Abstract
Haematogenous models of septic arthritis have some inherent disadvantages, such as the manifestation of arthritis relies on chance, the size of the inoculum is unknown and the number of animals to be studied cannot be reduced because the animals cannot serve as their own controls. This study aimed to develop a rat model of knee septic arthritis by injecting a known inoculum of
Introduction
Septic arthritis is joint inflammation caused by the presence of one or several pathogenic micro-organisms in the joint. Rapid diagnosis and treatment are required because delays can lead to more joint damage and even higher mortality rates.1,2 The incidence rates of septic arthritis are increasing; for instance the incidence of knee arthritis has increased fivefold since the second half of the 20th century, 3 and multi-resistant pathogens are being increasingly implicated.4,5 Therefore, it is important to search for new treatment alternatives, which usually have to be tested in animal models first.
Many rodent models of septic arthritis are based on the injection of pathogens through the haematogenous route,1,2,6–11 but these models have several obvious disadvantages. For instance, it is impossible to anticipate precisely which joint will become infected, even if the targeted joint is previously damaged to improve the success rate. 12 The bacterial load reaching the joint cannot be predicted and remains unknown. 12 In addition, animals are exposed to more suffering or even death when they suffer from a generalised septic process.1,9
Theoretically, all the above-mentioned shortcomings could be avoided by inoculating the pathogen directly into the targeted joint. Knee septic arthritis by direct inoculation has been studied mostly in rabbits,13–17 as well as in hamsters, guinea pigs 18 and mice, 9 but studies on rats are lacking. In one study in rats aimed to produce bacterial endocarditis after causing septic arthritis by direct inoculation, the joint selected was not the knee but the temporomandibular joint. 12
The main goal of this research was to develop a new model of septic arthritis by direct inoculation of a strain of
Methods
All experiments were performed following the European Directive (2010/63/UE) and Spanish law (Real Decreto 53/2013) for the protection of animals used for scientific purposes. The animal experiments were approved by the Animal Experimentation Ethics Committee of Castilla-La Mancha University (PR-2016-11-17). The ARRIVE reporting guidelines were followed. 19
Animals
Thirty-six adult male and female Sprague Dawley rats were obtained from colonies established at the authors’ animal facility. The two pairs of founders were purchased from a commercial breeder (Charles River Laboratories, Barcelona, Spain). As recommended by the Federation for Laboratory Animal Science Associations, rats in our animal facility are tested annually to ensure that the colony remains free of pathogens.
The sample was made up of 19 males, aged 3–5.5 months and weighing 382–481 g, and 17 females, aged 3.5–10 months and weighing 235–429 g. Animals were housed in groups of two to three individuals per cage in climate-controlled individually ventilated, type 1500U cages (Tecniplast, Varese, Italy) containing aspen-chip bedding material (2HK10Kg; Tapvei, Harjumaa, Estonia), with a 12-hour/12-hour light/dark schedule. They were maintained under controlled ambient conditions (temperature: 22±2°C; relative humidity: 30–70%) and had access to water and standard rat food ad libitum (Envigo Laboratories, Barcelona, Spain).
Study design
This research consisted of two consecutive experiments in which the initial procedures were modified as needed to find the final model (Figure 1). The first pilot study was aimed at creating a preliminary model. Nine animals were used: five to learn the inoculation technique, and another four to test the validity of the model. The second experiment – the main study – was aimed at creating the definitive model. A total of 27 rats were used, which were grouped according to inoculum size. For all experiments, invasive procedures were performed in the morning, and the rats were euthanised in a CO2 chamber after the experiments. The chamber was filled with CO2 at a low rate, 30% of chamber volume per minute. Animal welfare was supervised daily by a veterinary. Clinical variables were assessed by two researchers who were aware of infected knees, whereas microbiological and histological variables were assessed by a researcher who was blinded to the procedures performed.
