Abstract
The absence, in many nations, of appropriate and corresponding legislation for the protection of experimental animals as well as continual management education programs, significantly affects the inclusion and recognition of experimental results, worldwide. For more than a decade, researchers from Latin American countries have unsuccessfully struggled to get proper legislation. Until today, not many effective results have been seen. After reviewing previous literature and carefully analyzing the available methodologies and practical examples, this paper aims at redesigning the actions and strategies of the members of the research facilities to implement an effective laboratory animal care and use program, and permit the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) accreditation, independent of national legislative network .This paper also suggests a domestic working method for the teamwork to assume international harmonized legislation, through the application of the Five Disciplines stated by Senge, as methodological process linked with laboratory animal science as scientific background.
Introduction
The evolution of laboratory animal science and its corresponding regulations in Europe, Canada and the United States provide valuable study material for those who intend to change their animal facility environment. The strategies for applying new scientific–technical knowledge and the trial and error processes have led them to successful research programs within appropriate systems for the protection of experimental animals.
Understanding their experiences and strategies could help other researchers to avoid mistakes and apply creative solutions, taking into consideration the characteristics and peculiarities in developing countries of the Latin American region.
The developed countries apply very similar regulations on the protection of experimental animals. Although they are based on the fundamental criteria, differences in their implementation and control do not influence the essence of the fundamental principles accepted worldwide. 1
The purpose of this paper is to suggest a domestic working method for the teamwork to assume international harmonized legislation for laboratory animal care and implement a management program for human resources, through the application of the Five Disciplines stated by Senge, to reach excellence in laboratory animal protection programs.
In the present article, Directive 2010/63/EU 2 is used as a standard procedure as it is designed to be applied by different countries. Also, its characteristic of conciseness makes it a useful tool.
A brief look at the context
It might be thought that recognition of an ethical and moral responsibility in the use of animals for scientific and educational purposes will move everybody towards efficient legislative processes. Stated principles for the protection of experimental animals from international organizations such as the International Council for Laboratory Animal Science, 3 the Council for International Organizations of Medical Sciences 4 and Organización Mundial de Sanidad Animal (OIE) 5 provide significant criteria on the importance of government consideration for relevant legislation.
Countries from the Latin American region face very complex challenges and experience quite particular and unique conditions in their economic and political environment. However, OIE 5 has reaffirmed the necessity for appropriate legislation in which the governments demonstrate their commitment for the protection of experimental animals. This will be an essential prelude to facilitate national and international scientific collaboration among different countries. For more than 15 years, researchers in the Latin American region have been working on the promotion of an understanding on the importance of this regulation. They have made great efforts to present the proposals to legislative authorities, but only a few of them have achieved this goal.
It is time, perhaps, to modify the approach and demand more attention from the correspondent authorities. A look inside the animal facilities would give rise to some relevant questions and would allow changing the existing strategies.
If, in the hypothetical case, those countries without legislation could have an appropriate legal framework, addressing the following questions, in a short period of time:
Are the animal facilities and personnel prepared to meet the corresponding legislation? At which level of the domestic organization are such facilities? Do they meet the criteria of continuous assimilation of scientific advances and requirements from laboratory animal science and legal regulations? At which level of the external organization has a continuous system review for laboratory animal protection and care culture been implemented?
There are many available documents which identify the requirements to have efficient programs.6–8 They include also specific recommendations for technical training and specific skills for personnel,2,9 supplies for facilities,2,8 mandatory programs and systems2,8,10 and culture of care, 11 but how could these goals be achieved?
During the third OIE World Conference held in Malaysia in 2012, Judy McArthur Clark, 12 Head of the Animals in Science Regulation Unit of the United Kingdom, identified in her lecture three main categories in the evolution of animal welfare legislation between 1876 and 2012. The categories read as follows: evaluation of the justification for animal uses and performing of harm–benefit analysis, the promotion and implementation of the 3Rs; and the certainty of public confidence based on an appropriate balance between quality of science and animal protection.
