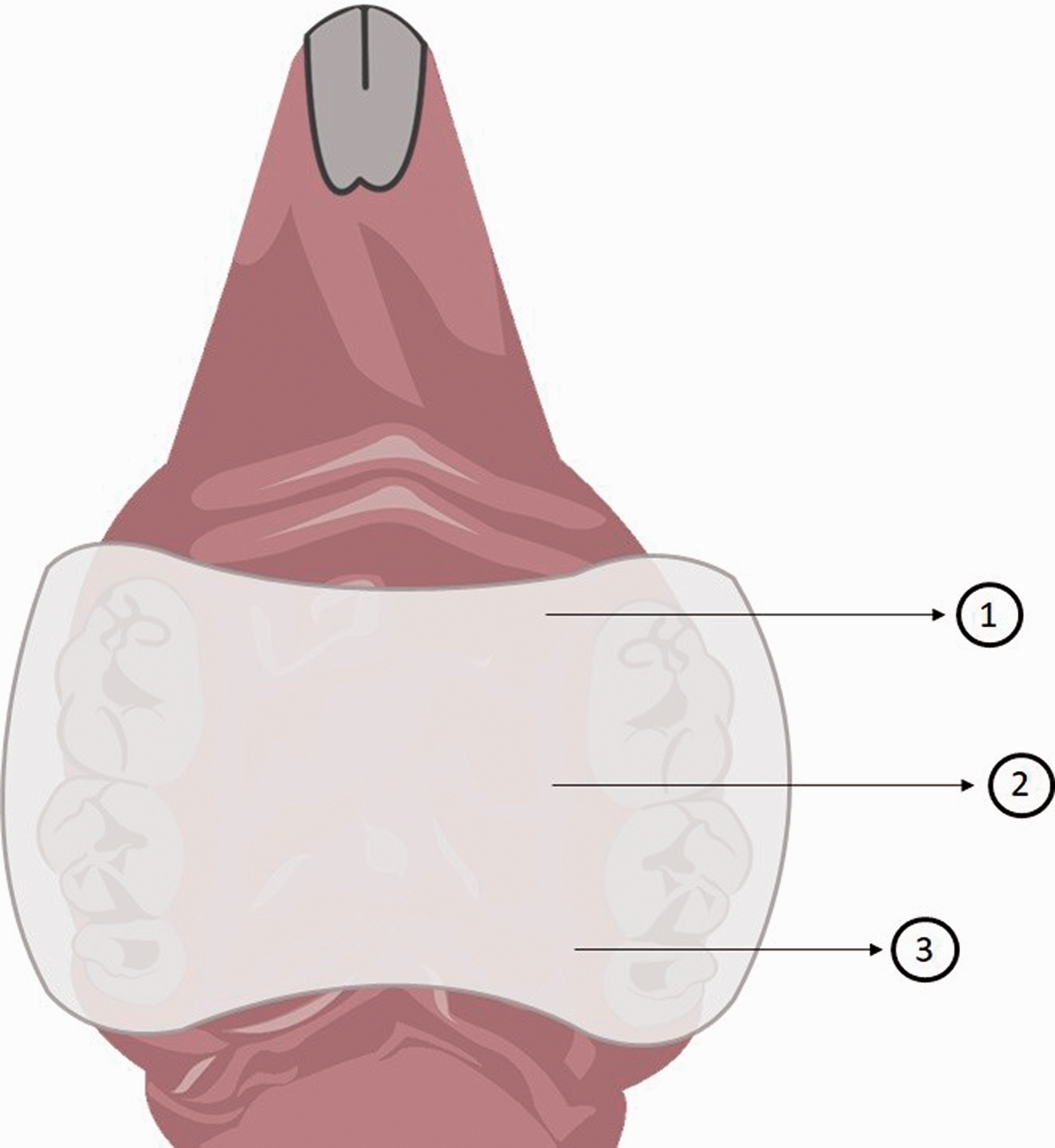
Schematic illustration of the palatal device developed by Nett et al.34 1: There was less than 1 mm of space between the hard palate and the device for Candida inoculation; 2: acrylic device covering only the posterior portion of the hard palate; 3: the autopolymerizing acrylic resin was applied directly in the animal’s mouth, without a previous palatal impression.
