Abstract
The main microbial contaminants of rooms in which laboratory rodents are housed are bacteria and fungi. Restriction of microbial growth to below threshold levels requires the application of various sophisticated antimicrobial techniques that must be effective and safe for the animals. Some of the most commonly used techniques, including chemical disinfection, ventilation, filtration, sterilization and radiation, are not always sufficiently effective. The aim of the current study was to evaluate the efficacy of a modern technique (i.e. radiant catalytic ionization (RCI)) on the microbiological status of an animal care facility, and the health of the mice housed therein. The experiment, conducted over seven days, compared an experimental room with an RCI system permanently turned on with a negative control room. At the completion of the experiment, the number of bacteria in the RCI room air and on its walls was lower than that in the control room (p < 0.01 in both cases). Values of the basic prooxidative parameter, thiobarbituric acid reactive substances concentration, in tissues of mice from the RCI room were within allowed boundaries. Hence, application of an RCI system proved to be an ideal technique to ensure high hygienic standards in animal rooms without any adverse effects on the animals housed therein.
Introduction
The microbiological conditions in animal rooms significantly influence the welfare of laboratory animals, and the validity and repeatability of experimental results.1–3 Accidental infection among rodents used in biomedical experiments constitutes a considerable problem, even affecting animal facilities with high hygienic standards. Sources of accidental infection vary, but can include incoming animals, food sources and visitors. 4
For the aforementioned reasons, most laboratory rodent colonies are maintained in specific-pathogen-free animal barrier facilities. 5 A barrier is a housing system that combines construction features, equipment and operating methods to provide a stabilized enclosed environment, minimizing the probability of pathogens or other undesired organisms coming into contact with laboratory animals living within the barrier. Barriers may be installed at facility, room, rack and cage levels. 6 From a safety viewpoint, the most important barrier control systems are ventilating and decontaminating devices. Accordingly, several air-cleaning techniques have been employed to remove microorganisms. Filtration is one of the most effective and reliable means of removing particulate matter and bioaerosols from a gas stream. Ventilation systems in Animal Biosafety Level 3 animal houses produce cascade pressure that allows air to flow in only one direction (i.e. from outside to inside the room), ensuring a constant level of all microorganisms. In every room, the entire air volume is exchanged at least 12 times per day. To prevent microorganisms from entering a room, ventilation ducts are equipped with class H13 or H14 high-efficiency particulate air (HEPA) filters. H13 filters remove 99.95% of particles 0.3 µm in size, while H14 filters remove 99.99% of particles 0.3 µm in size.
Biological safety in laboratory rodent facilities increases when individually ventilated cage (IVC) rack systems are used. In a typical IVC rack, HEPA-filtered air is provided to each cage via a pressurized air system. This protects the animals from airborne infections and other noxious environmental particulates.3,7 IVCs can be equipped with HEPA filters at both incoming and outgoing ventilation ducts. However, microorganisms may grow on the surfaces of these filters and emit metabolites (e.g. harmful, volatile organic compounds, or biotoxins).8,9 Filters can be pre-treated with antibacterial agents (e.g. tea tree oil, 10 silver nanoparticles 11 or flavonoids 12 ) to inactivate microorganisms. Another method for protecting filter surfaces from microorganisms involves combining HEPA filters with photocatalytic systems. 13
Temporary sterilization systems that use hydrogen peroxide (H2O2) at high concentrations are also used. To decontaminate laboratory rooms, modern systems use highly concentrated (i.e. 30–35%) gaseous (vaporized) H2O2 in addition to classic UV-C (ultraviolet radiation within the wavelength range of 280 to 100 nm) radiation. The application of such gas or ultraviolet (UV) radiation requires that no animals or humans be present in the treated room. Compared with other surface sterilants, such as chlorine dioxide (ClO2) or formic acid (HCOOH), vaporized hydrogen peroxide has a better safety profile and a higher permissible exposure limit. It is safe on many surfaces, and results in no substantial changes in their physical or chemical properties. 14