From the previous considerations, the present work objects are:
Management of human talent; organizational behavior,
13
intelligent organizations
14
and working groups inside the facility;6,10 Collaboration with quality systems existent at the institutions;
7
Application of the instructions described in the Animal Care and Use Program presented by AAALAC,
15
as a tool for evaluating the measures at the domestic level and assessing the progress, prior to external inspections.
Figure 1 represents the influence of human resources management in different systems, as part of the organizational transformation to systemic vision.
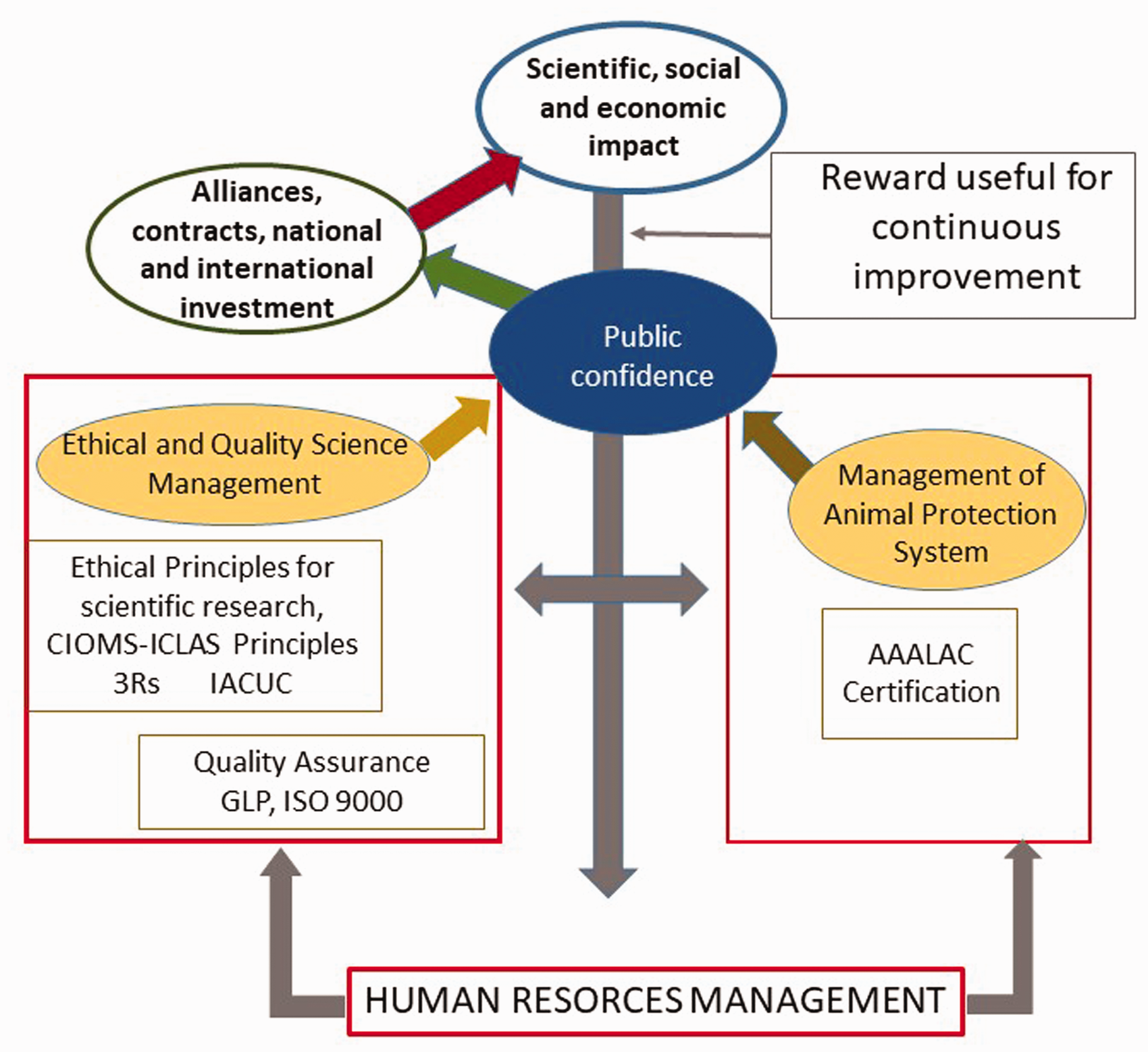
Influence of human resources management on different facility systems.IACUC: IACUC Institutional Animal Care and Use Committee; GLP: Good Laboratory Practice; AAALAC: Association for Assessment and Accreditation of Laboratory Animal Care International; ICLAS: International Council of LaboratoryAnimal Science; CIOMS: Council for International Organizations of Medical Sciences.