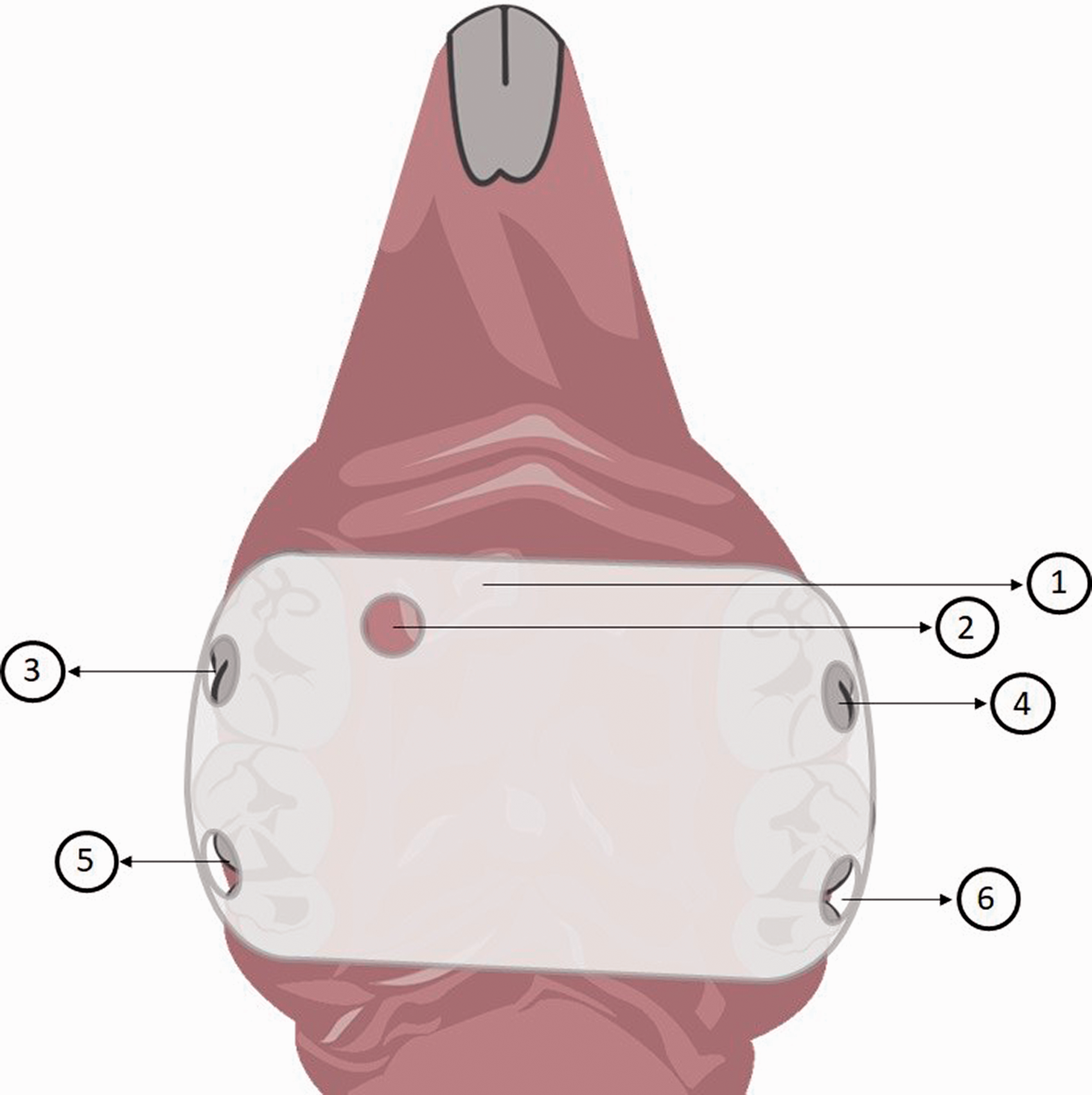
Moraes et al. 4 modified the design developed by Hotta et al. 33 by proposing the use of a colorless autopolymerizing acrylic resin to fabricate the palatal devices, in order to allow visibility during adaptation and fixation (Figure 6). The authors stated that visibility during experiments is essential, since any defect on the surface of the devices might compromise their adaptation and stabilization in the oral cavity. For instance, the opacity of the miconazole-supplemented acrylic resin devices made by Norris et al. 35 compromised the observation of irregularities on their surfaces, and nine appliances were lost during their experiments. These authors also tested different fixation methods for the devices (two autopolymerizing acrylic resins or a self-adhesive resin cement) and concluded that the latter was the best stabilization material, especially because it does not promote any thermal reaction that may compromise the palatal mucosa of the animals. 4
Schematic illustration of the palatal device developed by Moraes et al.4. 1: Acrylic device covering only the posterior portion of the hard palate; 2: the devices were fabricated with a colorless heat-polymerizing acrylic resin; 3: the devices were cemented into position with a self-adhesive resin cement, which was inserted in the molar region bilaterally.
