Abstract
This report describes transurethral catheterization with a Foley catheter in 19 anaesthetized, laterally recumbent female pigs with a chest circumference of 72.1 ± 4.2 cm and weighing approximately 40 kg. Catheterization within five minutes was successful in 78.9% of the pigs. Catheterization depth, measured as the distance between the bladder neck and the mucocutaneous junction of the vulva, was 14.5 ± 1.3 cm and correlated with chest circumference but not body length. Measurement of catheterization depth aids in optimal positioning and may prevent inflation of the Foley catheter in the urethra.
Introduction
Transurethral catheterization is necessary in various research and clinical applications but is not performed in male pigs because of the spiral end of the glans penis.1,2 The external urethral opening in female pigs is located ventrally at the junction of the vagina and vestibule. 3 The suburethral diverticulum (Figure 1C) caudal to the external urethral opening may complicate the catheterization.1,4,5
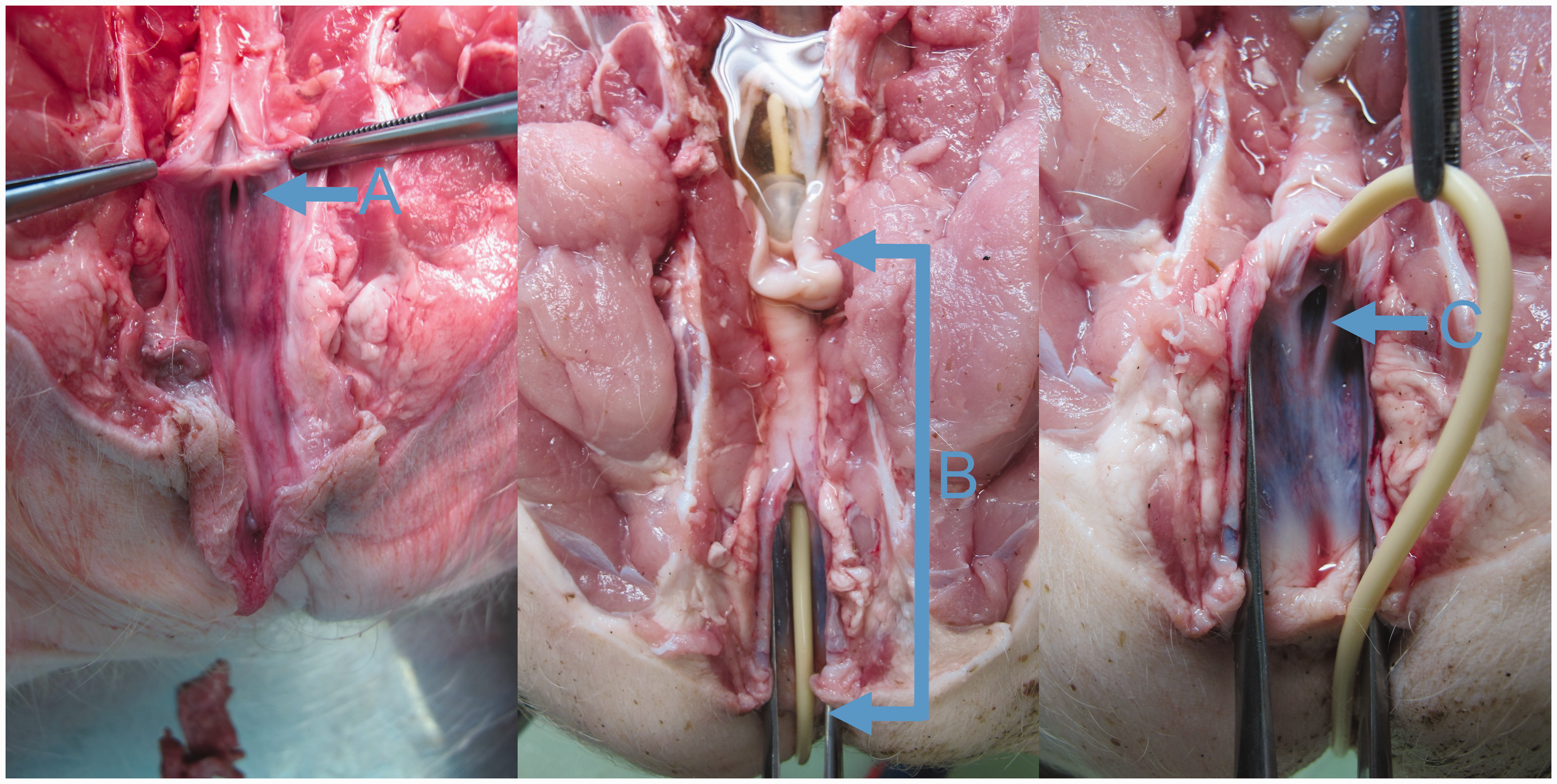
Postmortem dorsoventral view of the urethral opening (A), with the spine and colon removed (the cadaver was submitted for pathoanatomical assessment of cause of death and was not part of this study). The catheterization depth was defined as the distance between the bladder neck, where the end of the inflated baloon of the catheter stopped, and the ventral mucocutaneous junction of the vulva (B). The suburethral diverticulum (C) may complicate catheterization.
This report aims to provide information on transurethral catheterization in anaesthetized, laterally recumbent female domestic pigs. To clarify the anatomical conditions, the distance between the mucocutaneous junction of the vulva and the bladder neck was measured, which was determined as the catheterization depth.
Methods
This study was approved by National Ethics Committee and National Veterinary Administration (U34401-20/2016/4, U34401-20/2016/9, and U34401-20/2016/13). Transurethral catheterization was performed as a standard protocol during anaesthesia for a primary study. 6