The management of human talent and teamwork in animal facilities becomes an important factor for the continuous improvement and transformation of the environment. It should influence both the animal protection system and the program quality system. Quality systems at facilities should collaborate with each other, in spite of the fact that they have specific and different purposes.
The reference documents and understanding the standards
An animal protection program can be defined as ‘activities conducted in and by an institution that has a direct impact on the welfare of animals. They include animal and veterinary care, policies and procedures, personnel and program management and supervision, occupational health and safety, Institutional Animal Care and Use Committee (IACUC) [ethical oversight body] functions, design of animal facilities and their management’. 8 Explicit responsibilities that must be fulfilled for such a program are defined. 11
The animal protection program includes multiple systems and processes that are interrelated and must meet established standards. How will each standard be performed, in the context of Latin American facilities, and how will they be assimilated?
In 2015, Dr P Turner
16
described the interpretation of different standards that users of the National Research Council (NCR)
8
must consider:
Engineering standard. A guidance that specifies in detail a method, technology or technique to achieve an expected result. It does not allow modifications in the procedure, nor acceptable alternatives due to its prescriptive nature. Engineering standards are useful for limited processes. Performance standard. A guidance that describes the desired result while providing flexibility in the scope of the result. When developing a performance standard, it is essential to clearly define the expected result and regularly monitor the results to verify success.
And finally, she emphasized
16
that a well-established performance standard must follow the criteria:
Be supported by objective scientific criteria; Protect the health and welfare of animals; That the expected results have previously associated criteria to evaluate them; The process is regularly monitored.
While strict adherence to engineering standards should lead to an expected result, consideration of a performance standard necessarily involves teamwork. This requires: knowledge by the subject; ability to critically analyze the literature; creation of a mental model; realistic evaluation of the environment; design of performance evaluation criteria for the expected results from the implementation; and finally, a monitoring process that should be carried out by people other than the implementers, to assess that the intended results are being achieved and then to evaluate the efficiency that the new standard has demonstrated retrospectively.
Introduction of a specific score sheet from each protocol, in the established Standard Operation Procedure (SOP), could be an example of interaction between an engineer and performance standards which leads to a qualitative change in animal care management.
A glossary for clinical sign description has been identified as a critical problem of communication inside laboratories and when reporting the results. 10 Each protocol, according to its purpose and procedures, could have specific expected clinical signs and consequently a score sheet to make appropriate end point decisions by. Then SOP, classified according to the concepts of the Principles of Good Laboratory Practice, 17 as one engineering standard, established the procedures for clinical evolution, implementation and use of a glossary of clinical signs.
Teamwork needs to create a space inside the SOP in order to include this variable sheet, according to the protocol characteristics; so that it is, essentially, a performance standard. Thus, it requires to achieve the four criteria mentioned by Turner. 16 When the components are added to the SOP, they are automatically part of the quality assurance review, supervision and corrective action, if any is needed.
Furthermore, they enable good laboratory practice (GLP), as part of the quality assurance process, to indirectly stabilize the process related to refinement and a humane end point. Thus, there is an interrelation between the GLP system and the care and use program, despite their respective essence as systems. A clear comprehension of the different standards permits recognizing options for making improvements.
The previous approach demands reliance upon people trained in complementary activities, such as fluent communication, and the ability of sharing ideas and models and their consequent execution for the effective functioning of the laboratory. That is, changes in the management of human talents are needed to promote the improvement of learning organization. 18
This process of maturing to a learning organization takes place in five steps (Table 1) which Peter Senge called the ‘Five Disciplines’.19,20
The Five Disciplines by Senge.