Laboratories that conduct investigations with immunodeficient animals require efficient prophylactic hygiene strategies. The biological balance in rooms with animals can be ensured by continuous cycle systems that safeguard the active (i.e. not passive) reduction of undesired airborne or surface-borne microorganisms. The only antimicrobial technology allowed in animal facilities in the presence of living organisms is radiant catalytic ionization (RCI), which produces H2O2, which inhibits the growth of airborne bacteria, fungi and viruses. RCI has been proven to act against biofilm formation. 15 It can operate safely in occupied spaces, preventing recontamination in real time. In 2020, a healthcare-specific air purifier based on RCI technology was registered and approved by the US Food and Drug Administration as a Class II Medical Device. 16 The RCI technique emits a concentration of H2O2 that does not exceed 0.0328 ppm, which is safe for humans and animals. 17 The permitted concentration of H2O2 in the air is 1 ppm. 18 The primary adverse affect associated with high dose H2O2 is local irritation, which has been found to occur in workers after daily exposure to H2O2 concentrations of 1.7–3.4 mg/m3. Exposure to H2O2 at concentrations of 10 mg/m3 for 28 days was found to cause necrosis in rat nasal passages; however, no effects were found to occur at 2 mg/m3. 19 The small doses of reactive oxygen species generated during RCI activity can cause oxidative damage to viral and bacterial genetic material, and the photocatalysis process damages viral capsid proteins and oxidizes coenzyme A molecules in bacterial cells, resulting in inhibition of cellular respiration pathways. RCI can also oxidize unsaturated phospholipids and damage cell membranes.20–22 Although RCI is a very modern and useful approach to disinfection, it requires further investigation with regard to its efficacy and animal safety to achieve worldwide acceptance. In another separate study from our group, the results of the total number of airborne microorganisms evaluated by the sedimentation method and the ammonia and dust concentration in the air were promising (unpublished data). This data can be significant for scientists dealing with both laboratory animals and farm animals. Therefore, they have been prepared in a separate manuscript which is currently under revision. The study presented here aimed to determine whether the use of an RCI system in a rodent housing facility could improve existing environmental conditions, especially with regard to the overall number of microorganisms, without affecting the health of laboratory mice. This article describes the results of an investigation of the total number of airborne microorganisms determined by the ‘impact method’.
Materials and methods
Animals and housing
A seven-day study was carried out in an animal facility, using two identical rooms (58.8 m3), as negative control and RCI treatment rooms. Eighty male and 80 female four-week-old mice from an outbred colony of L and C selection lines derived from inbred A/St, BALB/c, BN/a and C57BL/6J strains were allocated to each room. The average weights of mice in the control and RCI groups were 11.58 g ± 2.3 SD and 11.26 g ± 1.73 SD, respectively.
The specified sex and number of mice was determined by colony size and structure. This colony was registered with the Polish Ministry of Science and Higher Education (No. 039) and has been under constant supervision by the Veterinary Inspectorate and the Warsaw University of Life Sciences Welfare Commission. This experiment did not include any procedures that required approval from the Ethical Committee for Animal Experimentation of the Polish Ministry of Science. At the end of the experiment, 20 animals (five males and five females per room group) were randomly selected (simple random sample) and euthanized as part of the routine culling of animals from the breeding herd.
Mice were housed in groups of four animals per conventional plastic open top cage with dimensions of 365 mm × 207 mm × 140 mm on a stainless-steel rack (Tecniplast, Milan, Italy). TierWohl Super (Rettenmaier) bedding was used. This bedding is thermally sterilized and is certified dust-free. For environmental enrichment, paper tubes and additional bedding materials were supplied to each cage. The animals were housed in humidity- and temperature-controlled rooms (45–65% RH and 20–24°C) under a 12-h light cycle with free access to water and complete food (Labofeed H, Morawski, Poland). Both rooms were surfaced with ceramic tile on walls and floors. Room ventilation provided 18 complete air exchange cycles of 1100 m3 of fresh air per hour. The intake duct of the ventilation system was equipped with standard F8 fine filters (EN 779:2002). 23 One day before the start of the experiment, routine maintenance was carried out in the rooms (i.e. litter replacement, wall and floor washing). Laboratory staff came into the room once a day to replace water and food and perform an animal health check.
