Abstract
This report describes two cases of dextran-induced anaphylactoid reaction (DIAR), with and without cutaneous signs that appeared after administration of dextran 40 during general anesthesia in domestic pigs. Dextran was administered intravenously to 59 pigs that underwent general anesthesia for routine medical device training, and two pigs developed sudden, severe hypotension and decreased end-tidal CO2 level and arterial oxygen saturation within a few minutes after the start of dextran 40 infusion. A systemic cutaneous lesion presenting as flushing and urticaria was observed in case 1, whereas no skin lesions were found despite persistent observation in case 2. Cases 1 and 2 recovered with thoracic wall compression and an intravenous injection of epinephrine (0.02 mg) and hydrocortisone sodium succinate (250 mg). After DIAR treatment, no complications were observed, and the pigs were euthanized with an intravenous injection of thiopental sodium (1.0 g) when the training program was completed. This case report discusses the appearance of DIAR after dextran 40 administration during general anesthesia in domestic pigs, which is similar to DIAR in humans. However, the incidence of DIAR in this study was much higher than that in humans, suggesting that dextran 40 should not be used in domestic pigs.
Case report
Dextrans are colloid plasma volume expanders used in human general anesthesia and occasionally in veterinary medicine.1–3 However, life-threatening dextran-induced anaphylactoid/anaphylactic reaction (DIAR) has been reported in human medicine. 3 DIAR is a serious anesthetic adverse event that is clinically diagnosed using cardiovascular, respiratory, and dermatologic criteria.3–5 However, DIAR has not yet been reported in pigs.
In our facility, dextran was administered intravenously to 59 pigs that underwent general anesthesia for medical doctors to learn the proper usage of medical devices (e.g., stents, rotational atherectomy devices, and abdominal coils) between February 2018 and November 2019. All training procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the Institute for Advancing Science Miyazaki (IAS Miyazaki), Boston Scientific Japan, and were conducted in accordance with the Guidelines for Proper Conduct of Animal Experiments by the Science Council of Japan. All pigs were female, and the median age was 15 weeks (range, 12–20 weeks). The breeds of the pigs were as follows: Duroc, 7; Landrace, 22; Large White, 22; Large White/Landrace, 7; and Landrace/Large White, 1. All pigs were obtained from the same breeder (Farmtech, Miyazaki, Japan). DIAR occurred in 2 of the 59 pigs within 5 min of dextran 40 administration.
Case 1
A healthy 14-week-old female Large White pig weighing 46 kg was anesthetized by intramuscular injection of ketamine (5 mg/kg) (Ketalar™; Daiichi-Sankyo, Tokyo, Japan), medetomidine (60 μg/kg) (Dorbene™; Kyoritsu-Seiyaku, Tokyo, Japan), and butorphanol (0.2 mg/kg) (Vetorphale™; Meiji-Seika-Pharma, Tokyo, Japan) followed by tracheal intubation. After moving the pig to the operating room, anesthesia was maintained using 1.5%–2.0% isoflurane (Isoflurane for animals; MSD Animal Health, Tokyo, Japan) and 50% O2 mixed with air. By means of positive pressure respiration, the end-tidal CO2 (EtCO2) was adjusted to 30–40 mmHg. A sheath-introducer (8F, Super Sheath™; Medikit, Tokyo, Japan) was placed in the left femoral artery to measure the arterial blood pressure (ABP) and also in the right femoral artery to deliver the devices necessary for the procedure. A sheath-introducer (6F, Super Sheath™) was placed in the left femoral vein to administer saline or medications. Electrocardiography (ECG), heart rate (HR), ABP, and arterial oxygen saturation (SpO2) were monitored using a CARESCAPE™ Monitor B650 (GE Healthcare, Little Chalfont, UK). Monitored data were automatically recorded using an e-Anereco Lite AR-U900 (Fukuda Colin, Tokyo, Japan) every 30 s. During the training for medical devices, dextran 40 solution (7 ml/kg/h) (low molecular dextran L injection, Otsuka Pharmaceutical, Tokyo, Japan) was administered into the femoral vein because the anesthesiologist noticed blood leakage from the catheter and a mild decrease of <10 mmHg in mean ABP. Prior to the infusion, HR, ABP (systolic/diastolic (mean)), EtCO2, and SpO2 were 116 beats/min, 86/57 (67) mmHg, 36 mmHg, and 98%, respectively. Approximately 4 min later, the anesthesiologist noticed a sudden decrease in ABP (39/32 (34) mmHg) and EtCO2 (20 mmHg), immediately followed by a decrease in SpO2 (66%) (Figure 1). HR did not change at this point (114 beats/min). Complications associated with interventional procedures such as perforation were not observed under fluoroscopy; however, the sudden change in vital signs alerted the anesthesiologist to a potential case of DIAR. Dextran was immediately discontinued. Simultaneously, epinephrine (0.02 mg) (Bosmin™ injection, Daiichi-Sankyo) and hydrocortisone sodium succinate (250 mg) (Solu-Cortef™; Pfizer, New York, NY, USA) were administered with a bolus of saline (20 ml) in the femoral vein, and the clamp of the saline infusion line was fully opened. The inspired oxygen concentration was increased to 100%. The anesthesiologist requested the surgeon to stop the procedure, and the drape was removed. Systemic skin flushing and urticaria were observed by the anesthesiologist. The ABP had dropped to 13/13 (13) mmHg at this point, and chest compression was started at a frequency of approximately 100/min. At 2 min after administration of epinephrine, ABP increased to 195/121 (146) mmHg, followed by an increase in EtCO2 (44 mmHg) and SpO2 (98%). HR also increased to 141 beats/min. The ABP and HR gradually decreased and returned to their former state within 20 min. After the recovery of ABP, the O2 level gradually decreased to 50%. The draping was repositioned on the pig, and training was continued. The anesthesiologist checked the skin symptoms every 10 min, and systemic flushing and urticaria were gradually reduced at 20 min after the DIAR reaction and disappeared completely 40 min later. No further complications were observed during the rest of the training. The pig was then euthanized with an intravenous injection of thiopental sodium (1.0 g) (Ravonal™; Nipro ES Pharma, Osaka, Japan). No autopsy was performed.