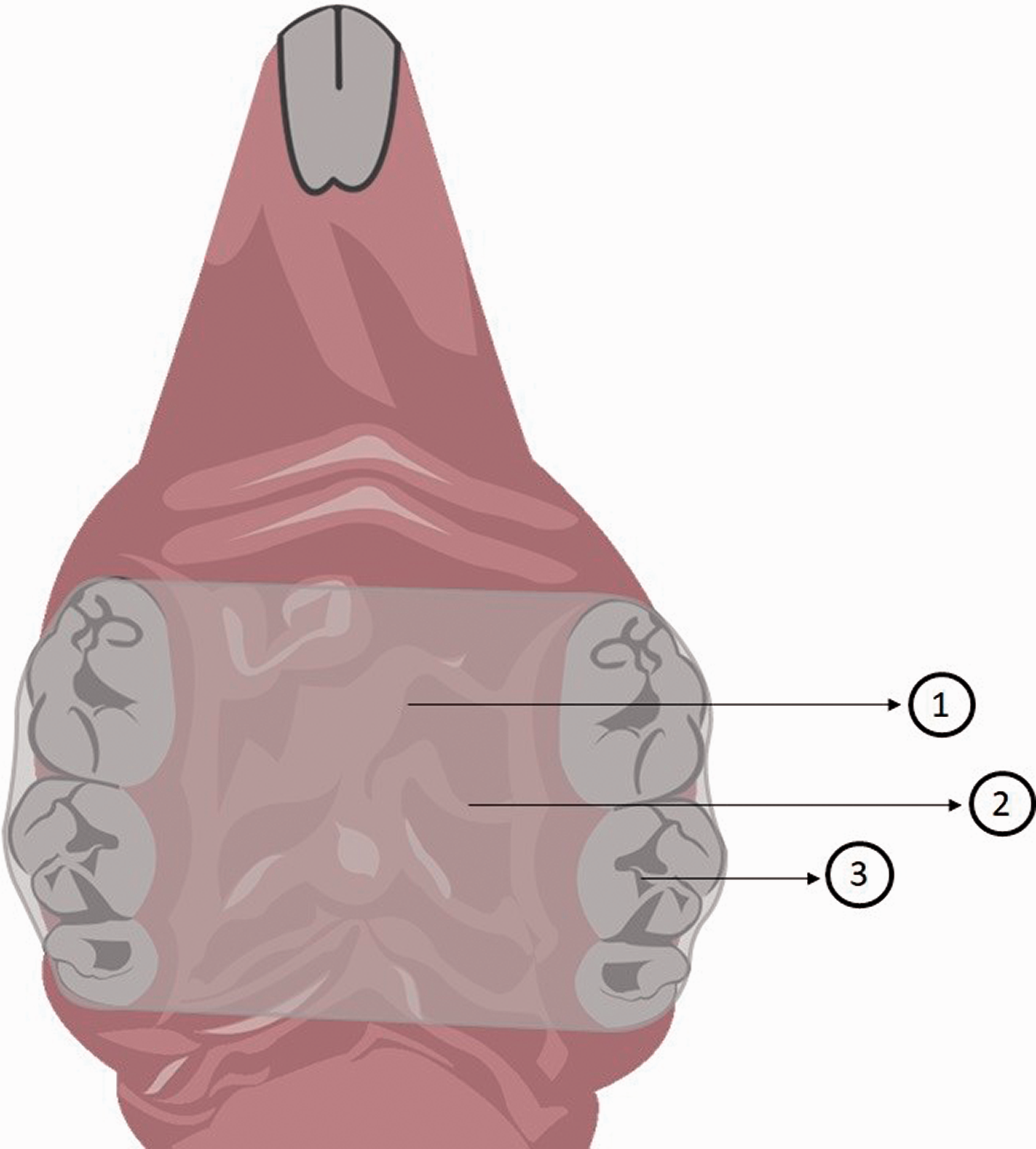
Lee et al. 36 developed a more complex device consisting of a fixed part that is anchored to the posterior palate by orthodontic wires and acrylic resin and a removable part fitted to the anterior palate that is retained by magnets embedded in the fixed part (Figure 7). One of the main advantages of this type of device is the ability to monitor fungal colonization longitudinally. This design has been successfully used in other studies.37–39
Schematic illustration of the palatal device developed by Lee et al.36, Johnson et al.39 and Yano et al.37,38. 1: The removable portion was connected to the fixed portion by magnetism; 2: embedded metal bar; 3: acrylic device divided into two portions covering the entire hard palate; 4: fixed portion; 5: two magnets were embedded in the fixed part of the device.
