Abstract
Maxillary nerve blocks (MNBs) commonly facilitate dental surgeries in standing horses. The goal of this prospective, blinded, cross-over design trial including 15 client-owned horses was to evaluate 3 methods of sensory function testing for confirming a successful MNB. Testing was performed bilaterally before sedation, 5 min after sedation, and 15 and 30 min after MNB with 0.5% bupivacaine and involved a needle prick dorsal to each naris, hemostat clamping of each nostril, and gingival algometry (measuring sensitivity to pain). Responses to stimulation were numerically scored and scores were summed up to a total score. Total score increases on the blocked side by ≥ 2 between baseline and 30 min Post MNB recordings signified a successful MNB. Sedation in the preceding 6 h, presence of sino-nasal disease, side of dental pathology, age, butorphanol administration, and detomidine dosing (µg/kg/min) throughout the tooth extraction procedure were recorded. In 73% of horses, MNB was successful. Sedation in the preceding 6 h (P = .732), age (P = .936), side of pathology (P = .516), and sino-nasal disease (P = .769) were not associated with total scores. Detomidine dosage and butorphanol use did not differ between horses in which the MNB was considered successful and for those in which it was not (P = .967 and P = .538, respectively). Scores obtained with gingival algometry were less closely associated with total scores (rho = .649) than those obtained with needle prick and nostril clamping (rho = .819 and .892, respectively). Therefore, needle prick and nostril clamping are considered the more reliable methods for use in clinical practice to determine the success of an MNB.
Introduction
Surgical procedures performed on sedated, standing horses do not have the risks or the cost of general anesthesia.1–3 Regional anesthesia is often used for surgical procedures to reduce both the sedation dosage and the amount of inhalant anesthetic as well as to reduce postoperative pain.4,5 In equine dentistry, oral tooth extraction on a standing, sedated patient has been shown to have lower complication rates than surgical extractions performed under general anesthesia.2,6–8 Other benefits of standing dental procedures include anatomic orientation, reduced hemorrhage, more ergonomic positioning for both the patient and clinician, and equal access to both sides of the head. 9 New, minimally invasive surgical techniques make it possible for more procedures to be performed on standing, sedated patients.10,11
The maxillary nerve is a sensory branch of the trigeminal nerve. It provides sensory function to the ipsilateral maxillary dental quadrant, soft tissues, and paranasal sinuses. Regional anesthesia of the nerve is utilized for oral and dental surgeries 12 and in diagnostic anesthesia of headshaking syndrome.13,14
There have been multiple studies describing the various approaches to maxillary nerve blocks (MNBs),12,14–21 some including ultrasound-guided techniques.18,19,21 Cadaveric studies have been used to assess accuracy levels.13,16,17,22 It has been suggested that the extraperiorbital fat body insertion (EFBI) approach can decrease the complication rate by reducing the risk of puncturing the deep facial vein, the infraorbital artery and the descending palatine artery. This approach can also decrease the risk of retrobulbar hematoma, collapse, and blindness. 17
Accurate placement of the MNB and anesthesia of the nerve is important for all of the reasons listed above as well as for the safety of the patient, support staff, and clinician. 15 Assessment of regional anesthesia prior to surgical stimulation by nociceptive threshold testing is a well-established practice.23–25 While nociceptive threshold testing of the equine head including tactile sensory, mechanical nociceptive (which includes pressure algometry), and thermal nociceptive threshold testing have been performed in the past, 26 gingival algometry is still not widely used in equine dentistry other than in one recent study, 22 despite the fact that studies in humans27–29 are promising. This assessment can be made ideally in horses without the effect of sedatives and/or analgesics. However, sedation is commonly required for the placement of a MNB and exodontia. 12 Sedation decreases the response to stimuli, which can confound the surgeon's assessment of effective regional anesthesia.
Knowledge of accurate MNB placement and confirmation of proper regional anesthesia prior to starting a dental procedure is advantageous. The authors hypothesized (i) that sensory function testing is an effective method for predicting a successful MNB, (ii) that sedation in the 6 h prior to sensory function testing and factors such as sino-nasal disease, patient age, and the side of the dental pathology do not have any significant effect on sensory testing or MNB success, and (iii) that overall doses of analgesic drugs used can be lower in animals with successfully performed MNB. Accordingly, the specific aims of this study were as follows: (a) to evaluate the adequacy of 3 different methods of sensory function testing in forecasting a successful MNB, (b) to determine the effect of sedative administration having been performed 6 h prior to sensory function testing, (c) assess the dose of detomidine administered throughout the exodontia procedure and the use of butorphanol and finally (d) to assess the impact of sino-nasal disease, patient age, and the side of the dental disease on the outcome of sensory function testing.
