Abstract
Oral administration of medication to experimental animals is a cause of significant stress. When coupled to animals who are already under strenuous circumstances due to the disease being modelled, there is a significant risk for increased morbidity and mortality, thus influencing the results. Faced with these constraints, a low-intensity method for oral administration was developed, based solely on the natural behaviour of the animals and minimal conditioning, in which precise doses of medication were administered in a locally available, standard wheat cookie fragment, providing both a palatable vehicle and an absorbent matrix for the medication. Fast administration to large numbers of animals was thus achieved, safeguarding the animals’ welfare and ensuring ease of handling. This method is a promising alternative to oral gavage in pre-clinical drug studies with laboratory mice.
Introduction
Mice models have emerged as the tool of choice for basic research. 1 They are easily stressed, and stress per se may modify food intake, energy expenditure, physical activity, and body composition.2–5 The degree of stress experienced by the animals may be influenced by the number of researchers involved in the animals’ manipulation, as well as their skill and experience working with mice. 6
When housed under controlled conditions of light–dark cycle (12h light/12h dark), mice consume most of their food (almost 70%) during the dark period, with short feeding bouts during the light period. Water consumption is linked to food intake and declines in fasted or food-restricted animals. Additionally to the light–dark cycle, mice feeding behaviour may be also influenced by other variables, like room temperature, humidity, noise, biological differences, and social stratification. 7
Oral administration of investigational medicinal products to mice is usually performed. Some compounds may affect the odour or flavour of food or water, causing animals to avoid consuming them and changing the amount of the test compound ingested by each animal. 8 Oral gavage is a widely used alternative method for administering test compounds to animals by introducing them in liquid form directly into animals’ stomachs using a special feeding needle.9–11 While experienced researchers are able to minimize the possibility of injury to the laboratory animal, the stress induced by the manipulation and insertion of the tubing cannot be ignored. 12
Several pre-clinical studies have been conducted using K14HPV16 mice, 13 a transgenic murine model which expresses the early region genes of the human papilloma virus type 16, featuring lesions that closely resemble those of human papillomavirus (HPV) infection.14,15 Considering the highly stress-prone nature of this model, we aimed to develop an effective method for the oral administration of water or oil-soluble compounds that minimizes handling-induced stress in laboratory mice.
Methods
Animals
The generation of K14-HPV16 mice on a FVB/n background was previously described. 16 K14-HPV16 mice were kindly donated by Drs Jeffrey Arbeit and Douglas Hanahan (University of California) through the USA National Cancer Institute Mouse Repository. The study was approved by the University de Trás-os-Montes and Alto Douro Ethics Committee (approval number 10/2013) and the Portuguese Veterinary Directorate (approval number 0421/000/000/2014). Animals were maintained in groups of four or five animals per cage (1284L Eurostandard Type II L, Tecniplast, Italy) in a ventilated room. They were bred according to the Portuguese (Decree-Law 113/2013) and European (Directive 2010/63/EU) legislation and maintained under controlled conditions of temperature (23 ± 2°C), light–dark cycle (12h light/12h dark), and relative humidity (50 ± 10%), using corncob for bedding and plastic tubes as environmental enrichment. Cages were cleaned once a week, and water was changed weekly. All animals had free access to a basic standard diet (4RF21, Mucedola, Italy) and tap water was supplied through capped water bottles ad libitum throughout the study. Animals’ body weight, food, and water were weighed once a week using a top-loading scale (KERN® PLT 6200-2A, Dias de Sousa SA, Alcochete, Portugal). The food and water consumption for each cage was calculated by subtracting the weight of the food container or water bottle at the end of the week from its weight at the beginning of the week. The general health status of all mice was monitored once a day for signs and symptoms of toxicity. For this, the following indicators of animal welfare were assessed: body condition (well, under conditioned, or emaciated), alertness state (normal, lethargic, stupor, moribund), response to external stimuli (normal, mild, moderate, or violent), posture (normal or changed), mucosal (normal, mild, moderate, or severe), and coat and grooming (normal, lack of grooming, rough coat, piloerection).