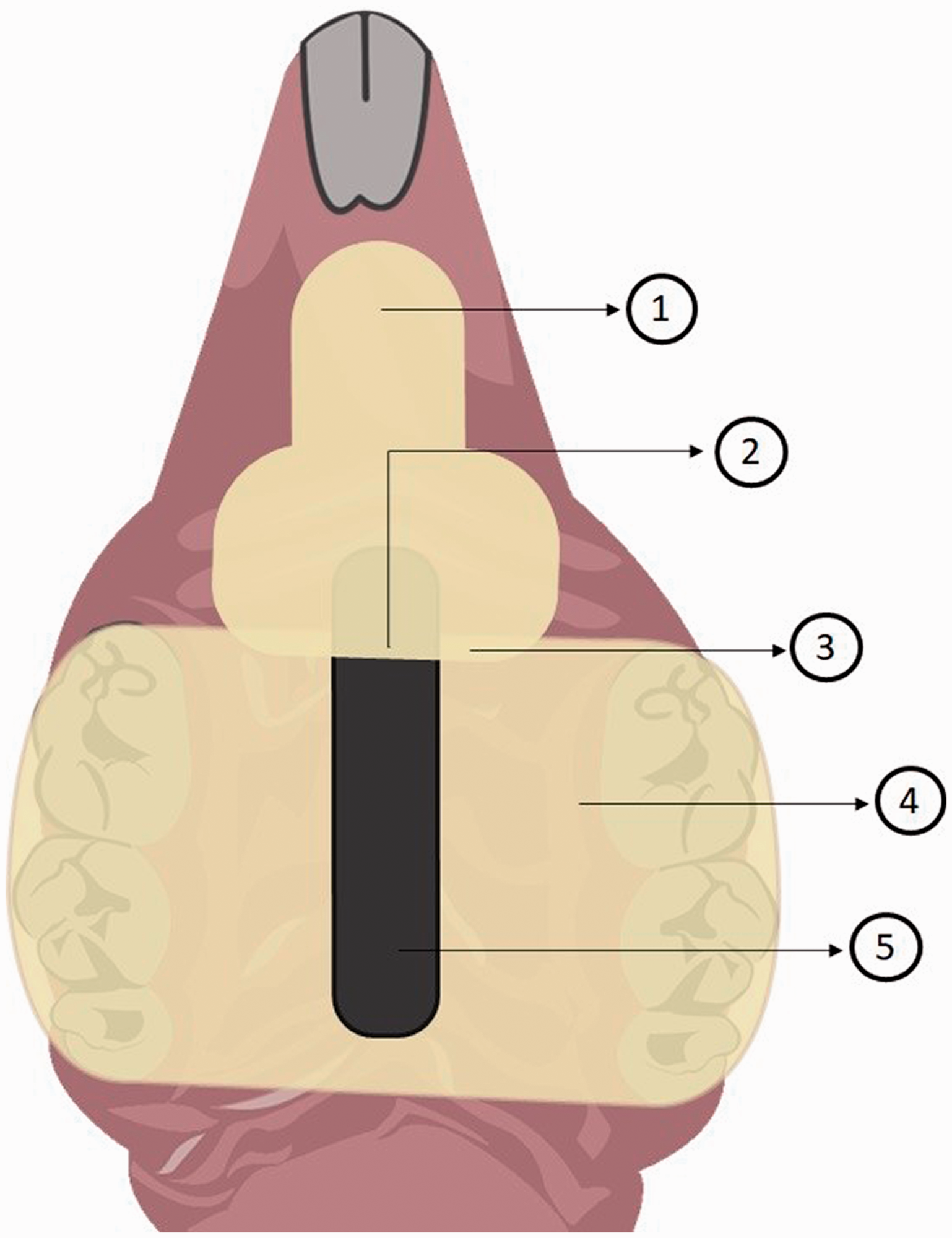
Recently, an intraoral device was developed by using a digital design and three-dimensional technologies (Figure 8). 40 In this study, a master cast of a rat palate was scanned, and the intraoral device was designed using software and printed in translucent resin. A reservoir was incorporated on the device surface to provide space between the device and the palate for the C. albicans inoculation. The devices were fixed with Optibond and a flowable composite.
Schematic illustration of the palatal device developed by Sultan et al.40. 1: Three-dimensional-printed acrylic device covering only the posterior portion of the hard palate; 2: reservoir for the administration of the Candida suspension and treatment; 3–6: slits through which flowable composite could be introduced to secure the installed device to the palate.
Device use time
There is a wide range of times described in the literature, from 6 h 27 to eight weeks.35,39 This diversity is strongly related to the aim of each study, as different experiments demand different device use times. For example, some authors aimed to analyze the biofilm formation on the palate after the device placement, 34 while others compared the virulence of different Candida strains. 37
Although most studies performed euthanasia right after the device removal, few authors21,41 investigated for how long the infection could be maintained without the use of the devices. According to these authors, removal of the appliances for two weeks resulted in complete clinical and histological resolution of denture stomatitis.21,41 The assessment of the longevity of denture stomatitis signs is important in order to determine for how long a new treatment could be tested without the spontaneous resolution of the infection.
Changes in palatal tissues resulting from the use of devices
Jennings and MacDonald 30 assessed the changes resulting from the use of palatal acrylic devices for 28 days. In the microscopic evaluation, desquamated keratin and food debris were found under the devices, and the palatal epithelium in the experimental group was thicker than that of the control group. In contrast, Barclay et al. 42 noticed that the use of the devices did not change the length and thickness of the palatal epithelium or the length of the basal membrane.
Hotta et al. 33 also investigated the best method to standardize the histological analysis: using only the palatal mucosa or using the palatal mucosa with the supporting hard tissue. After seven or 14 days of device use, the animals were euthanized and histological samples from their palates were obtained. According to the authors, it was not possible to analyze samples that contained only the mucosa, as they were very discrepant. Thus, samples of the palatal mucosa with the supporting bone tissue were selected, as they presented fewer variations in the epithelium and connective tissue. At 14 days, an increase in the compression of epithelial cells and connective tissue structure of the lamina propria was observed in the experimental groups compared with the negative control. The authors concluded that this finding could be explained by the continuous device use. Over the 14 days of evaluation, no histopathological changes in the connective tissue were observed.