Materials and Methods
This study was designed as a prospective, blinded, cross-over trial and was approved by the University of Pennsylvania's Institutional Animal Care and Use Committee (No. 806340). Clinical cases presented to the University of Pennsylvania's New Bolton Center for oral extraction of a maxillary cheek tooth (Modified Triadan tooth numbering system: #106-111 or 206-211) were included in this study. Owner consent was obtained. To be included in the study, the horses must have had evidence of a nonvital maxillary cheek tooth on oral examination and evidence of apical disease, on either radiographs or robotic, cone beam computed tomography (CT). Horses had to be between 3 and 25 years of age and be between 200 and 650 kg body weight. Cases were excluded if they received detomidine in the 50 min prior to “Baseline” testing or had primary periodontal disease. Historical data, such as presence of sino-nasal disease, the side of the pathology, age, and administration of sedatives in the previous 6 h were recorded.
Patients were sedated with xylazinea (0.5 mg/kg) prior to performing the MNB. The MNB was performed utilizing the EFBI technique. Both sides of the head at the proposed injection site were aseptically prepared so that the blinded investigator could not recognize which side was blocked. After aseptic preparation, a Tuohy needleb was slowly inserted on the ipsilateral side of the head as the dental disease, at an of angle 90° to the surface of the skin, approximately 1 cm ventral to the zygomatic arch at the level where the arch curves medially. The needle was advanced through the masseter muscle and the medial fascia. When a sudden change in insertion resistance was noted, the needle was advanced 1 cm further into the extraperiorbital fat pad. A syringe was attached to the needle and gently aspirated. Twenty mL of 0.5% bupivacainec were injected in all patients regardless of body weight. Eye lubricantd was applied topically to the ipsilateral cornea of the MNB side after sensitivity testing.
Standing sedation for exodontia was initiated following the final MNB testing with an intravenous (IV) bolus of detomidinee (10-22 µg/kg) followed by IV boluses of detomidine (5-10 µg/kg) and/or butorphanolf (6-16 µg/kg) as needed to maintain adequate patient sedation and compliance during the exodontia procedure. The procedure length was recorded in minutes. The dose of detomidine in µg/kg/min was calculated according to the length of the procedure.
Patients were tested prior to sedation (Baseline), 5 min after receiving xylazine IV but prior to the MNB (Post Sedation), 15 min (15 min Post MNB), and then again 30 min after the MNB was performed (30 min Post MNB). The “Baseline” testing was performed in a stall. The remainder of the testing was performed in standing stocks. The patients were not stimulated and no additional sedation was administered between testing periods. Horses were tested by a blinded investigator (LZ). Any features that might have identified the diseased side, such as nasal discharge, draining tracts, or swellings were cleaned or covered. The horses were blindfolded during testing. The investigator started testing on the left side of each horse. The side not affected by dental disease served as the control when the investigator bilaterally performed the following 3 sensory function tests in the order listed below:
Clamp with a hemostat on the dorsum of each nostril. Prick with a 20-gauge needle 1 cm dorsal to each nostril. Gingival algometry pressure was measured with an analog dial force gaugeg on the gingiva 0.5 cm dorsal and 0.5 cm distal to the ipsilateral maxillary canine (Tooth #104 and 204).
The response to a needle prick and nostril hemostat clamping were scored by full response to stimuli: the patient immediately moves away from stimuli (score of 0); minor response to stimuli: the patient changes ear positioning or facial expression but does not immediately move away from stimuli and without noticeable difference compared with the contralateral side (score of 1); minor response to stimuli: the patient changes ear positioning or facial expression but does not immediately move away from stimuli and there is a noticeable difference compared with the contralateral side (score of 2); and no response to stimuli (score of 3). Gingival algometry force in kg/cm2 was recorded for each side. The gingival algometry force for the non-diseased (non-blocked) side was subtracted from the diseased (blocked) side. This difference in response to force was categorized as follows: less than 1 kg/cm2 (score of 0), 1 to 1.5 kg/cm2 (score of 1), 1.6 to 2 kg/cm2 (score of 2), and greater than 2 kg/cm2 (score of 3). A total score was determined by summing the individual scores for response to needle prick, nostril clamping, and gingival algometry.