Genotyping
Animals were genotyped at weaning, using tail-tip samples as previously described.13,17 Briefly, nucleic acids were extracted, and DNA quality and purity were assessed. HPV16-E6 and E2 genes were amplified to confirm the presence of HPV DNA and a fragment of mouse β-globin was also amplified and endogenous control. Lengths of the amplicons were confirmed by agarose gel electrophoresis. Only hemizygous females were used for the transgenic groups of the experiment.
Experiments
Thirty-four female mice at four weeks of age were used. Animals were allocated to three groups: group I (n = 15) – wildtype mice not treated; group II (n = 9) – K14-HPV16 mice not treated; and group III (n = 10) – K14-HPV16 mice treated with celecoxib. All animals received a piece of cookie, five times a week, for four consecutive weeks. Animals from group III were treated with celecoxib at a dose of 2.5 mg/mouse/day. Each week, 125.0 mg celecoxib were dissolved in 10:90 (v/v) ethanol/distilled water and the individual 2.5 mg dose (µL ethanol) was added to a standard wheat cookie. Administration was performed by isolating each animal in a small cage, to absorb urine and avoid soiling the cookie. The animals were under permanent observation by a single researcher until completely consuming the cookie. Upon consumption, the animals were returned to their cages. During the experiment, the animals’ body weight, food, and water consumption were assessed once a week using a scale. The study lasted four weeks, beginning at the time of the first cookie administration and ending when the animals were humanely euthanized at eight weeks old by intraperitoneal pentobarbital overdose, followed by exsanguination by cardiac puncture.
Statistical analysis
Data were statistically analysed with Statistical Package for Social Sciences (SPSS) version 25 (Chicago, IL, USA) using ANOVA with the Bonferroni correction for multiple comparisons. Data are presented as mean ± standard deviation. P-values lower than 0.05 were considered statistically significant.
Results and discussion
Oral gavage is the most straightforward approach to achieve precise enteric administration in rodents, but it is associated with potential adverse consequences.12,18 Although the animal stress and mortality related to oral gavage can be minimized when the procedure is carried out by an experienced researcher, this procedure requires animals’ restraint, which may cause systemic stress response and consequently increase blood pressure, heart rate, and blood glucocorticoid levels.19,20 Stress may influence animal behaviour and confound experimental results. In some cases, serious complications have also been associated with oral gavage, including reflux, tracheal administration, aspiration, trauma, oesophageal irritation and perforation, pneumonia, and death.9,12,21–27
Our approach was to take advantage of the normal feeding behaviour of mice. Several food products were considered as candidate vehicles for the incorporation of drugs of interest in them and tested alone to assess their palatability. Of these, the most promising one was an easily obtainable, affordable, and commercially available variety of standard wheat cookie. 28 The cookie was found to be highly palatable, capable of holding structural integrity while being manipulated by the mice. A conditioning effect was also observed in which animals took progressively less time to find the fragment and consume it. In the first week the animals took 5–10 minutes to consume the cookie, and the consuming time decreased overtime. In the last week of the experiment, the cookie was immediately found and consumed, taking less than one minute. No changes in the indicators of animal welfare were observed.
Measuring approximately 5.0 cm in diameter and 0.4 cm in thickness, with a weight of about 6.3 g per unit, these cookies have a porous structure that allows the retention of a certain amount of fluids without compromising structural integrity. For standardizing purposes, the cookies were manually broken up into uniformly sized fragments (150 mg ± 10%), which were then used to retain commonly used solvents. The cookie’s energy value is 17.94 KJ per gram (∼270 J per dosing fragment), which was considered to have a negligible effect on energy intake, even assuming a daily administration. 29 The proposed solvents (distilled water or ethanol) were applied to the fragments by pipetting a maximum of 1 µl/mg, which allowed the fragment to remain solid, and dried overnight in an oven, at 40°C before being administered to the animals. Care is needed to apply the solvent slowly, ensuring that it is absorbed and does not fall off the surface. For heat-sensitive drugs, drying the cookies at room temperature may be advisable. Additionally, the impact of the cookie on pharmacokinetic parameters for each test compound may vary and its interference with celecoxib was not tested in the present study.