RCI device (AP 3000) description and action
The structure of the RCI cell was optimized for purified air flow. In the AP 3000 (ActivTek Sp. z o. o., Kielce, Poland), air flow was produced by a fan (220 V, 50/60 Hz, 50 W) with a 120 mm diameter. The maximum air capacity of the device was 233 m3 h−1. The RCI device consisted of a matrix of elongated tubular polycarbonate components arranged in parallel in a honeycomb-like pattern. The ActivTek device was equipped with two matrices consisting of 406 tubular components. Each tube had a 4 mm diameter and a 15 mm length. The total active surface area of both matrices was 763.28 cm2. The coating of the basic matrix elements showed hydrophilic properties and contained TiO2. Opposite to this system, an 8-W power UV lamp GPH118T5L/4 was the source of wide-spectrum UV radiation. The UV lamp used argon gas with mercury and carbide fibres (100 and 367 nm). This UV lamp generates UV rays with wavelengths of 185 and 254 nm. As a result of catalytic oxidation stimulated by UV radiation, reactive oxygen forms were generated at the boundary of the heterogeneous phases. The total number of generated ions was ∼50 × 104 ions per cm3 of air. Detailed information on RCI devices and their technology is available in Patent No. US 8,585,979 B2. RCI cells produce low levels of H2O2 gas and superoxide (O2–). Ozone production was turned off during this study. The Ap 300 device was placed in the experimental room 1 m above the floor, facing the entrance door (Figure 1). During the seven days of the experiment, the unit was permanently turned on.
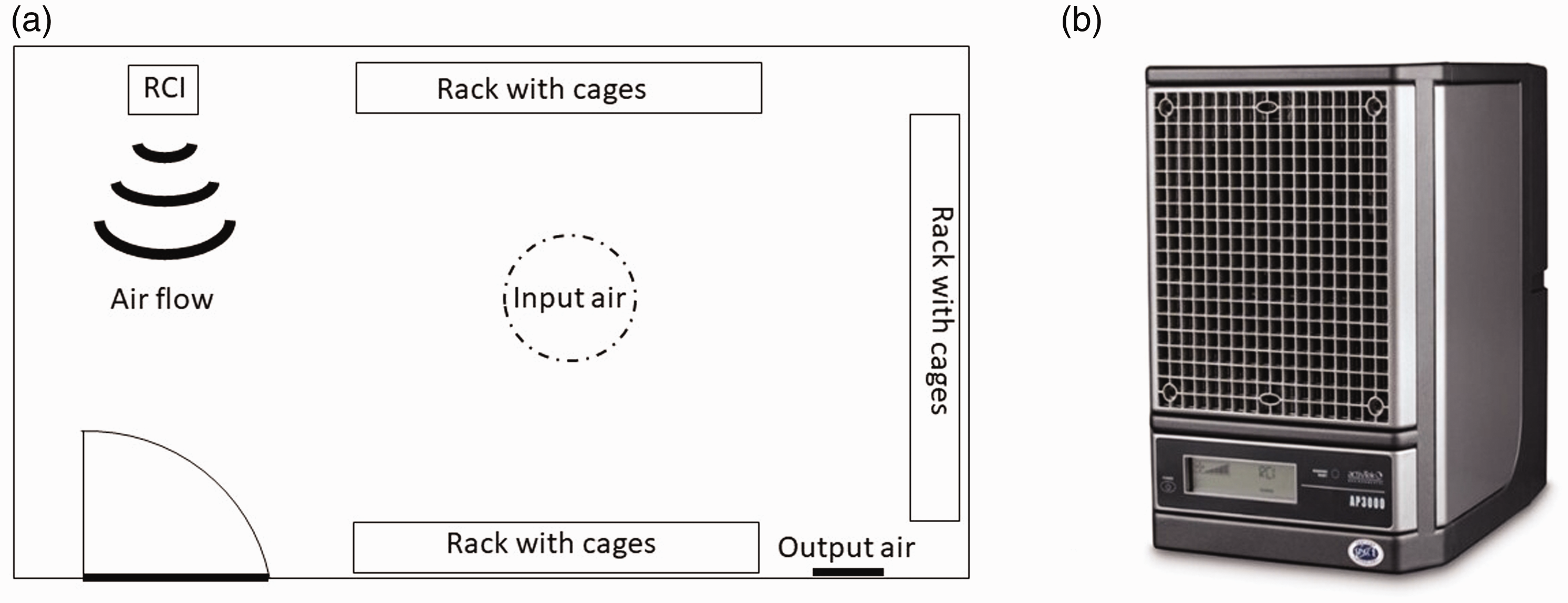
(a) RCI room projection with location of all elements, that is, door, racks and RCI instrument, as well as air flow input and output. (b) A depiction of the RCI device located in the upper left corner of the projection.
Sample collection
On the first and seventh days of the experiment, air and surface samples were collected to determine total numbers of bacteria and fungi. Animal tissue samples for morphological and biochemical analyses were collected on day 7 of the experiment.