The MNB was considered successfully performed if the total score on the blocked side had increased by ≥ 2 between “Baseline” or “Post Sedation” testing and at 30 min Post MNB. This threshold was determined with regard to the high variability of responses to nociceptive stimulation noted in the literature.24–26,30–32 Differences in total scores at “Baseline” and “Post Sedation”, but prior to MNB were also analyzed.
All analyses were conducted with Stata 16.1MPh, with two-sided tests of hypotheses and a P value < 0.05 as the criterion for statistical significance. Descriptive statistics were presented as medians and ranges of continuous variables in Figure 1 and categorical variables were tabulated. Test of normal distribution (Shapiro–Wilk test) was performed to determine the extent of skewness of data. Frequency counts and percentages were used for categorical variables. Fisher exact test was used to compare the pathological side as a binary variable and other independent categorical variables. Pairwise comparisons between needle prick, nostril hemostat clamping, and gingival algometry with total score were conducted using Spearman rank correlation. For the purpose of Inference statistical analysis, mixed-effects (ME) linear and Poisson regression were used. ME linear regression was used for assessing the gingival pressure force (in kg/cm2) outcome and ME Poisson regression was used for the hemostat clamp, needle prick, and gingival algometry score outcomes. For both models, the fixed effects were set as the interaction between time period and pathologic side. Random effects were set on the level of individual animal. Post hoc pairwise calculations were conducted in order to estimate marginal (model adjusted) means and differences. To adjust for multiple comparison, the least significant difference method was used.