The administration by isolating each animal in a small cage allowed direct observation of complete consumption of the fragment (Figure 1). The technique allowed near simultaneous administration of the fragment to large numbers of individuals (up to 30 animals) in a very short time span, providing considerable time savings over gavage. Over 80% of the animals readily accepted the cookie fragment, and these were the ones chosen for incorporation into studies. We found that the addition of unmedicated cookie fragments had no impact on food and water consumption for the control groups (Figure 2). The transgenic mice presented a lower body weight when compared with wildtype mice, which has been associated with their health condition (Figure 3).30–32

K14HPV16 mouse ingesting the cookie fragment. Single frame from a video recording, with the mouse fully consuming the cookie fragment within 15 seconds.
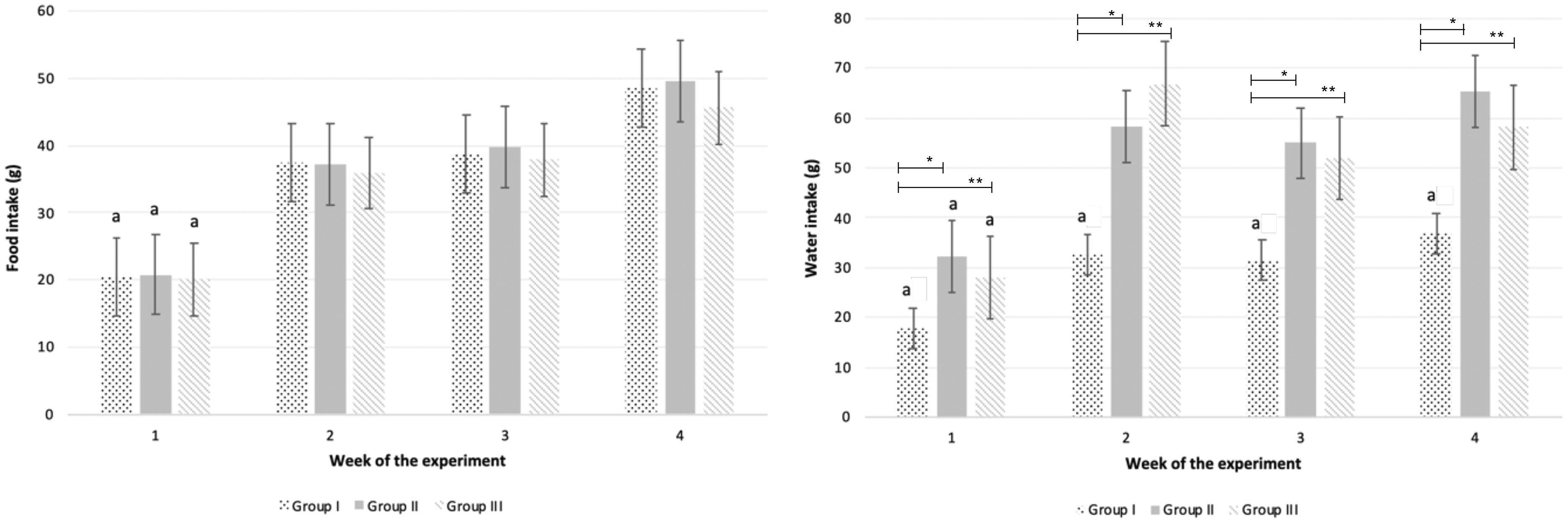
Variation of food and water intake per animal per week was addressed throughout the study in all experimental groups (group I – wildtype not treated; group II – K14-HPV16 not treated; group III – K14-HPV16 treated with celecoxib).