Microbiological approach
Air
Microbiological analyses of air samples were carried out twice, once before the RCI was turned on, and once after completion of the seven-day operation period. Samples were collected from three sampling points located 1 m above the floor using a microbial air sampler MAS 100 Eco (MERCK, Burlington, USA). Air samples were cultivated on Columbia agar with 5% sheep’s blood (CAB; Becton Dickinson) and on Sabouraud agar (SAB; Becton Dickinson). Plates arrived at the laboratory after 4 h at 25°C. CAB plates were incubated for 48 h at 37°C, and SAB plates were incubated at 37°C for 24 h, then transferred to 25°C for four days. Colonies were counted using a Galaxy 330 colony reader. Results are expressed as numbers of colony-forming units (CFUs) in 1 m3 of air.
Surfaces
For every space/surface evaluated, microbiological analysis was carried out twice, once before the RCI was turned on and once upon experiment completion. Samples were collected using swabs from the floor (five points, 25 cm2 each, in the corners and at the intersection of room diagonals), walls (two points of 25 cm2 each on every wall at a height of approximately 1 m above the floor) and the joints between the floor and walls (one point halfway along the length of the wall, 25 cm above the floor). Samples were collected in duplicate. Swabs were delivered to the laboratory within 4 h. Swabs were immersed in 10-cm3 sterile solutions of phosphate buffered saline (PBS; BTL) and shaken thoroughly for 15 min. Next, serial dilutions (100–10−6) were carried out using sterile PBS. Two independent series were produced (100 µl each) for each sample. The resulting dilutions were cultivated on CAB and SAB plates and incubated as previously described. The results are expressed as numbers of colonies per 1 cm2 of the examined surface.
Lipid peroxidation status
On completion of the experiment, selected mice were euthanized by cervical dislocation, conducted by experienced staff. The animals were euthanized during routine thinning of the breeding colony. Blood samples were collected into heparinized tubes and centrifuged to separate erythrocytes from the blood plasma. Liver, heart, brain and lung tissue samples were collected using standard techniques. Both blood plasma and tissue samples were frozen and stored at −80°C for further analysis.
Lipid peroxidation indices
The level of malondialdehyde, the most abundant lipid peroxidation product, was measured using thiobarbituric acid (TBA) according to the technique described by Uchiyama and Mihara. 24 Absorbance at 535 nm was measured with a Tecan Infinite M200 (Mannedorf, Switzerland) analyser and represents the concentration of TBA reactive substances (TBARSs) in the samples. Tissues were homogenized in 1% KCl and centrifuged at 2000 × g for 15 min at 4°C. Supernatants were used for analyses and tissues were placed in a reaction solution (1% phosphoric acid, 2% butylated hydroxytoluene, 1% potassium chloride and 0.4% TBA). The solution was stored at 95°C for 60 min prior to analysis.
Statistical analyses
The software packages Statistica 9.1 (Statsoft, Tulsa, OK, USA) and Statgraphic 4.1 (StatPoint, Inc., Herndon, VA, USA) were used for microbiological and health status data processing, respectively. The microbiological data did not display normal distribution; hence, non-parametric median tests (Mann–Whitney U and Kruskal–Wallis) were used to compare experimental groups. Health status data (Gaussian normal distribution) were processed by one-way analysis of variance (ANOVA) and Duncan’s multiple range test. Differences were considered significant at p < 0.01.
Results
Microbiology of the environment
Numbers of CFUs in control and RCI rooms are presented in Figures 2–4. In the experimental RCI-exposed room, a statistically significant (p < 0.01) reduction in airborne bacteria and fungi was recorded (see Figure 2(b) and (d) for bacteria and fungi, respectively). In contrast, the total number of airborne microorganisms increased in the control room throughout the experiment, although the increase was not statistically significant (p < 0.01; Figure 2(a) and (c)).