Nineteen female Landrace and Large White hybrid pigs were obtained from a commercial farm three days before they were anaesthetized. Pigs were free of classical and African swine fever and Aujeszky’s disease. They were vaccinated against Mycoplasma hyopneumoniae and porcine circovirus type 2. The body mass was determined by chest circumference using the Rondo Combi Weight Measuring tape (Kruuse, Langeskov, Denmark). They were housed in groups of two or three in straw-bedded pens at an ambient temperature of 20–28°C (mean 24.6 ± 2.0°C) and humidity of 57–82% (mean 68.7 ± 5.6%), fed twice daily (BEK-1, Jata Emona, Ljubljana, Slovenia) and had unrestricted access to water. Animals were exposed to the natural light–dark cycle except for the first 48 h after anaesthesia. They were clinically examined 12 h before anaesthesia to ensure their adequate health status.
The pigs were anaesthetized as described elsewhere. 6 They were placed in left lateral recumbency and body length was measured from the nuchal crest to the base of the tail. A surgical tampon was inserted into the rectum to prevent contamination, and the perineal area was cleaned with an antiseptic scrub. The mucosa of the vulva was treated with an antiseptic (Skinsept mucosa, Ecolab Deutschland GmbH, Mannheim am Rhein, Germany) and lidocaine gel (Xylocaine 2%, AstraZeneca, Cambridge, UK) was applied. An assistant inserted a Killian septum speculum (Surgiway, Paris, France) with 95 mm long blades into the vestibule of the vagina and spread the blades. The surgeon inserted a laryngoscope with a standard Miller blade (size 1, Midmark, Dayton, USA) into the vestibule and retracted it ventrally to visualize the urethral opening (for clarity, the postmortem section is shown in Figure 1A). A siliconized latex CH 8 Foley catheter with a plastic stylet (Securmed, Thiene, Italy) was carefully secured between the jaws of curved Rochester Pean forceps (Integra Lifesciences, Princeton, USA) with the catheter tip protruding approximately 2 cm from the tips of the jaws (Figure 2). The surgeon gently inserted the catheter tip into the urethral opening and repositioned the forceps to push the catheter deeper into the urethra. When the catheter was inserted all the way, the balloon was filled with 3 ml of saline and the stylet was removed. The catheter was withdrawn until the balloon stopped at the bladder neck, and a mark was made on the catheter at the level of the ventral mucocutaneous junction of the vulva. The catheterization depth was defined as the distance between the bladder neck where the inflated balloon of the catheter stopped and the mucocutaneous junction of the vulva (Figure 1B). After emptying the bladder, the catheter was deflated, removed, and the catheterization depth was measured. Catheterization was discontinued if it was not successful within 5 min.