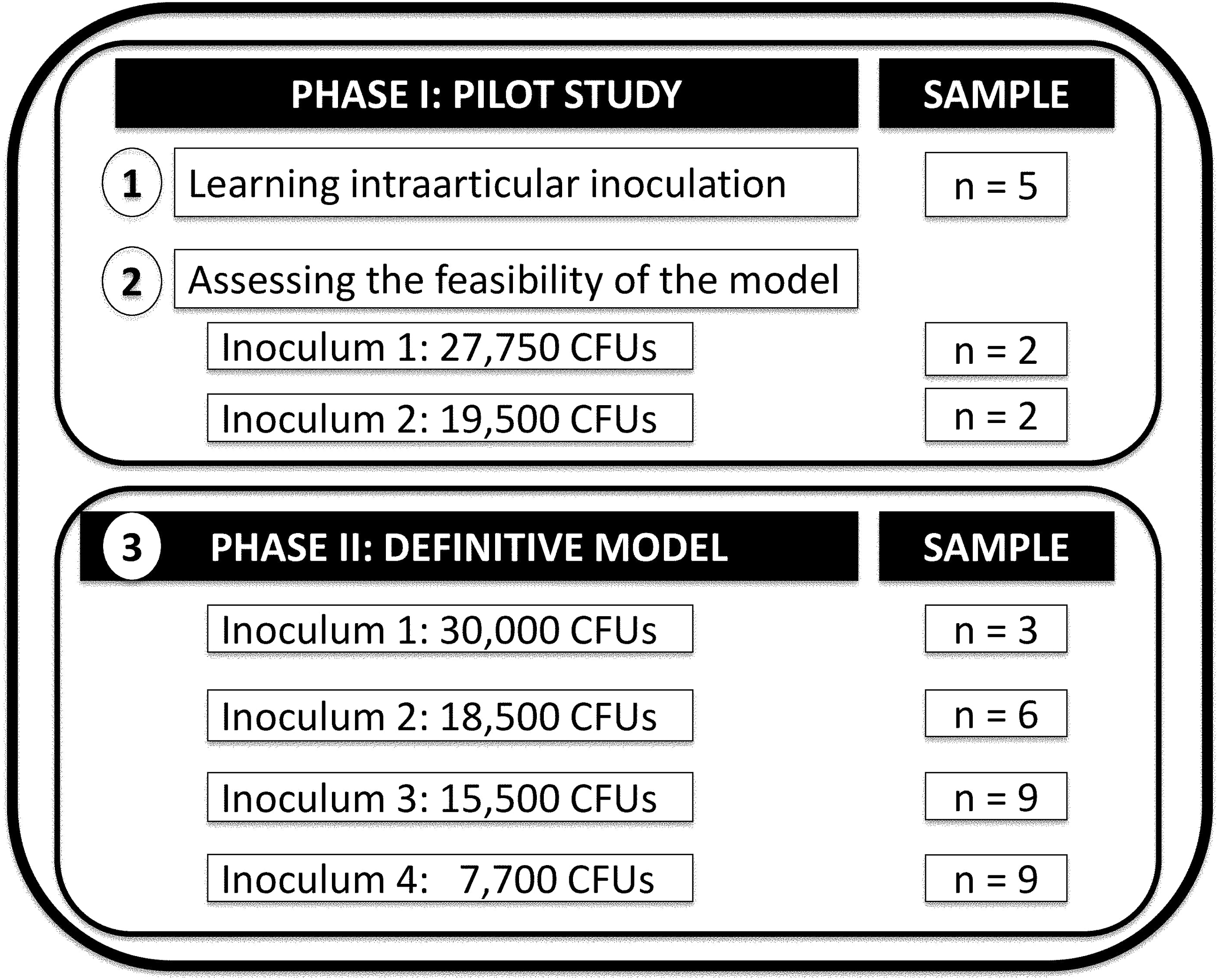
Distribution of the 36 rats in the experimental groups.
Anaesthesia
Anaesthesia was induced with a mixture of 5% vol. of sevoflurane in 2 L/min oxygen for four to five minutes and then maintained with 2–3% vol. of sevoflurane in 1 L/min oxygen. All invasive procedures were performed under general anaesthesia and analgesia (a single dose of 12.5 mg/kg of intraperitoneal tramadol prior to each invasive procedure), including arthrocentesis and blood extraction from the heart.