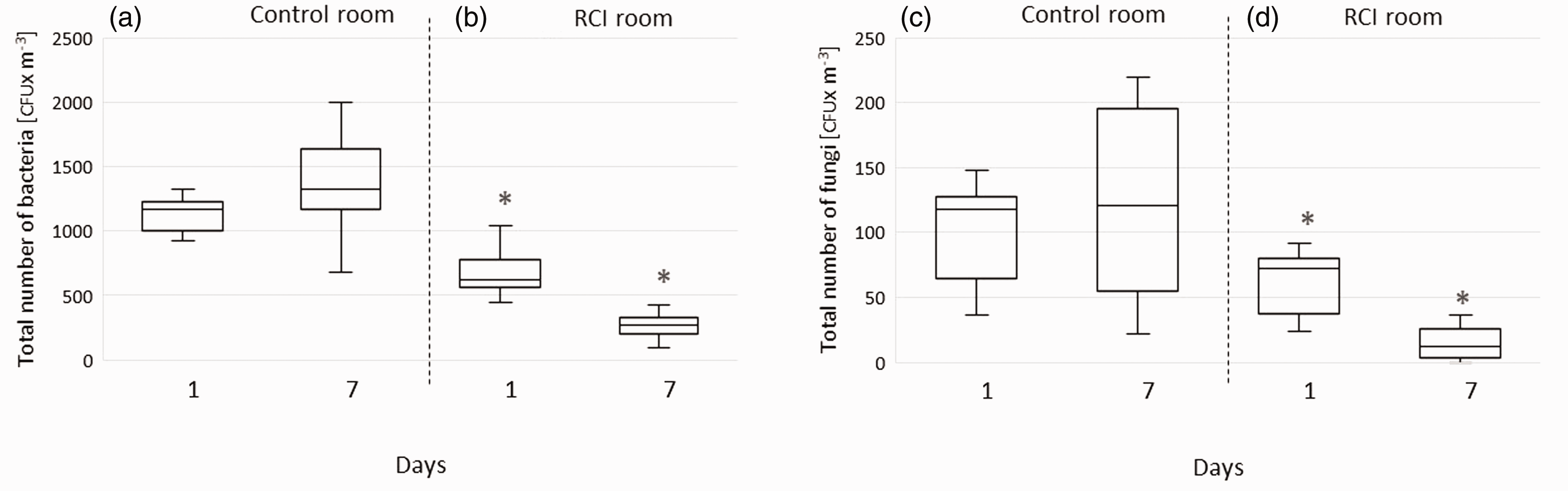
CFUs for airborne bacteria and fungi in control and RCI rooms on the first and seventh days of the experiment.
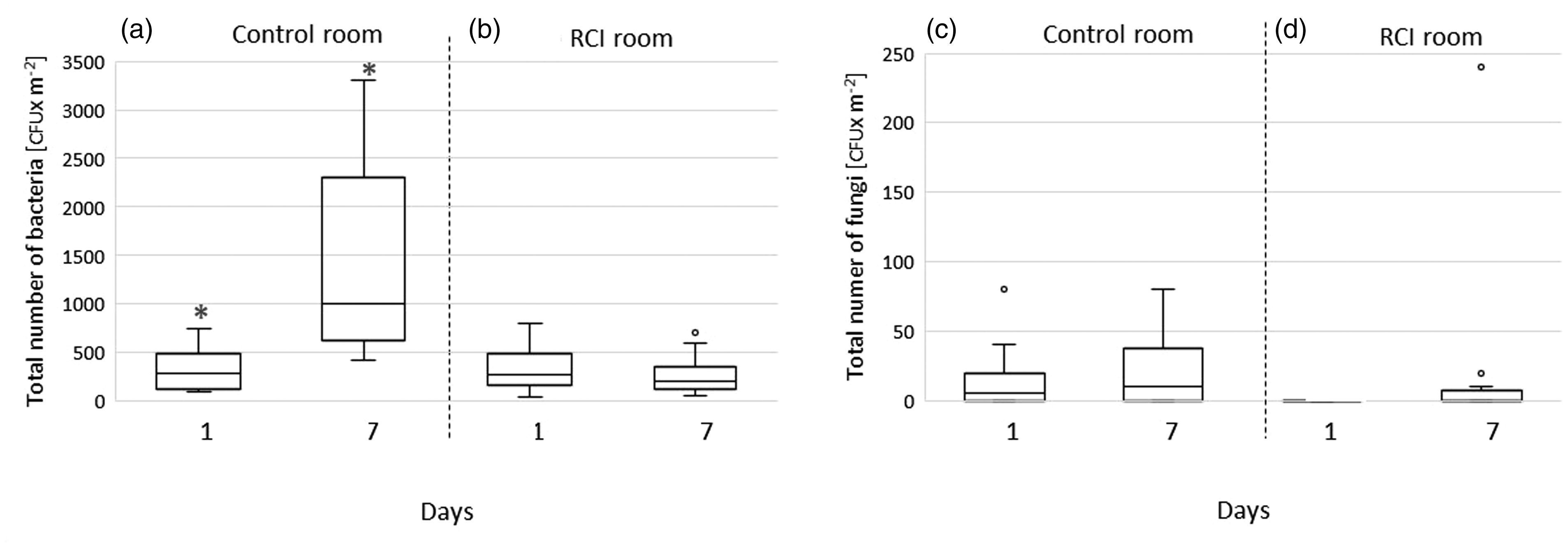
Bacterial and fungal CFUs on control and RCI room floors on the first and seventh days of the experiment.