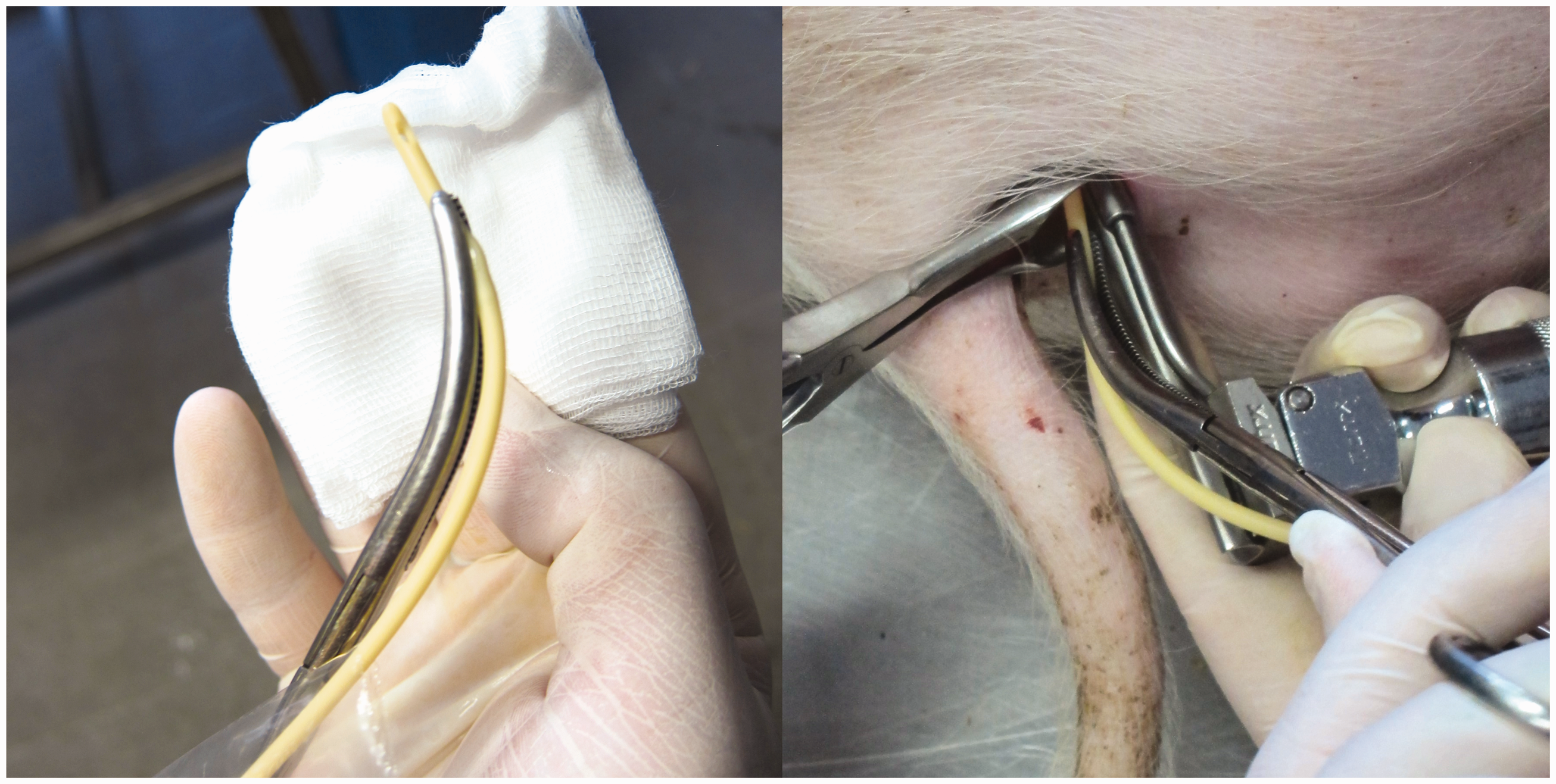
A CH 8 Foley catheter was secured between the jaws of a curved Rochester Pean Forceps with the catheter tip protruding approximately 2 cm from the tips of the jaws (left). With the Killian speculum in place, the surgeon inserted a laryngoscope blade to visualize the urethral opening and inserted a Foley catheter all the way (right).