Diet and animal welfare
Given that the intraoral device use is essential for the development of denture stomatitis in animals, diet adequacy is important to keep devices in position, avoid debris accumulation and ensure the animals’ well-being during experiments. Some studies used the conventional pellet diet21,35,43,44 but this approach may result in significant amounts of debris accumulated between devices and the epithelium,30,31 which can compromise the experiments.
Three different dietary regimens were compared by Barclay et al.: 42 a standard rodent pellet diet provided ad libitum and a nutritionally complete powdered diet provided as a thick paste mixed with tepid water or as a liquid diet. The animals from these two latter groups received 20 g of powdered diet made up freshly each day. The rats were randomly allocated to one of three experimental groups, each on a different diet, for seven or 14 days. The authors investigated debris accumulation under the intraoral devices and measured epithelial parameters, such as epithelial surface and basement membrane lengths, by computerized planimetry. The animals that received a pasty diet showed less accumulation of food debris without significant changes in their weights. This finding agrees with Hotta et al., 33 who also evaluated different diets: a pasty, a liquid and a powdered diet. The pasty diet was prepared in the same way as described by Barclay et al., 42 the liquid diet was composed of whey protein, maltodextrin and lipid, and the powdered diet was obtained by grinding the standard pellet diet. These authors concluded that the pasty diet is the ideal option, since less food debris was found and fewer animals showed weight loss, darkening of hair, prostration and death. In contrast, some authors32,34,40 used supplemented liquid diets and found no significant weight loss. The gel diet 76A seems to be another viable option, as it managed to avoid debris gathering under the devices and did not promote significant weight loss in different studies.36–39
Inoculum
Although the etiology of denture stomatitis is multifactorial, Candida infection has been found in approximately 93% of individuals with clinical signs of inflammation in the denture-supporting tissues. 45 C. albicans is the predominant species in denture stomatitis, identified in 50% to 98% of cases.45,46
Yano et al. 38 investigated whether C. glabrata could help to establish colonization and cause palatal tissue damage and inflammation but found no apparent role for it in the induction/enhancement of C. albicans pathogenesis in denture stomatitis. A similar finding was reported by Shakir et al., 47 who tested two strains of C. tropicalis and claimed both were not able to cause pathological changes in the palatal mucosa of rats. In this sense, most denture stomatitis animal models have used C. albicans as the species of choice.
Regarding the strains, there is a predominance of C. albicans 3091,21,35,41,44,47–50 but other strains, such as SC5314 and its derivatives,32,34,37–40 3118C, 47 CJN702 34 and ATCC 18804,43,51 have also been used. Knowing the differences between the C. albicans strains is important because various intrinsic characteristics can be responsible for the pathogenicity in vivo. 52 For instance, Shakir et al. 47 compared strains 3091 and 3118C and reported that strain 3091 consistently induced denture stomatitis whereas strain 3118C did so in only two of five animals, a finding that indicates a difference in their pathogenicity.
Another crucial variable is the concentration of the inoculum used to induce the disease. However, there is no consensus regarding how concentrated the inoculum should be, as concentrations range from 1 × 10650 to 1 × 1010 colony-forming units (CFUs). 39 Chandra et al. 53 tested different inoculum sizes for the contamination of acrylic resin specimens and reported that concentrations over 107 cells/mL did not enhance biofilm formation. It is important to note that biofilm formation occurs differently in vivo, so the ideal inoculum concentration should be investigated.