Microbiological procedures
Preparation of inocula for injection
An oxacillin-sensitive strain of
Sample collection for cultures: synovial fluid
Arthrocentesis was performed under aseptic conditions, and the right knees (controls) were punctured before the left knees (cases). Intra-articular synovial fluid was obtained by aspiration through a 27G needle and plated on a sterile surface. Then, a 1 µL sample was collected with a calibrated loop and cultured in blood agar medium. 14 Samples of synovial fluid from both legs were cultured in liquid enrichment medium (thioglycollate broth), and the results were qualitatively expressed as positive or negative. Samples from the left legs (1 µL) were also cultured in solid medium (blood agar), and the results were quantitatively expressed as counts of CFUs/mL.
Sample collection for cultures: blood
To study bacteraemia, 1.5–2 mL of blood was drawn from the heart after puncturing it with a 25G needle (preliminary and definitive models), or 0.5 mL was drawn from the tail vein through a 23G needle (definitive model). Blood samples were always cultured in aerobic conditions for 10 days, and the results were qualitatively expressed as positive or negative.
Clinical assessment
Limp
Two researchers assessed the limp daily by observing animals walking for five minutes. According to the percentage of non-weight-bearing steps when walking, the limp intensity was classified into two categories: mild or moderate to severe. Discrepancies were resolved by agreement between the same observers.
Knee diameter
In addition, mediolateral diameters of 40°-flexed knees were measured in millimetres by using a calibrated calliper at day 0 and at sacrifice (day 2 or 3, depending on the study phase).
Animal welfare
The clinical assessment was based on the general appearance of the animal, body posture, mobility (limp), body weight, coat and grooming, ocular appearance, indications of self-harm (joint knee) and behaviour and sociability. Humane end points considered were as follows: weight loss >20%, severe lethargy, very rough coat/piloerection or excessive chromodacryorrhea.
Histological procedures
Sample collection
Knee joints were stored after fixation with 4% formaldehyde. The joints were decalcified for 10 days in a solution with 15% formic acid. On day 7, they were longitudinally sectioned to facilitate the decalcification process. After decalcification, the samples were embedded in paraffin. Then, histological sections 3 µm thick were cut and stained with hematoxylin and eosin.
Histological variables
One pathologist assessed the following histological variables: (a) synovial membrane hypertrophy, categorised as none, slight, moderate or intense; (b) cartilage damage, expressed as present or absent in the pilot study, and later expressed as a percentage in the definitive study; and (c) bone damage, expressed as present or absent. The presence of neutrophils as indicators of acute inflammation was categorised according to the number of neutrophils per high magnification field as none, slight (<500 cells), moderate (501–1000 cells) or intense (>1000 cells). The presence of lymphocytes as indicators of chronic inflammation was categorised according to the number of lymphocytes per high magnification field as none, slight (<250 cells), moderate (250–500 cells) or intense (>500 cells).
Diagnoses of acute septic arthritis
For this study, the main microbiological diagnostic criterion for confirming the development of acute septic arthritis was the growth of
Experiment 1: pilot study – preliminary model
Nine animals were used for the pilot study. No statistical analysis was performed for this phase, and individual results are shown, along with their methodological description (Table 1).
Preliminary model.