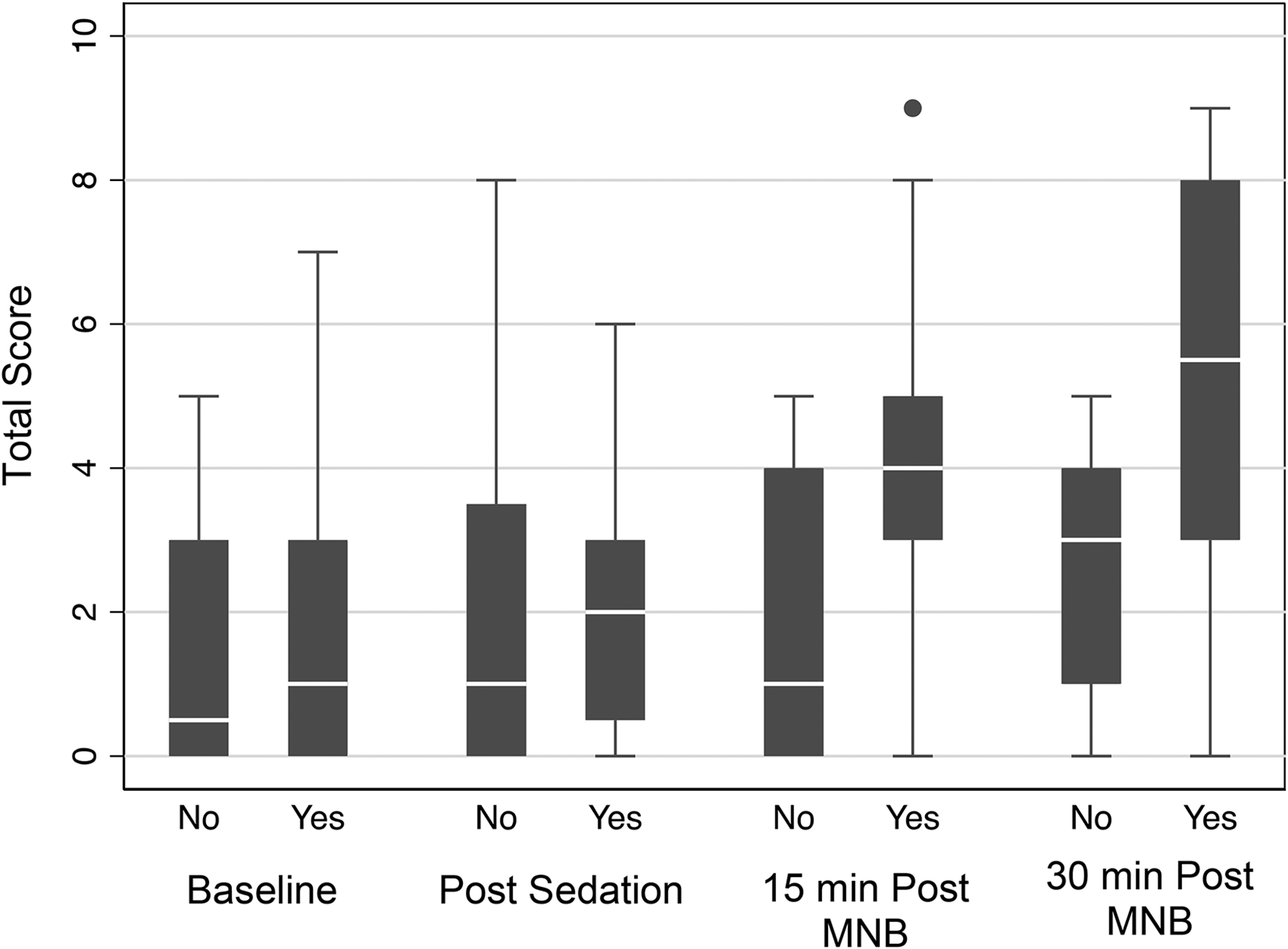
Median total scores at evaluation times for the nonpathologic/nonblocked (No) and pathologic/blocked (Yes) sides. The dot in the 15 min post maxillary nerve block (MNB) column at 9 represents an outlier.
Results
Perineural injection for the maxillary nerve via the EFBI technique was attempted 15 times on 15 patients. Six Thoroughbreds, 4 Warmbloods, 2 Crossbreds, 1 pony, 1 Andalusian, and 1 Haflinger were enrolled in the trial. Horses ranged in age from 3 to 25 years old and in body weight from 204 to 613 kg.
The study results are shown in Table 1 and Figure 1. There were no significant differences in total scores, at “Baseline” and at “Post-Sedation” on the diseased, ipsilateral side (P = .640) or the nondiseased, contra-lateral side (P = .406). In one of the horses, “Baseline” data was not obtained but “Post-Sedation” testing was performed. In 3 other horses, “Baseline” testing was performed but not “Post-Sedation” testing. In all remaining horses (11 of 15) complete sensory function testing was performed at all time points. On the contralateral, nondiseased, nonblocked side repetitive sensory function testing yielded similar results, with no significant differences in total scores between the 4 recordings (“Baseline”, “Post-Sedation”, “15-min Post MNB”, and “30-min Post MNB”).
P Values Determined for Comparisons of Scores Determined for Each Time Point. Bold Values Signify Statistically Significant Differences Between Total Scores Obtained at Different Time Points or for Different Sides (Pathologic vs nonpathologic Side).
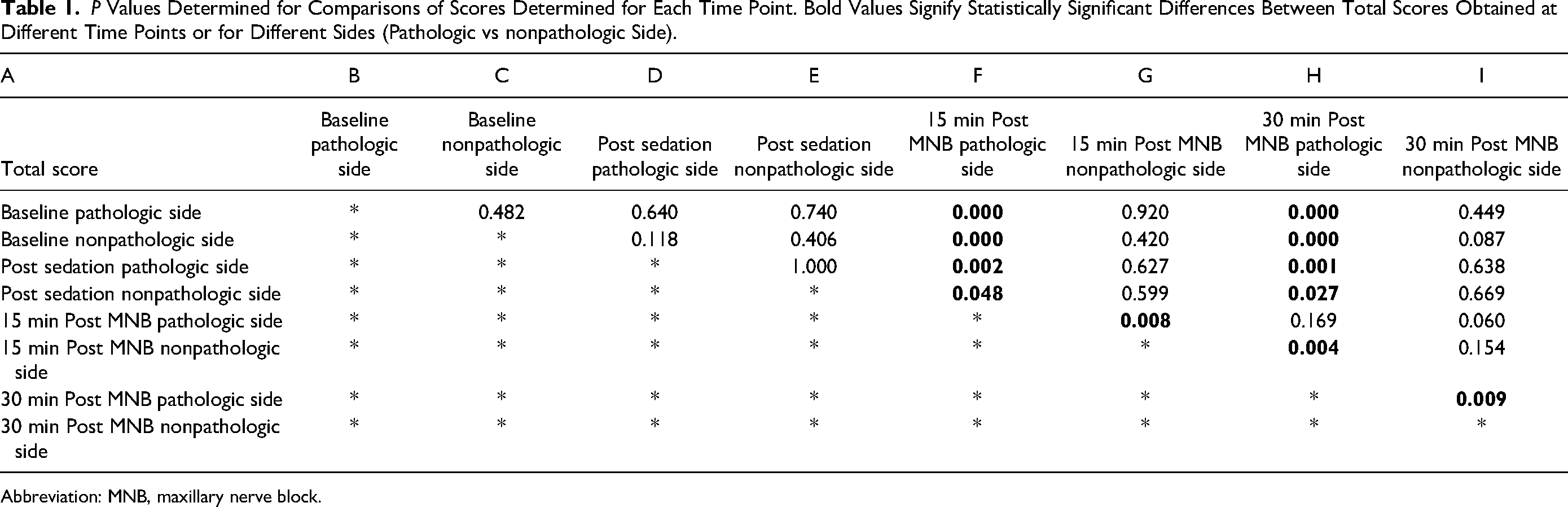
Abbreviation: MNB, maxillary nerve block.