Denture stomatitis models
Figure 9 presents a flowchart with the main characteristics of each model.
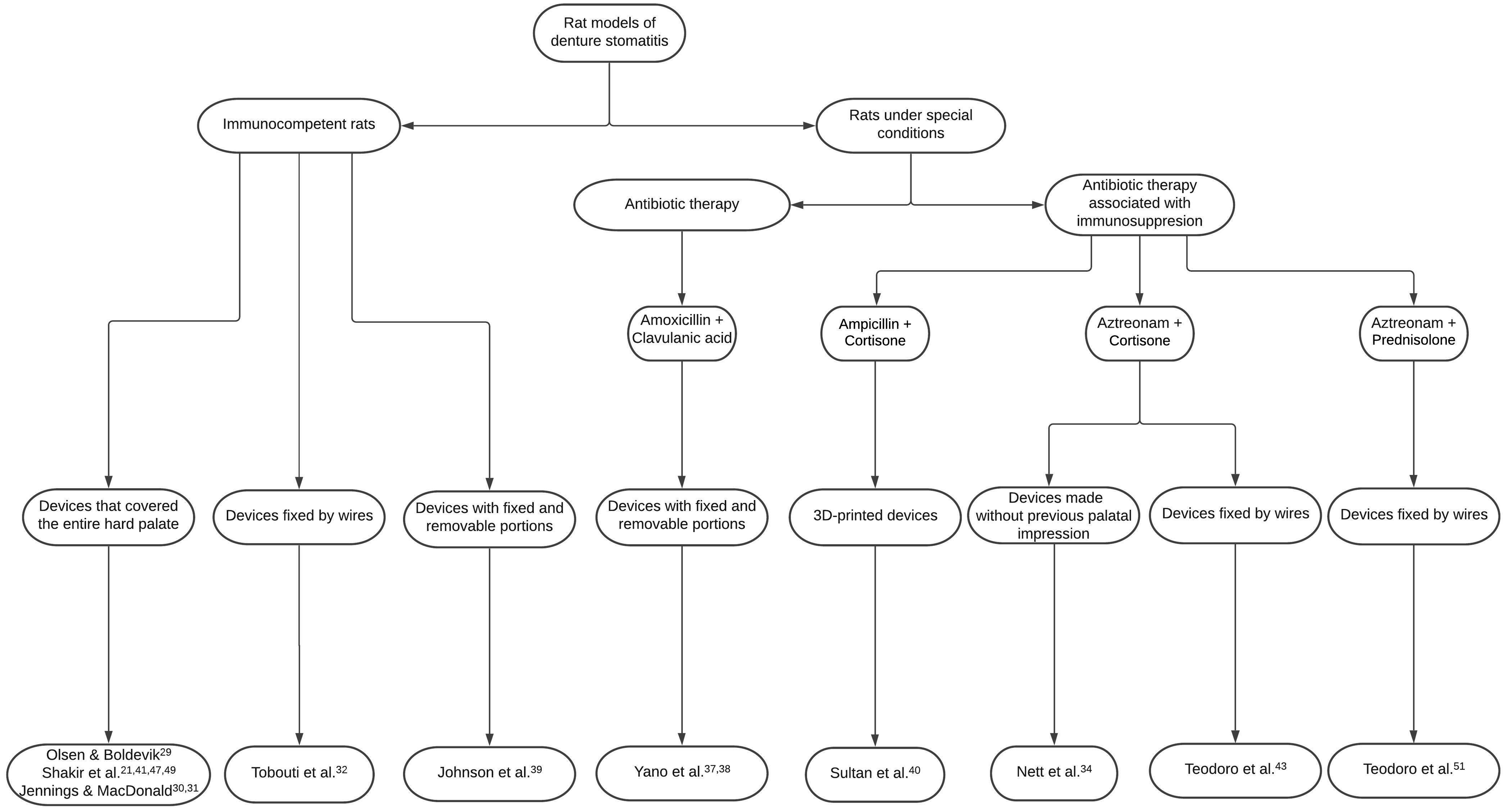
Main characteristics of the existing denture stomatitis models in rodents.
In immunocompetent rats
Some studies report that denture stomatitis might be associated with a systemic cell-mediated inflammatory response. 54 Considering this hypothesis and the fact that denture stomatitis can occur in healthy patients, it would make sense to develop a model using immunocompetent animals.
Most studies induced denture stomatitis in immunocompetent rats through the use of intraoral devices and C. albicans inoculations.21,29,37,39,48,50 Colonization cannot be observed in uninoculated animals with a device and in inoculated animals without a device.21,39 On the other hand, when the intraoral devices were associated with the inoculation of C. albicans, clinical and histopathological changes were evidenced, proving that this association is fundamental for the development of denture stomatitis in animals. 21
In contrast to the aforementioned studies, Tobouti et al. 32 developed a model of denture stomatitis using a device contaminated with C. albicans biofilm, to simulate the actual clinical conditions of a contaminated denture. The rats that were fitted with a contaminated device showed clinical, microscopic and molecular changes in comparison with the other groups.
In rats under special conditions
Antibiotic therapy
The oral microbiota of humans and rats have similarities,55,56 as both showed no significant differences in the number of Gram-positive and -negative cocci, Gram-positive and -negative rods, lactobacilli, Gram-negative anaerobic rods, staphylococci, yeasts and coliforms in a study conducted by Hall et al. 55 However, rodents are coprophagic, and this habit might introduce microorganisms associated with fecal material, including Escherichia coli, into their oral cavity.55,57 Considering this issue, some authors have injected broad-spectrum antibiotics in order to reduce the enteric oral colonization prior to device placement34,43,51 and/or have housed the animals in special conditions to avoid feces consumption or any contamination.32,34,40 Most studies,21,29,35,36,38,41,44,47–50 however, did not address this concern.
Broad spectrum antibiotic therapy is one of the predisposing factors for the occurrence of denture stomatitis in humans, 58 as the use of antibiotics can help Candida in the transition from commensalism to parasitism. 59 The administration of antibiotics in the drinking water is frequently described in animal models of oral candidiasis,22,60–68 but is rarely reported in denture stomatitis models.37,40 This approach can significantly increase the oral fungal burden67,69 and favor the infection, 19 although some divergences can be found in the literature. 70
Most oral candidiasis models add tetracycline to the drinking water, which is usually provided to the animals ad libitum. Previously described concentrations include 0.1% (wt/vol.),22,60–62,71 15 mg/mL66 and 0.83 mg/mL,64,66,72 and treatment-time regimen differs greatly among studies. Some authors administered it for 24 h only, 66 while others 61 began the antibiotic therapy seven days prior to oral inoculation and maintained it up to the end of the experiment. However, some studies73–75 report that tetracyclines have non-antimicrobial properties and are effective in reducing inflammation by inhibiting matrix metalloproteinases, preventing excessive angiogenesis and inhibiting apoptosis. These properties could compromise the development of an inflammatory condition such as denture stomatitis. In this regard, other antibiotic agents, including amoxicillin 37 and ampicillin, 40 could be better options. However, there is no study comparing the impact of these antibiotics in the development of denture stomatitis.