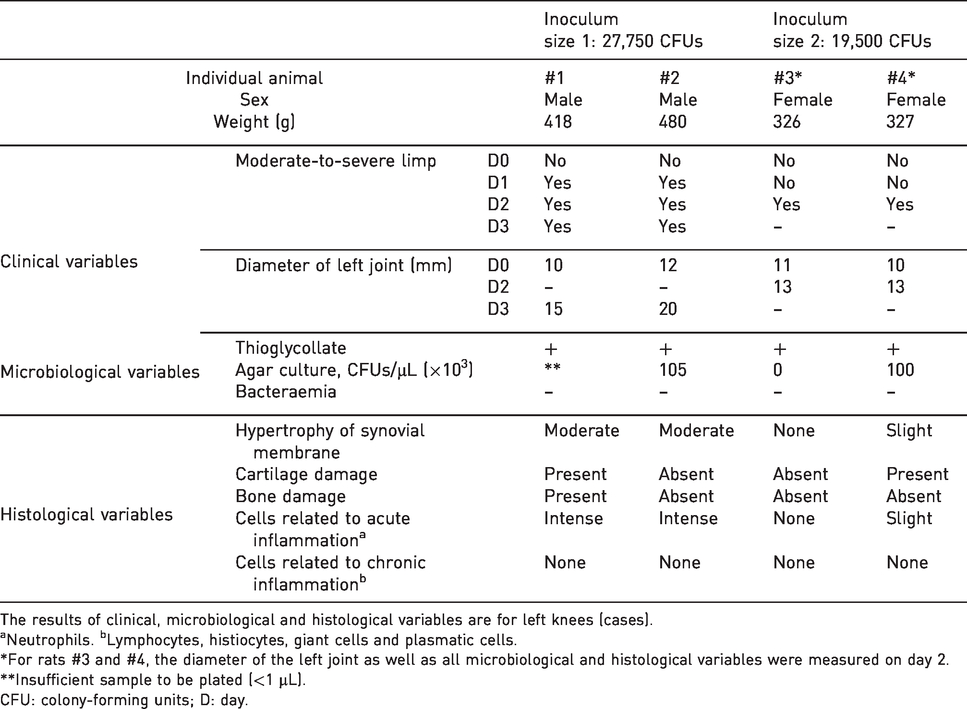
The results of clinical, microbiological and histological variables are for left knees (cases).
aNeutrophils. bLymphocytes, histiocytes, giant cells and plasmatic cells.
*For rats #3 and #4, the diameter of the left joint as well as all microbiological and histological variables were measured on day 2.
**Insufficient sample to be plated (<1 µL).
CFU: colony-forming units; D: day.
Intra-articular puncturing for injection and extraction
The goal was to learn the skills needed to puncture the knees properly for both inoculating the micro-organism and obtaining synovial fluid. For this, 10 knees corresponding to five animals were punctured. After shaving the knees, they were punctured with a 30G needle at the level of the external upper third of the kneecap, near the suprapatellar bursa.14,17 The needle was positioned perpendicular to the leg axis and parallel to the ground. Once punctured, methylene blue-stained saline was gently and progressively injected until a sharp fall in injection pressure occurred, which was attributed to the rupture of the articular capsule. The volume injected until then was assumed to be the maximum allowed intra-articular volume. In addition, a surgical dissection was performed to confirm that the inner part of the joint was blue-stained, indicating that the tip of the needle had been placed into the intra-articular space. By following this technique, knee injections were successfully achieved in all 10 attempts, and capsular rupture was considered to occur after the injection of a volume of approximately 250 μL.
Preliminary models of septic arthritis
The goal was to test the feasibility of producing septic arthritis by direct inoculation of a known inoculum of
Experiment 2: main study – definitive model
The goal was to find an inoculum capable of producing septic knee arthritis without causing bacteraemia severe enough to impair the general condition of the animals during the experiment. Starting with the highest inoculum, each inoculum was tested sequentially in groups of three animals until a total of nine animals had been reached. In the case that the inoculum caused severe impairment to the animals, no more animals were exposed to it, and a lower inoculum was selected for further experiments. Clinical variables were assessed on days 0, 1, 2, 3, 4 and 7, and microbiological variables were assessed on days 2 and 7. Blood samples for bacteraemia were drawn on day 2 from the tail vein and on day 7 from intracardiac puncture before euthanasia. On this day, samples for the histological study were also obtained.
Statistical analysis
The results are shown as median and absolute ranges or as absolute values and percentages, as appropriate. The non-parametric Mann–Whitney
Results
Flow chart of study groups according to inoculum size
Four inoculum concentrations were tested in descending order in a total of 27 rats. Just like the preliminary model, the first inoculum was 30,000 CFUs (later determined to actually be 30,000 CFUs).2,14 This inoculum was discarded after the first attempt because all three rats suffered from severe clinical impairment. Thus, group 1 comprised three rats.