In 73% (11 of 15) of all horses studied, the MNB performance was considered successful because sensory function testing on the pathologic, ipsilateral side revealed an increase by ≥ 2 in total scores when data recorded at “Baseline” or “Post Sedation”, were compared with those obtained at 30 min Post MNB. There were 2 horses that had an increase by ≥ 2 in total scores on the pathologic, ipsilateral side between “Post Sedation” and 15 min Post MNB later had a total score difference of < 2 at the time of 30-min Post MNB testing. These 2 cases were not considered to have a successful MNB per the predetermined criteria because they did not have a score difference of ⪴ 2 at the time of 30 min Post MNB testing. Two horses, in which a total score difference of < 2 was noted between “Post Sedation” and 15 min Post MNB testing, exhibited a total score difference of ≥ 2 at the 30 min Post MNB time point and were deemed to have a successful MNB by the predetermined criteria.
In 4 of the 15 horses, in which the MNB was considered unsuccessful, the average time required for the extraction procedure to be completed was 100 (35-173) min, while in the remaining 11 horses, in which the MNB was considered to be executed successfully, the extraction procedure lasted on average 63 (25-97) min. Still, the difference in the duration of the extraction procedure was not statistically significant between the 2 groups (P = .159). Likewise, there was no significant difference noted in the dose of detomidine per time unit that animals in either group received throughout the entire dental procedure period (P = .967). In the group with failure of the MNB, the mean detomidine dose amounted to 0.39 (0.23-0.59) µg/kg/min, and in the group of horses with successfully performed MNB it was 0.39 (0.24-0.52) µg/kg/min.
Butorphanol was administered during the exodontia procedure in 9 of 15 horses, including 2 of 4 horses where the MNB was considered unsuccessful and 7 of 11 of the horses where the MNB was considered successful. There was no significant difference in patients that received butorphanol in either group (P = .538).
Forty percent (6 of 15) of horses had a history of sedation in the 6 h prior to testing. One horse had received xylazine (1 mg/kg IV) 24 min prior to testing. Five horses were administered detomidine (13-18 µg/kg IV) a minimum of 50 min prior to testing (50-150 min). A history of sedation in the 6 h prior to testing had no significant effect on response to stimuli (P = .732).
Sixty percent (9 of 15) of cases had concurrent sino-nasal disease. Both sino-nasal disease (P = .769) and age (P = .936) were not associated with sensory function testing scores. There was also no significant difference regarding the side of pathology and MNB success (P = .516).
While all 3 sensory function testing methods, needle prick (rho = .819), nostril hemostat clamping (rho = .892), and gingival algometry (rho = .649), were associated with the total score, the methods with the highest rho values were needle prick and nostril clamping.
Discussion
The present study revealed 2 key findings: (i) sensory function testing is a suitable method for predicting the failure or success of an MNB, even if individual responses to nociceptive testing are quite variable among equine patients and (ii) a successful MNB does not necessarily help to reduce sedative drug requirements for safely performing a dental procedure (in this study detomidine and butorphanol).