Senge stated that, ‘System Thinking is the umbrella concept that encompasses all of the other disciplines to enable learning organization to be actualized. It is the understanding of that cause and the effect are connected, being able to see big picture and patterns in organizations’ (Figure 2).
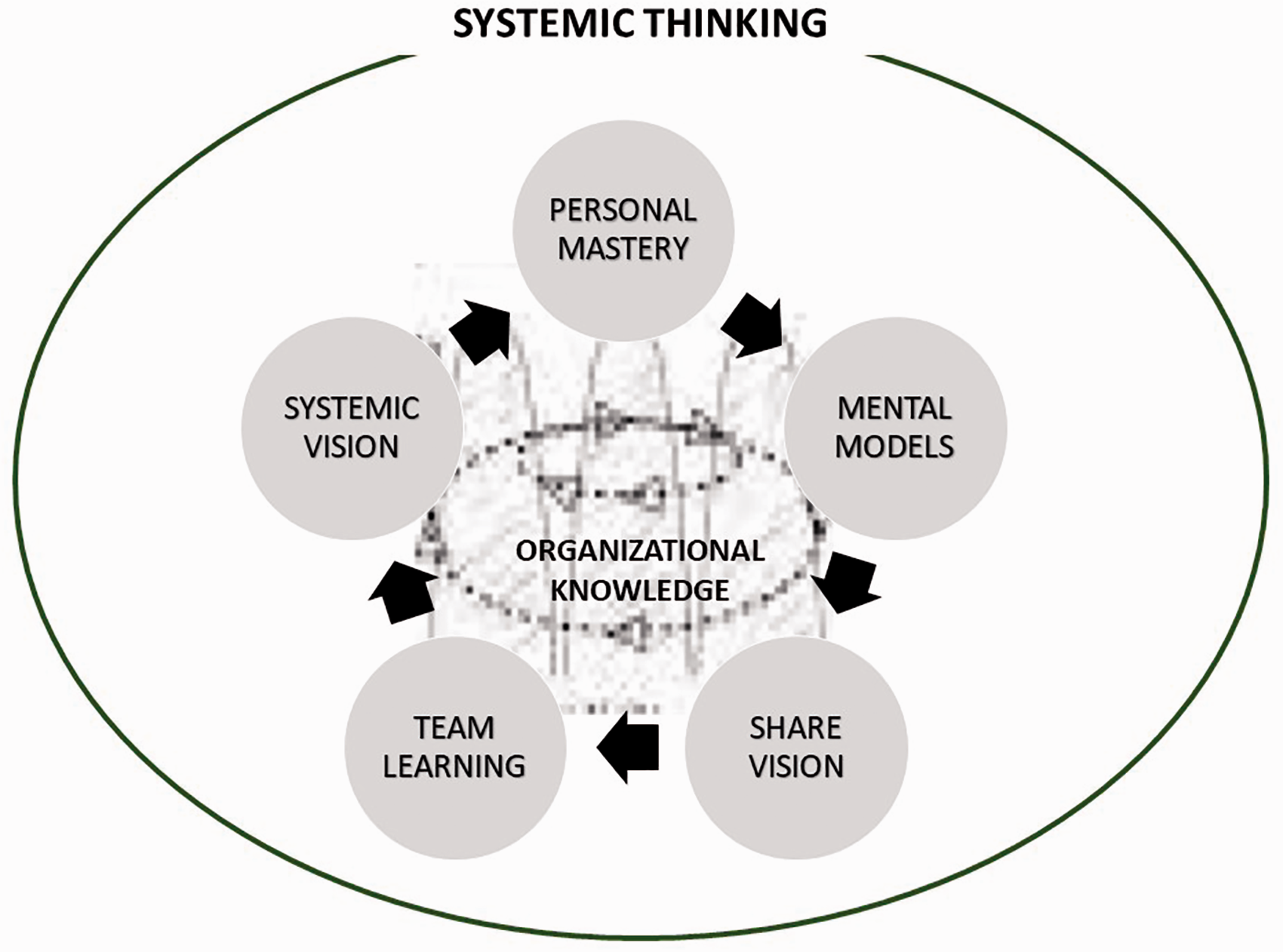
The Five Disciplines (5 DISC) as a cycle.