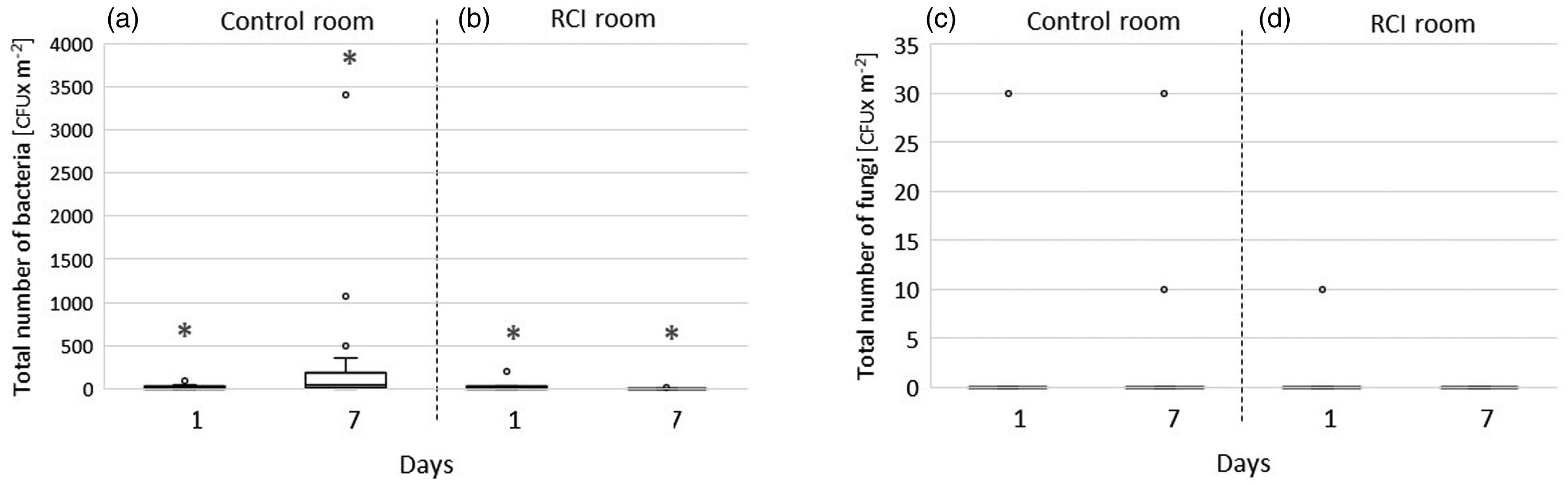
Bacterial and fungal CFUs on control and RCI room walls on days 1 and 7.
Bacterial counts on the floor of the control room were significantly higher on day 7 than on day 1 of the experiment (p < 0.01; Figure 3(a)). Floor fungal counts also increased in the control room during the experiment; however, this increase was not statistically significant (Figure 3(c)). The number of floor bacteria in the RCI room slightly decreased over the seven-day period, but the decrease was not statistically significant (see Figure 3(b)). No effect of the RCI system on floor fungi was observed (Figure 3(d)).
The median number of bacterial CFUs on the walls of the control room on day 7 was significantly higher (p < 0.01) than that on day 1 (Figure 4(a)). No difference was found between medians for total fungal CFUs on the walls between days 1 and 7 in the control room (Figure 4(c)). In the RCI room, the average number of bacteria on the walls was significantly reduced over the course of the experiment (p < 0.01; Figure 4(b)). No changes in wall fungi were observed (Figure 4(d)).
Prooxidative status of mice
TBARS concentrations in mouse blood plasma, liver, brain and lungs (Table 1) were very similar between control and RCI groups. The data were normally distributed with equal variances. No effect of RCI on these parameters was observed.
The effect of RCI treatment of TBARS concentration in the plasma, liver, brain and the lungs of mice.