Antibiotic therapy associated with immunosuppression
Immunocompetent rodents are capable of eliminating Candida yeast and hyphae three to eight days after oral inoculation,26,27 probably due to their immune system. 26 For this reason, some studies34,40,43,51 have administered corticosteroids in order to induce an immunodeficient condition and facilitate Candida infection. Even though immunosuppression is a risk factor for denture stomatitis in humans, it has been reported that immunosuppressed mice can become severely ill, with significant weight loss and decrease in grooming. 27
The most commonly used protocol is the injection of one single dose of cortisone (200 mg/kg subcutaneously) on the day of infection, prior to placement of denture appliances.34,40,51 However, Teodoro et al. 43 chose to administer prednisolone (100 mg/kg intradermally) every other day until euthanasia.
Sultan et al. 40 stated that even though denture stomatitis occurs in healthy individuals, the disadvantage of not inducing immunosuppression in animals is that colonization and infection would require an extremely high inoculum of C. albicans, which is not reflective of the normal environment in the oral cavity and would result in overwhelming infection. These authors managed to find marked edema, erythema and inflammation of the denture-bearing palatal mucosa and dense white/yellow curdy plaques. However, there is no study directly comparing immunocompetent and immunocompromised animals to establish whether this approach is indeed necessary in the development of denture stomatitis.
Methods for the evaluation of denture stomatitis in animal models
CFU/mL count
Several methods are described in the literature to verify the presence of denture stomatitis and/or the effectiveness of a new therapy. The counting of CFUs aims to estimate colonization by C. albicans on the palates of rats. Determination of microbial cell number is one of the most fundamental procedures in microbiology. 76 CFU counting on agar plates is considered the gold standard method given its three main advantages: (a) an unlimited range of detection, as it allows the counting of any number of microorganisms; (b) only viable microorganisms are counted; and (c) no special equipment is necessary. 76
Biofilm samples are usually collected from the palatal mucosa using sterile swabs, which are subsequently diluted in sterile PBS or saline solution and seeded in culture medium – mainly Sabouraud Dextrose agar or ChroMagar®, a specific chromogenic medium for isolation and presumptive identification of Candida species. Then, the Petri dishes are kept at 35–37°C for 24–48 h.
Despite being a well-established method, few authors have used the CFU/mL count as a parameter for the presence of denture stomatitis or for the success of treatment. In fact, some authors did not even perform this analysis,32,43,44,49 while others did not perform any type of inferential statistics with the data obtained from this method.21,29,34,39,41,47,48,50 Perhaps this finding can be justified because significant variation in fungal burden is frequently noted in animal models of candidiasis. 27
Visual analysis
The first rat model of denture stomatitis was proposed in 1978. 29 Over 40 years later, only three studies32,39,40 have presented clinical images of the lesions, making it hard to make comparisons between studies or to compile evidence. Johnson et al. 39 and Tobouti et al. 32 used the Newton classification to evaluate the palates of Wistar rats. Clinical signs were scored as type I: pinpoint erythema; type II: diffuse erythema/edema; and type III: diffuse or generalized erythema with papillary hyperplasia. Tobouti et al. 32 found only lesions consistent with type I, while Johnson et al. 39 reported lesions corresponding to types I and II, which were expressed as percentages. On the other hand, Sultan et al. 40 described their main findings without using scores.
Some authors21,41,51 reported their clinical findings, but for some reason did not provide figures to illustrate such features. Olsen and Boldevik 29 described their model through scores but claimed that the photographic assessment of the palatal mucosa did not provide any additional information. They used a three-point score for palatal erythema based on severity (No = 0; Slight = 1; Moderate = 2; and Severe = 3) and presented fractions of accumulated score/maximum possible score for each group.
Histopathological analysis
This is the most common type of analysis in this field, performed by 16 out of the 19 studies reviewed. Histological analysis allows the characterization of processes occurring in cells and tissues and their structural features. 77
The palatal sections are usually stained with hematoxylin-eosin or Schiff's periodic acid (PAS), but other stains, such as Gomori’s methenamine silver, can also be used to evidence tissue invasion by Candida. 34 The most common histopathological changes observed in the studies are the presence of hyphae and yeasts of C. albicans in the epithelial layers, microabscesses, epithelial hyperplasia or atrophy, hyperparakeratosis, disorganization of the basal layer of the epithelium, spongiosis, exocytosis and inflammatory infiltrate.