The second inoculum selected was 20,000 CFUs (actually 18,850 CFUs). It yielded favourable results in terms of animal welfare and the rate of septic arthritis, but after two sets of experiments, it was found to produce a high rate of bacteraemia. Therefore, we decided to lower the inoculum concentration further in an attempt to reduce the rate of bacteraemia. Therefore, group 2 comprised six rats.
The third inoculum selected was 15,000 CFUs (later determined to actually be 15,500 CFUs). Since there was no reason to stop the experiments with this inoculum, group 3 comprised nine rats as planned. Compared to group 2, the rate of bacteraemia was lower, whereas the rate of arthritis, although slightly lower, was assumed to be acceptable.
The fourth inoculum was intended to be 7500 CFUs (later determined to actually be 7700 CFUs) in a final attempt to reduce the rate of bacteraemia, but it failed. Besides, the clinical symptoms of arthritis appeared to abate spontaneously during the study week. So, it was decided not to test additional lower inoculum concentrations. Group 4 also comprised nine rats.
None of the rats reached the pre-established humane end points.
Clinical variables
Baseline characteristics were similar among groups as well as the weekly evolution of weight and diameter of the left joints (Table 2). All groups showed weight loss and a noticeable increase in left joint diameter in the experiment (Figure 2). All rats in groups 1 and 2 suffered from a moderate-to-severe limp at days 2 and 7. Limp rates were lower for group 3 and 4, but the differences were not statistically significant (Table 2). Both observers shared the clinical impression that rats in group 4 experienced a higher degree of spontaneous recovery than rats in any other group. Overall, rats in which arthritis was later diagnosed by microbiological criteria presented consistently with a moderate-to-severe limp, and their left joints increased to at least 3 mm in diameter.
Clinical variables of the definitive model.
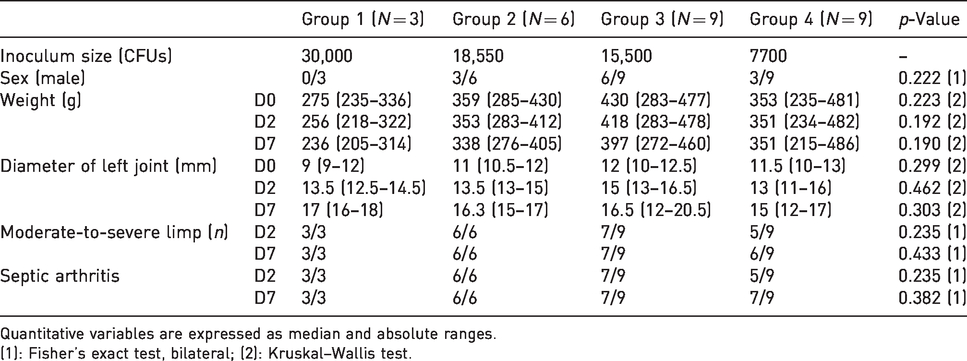
Quantitative variables are expressed as median and absolute ranges.
(1): Fisher’s exact test, bilateral; (2): Kruskal–Wallis test.