This process transforms the level of knowledge from personnel knowledge to organizational knowledge. This is a relevant transformation. Laboratory animal science is a young scientific discipline with accelerating growth. The results are usually transformed into committed requirements. Equivalent growth in organizational knowledge is an intelligent strategy to maintain competence at the facility.
Thus, the importance of the following is recognized:
Laws, guidelines and recommendations from expert groups, offering enough information on the requirements of an efficient system of experimental animal protection, and these documents must be part of institutional information. Training programs to establish the theoretical knowledge, skills and abilities needed to properly perform their function. People involved in this activity should be aware of compulsory changes in order to efficiently meet the international standards. Recognition of the opportunities and possible internal collaborations. Interchange of ideas (brainstorming, knowledge transfer, consultation, among others), which permits the improvement of procedures. Transformation of laboratories according to the harmonized international criteria, which allow programs in concordance with the state of the art.
Teamwork is a natural consequence when previous considerations are assimilated. It is the ‘team learning phase’, where role connections and shared responsibilities are identified. Thus, a fluent communication is compulsory among researchers, supervisors, veterinaries, IACUC, technicians and care-takers involved in the process of animal protection.
6. Management of human talent is an essential tool in this process. Shared vision and teamwork are integrated in the culture of animal care conception, while ‘organizational knowledge’ is accomplished as an intangible advantage. 7. When systematic thinking becomes a method of transferring organizational knowledge to action, the organization is considered to have reached the Fifth Discipline.
21
At this level, the organization members of the animal research facility must achieve the ability to understand and handle the different activities inside the facility as a whole, and develop the ability to both recognize the interactions between the parts and implement critical points for the evaluation and improvement.
Systematic thinking helps to constantly update the organization and keep it in harmony with its environment. For this reason, at this stage, ‘the Fifth Discipline’ 22 is considered the base and fundamental characteristic of a learning organization.
Process of clinical evaluation of animals in a research project
Protocol design for safety evaluation of new substance must consider: literature review and in vitro studies, pharmacological and toxicity information, experimental procedures and their refinement options, type of animal model and pilot study results.
Researchers responsible for animal protocol design should have an idea of expected clinical signs. They are responsible for applying the 3Rs criteria23,24 and perform the appropriate assessment of the procedures. 25 Note that the project design group usually includes an animal science specialist, a project manager and a designed veterinary. All of them must have completed the training program required for the functions. Yet that may not be sufficient for effective animal protection; unless the team involved in the study (technicians, care-takers, on-site supervisor, including IACUC) have the proper training and employ the same descriptive language. 10 Thus, the supervisory score sheet 26 and the decision for humane end point would be more effective. In particular, the correct design and clinical sign choices for a scoring system could allow assessment of the real and cumulative severity.25,27 Score sheets must be objective and understood by the personnel involved in the study. Its complexity must not affect efficiency or it will be too awkward for technicians; 6 and it must allow retrospective evaluations.
Collective analysis improves score sheets and simultaneously exercises communication and contributes to increasing the organizational knowledge. It is important to promote informal debate to create opportunities for sharing ideas, improving communication and the learning process as teamwork.
The quality assurance program 28 is a defined system inside GLP, personnel included. As previously mentioned, it could be an excellent component to stabilizing the culture of care.
Figure 3 describes the combination of laboratory animal science as scientific platform and the Five Disciplines as methodological process to achieve excellence.