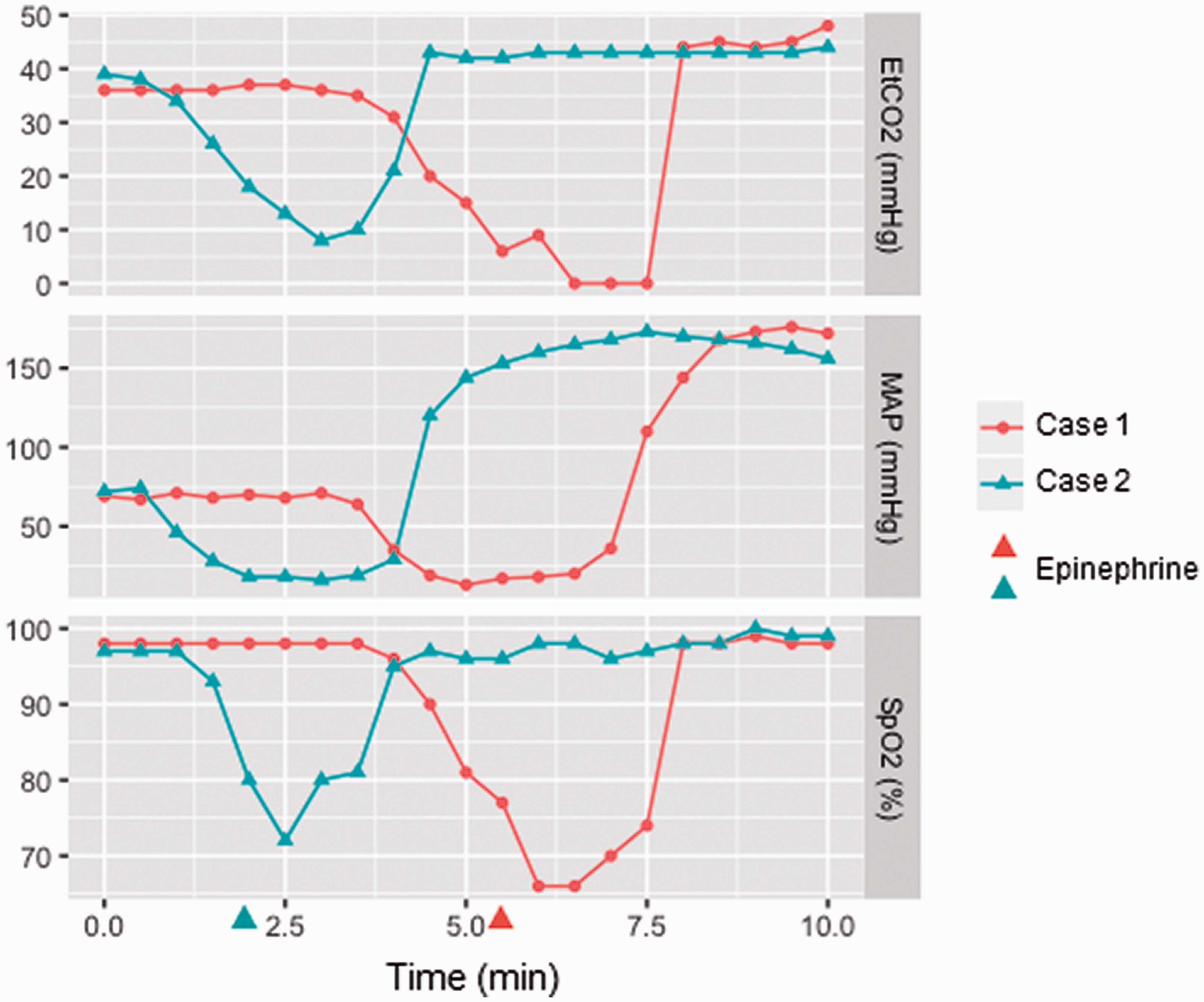
The end-tidal CO2 (EtCO2), mean arterial blood pressure (MAP), and arterial oxygen saturation (SpO2) after administration of Dextran 40.