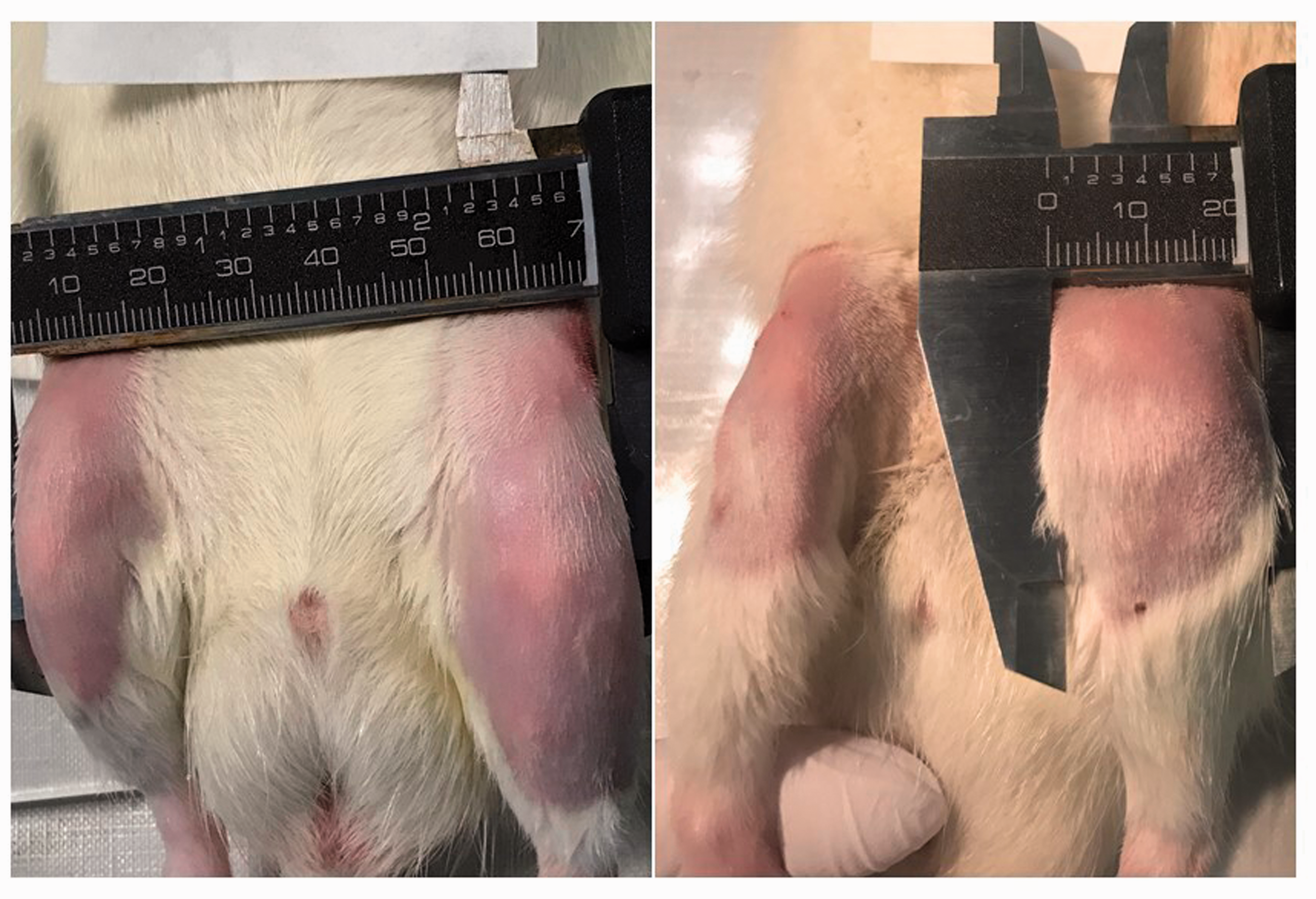
Inflammatory aspect of the left knee (case) compared to that of the right knee (control) on day 2 (left) and day 7 (right).
Microbiological variables
Differences between groups were not statistically significant at any time point for any microbiological variable (Table 3). No bacteraemia was found on day 2, but on day 7, all groups showed animals with bacteraemia. Regarding synovial fluid, the CFU count in all groups showed a noteworthy reduction from day 2 to day 7, except for group 1. Thioglycollate cultures were positive for most animals that could be evaluated on day 2 (three samples in group 4 were lost); cultures were positive for all rats in groups 1 and 2, while the number of positive cultures was slightly lower for groups 3 and 4. Collectively, the diagnosis of acute septic arthritis was made for all animals, except for three of them (one animal in group 3 and two in group 4).
Microbiological variables of the definitive model.
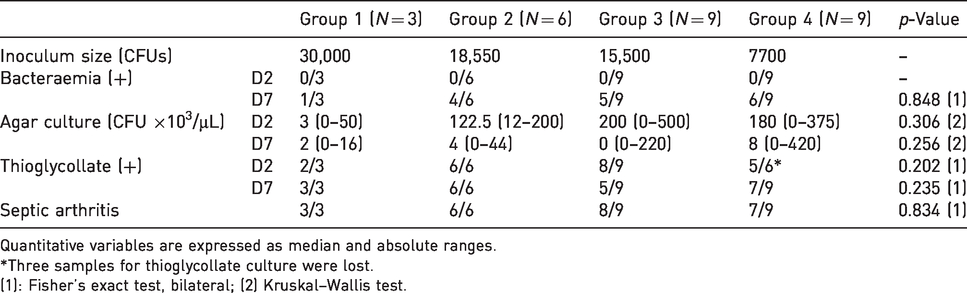
Quantitative variables are expressed as median and absolute ranges.