Alpha-2 adrenoceptor agonists, such as xylazine and detomidine, are commonly used in equine dental practice and have been demonstrated to exhibit dose-dependent sedative and analgesic effects. 33 Therefore, patients in this study cohort were tested at “Baseline” and “Post Sedation” to identify any substantial effect of these drugs on sensory function testing. Sedation is commonly required to safely perform an MNB and previous studies have demonstrated difficulty in determining the success of an MNB in sedated patients. 34 However, in this study group there was no significant difference between total scores obtained at “Baseline” and “Post Sedation” when assessing sensitivity in the pathologic side or at any time point when assessing sensitivity in the nonpathologic/nonblocked side. This different finding was likely due to the relatively low dose of xylazine administered in this patient cohort to limit the degree of central nervous depression and analgesia (0.5 mg/kg IV; at the low end of a dose range published in the literature: 0.5-1.0 mg/kg IV). 33 In the study previously mentioned, 34 detomidine was used, and this has been claimed to have more profound sedative and analgesic effects than xylazine, 33 and therefore detomidine may have suppressed responses to noxious stimulation more effectively. In the present study, 40% of horses did receive detomidine, in order to perform an oral examination and obtain diagnostic radiographs prior to “Baseline” testing, but this dose was administered a minimum of 50 min prior to testing such that sedative effects had largely subsided by the time testing was performed.
The lack of significant differences between total scores obtained at “Baseline” and “Post Sedation” allowed the authors to include 4 horses in the data analysis in which sensory function testing was missed either at “Baseline” or “Post Sedation” time points (but not at both time points). These data points were missed in the initial phase of the study, as the study protocol was being refined.
Based on the described criteria, in the present clinical trial, 73% of the MNB procedures were effective. This rate is consistent with data from other studies reporting success rates ranging from 50% to 100%, with experienced operators being more successful (success rates of 69%-100%)13,16–18,21,22 than inexperienced operators (success rates of 40%-83%).13,19
Being able to confirm the successful execution of an MNB prior to starting a painful dental procedure is very valuable to the clinician. Prior studies in cadaver specimens have included injection of radiographic contrast medium followed by CT scan or injection of dye followed by tissue dissection to assess accurate block placement.13,16–19,21,22 However, the spread of dye or contrast medium in cadaveric specimen may not accurately represent diffusion patterns of local anesthetic solution in live animals. 16 Further studies are needed to assess the diffusion patterns in vivo. In the present study, a more accurate method of assessing the accuracy of local anesthetic drug disposition might have been the injection of radiographic contrast medium together with the local anesthetic followed by CT imaging of the skull. However, this was not a realistic option in the cohort of clinical patients used in this study and likely will not be an option in most clinical settings. In clinical cases, clinicians are limited to sensory function testing. There are several methods reported for sensory function testing in horses, including thermal, tactile sensory, mechanical, and electrical stimulation.25,29,31,32 Assessment of maxillary nerve blockade in horses has been performed by needle prick, 21 loss of cutaneous sensation, 18 and mechanical and thermal stimuli. 34 A study that investigated sensory testing of the equine face found tactile sensory stimulation to be one of the most reliable and reproducible testing methods when compared to thermal and mechanical noxious stimulation. 26 The present study also revealed differences in responses, dependent upon which sensory function test was performed and in what location: needle prick dorsal to the nares versus nostril clamping versus pressure stimulation of the gingiva (gingival algometry). Responses to mechanical stimulation of the gingiva were substantially more variable so testing scores were not as closely associated with the total sensory testing score (rho = .649) than those obtained using the other 2 sensory testing techniques (rho = .819 and .892 for needle prick and nostril clamping, respectively). The authors suspect this is partly due to the location of the mechanical stimulation and the manipulation of the lip required to place the dial force gauge on the gingiva of the canine tooth. Further studies should utilize remote-controlled nociceptive testing devices to avoid such confounding issues. 31 A benefit of mechanical threshold testing is the ability to stimulate both nociceptive (C fibers) and mechanoreceptive (Aδ) nerve fibers. 32 C fibers mediate the delayed, diffuse, and dull pain from noxious stimuli, 35 that is likely present in dental disease.