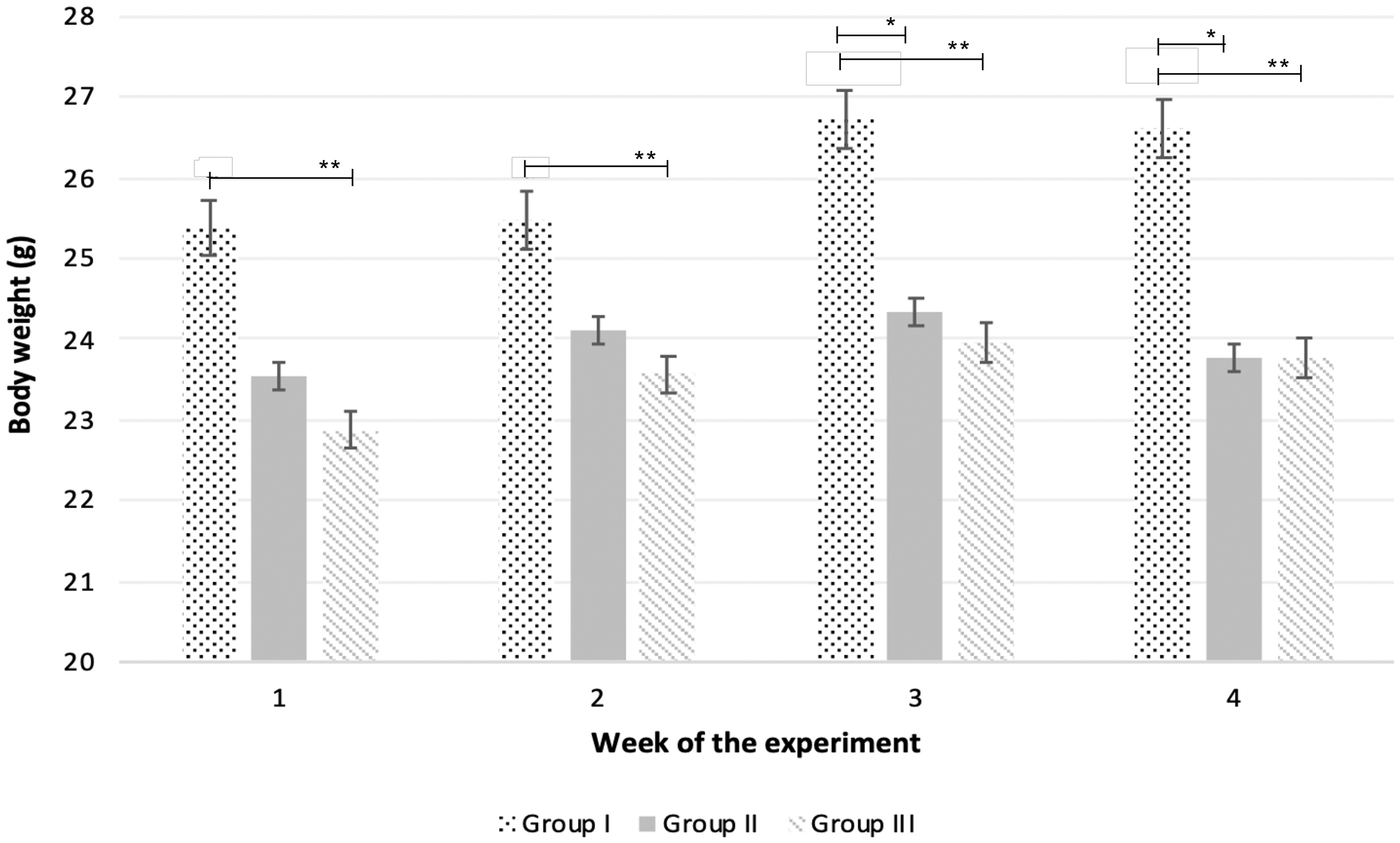
Variation of body weight throughout the study in all experimental groups (group I – wildtype not treated; group II – K14HPV16 not treated; group III – K14HPV16 treated with celecoxib). Differences between groups are represented by horizontal lines.
This work was only performed in female transgenic mice, which may constitute a limitation. More studies addressing the feed behaviour in males and in other mice models are warranted.
Conclusions
This method allows for the fast and efficient oral administration of compounds to mice, while inducing very little stress with animal manipulation. It constitutes a promising alternative to oral gavage in pre-clinical studies with laboratory mice.
Footnotes
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Research Center of the Portuguese Institute of Oncology of Porto (CI-IPOP 37-2016), by project POCI-01-0145-FEDER-006939 (Laboratory for Process Engineering, Environment, Biotechnology and Energy), projects POCI-01-0145-FEDER-006958 and UID/AGR/04033/2020, funded by FEDER funds through COMPETE2020 – Operational Competitiveness and Internationalization Program (POCI) – and by national funds through the Portuguese Foundation for Science and Technology (FCT). Rui M Gil da Costa received a grant from FCT, funded by the Portuguese Government and the Social European Fund (grant number SFRH/BPD/85462/2012).