*Three samples for thioglycollate culture were lost.
(1): Fisher’s exact test, bilateral; (2) Kruskal–Wallis test.
Histological variables
Histological study showed no evidence of damage affecting the right knees, suggesting that arthrocentesis performed on day 2 did not result in permanent damage to these control joints (Table 4). In contrast, the left knees were damaged. Remarkably, the synovial membrane, cartilage and bone were found to be severely damaged in all joints in which septic arthritis was confirmed according to microbiological criteria. Taking into account all the above, the inoculum intended to be 15,000 CFUs (actually determined to be 15,500 CFUs) was selected as the most suitable to produce septic arthritis reliably while preserving animal welfare.
Histological variables of the definitive model.
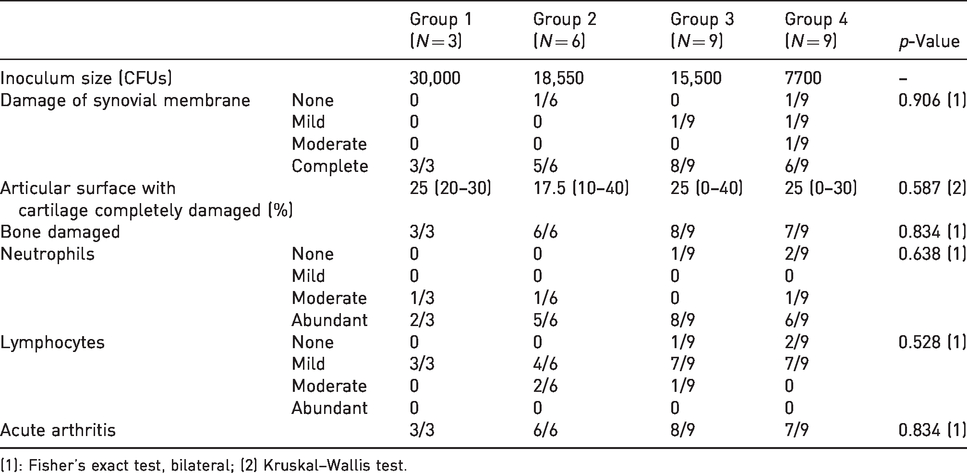
(1): Fisher’s exact test, bilateral; (2) Kruskal–Wallis test.
Level of agreement between the diagnostic criteria
On day 2, both clinical and histological criteria for arthritis showed a significant association with microbiological criteria (Fisher’s exact test, with
Discussion
This study shows that acute septic knee arthritis can be produced in rats by direct inoculation of a strain of
These limitations inherent to haematogenous models could be easily overcome by direct injection of a known inoculum into a selected joint. 20 However, most intra-articular models of arthritis have been developed for rabbits.6,13–18 Concerning rodents, a few models have been developed for hamsters and guinea pigs, 21 but rats were lacking such models until now. Perhaps the scarcity of intra-articular models for rodents could be explained by the anticipated technical difficulties in puncturing the joints of small animals such as rats. However, rats are the most frequently used animals in biomedical research, especially concerning models of infection and toxicity, 22 making it necessary to create such a model of intra-articular injection.
Provided the technical difficulties are overcome with practice, intra-articular models have several inherent advantages compared to haematogenous models: it is possible to select in advance both the specific joint to be infected and the bacterial load to be inoculated; the sample size can be reduced to half, since contralateral joints serve as controls; and animal welfare would be better, as bacteraemia rates inherent to haematogenous models could also be reduced in intra-articular models.