Limitations of sensory function testing are numerous and include variations in skin thickness and blood flow, local nociceptor distribution, stimulation of different nociceptors than those involved in clinical pain sensations, consistency in stimulus intensity both by operator and instrument, consistent grading of the response, and sensitization or habituation of the subject to the stimulus.23–26,30–32 Some horses may also respond violently to different forms of algometry. 32 All of these factors may have contributed to the substantial variability seen in total scores, as shown in Figure 1. This variation makes the interpretation of sensory testing scores as indicators of a successful MNB procedure rather challenging. Ideally, the periodontium and pulp horn structures could be tested for sensitivity, rather than just cutaneous or canine gingival tissues using algometry. In small animals, thermal and electric stimulation techniques have been used.36,37 In one study, horses were trained to tolerate an oral speculum without sedation. These horses were subsequently stimulated by the application of a sharp probe to the gingiva of the second and third maxillary premolars (106, 107, 206, and 207). They were also tested with swabs soaked in cooling spray and placed on the occlusal surface of the 106, 107, 206, and 207 teeth. These horses responded to both types of stimuli. However, when these stimuli were applied to sedated horses, they failed to produce any significant effect. 34
In 27% (4 of 15) of equine patients participating in the present study, the total score difference was < 2 at 15 min Post MNB (unsuccessful MNB). Two of those horses went on to show a total score difference of ≥2 at 30 min Post MNB (successful MNB). The authors assumed this development of increasingly less sensitivity to the stimuli was due to further diffusion of the local anesthetic toward the maxillary nerve but cannot exclude the possibility that the increase in total score between the 2 time points was just a manifestation of the sensitivity testing inconsistencies described above. Two horses showed a total score difference of ≥ 2 at 15 min Post MNB (successful MNB) that diminished to a difference of < 2 at 30 min Post MNB (unsuccessful MNB). Based on the predetermined criteria, those 2 horses were assigned to the group in which MNB was not successfully performed. Those cases, however, highlight the remaining difficulty in accurately determining MNB success.
There was no significant difference in detomidine dose administered per time unit, the use of butorphanol, or in length of time of the extraction procedure in horses in which the MNB was deemed successful versus those in which MNB was considered unsuccessful. However, there was a notable difference in average extraction procedure time between the two patient groups: 63 versus 100 min, even if it did not turn out to be statistically significant given the small number of patients in the not successfully blocked group. In addition, one has to consider that, independent of proper locoregional anesthesia, performance of surgical procedures in the standing horse, (especially in more fractious or stressed animals) requires an adequate level of chemical restraint, most commonly achieved by administration of an alpha-2 agonist, such as detomidine. Since detomidine is known to produce stronger and longer-lasting sedation compared to its analgesic effect, 38 it might not be surprising that the dose requirement per time unit did not differ between animals with or without successful locoregional analgesia. It is also still possible that in some cases that were categorized as not successfully nerve blocked that not enough time had elapsed between testing time points and that subsequent testing at 45 or 60 min Post MNB would have led to a different judgement.
Hypersensitization occurs when animals that are experiencing pain over extended periods of time become more sensitive to subsequent painful stimuli.23,25 Horses with sino-nasal disease secondary to dental pathology likely have a very chronic disease process. This study also attempted to determine whether horses with sino-nasal disease responded differently to sensory testing prior or subsequent to MNB. In this study cohort, the authors did not find any statistically significant association between sino-nasal disease and recorded testing scores.
Besides the limitations in sensitivity testing described above, a major limitation of this study was the small sample size and the lack of true control animals, in which saline instead of local anesthetic could have been injected into the extraperiorbital fat body. The use of saline instead of local anesthetic was not an option in this clinical trial and this was the reason why the side not affected by dental disease served as the control.
In conclusion, sensory function testing is useful for determining successful MNB in lightly sedated horses, with the understanding that there is much individual variability in response to testing. Based on the findings in the present study, needle prick and nostril clamping appear to be more reliable in detecting a significant decrease or complete loss of sensation following MNB. Both needle prick and nostril clamping with a hemostat are easily performed tests that only require materials that are readily available. Performing sensitivity testing bilaterally prior to the MNB would give the clinician a reasonably accurate assessment of pain sensation with which to compare responses obtained by testing Post MNB.
Materials
XylaMed, 100 mg/mL, Henry Schein, Dublin, Ohio.
BD Tuohy Epidural Needle, 9 cm, 20 g, Becton and Company, Belgium.
Bupivacaine HCl, preservative free, 5 mg/mL, Hospira Inc, Lake Forest, Illinois.
Puralube Vet Ointment, Dechra, Overland Park, Kansas.
Dormosedan, 10 mg/mL, Zoetis, Kalamazoo, Michigan.
Torbugesic, 10 mg/mL, Zoetis, Kalamazoo, Michigan.
FDK-20 Analog Dial Force Gauge, Cole-Parmer North America, Vernon Hills, Illinois.
Stata 16.1MP, StataCorp LLC, College Station, Texas.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