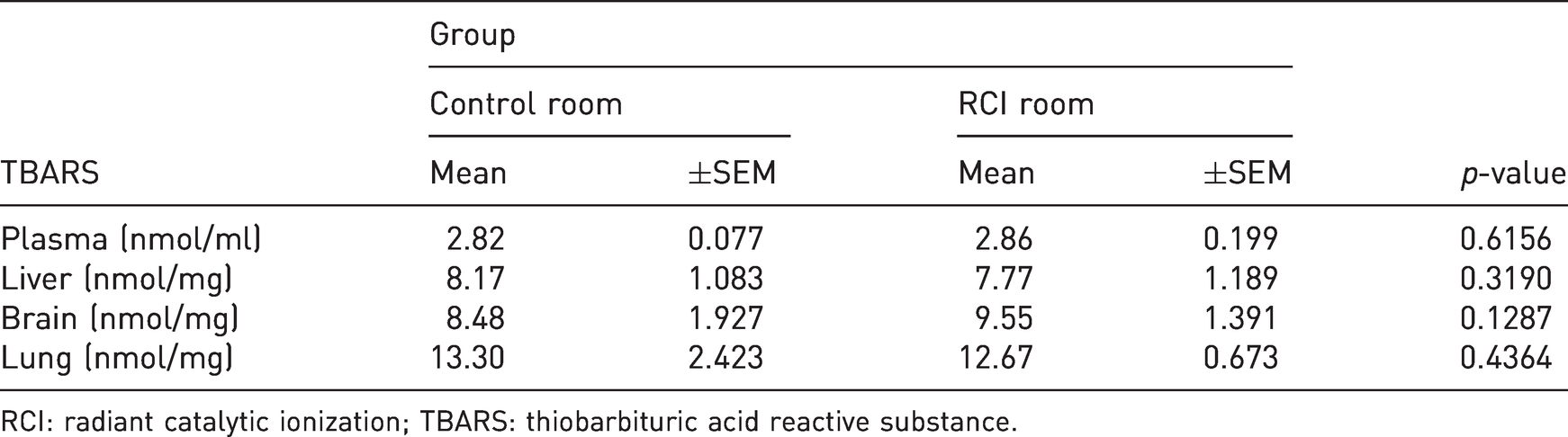
RCI: radiant catalytic ionization; TBARS: thiobarbituric acid reactive substance.
Discussion
State of the microbiological environment
Infectious diseases among laboratory animals have been extensively discussed in the literature, mainly in the context of surgical, respiratory and intestinal tract infections, which are typically spread between animals.25,26 The results obtained in this study definitively confirm the efficacy of the RCI system. Specifically, the RCI system decreased numbers of bacteria and fungi in the air. In the current experiment, after seven days of RCI activity in a conventional animal room, the number of bacteria decreased by 63%, from 684 CFU × m−3 to 256 CFU × m−3. Similar results were obtained when investigating airborne microorganisms using the sedimentation method (unpublished data). Stocks et al. 27 evaluated the use of a system that delivers a small field of local, directed air from a HEPA filter to reduce airborne particulates. A comparison of the maximum CFUs × m−3 in experimental and control space showed that the filtration system decreased the number of bacteria in the air by approximately 82%. At the same time, 60% of the samples from the HEPA room showed a complete absence of airborne CFUs. Kujundzic et al. 28 investigated the performance of a ceiling-mounted HEPA filter with internal UV lamps. Total airborne bacteria were reduced by 12% and 76% after three and 18 months, respectively, with the device being constantly active. The UV source in an RCI unit is either in a device case or in its ventilation system, and is only used for catalytic formation of H2O2. In animal rooms, whole room UV-C is only on when neither animal nor staff are present. In the current investigation, the number of bacteria on the floor and walls of the control room increased by 85% and 94%, respectively, over the seven day period. In the RCI room, the number of bacteria on the floor remained constant, and the number of bacteria on the walls was reduced by 11%. The antifungal effects of RCI on the walls and floor did not register, probably due to the very small absolute number of fungi. The small overall number of fungi was most likely due to regularly performed quarterly disinfections. Such a disinfection procedure is strongly recommended due to the specific structure of the floors and walls in the animal rooms. The presence of grout between tiles provides a very good environment for development of microbial biofilms. Besides disinfections, there are other techniques to maintain low microbial presence on walls and floors. Although they were not applied in the current experiment, it is worth discussing them at this point.