Exfoliative cytology of mucosal smears
In this technique, smears are taken with a metal spatula from the palate and immediately smeared onto glass slides, fixed with a fixative spray, stained with PAS and hematoxylin, and then evaluated for the presence of yeast-like fungi. 29
Scanning electron microscopy (SEM)
SEM is an excellent tool for investigating ultrastructure and morphological features of microorganisms. 78 This technique was used by Martin et al. 44 to evaluate whether the permeability barrier of the palatal mucosa was infected by C. albicans. The authors also evaluated the permeability after mucosal healing. For this purpose, the right jugular vein was exposed by blunt dissection and the animals were perfused with lanthanum. In this study, the epithelium of rats that were inoculated with C. albicans under an acrylic appliance became permeable to perfused lanthanum.
SEM micrographs can also be of value to study C. albicans biofilm formation and architecture on acrylic devices4,34,37,39 and on the palatal mucosa.37,39
Confocal laser scanning microscopy
Confocal microscopy has many applications, including the evaluation of cell viability on biofilms and contaminated surfaces. Therefore, it is possible to visually assess C. albicans contamination in animal models through this technique. 34 Furthermore, quantitative data can be obtained from the confocal micrographs and be expressed as total, viable and non-viable biovolume, percentage of cell viability, and area covered by C. albicans biofilm. 4 Some studies performed this analysis to evaluate the contamination on the palatal device surfaces4,34,39 and/or to observe 39 and quantify the biofilm 37 on the palatal tissue of rats after denture stomatitis induction.
Different dye sets have been used in this field, including calcofluor white and concanavalin,37,39 SYTO-9 and propidium iodide, 4 and calcofluor white, SYTO-9, FUN-1 and ConA. 34 These stains are able to provide important information regarding the fungal cell wall and the extracellular matrix34,37,39 and the viability of cells4,34 and can also help to visualize bacteria in mixed biofilms. 34
Gel electrophoresis
Martin et al. 44 also used electrophoresis gel as a biochemical method to evaluate the permeability of the palatal mucosa after infection. This technique is used to separate and identify macromolecules as DNA, RNA or proteins based on size, form or isoelectric point. The separation of molecules is based on the fact that charged molecules migrate through a gel matrix upon application of an electric field. The palatal tissues were irrigated to remove debris with sterile water and the mucosa was gently dried with a sterile swab. A cellulose acetate filter was applied to the intermolar area and held in position to collect transudates. The filters containing the transudates were kept at –20°C in airtight containers and then analyzed by electrophoresis.
Lactose dehydrogenase (LDH)
Another method that can be used to measure tissue damage is the LDH assay. The enzyme LDH is present in several different tissues in the body, and an increase in its levels indicates tissue damage or disease. 79 This assay was used by only one research group37,38 for the assessment of tissue damage in rats with denture stomatitis. These authors measured the LDH levels in the supernatants of palate swab suspensions with a colorimetric probe. Through this technique, they observed two main findings: (a) rats inoculated with C. albicans mutants defective in biofilm formation by deletion of the Efg1 or Bcr1 transcription factors have reduced oral tissue damage; 37 and (b) C. glabrata has no inducing or enhancing effects on C. albicans virulence. 38
Mitotic activity
An increase in the mitotic activity is a common tissue response to physiological or pathological stimuli. There is only one study 49 that investigated the mitotic activity in an animal model of denture stomatitis. These authors evaluated cells of different epithelial layers from slides stained with hematoxylin and eosin and observed that the palatal epithelium of animals infected with C. albicans became thicker and had more mitotic figures than that of control animals. Nowadays, this histological technique is no longer considered the most accurate method given the many immunohistochemical stains that can evidence cells in proliferation in a more precise manner.
Aspartyl proteinases (SAPs) expression
C. albicans invasion leads to tissue destruction. In this process, SAPs, which are extracellular enzymes considered key virulence factors of C. albicans, are secreted. So far, only one study 32 has evaluated the SAPs gene expression in an animal model of denture stomatitis. These authors concluded that the use of palatal devices contaminated by C. albicans resulted in high SAPs expression, which was consistent with their clinical, microscopic and serum leukocyte findings and with the virulence capacities of C. albicans and epithelial invasion.
Conclusion
The development and use of animal models are critical for the assessment of new therapeutic alternatives for denture stomatitis. However, as discussed in our review, this is not an easy task, as there are still many unanswered questions and issues to be addressed. Over the years, different models have been developed with different aims and evaluated by different methods, making it hard to compare their strengths and weaknesses. Perhaps one of the reasons why studies differ so much is the lack of proper diagnostic criteria. What is essential to induce a disease and what determines whether a model succeeded in doing so? Is the clinical aspect of the palatal mucosa, the fungal load or the histopathological findings? And for how long should these findings remain present? Considering what is known about denture stomatitis in humans, we believe that an animal model should present a combination of these factors during a reasonable period of time in order to reproduce the human clinical condition more accurately.
Research data availability statement
The studies used in this literature review are available and will be provided by the corresponding author through email (
Footnotes
Acknowledgements
The authors would like to thank Christina Desnoyers for her writing assistance. This manuscript is a literature review. Therefore, it does not require an ethical statement nor the completion of the ARRIVE checklist.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Coordination for the Improvement of Higher Education Personnel – Brazil (CAPES) (finance code 001) and the São Paulo Research Foundation (FAPESP) (grant numbers 2017/22996-1 and 2017/07314-1).