In this study, septic arthritis was precisely diagnosed. Usually, the diagnosis of septic arthritis has been based on clinical criteria such as swelling, 3 as arthrocentesis and culture of synovial fluid were not routinely performed in previous haematogenous models. In this study, a more precise diagnosis of arthritis was possible because synovial fluid obtained through arthrocentesis was cultured, and CFUs were also counted. Interestingly, microbiological criteria of arthritis showed a significant level of agreement with clinical and histological criteria, although the agreement was lower for clinical criteria. Expressed in pragmatic terms, a rat presenting with a moderate or intense limp at day 2 after inoculation is expected to truly suffer from acute septic arthritis. This could be of great interest to researchers testing drugs for the treatment of this pathological condition.
Our model is highly reproducible. Previous experimental models of arthritis were frequently developed by inoculating clinical strains of a variety of micro-organisms,2,14,17,18 which made it difficult to reproduce the experiment under the same conditions.1,5 On the contrary, we selected a single ATCC strain of
Finding an appropriate inoculum size was more laborious. In rabbits, a bacterial load between 140 and 400 cells has been proven to be high enough to produce septic arthritis. As only 10% of the initial inoculum was found to remain after 10 minutes because of the rapid articular bacterial clearance in rabbits, it is necessary to inject the joint with an inoculum size at least 10-fold higher than that needed to produce arthritis.
14
As for rats, bacterial clearance should be as rapid as that in rabbits if not more rapid and, besides, rats are usually resistant to infections caused by staphylococci,
22
making the inoculum size needed more difficult to predict. We aimed to find an inoculum size that was large enough to cause arthritis but which, at the same time, did not produce bacteraemia or interfere with animal welfare. However, the amount of information available was deemed insufficient to help us make such a decision because, as stated before, no model of acute knee arthritis in rats had been developed by direct injection of a strain of
In rodents, three models of knee arthritis produced after inoculating a strain of
This first inoculum was effective in producing acute septic arthritis, but it was discarded because associated animal suffering was considered unacceptable, and the same was true for the second inoculum. The lower inoculum studied, the fourth one, did not impair animal well-being, but it was also discarded for a completely different reason, as arthritis tended to self-heal during the week of study. Such a trend towards self-healing was thought to have the potential to interfere with the evaluation of drugs intended to heal the infection, so it was discarded. As a result, the third inoculum was selected.
Intra-articular models face some technical difficulties. In our model, we found different technical difficulties depending on the knee punctured. The right knees served as controls. The identification of the joints was very easy because there was no tumefaction. However, taking synovial fluid from these knees to be cultured was found to be a difficult task because the intra-articular fluid was sparse. The fluid obtained was haematic on all occasions, which was attributed to traumatic manipulation (even though the joint was not found to be damaged in the posterior histological analysis). The left knees were the cases, and soft tissues surrounding the knees were swollen. Identifying the joint was more difficult, but this problem was solved with practice. Once the joint was punctured, there were two different scenarios. In the best case, the intra-articular fluid was easily aspirated, since it was usually abundant. In the worst case, the intra-articular fluid was sparse, dense and haematic, making aspiration a difficult task. In such cases, later CFU counts were lower than expected. The progression of the septic process may have damaged the capsule, leading to fluid leakage, as this scenario was more frequently found at day 7 and for joints injected with higher inoculum concentrations.
Our model has two main limitations. First, the sample size is small, but this is a common limitation inherent to most animal research conducted following the 3R principles. Second, the clinical diagnosis of arthritis was mainly based on the presence of a limp. Unlike objective quantification of CFUs count, assessment of the degree of the limp is always somewhat subjective, and in addition, there was no previous research to guide us on how to categorise the limp.23,24 Despite the inter-observer variability, rats presenting with a mild-to-moderate limp from the third day onwards were later found not to suffer from arthritis. Thus, such categorisation seemed to be useful to distinguish between infected and non-infected rats.
Conclusion
Acute septic arthritis can be caused in rats by inoculating 15,500 CFUs of an ATCC strain of
Footnotes
Acknowledgements
The authors wish to thank Fernando Andrés Petrel (Clinical Research Support Unit, General University Hospital of Albacete, Spain) for his invaluable help with the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