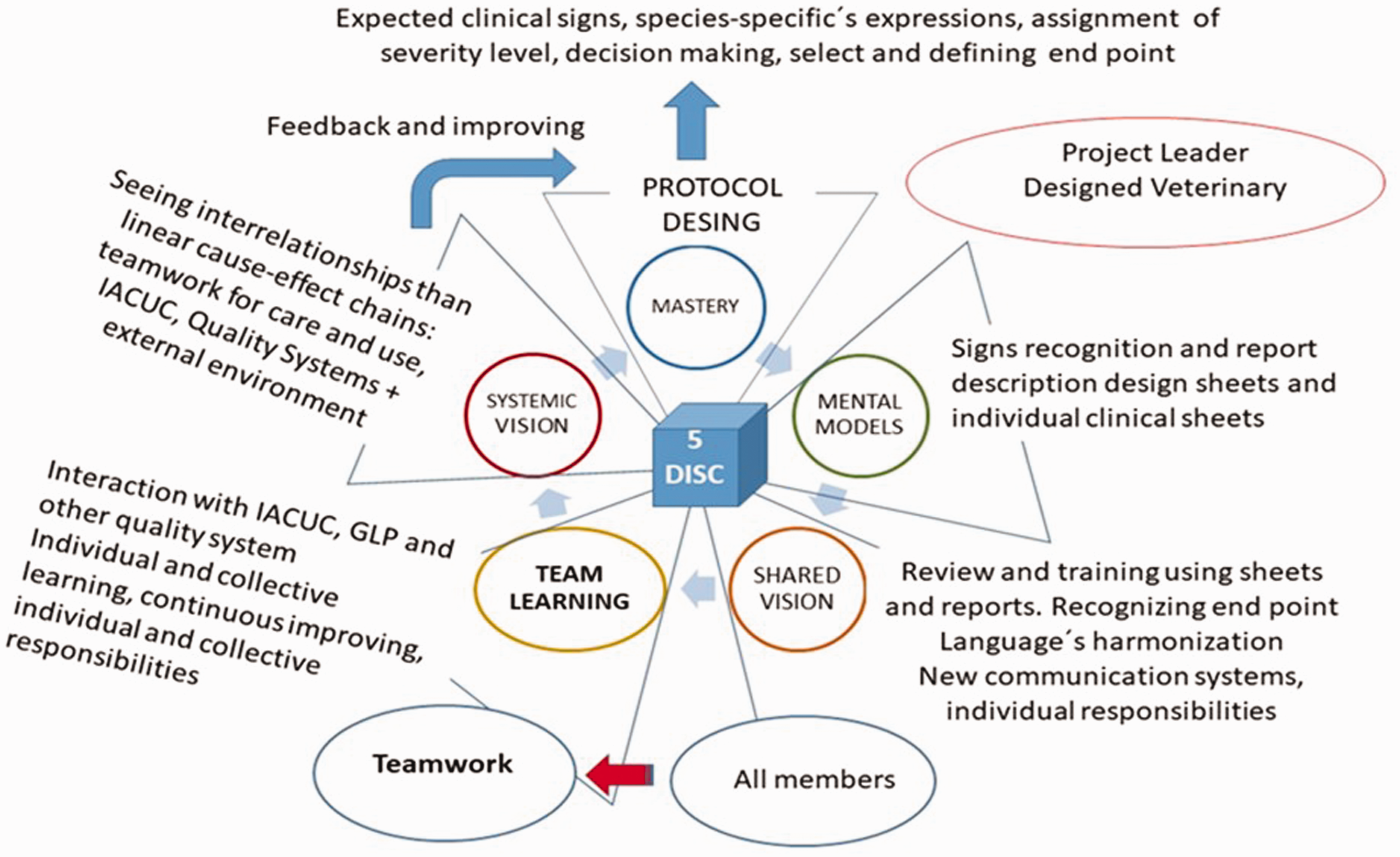
Example of Senge’s Five Disciplines in action in one animal facility.IACUC: Institutional Animal Care and Use Committee; GLP: Good Laboratory Practice.
The inclusion of researchers during project design and IACUC during protocol revision is mandatory in the analysis of each procedure of the project, refinement and analysis of harm–benefit balance.29–32 Their work should be focused on the 3R alternatives, the literature search and the discussion of the cumulative suffering of animals, in each protocol. All these components are susceptible to the Five Disciplines.
The AAALAC Instructions for completing and submitting the program description for the institutional animal care and use program 15 should be periodically applied in the analyses of each component of the animal care program, to gradually identify items to be improved and the application of the Five Disciplines to each identified aspect. This procedure is valid for physical plant, veterinary care, occupational health, safety of personnel and other activities. The program will be completed once every component is revised and improved, as well as achieving better quality in human resources.19,21
European Directive 2010/63/EU
2
establishes two types of individual that can contribute in the master programs for the personnel:
Persons responsible for the information on the species housed in the establishment (article 24, paragraph 1, letter b); Persons responsible for ensuring adequate training and access to continual professional development (article 24, paragraph 1, letter c).