Some specific paints designed for low microbial load rooms contain various biocides to protect surfaces against microorganisms, providing a protective dry film that impedes fungal and algal growth. The benefits of such paints are improved surface aesthetics 25 and a practical limitation on pathogens in livestock buildings. 29 Working examples of paint additives include nanoparticles (e.g. silver (Ag), titanium dioxide (TiO2) and zinc oxide).30,31 Guttman 29 reported that the total number of bacteria growing on a plate coated with paint containing Ag nanoparticles that was placed in a stable for seven days was 65% lower than that on a control plate coated with nanoparticle-free paint. Unexpectedly, the total number of fungi on the Ag nanoparticle plate was higher than that on the control plate. The application of nano-TiO2 stabilized with polyacrylic acid on interior walls resulted in an almost 100% reduction in Escherichia coli CFUs compared with control walls. 32 One disadvantage of nanoparticles in outdoor paints is their tendency to release ionic forms of active substances into the environment. In the case of Ag nanoparticles, approximately 30% of the initial mass of silver is released into the environment annually. 33 Furthermore, the ability to accumulate nanomaterials in animal bodies is well known.34,35
The results obtained in the current study are consistent with those obtained by Skowron et al., 36 who evaluated the influence of RCI systems on microbial contamination of various surfaces. The effects of photocatalytic treatments on fungal cells have been found to be weak and limited by cell type. In addition, studies have revealed that fungal cells and spores are more resistant to photocatalysis than bacteria, which is certainly due to different cell wall properties. 37
Prooxidative status in mice
The photocatalytic RCI system used in the current experiment functions via production of H2O2 at very low doses. 17 It is commonly used for disinfection, and acts by oxidizing organic structures in plant and animal tissues. The H2O2 concentration generated is central to its effectiveness. In every cell of every organism, superoxide dismutase catalyses conversion of superoxide anions to H2O2, reducing the amount of dangerous activity present. 38 H2O2 is a non-radical derivative of reactive oxygen species (ROS) with a long biological lifespan and higher stability than that of free radicals. 39 Moreover, in animal cells, and specifically in phagocytic cells after phagocytosis of bacterial cells, so called oxygen-dependent mechanisms are activated that kill dangerous intruders and microorganisms through lysis. These are non-specific immunological responses in organisms that constitute a first line of defence against pathogens. 40 Similar actions are performed by RCI systems, which emit low dose H2O2 to keep levels of environmental microorganisms low. The safety of RCI application in animal houses was evaluated by assessing the degree of lipid oxidation in tissues.
Studies of the effects of photocatalysis on microorganisms have shown that reactive oxygen species (including H2O2) break down the lipopolysaccharide layer of the cell wall, leading to peroxidation of lipid membranes and oxidation of membrane proteins.41,42 H2O2 is the most effective species for causing cellular injury. 43 ROS are known to damage proteins, nucleic acids and cell membranes, and have also been associated with cancer, aging and several chronic neurodegenerative diseases. 44 H2O2 is potent enough to induce lipid peroxidation in cultured human cells in in vitro models. 45 Peroxidation of cell membrane lipids by ROS leads to production of malondialdehyde (MDA). Lipid peroxidation products are well-established lipid markers of oxidative stress and tissue damage. We estimated MDA levels based on tissue chemical reactivity with TBA.
RCI use for one week did not influence lipid oxidation levels in any tissues studied. This was confirmed by similar TBARS results obtained from control and RCI groups. We conclude that the small doses of H2O2 produced by RCI efficiently decreased numbers of microorganisms, but did not induce adverse effects in mice. Eliminating air and wall pathogens seems to be the best solution for minimizing unpredictable contamination threats from animal house staff. Hence, application of an RCI system as presented in the current paper is a perfect solution to reduce or even eliminate pathogens in animal houses, including those introduced due to human error.
Conclusions
Seven days of RCI system antimicrobial action in a rodent facility significantly improved the environmental status of the experimental room by decreasing the overall number of microorganisms (see Figure 2(b) and (d) and Figure 4(b)). The antifungal air clearing effect of RCI was proven. However, the antifungal effect on surfaces was not, most probably due to the relatively small absolute number of fungi on floors and walls of the experimental room. As the RCI system did not induce adverse effects as assessed by examining lipid peroxidation in a range of visceral tissues, it is concluded to be a safe technique for animals living in laboratory rooms in a photo-catalytically controlled environment (Table 1). Additional studies on the effect of RCI in other tissues, especially nasal mucosae, are planned. Further, RCI has the potential to be successfully applied elsewhere, for example, in livestock buildings with other animals. It may be a very good solution for microbiologically contaminated environments, especially when drug-cross-resistant pathogens are present. Only short-term investigations have been conducted so far; hence, long-term studies are required to confirm the safety of the long-term use of RCI systems in facilities with animals and humans. This requires further studies that should include health indicators, specific for evaluation of the health of organisms living in the environment with ultra-trace concentrations of H2O2 in air.