The research members evaluated the pigs hourly for the first 12 h after the anaesthesia, and then every 4 h until 48 h. For the remaining 19 days of the study, the animals were observed once daily. After that they were euthanatized due to the requirements of the primary study. 6
Results are presented descriptively. Continuous variables are presented as mean ± standard deviation of the mean (SD) and discrete variable as n and its rate in %. The correlation between catheterization depth and chest circumference or body length was tested using Pearson’s two-tailed correlation coefficient. The significance was set at p < 0.05. Statistical analyses were performed using SPSS 25 for Windows (SPSS Inc, USA).
Results and discussion
Four pigs were not catheterized within 5 min and were excluded from further statistical analysis (78.9% success rate). Chest circumference was 72.1 ± 4.2 cm, body mass determined by chest circumference was 42.1 ± 4.0 kg, body length was 82.1 ± 3.3 cm, and catheterization depth was 14.5 ± 1.3 cm. Catheterization depth was positively related to chest circumference (r = 0.556, p = 0.031) but not to body length (r = 0.351, p = 0.2).
In our study, catheterization was successful in 15 of 19 pigs, but we set a relatively short maximum catheterization time of 5 min. Musk et al. 4 reported a higher success rate in pigs of similar weight, but catheter placement took up to 60 min in some animals. On the other hand, Li et al. reported successful catheterization of all miniature Bama pigs in 15 min. 1
Difficulties encountered during catheterization have been well described 4 and were also observed in our study. During the procedure, the pigs were positioned in the lateral recumbency on a fixed-height table. Although such positioning may be necessary in certain study protocols, 6 it proved uncomfortable for the surgeon and could be mitigated with pigs in the supine position.1,4,5 Nevertheless, the lateral recumbency did not appear to affect the success rate in our study, as the surgeon (JŽ) regularly catheterizes female dogs in this position and is accustomed to doing so.
In some animals, repeated catheterization attempts resulted in trauma and minimal mucosal bleeding, 5 which affected the visibility of the urethral opening. An additional light source may be beneficial in these situations. When available, endoscopy can provide better visualization of the urethral opening and surrounding structures.
Determining catheter entry into the urinary bladder was problematic because urine outflow through the catheter was slow, so the catheter was inserted all the way before the balloon was inflated and the stylet removed. Despite the stylet, the softness of the Foley catheter made catheterization difficult. The plastic stylet proved to be too soft; therefore, replacement with a guidewire as a stylet may be considered as an alternative. Despite the described complications, no micturition abnormalities were observed in any pig during the recovery period and after anaesthesia in our study.
The limitation of our study is that it was designed in accordance with the 3Rs principle, and the number, size and weight of the pigs were predetermined by the primary study. 6 The estimated catheterization depth can only be used in pigs with similar body constitution and weight as in this study.
In conclusion, catheterization of anaesthetized, laterally recumbent female pigs weighing approximately 40 kg can be performed quickly and efficiently. The measured catheterization depth aids in optimal positioning of the Foley catheter and correlates with chest circumference, not body length.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by the Slovenian Research Agency (P4-0053, P3-0043) and Tertiary funding from the Clinical Department of Anaesthesiology and Intensive Therapy, University Medical Centre Ljubljana, Slovenia.
Data availability statement
Raw data were generated at Veterinary Faculty, University of Ljubljana, Slovenia. Derived data supporting the findings of this study are available from the corresponding author (