The selected individuals should be identified within the staff of the center and their duties should be included in the description of their respective working plans. It is essential that the personnel with greatest responsibility for animal care program quality and success (i.e. the veterinarian, the IACUC chairman, the technical managers and supervisors) receive high quality training to guarantee a full understanding of the program content. All of them working together would allow the teamwork to create an intelligent organization.
An animal care and use program has authoritative command for precise training and skill.2,9 Websites such as Animalaria (http://www.animalaria.org), NCR3 (https://nc3rs.org.uk), Norecopa (https://norecopa.no/education-training/films-and-slide-shows), AALAS (https://www.aalaslearninglibrary.org), Bioterios.com (http://www.bioterios.com), Canadian Council on Animal Care (https://www.ccac.ca/en/training), UFAW (https://www.ufaw.org.uk), Taconic (https://www.taconic.com), among others, provide important teaching resources that permit the creation of domestic training programs and on-site courses.
Domestic courses generated in labs should be recognized by their local universities as postgraduate programs and by their national society as part of continuing education program in National Society for Laboratory Animal Science. The promotion of and public access to these teaching activities and open lectures will increase consciousness in the population.
Discussion
Most of the papers from groups of experts implicitly suggest the necessity of achieving an intelligent organization that meets the international standards. In some papers the Five Disciplines are also suggested.
Lloyd et al. 33 explained that refinement in acquired knowledge and mental processes, applying mode of thinking and application to objective reality, could promote great and valuable changes. This process transforms unconscious incompetence to conscious competence, and mastery. They expressed that dissemination of information and shared vision are essential for the implementation of the 3Rs culture of care.
On the other hand, Lilley et al. 34 considered the development of care culture from the vision of the ‘working group’, in which openness and exchange of information form the fundamental basis for domestic exchange and strengthening of associated programs and responsibilities. A similar criterion is described,6,10,11 in which the team approach and collective actions are considered relevant to meet the appropriate standards.
The framework provided by the European Union identifies different levels and steps in the severity evaluation for the teamwork. Specifically, in retrospective evaluation, the systemic thinking discipline could be seen in the expressed intention of feedback improvement and opportunities for communication and offers transparency.
Love of animals and the necessity of a continual learning process will be the first motivation to start the spiral of change. The comprehension that to reach an organization of excellence requires that everyone has the opportunity to participate and contribute, 22 rather than to perform with quality individual specific responsibilities, could be a strong incentive. However, it is inevitable that some negative aspects could emerge during the process of change.
Organizational behavior in a research facility presents other complexities 13 that have influence during the process of transformation. Lloyd and collaborators 33 have identified obstacles which affect the implementation of the animal care culture and Senge, in his papers,21,22 has advised about searching for and understanding the original causes of such resistances and obstacles. He also provided suggestions that are useful in the animal facility context.
Conclusions
The Latin America region faces environmental and health challenges; hence it is important to achieve new strategies to eradicate the current restrictions that prevent the development of a legal framework for laboratory animal protection. Non-compliance absolutely limits national and international cooperation and acceptance of the research results. Information and methodological tools, accessible training and learning lessons are available to find and to create solutions.
Clearly evident, in many Latin American countries, is the crucial concern to have an animal welfare legal network in order to achieve the appropriate regulations. At the same time, other strategies should be developed that guarantee high quality animal care and use programs, and a sustained movement on behalf of the laboratory animal science communities.
Footnotes
Acknowledgments
Thanks are due to Dr Cynthia Pekow and reviewers for their kindly review and advice. Thanks also to specialist Janet Perodin Hernández, translator and editor, for her gentle review and suggestions.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from LA Ltd (LAL BA 371 2017 and LAL BA 475 2018) and ICLAS (ARC 2016 and ARC 2019) that allowed members of the Cuban Society of Laboratory Animal Science to exchange experiences with recognized experts. This exchange also permitted a wide vision and comprehension for the new alternatives to develop culture of care in our context.