Case 2
A healthy 16-week-old female Landrace/Large White pig weighing 48 kg was anesthetized and maintained using the same methods as in case 1. The sheath introducers were inserted in the same position as in case 1. During anesthesia, the administration of dextran 40 solution (7 ml/kg/h) was started to compensate for blood leakage from the catheter. Prior to the infusion, HR, ABP, EtCO2, and SpO2 were 107 beats/min, 98/54 (69) mmHg, 39 mmHg, and 97%, respectively. Approximately 2 min later, ABP, EtCO2, and SpO2 suddenly decreased to 31/25 (27) mmHg, 26 mmHg and 80%, respectively (Figure 1). The anesthesiologist suspected DIAR and immediately discontinued the administration of dextran 40. Epinephrine (0.02 mg) and hydrocortisone sodium succinate (250 mg) were administered with a bolus of saline (20 ml) into the femoral vein, and the saline infusion line was fully opened. The drape was removed, and chest compression was started at a frequency of 100/min. No cutaneous signs associated with DIAR were observed. Approximately 3 min after chest compression was initiated, ABP, EtCO2, and SpO2 increased to 194/96 (129) mmHg, 43 mmHg, and 97%, respectively. Although the training was resumed, the surgical drape was not replaced in the upper half of the body to monitor for skin lesions. No additional complications were observed, including cutaneous lesions, for the duration of the procedure (7.5 h after DIAR). The pig was then euthanized with an intravenous injection of thiopental sodium (1.0 g). No autopsy was performed.
Discussion
This case report demonstrates that DIAR occurs rapidly after the administration of dextran 40 in two domestic pigs. Anaphylaxis is usually diagnosed based on the appearance of cardiovascular, respiratory, and dermatologic lesions in human medicine.4,5 One case report that described suspected anaphylaxis caused by ketamine in three pigs showed that all cases presented with signs similar to those in humans, such as cutaneous lesions, respiratory disorder, and hypotension. 6 These signs are also believed to be important in diagnosing anaphylactoid reaction in pigs. In the present case report, sudden decreases in ABP and EtCO2 were observed without a change in HR. This shows that the cardiac output was diminished because EtCO2 is strongly related to a logarithmic transformation of the cardiac output. 7 However, respiratory signs such as tachypnea or dyspnea did not occur because breathing was maintained by positive-pressure respiration in both pigs. To detect respiratory signs such as bronchospasm, spirometry and/or changes in ventilator performance should be recorded. Regarding skin lesions, case 1 showed systemic flushing and urticaria, whereas case 2 did not show any skin lesions. Skin manifestations are important in diagnosing anaphylaxis in humans. 5 However, cutaneous manifestations occur in only approximately 70% of anaphylactoid reaction during anesthesia. 8 The most common signs initially observed are pulselessness, ventilation difficulty, and oxygen desaturation. 8 When the shock occurred in case 2, administration of epinephrine and hydrocortisone sodium succinate was performed immediately. Although it seems unlikely, the epinephrine or hydrocortisone may have masked the development of cutaneous lesions. This report showed that there are other ways to identify anaphylaxis during general anesthesia in addition to skin manifestations in domestic pigs. In fact, the anesthesiologist noticed DIAR due to severe hypotension, low EtCO2, and low SpO2 in both cases. Moreover, because the skin is often covered with a surgical drape, the anesthesiologist cannot always observe the skin. 8 Therefore, blood pressure monitoring, capnography, and pulse oximetry measurements are also important sources for detecting DIAR during general anesthesia.
A comprehensive study examining the rate of DIAR in humans under general anesthesia reported that the incidence rate was 13/100,000 (0.013%). 4 The incidence of DIAR in pigs at our facility was 2/59 (3.4%). Most piglets are treated with iron dextran to prevent the development of anemia. 9 In fact, all of the pigs in this study were treated with 200 mg of iron dextran (Tonkey 200; Fujita-Seiyaku, Tokyo, Japan) after birth according to the breeder. Moreover, all pigs used in our facility were brought from the same breeder and were expected to be genetically related. Genetic factors may also have been involved in the development of DIAR in the two pigs because anaphylaxis is affected by genetic factors. 10
Obtaining pigs that have received other iron supplements may prevent DIAR. However, it is important to avoid the use of dextran 40 altogether in pigs. In humans, hydroxyethyl starch (HES) is used widely instead of dextran for volume expansion. 11 Conversely, HES is used in pigs to evaluate resuscitation in an animal model. 12 No serious adverse effects of HES have been reported; however, the use of HES cannot be recommended because of limited information. To prevent adverse effects, the administration of crystalloids (e.g., ringer-acetate solution) is recommended.
This case report highlights two cases of DIAR in pigs. Monitoring the patient/animal during general anesthesia is critical for detecting DIAR. However, it is important to avoid using dextrans.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
